Abstract
The effects of human amniotic fluid stem cells (hAFSCs) transplantation on bladder dysfunction after pelvic nerve transection (PNT) remain to be clarified. Five groups of female Sprague-Dawley rats were studied including sham operation, unilateral PNT alone or plus hAFSCs transplantation, and bilateral PNT alone or plus hAFSCs transplantation. hAFSCs were injected at the site of PNT. Cystometries, neurofilament density within bladder nerves, and the expressions of bladder protein gene-product 9.5 (PGP9.5), growth-associated protein 43 (GAP-43), nerve growth factor (NGF), p75 (NGF receptor), CXCL12, CCL7, and enkephalin were studied. Compared to sham-operation group, bladder weight increased and neurofilament density decreased at 10 and 28 days after unilateral and bilateral PNT, but all improved after hAFSCs transplantation. Unilateral PNT could increase bladder capacity, residual volume, and number of nonvoiding contractions but decrease peak voiding pressure and leak point pressure. Bilateral PNT caused overflow incontinence and increased the number of nonvoiding contractions. These cystometric parameters improved after hAFSCs transplantation. After PNT, bladder PGP9.5 mRNA and immunoreactivities decreased at 10 and 28 days, GAP-43 mRNA and immunoreactivities increased at 10 days and decreased at 28 days, both NGF and p75 mRNAs and immunoreactivities increased at 10 and/or 28 days, and enkephalin immunoreactivities decreased at 10 and 28 days, but these were all improved after hAFSCs transplantation. Our results showed that bladder dysfunction induced by PNT could be improved by hAFSCs transplantation, and PGP9.5, GAP-43, and neurotrophins could be involved in the mechanisms of nerve regeneration after hAFSCs transplantation.
Introduction
Radical hysterectomy for early-stage cervical cancer carries a good outcome with a 5-year survival rate over 80% 1 . However, during parametrial dissection in radical hysterectomy, damage of the pelvic nerve innervating the bladder may occur and result in postoperative lower urinary tract dysfunctions including frequent urination, urinary incontinence, voiding by abdominal straining, and urinary retention 2 . Nowadays, no effective treatment has been developed for the bladder dysfunction after radical hysterectomy. Human amniotic fluid stem cells (hAFSCs) can be a noninvasive stem cell source and are multipotent embryonic stem cells. hAFSCs are readily available from routine amniocenteses, grow easily in culture, appear phenotypically and genetically stable 3 , and can mediate therapeutic effects in some animal models of bladder dysfunction 4,5 . Our previous study showed that hAFSCs transplantation into rat bladder could improve bladder dysfunction induced by focal cerebral ischemia 4 .
Animal studies demonstrated that denervation of rat pelvic nerve could cause bladder hypersensitivity and hypertrophy 6 . However, whether hAFSCs treatment can improve the bladder dysfunction caused by pelvic nerve damage or not remains unknown. Growth-associated protein 43 (GAP-43), nerve growth factor (NGF), and p75 (NGF receptor) were found to be involved in nerve regeneration in the bladder with pelvic nerve injury 7 . Protein gene-product 9.5 (PGP9.5) 8 is a cytoplasmic neuron-specific protein, and encephalin 9 can be eliminated after transection of the pelvic nerve; both may be used as markers to examine the bladder status after pelvic nerve injury. CCL7, a monocyte chemoattractant protein, and CXCL12, which regulates mesenchymal stem cell survival and differentiation, are suggested to be involved in the process of stem cell treatment 9 . This study wished to investigate the effect of hAFSCs transplantation and the possible roles of GAP-43, neurotrophins, PGP9.5, and encephalin in the functional improvement of bladder dysfunction induced by pelvic nerve transection (PNT).
Materials and Methods
Animal Preparation
Animal experiments were approved by Institutional Animal Care and Use Committee of our institution (No. 2016121515). Female Sprague-Dawley rats (10 to 12 weeks old) were given tap water ad libitum and maintained in temperature and humidity-controlled room on a 12-h light/dark cycle.
Experiment Design
Rats were subjected to sham operation, and unilateral or bilateral PNT. Experimental groups included (1) sham-operation group, (2) unilateral PNT rats with injection of phosphate buffer solution (PBS), (3) unilateral PNT rats with injection of hAFSCs 1 × 106, (4) bilateral PNT rats with injection of PBS, and (5) bilateral PNT rats with injection of hAFSCs 1 × 106. hAFSCs were injected at the site of nerve transection (Figure 1). Cystometries were studied, neurofilament density within bladder nerve axons were assessed with immunohistochemistry, and expressions of PGP9.5, NGF, p75, GAP-43, CXCL12, CCL7, and enkephalin were studied using immunohistochemistry, immunofluorescence, and real-time reverse transcription polymerase chain reaction (RT-PCR) at 10 and 28 days after sham operation or PNT (n = 10 per group).

Schema of the experimental procedure. EdU: 5-ethynyl-2′-deoxyuridine; hAFSCs: human amniotic fluid stem cells; MPG: major pelvic ganglion; PCR: polymerase chain reaction; PNT: pelvic nerve transection.
hAFSCs Isolation and Culture for Transplantation
Institutional review board of our hospital approved this study (No. 201601690A3). hAFSCs were obtained after written informed consent from freshly collected amniotic fluid by routine amniocentesis in the second trimester of healthy pregnant donors. Cells were cultured in StemPro® MSC SFM supplemented with 10% fetal bovine serum (Invitrogen, Carlsbad, CA, USA) and incubated at 37°C with 5% carbon dioxide. Specific surface antigens of hAFSCs had been examined using flow cytometry in our previous study 4 . Cultured cells were trypsinized and stained with phycoerythrin-conjugated antibodies against CD44, CD45, CD73, CD90, CD105, and CD117 (BD PharMingen, San Diego, CA, USA). Cells were analyzed using Calibur flow cytometer (Becton Dickinson, Heidelberg, Germany). Passage 4 to 6 hAFSCs were collected and prepared to a final concentration of 1 × 106 cells/0.3 ml PBS. hAFSC dose was determined according to our previous work that used hAFSCs to treat rat bladder dysfunction after focal cerebral ischemia 4 .
Pelvic Nerve Transection
Under isoflurane anesthesia, a 4-to-5-cm-long midline incision was made in the lower abdomen 10 . Preganglionic pelvic nerves were exposed and separated from surrounding muscles, and a 2 to 4 mm portion of the nerves were removed at 5 mm central to major pelvic ganglion (Figure 1). Sham-operation group received the same operation without removal of the nerve. After completion of PNT, a silicone tube with 0.635 mm in diameter (A-M Systems, Sequim, WA, USA) was surgically implanted using a 6.0 suture (Ethicon, Somerville, NJ, USA), followed by the injection of either PBS or hAFSCs with Matrigel (Becton, Dickinson and Company, Franklin Lakes, NJ, USA) at the site of transection. Subsequently, the abdominal wall was closed. Urine was manually expelled out twice daily by gently compressing the lower abdomen. The surgical wound was checked for signs of infection every day, and gentamycin was administered if there was evidence of wound infection. In addition, 0.3 to 0.5 mg/100 g of meperidine was injected intramuscularly if any symptoms of pain were observed.
Conscious Cystometry
Rats received suprapubic tube implantation under 1.5% isoflurane inhalation at 2 days before cystometry implementation. Conscious cystometries 4 were performed with rats placed in special metabolic cages (Med Associates Inc., St. Albans, VT, USA). Bladder pressure was referenced to air pressure at the bladder level. Bladder was filled with 0.9% saline at 5 ml/h through the bladder catheter while bladder pressure was recorded. Saline infusion was continued until rhythmic bladder micturition contractions became stable. The mean data of peak voiding pressure, bladder capacity, residual volume, and nonvoiding contraction were collected in five micturition cycles for analysis 4 .
Under general anesthesia, rat bladder was filled with saline at 1 ml/h through bladder catheter. Average bladder capacity was determined after five micturition cycles in each rat. When half level of bladder capacity was reached, pressing was gently applied on rat’s abdomen using one finger. When bladder pressure was over 30 cmH2O, externally applied pressure was stopped to prevent from damaging bladder. Peak bladder pressure at the level that urine would leak out was taken as the leak point pressure (LPP) 11 . Cystometry Analysis Version 1.05 (Catamount R&D Inc., St Albans, VT, USA) was used for analysis.
Immunohistochemistry and Immunofluorescence
After cystometry, rats were euthanized with 3% isoflurane and then decapitated. Bladders were transected at the level of ureteral orifices, fixed in optimal cutting temperature compound, frozen in liquid nitrogen, and stored at −80°C. The 4,000-µm fresh-frozen bladder dome was cut into 10 cycles at −20°C. Each cycle contained 10 sections (each 10 µm thickness), which were mounted on saline-coated slides (Muto Pure Chemical Co., Ltd, Tokyo, Japan) for immunohistochemistry and immunofluorescence and one section (300 µm thickness) for real-time RT-PCR. The optical density of immunoreactivity on 10 slices of each animal was counted and summed, and the mean ± SD at each time point was calculated in every group.
Bladder sections were immunostained against PGP9.5, GAP-43, NGF, p75, and neurofilament (supplemental Table S1) using the avidin−biotin peroxidase method. First, fresh-frozen sections were fixed for 10 min in acetone, air-dried, and rinsed with PBS. After blocking with Dako REAL peroxidase blocking solution (code S2023, DAKO Corp., Carpinteria, CA, USA) for 20 min, sections were washed and incubated with primary antibodies for 18 to 20 h at 4°C and then, incubated for 1 h using biotinylated secondary antibodies (1:500, Vector Laboratories, Burlingame, CA, USA). Staining was developed with 3,3′-diaminobenzidine plus hydrogen peroxide as a chromogen. Sections from each experimental group were put on the same slide to keep same incubation time for the antibodies and chromogen.
Immunofluorescence against CXCL12, CCL7, and enkephalin (supplemental Table S1) was done on sections of major pelvic ganglion. These sections were first immersed in 4% paraformaldehyde, replaced with 30% sucrose, airdried, and then rinsed with PBS. After incubation with blocking agent and 0.2% Triton X-100 for 30 min, sections were washed and incubated for 18 to 20 h at 4°C with primary antibodies and then incubated with secondary antibody. For tracking 5-ethynyl-2′-deoxyuridine (EdU)-positive cells, sections were incubated with Click-IT reaction cocktail (Invitrogen) for 30 min at room temperature. Nuclear staining was performed with 4′,6-diamidino-2-phenylindole (DAPI). Immunoreactivity/immunofluorescence was analyzed with Image-Pro Plus Software (Media Cybernetics, Silver Spring, MD, USA) under Olympus BX-51 microscope.
Nerve Calculations
Nerve size and quantity were acquired from the stained sections using Image-Pro Plus Software. Images for analysis were obtained using a Spot charge-coupled device of color digital camera under Olympus BX-51 microscope. Transverse section of each specimen was captured in four frames at 200× magnification. The nerves in each image were highlighted using Image-Pro Plus Software by manually selecting the pixel value within the stained nerves using color cube-based tool in count/size application of the program. Measurement area was selected for all the highlighted nerves and compared for all groups.
Quantitative Real-Time RT-PCR
The 10 sections (each 300 µm thickness) obtained from bladder dome were used for RT-PCR according to the manufacturer’s protocol. Total RNAs were prepared using a Trizol reagent (Invitrogen) and incubated in reverse transcription mixture at 25°C for 5 min, 50°C for 1 h, 70°C for 15 min; finally, the tubes were cooled to 4°C for 5 min. Gene expressions for PGP9.5, GAP-43, NGF, and p75 were analyzed by real-time RT-PCR using inventoried TaqMan assays from Applied Biosystems (Life Technologies, Grand Island, NY, USA). The PGP9.5, GAP-43, NGF, and p75 assay codes were Rn00568258-m1, Rn01474579-m1, Rn01533872-m1, and Rn00561634-m1, respectively (Applied Biosystems, Oster City, CA, USA). GAPDH assay codes (Rs99999916-s1) were used as endogenous control. Thermal cycling and fluorescence detection were performed using an ABI Prism 7900HT Sequence Detection System (Applied Biosystems). PCR conditions were 50°C for 2 min, 95°C for 10 min, followed by 40 cycles at 95°C for 15 s and 60°C for 1 min. The data were calculated using the 2[-ΔΔC(T)] method.
Statistical Analysis
The immunoreactivity and mRNA ratio of PNT rat with or without hAFSCs transplantation to that of sham-operation rat was calculated, summed, and expressed as mean ± SD at each time point in every group. Data were analyzed with Prism 5 (GraphPad Software, San Diego, CA, USA). Continuous data in mean ± SD were compared statistically among the groups and among different time points in each group using one-way analysis of variance. The Tukey–Kramer test was used for post hoc comparisons. For categorical data, chi-squared test was performed with Fisher’s exact test. Probability value of <0.05 was considered statistically significant.
Results
hAFSCs Transplantation Improved Bladder Weight
There was no significant difference in body weight between groups. At 10 and 28 days after PNT, bladder weight increased after unilateral and bilateral PNT but improved after hAFSCs transplantation (Table 1).
Body Weight and Bladder Weight in Rats with Sham Operation (Sham), Pelvic Nerve Transection (PNT) Alone, and PNT Plus hAFSCs Transplantation (PNT + hAFSCs).

# P < 0.05 vs. bilateral sham (10d and 28d).
d: days; hAFSC: human amniotic fluid stem cell; PNT: pelvic nerve transection.
N = 10 rats in each group.
Initial body weight: body weight measured before sham operation, PNT alone, or PNT plus hAFSCs transplantation.
Final body weight: body weight measured 10d and 28d after sham operation, PNT alone, or PNT plus hAFSCs transplantation.
Bladder weight: bladder weight measured 10d and 28d after sham operation, PNT alone, or PNT plus hAFSCs transplantation.
hAFSCs Transplantation Improved Bladder Dysfunction
Unilateral PNT caused increase of bladder capacity and residual volume and increased number of nonvoiding contractions but decrease of peak voiding pressure and LPP at 10 and 28 days after PNT. Bilateral PNT caused overflow incontinence in cystometric study including peak voiding pressure, bladder capacity, residual volume, and LPP at 10 and 28 days after PNT and increased number of nonvoiding contractions. These cystometric parameters improved after hAFSCs transplantation (Figure 2).
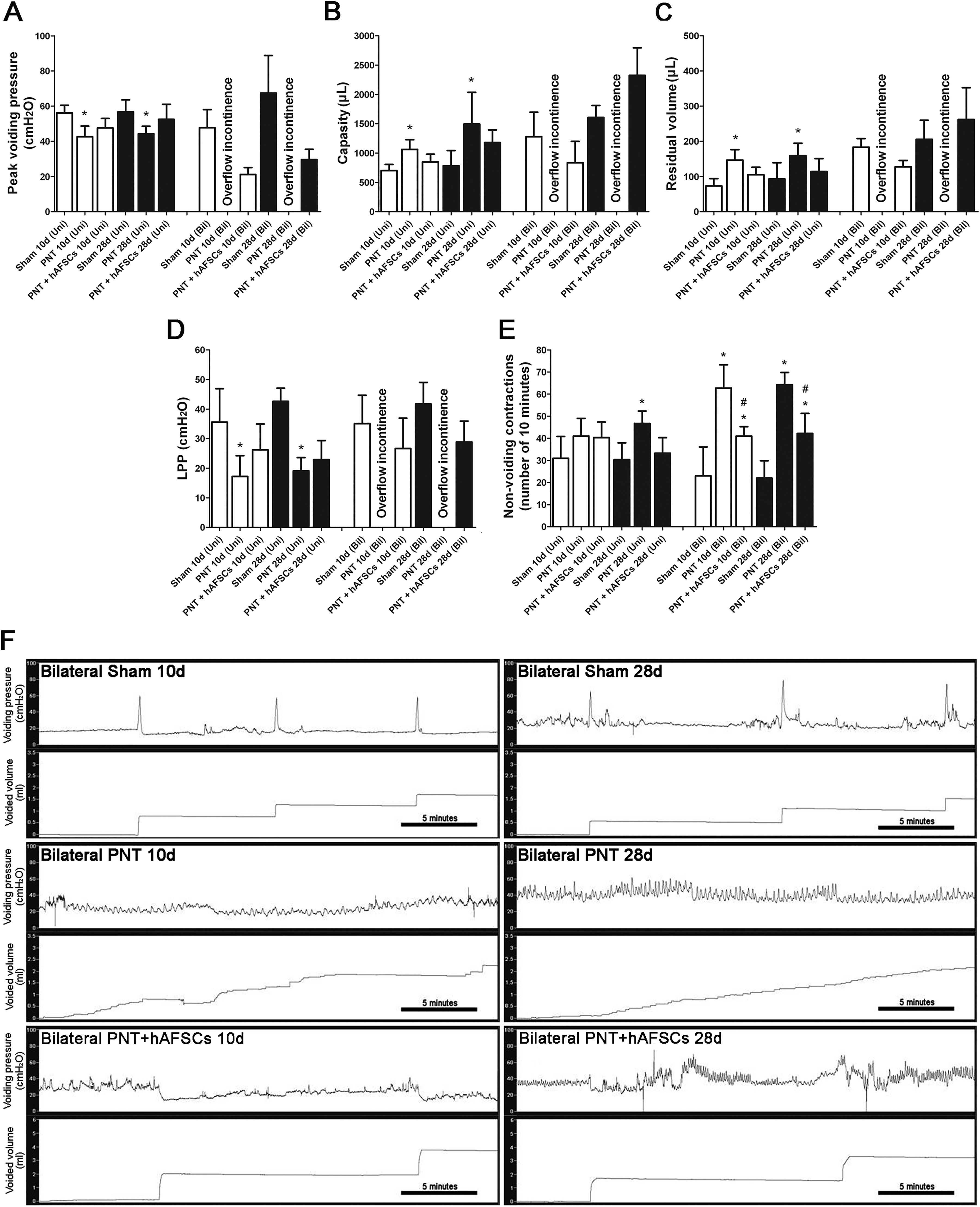
Urodynamic studies of all groups are presented. In upper panel, cystometric variables include (A) peak voiding pressure, (B) bladder capacity, (C) residual volume, (D) LPP, and (E) nonvoiding contraction. hAFSCs transplantation improves bladder dysfunction after PNT. Lower panel (F) depicts peak voiding pressure and voided volume at 10 and 28 days after sham operation, PNT alone, and PNT plus hAFSCs. Bilateral PNT causes overflow incontinence in all cystometric variables and increases the number of nonvoiding contractions. However, hAFSCs transplantation improves these cystometric parameters. *P < 0.05 vs. sham-operated group. #P < 0.05 vs. PNT rat group. N = 10 at each time point.
hAFSCs Transplantation Improved Nerve Density and PGP9.5
Neurofilament density within PNT bladders decreased at 10 and 28 days after unilateral and bilateral PNT but improved after hAFSCs transplantation (Figure 3). PGP9.5 mRNA (Figure 4) and immunoreactivities (Figure 5, S1) decreased at 10 and 28 days after unilateral and bilateral PNT but improved after hAFSCs transplantation.

Temporal change of neurofilaments in the rat bladder of sham operation, PNT alone, and PNT plus hAFSCs groups in unilateral (Uni) and bilateral (Bil) PNT rats. The density of neurofilaments within the bladders decreases at 10 and 28 days after unilateral and bilateral PNT, but hAFSCs transplantation may improve the nerve density. Bar indicates 50 μm. *P < 0.05 vs. sham-operated group. N = 10 at each time point.

Temporal change of mRNA expressions of PGP9.5 (A), GAP-43 (B), NGF (C), and p75 (NGF receptor, (D) in the rat bladder of sham operation, PNT alone, and PNT plus hAFSCs groups in unilateral (Uni) and bilateral (Bil) PNT rats. PGP9.5 mRNA decreases at 10 and 28 days after unilateral and bilateral PNT, and GAP-43 mRNA increases transiently at 10 days after unilateral PNT and decreases at 28 days after bilateral PNT. NGF mRNA increases at 10 and 28 days after unilateral and at 10 days after bilateral PNT, and p75 mRNA increases at 28 days after unilateral PNT. Most of them improve after hAFSCs transplantation. *P < 0.05 vs. sham-operated group. # P < 0.05 vs. PNT rat group. N = 10 at each time point.

Temporal change of immunoreactivities of protein gene-product 9.5 (PGP9.5, A), growth-associated protein 43 (GAP-43, B), NGF (C), and p75 (NGF receptor, D) in the rat bladders of sham operation, PNT alone, and PNT plus hAFSCs groups in unilateral (Uni) and bilateral (Bil) PNT rats. PGP9.5 (Figure S1) immunoreactivities decrease at 10 and 28 days after unilateral and bilateral PNT, and GAP-43 immunoreactivities (Figure S2) increase transiently at 10 days after unilateral PNT and decrease at 28 days after bilateral PNT. NGF immunoreactivities (Figure S3) increase at 10 days after unilateral PNT and at 28 days after bilateral PNT. p75 immunoreactivities (Figure S4) increase at 10 and 28 days after unilateral PNT. Most of them improve after hAFSCs transplantation. *P < 0.05 vs. sham-operated group. #P < 0.05 vs. PNT rat group. N = 10 at each time point.
hAFSCs Transplantation Improved GAP-43
GAP-43 mRNA (Figure 4) and immunoreactivities (Figure 5, S2) increased transiently at 10 days after unilateral PNT and decreased at 28 days after bilateral PNT, but these changes improved after hAFSCs transplantation.
hAFSCs Transplantation Improved NGF and p75 Expressions
NGF mRNA increased at 10 and 28 days after unilateral PNT and at 10 days after bilateral PNT, and p75 mRNA increased at 28 days after unilateral PNT. Both NGF and p75 mRNAs improved after hAFSCs transplantation (Figure 4). NGF immunoreactivities (Figure 5, S3) increased at 10 days after unilateral PNT and at 28 days after bilateral PNT. p75 immunoreactivities (Figure 5, S4) increased at 10 and 28 days after unilateral PNT. However, both NGF and p75 immunoreactivities improved after hAFSCs transplantation.
Cytokines and Enkephalin Were Involved in the Mechanisms of hAFSCs at the Site of PNT
CXCL12 immunofluorescences increased significantly at 10 days after unilateral and bilateral PNT but all improved after hAFSCs transplantation (Figure S5). CCL7 immunofluorescences increased significantly at 10 days after unilateral and bilateral PNT but all improved slightly after hAFSCs transplantation (Figure S6). There were no significant differences in CXCL12 and CCL7 immunofluorescences between PNT alone and PNT plus hAFSCs groups. Enkephalin immunofluorescences decreased significantly at 10 and 28 days after unilateral and bilateral PNT, but all improved after hAFSCs transplantation (Figure S7).
Colocalization of EdU-positive Cells and DAPI
hAFSCs were labeled with EdU overnight before local injection. These EdU-positive hAFSCs could colocalize with DAPI around major pelvic ganglion at 10 days after PNT (Figure S8).
Discussion
Our results showed that bladder weight increased after PNT but improved after hAFSCs transplantation. Bilateral pelvic nerve crush could cause detrusor muscle hypertrophy and increase collagen deposition 12,13 , and bilateral PNT caused bladder wall muscle hyperplasia and hypertrophy with increased bladder weight in rats 14 . However, Kwon et al. 15 reported that unilateral PNT caused no change of bladder weight in rats treated with muscle-derived stem cells and sham operation and also no change in normal controls.
PNT may accumulate connective tissues in bladder wall and cause detrusor dysfunction to empty bladder. Unilateral PNT can weaken bladder contractile force 16 , but bilateral PNT can abolish bladder contraction with consequent bladder distention and overflow incontinence 17 . The maximal intravesical pressure decreased significantly at 2 weeks after unilateral PNT, but no change occurred in bladder capacity or postvoid residual volume 15 . Bladder contractility was found impaired after bilateral pelvic nerve crush, which continued throughout 4 weeks 12 . In our study, bladder capacity and residual volume increased but peak voiding pressure and LPP decreased at 10 and 28 days after unilateral PNT. Our result is inconsistent with Kwon’s report 15 , which could be due to different study design (concurrent cut of hypogastric nerve and electrical stimulation during cystometric exam) and different study time points.
Bilateral pelvic nerve crush increased bladder size, impaired detrusor contractility, and caused decrease of smooth muscle and autonomic innervation 13 and also resulted in overflow incontinence and/or nonvoiding contractions 18 . At 10 days after bilateral pelvic nerve crush, overflow incontinence and nonvoiding contractions were more common than sham-operated rats 18 . Bilateral PNT could cause persistent bladder dysfunction and overflow incontinence within 6 weeks in rats 17 . However, none of the animals with PNT produced high-amplitude rhythmic contractions during the 6-week period of observation 17 . Our study showed that bilateral PNT caused overflow incontinence and increased number of nonvoiding contractions.
Mid-urethral sling has been reported to have an effect of more than 80% on urinary stress incontinence 19 but is ineffective for postoperative urinary incontinence after radical hysterectomy. Stem cells administered locally into patient’s urethra or periurethral areas may improve urinary stress incontinence in short-term follow-up 20 . Muscle-derived stem cell therapy has been assessed in animal model for bladder dysfunction induced by PNT and showed recovery of nerve conduction and improvement of maximal intravesical pressure at 2 weeks after unilateral PNT 15 . However, adult stem cells from muscle or bone marrow have the disadvantages of restricted differentiation potential, shorter life span, and difficulty to obtain 21 . Our study showed that hAFSCs transplantation could improve cystometric results and is a potential treatment in unilateral or bilateral PNT.
After nerve crush, PGP9.5-positive nerves in the operated side decreased at 3 and 7 days and returned to control levels by 60 days, indicating the presence of nerve regeneration after nerve crush 7 . Schwann cells possess p75-positive and GAP-43-immunoreactive characters 7 . The p75-labeled fiber density increased significantly from 3 to 30 days, and GAP-43-labeled fiber density increased significantly from 3 to 14 days after nerve crush. These data indicate that p75 and GAP-43 may be involved in the mechanism of bladder nerve regeneration. Our results found that the bladder neurofilament density and PGP9.5 expression decreased at 10 and 28 days, but GAP-43 and p75 increased at 10 and/or 28 days after PNT. However, hAFSCs transplantation may improve neurofilament density and PGP9.5 expression and reduce GAP-43 and p75 to sham-operation levels, indicating that hAFSCs could improve nerve regeneration in the process of recovery following pelvic nerve injury.
NGF is normally present in bladder muscle cells and urothelia and is absorbed by sympathetic and sensory fibers via its receptors to retrogradely transport to cell body. NGF binds to two types of receptors, tropomyosin receptor kinase A and p75 22 . The ability of NGF for proper neural growth and synaptic plasticity depends on p75 functions 23 . The p75 immunoreactivity is located on the cell surface of Schwann cells in rat bladders 24 . Schwann cells contain a rich source of NGF and P75, and NGF and p75 immunoreactivities could be upregulated in the bladders after pelvic nerve crush 7 . Our results demonstrated that bladder NGF and p75 immunoreactivities and mRNAs increased significantly at 10 and/or 28 days after PNT, and hAFSCs transplantation reduced the expressions of NGF and p75 to sham-operation levels.
Kawatani et al. 9 reported that encephalin could work as a cotransmitter with acetylcholine in the sacral preganglionic pathways to the urinary bladder. Our study found that encephalin decreased after PNT and improved after hAFSCs transplantation. Also, the expressions of CCL7 and CXCL12 were upregulated after PNT, which might attract hAFSCs to the site of transection, and thereby promote pelvic nerve repair and increase encephalin neurons to improve bladder dysfunction. The aggregation of EdU-labeled hAFSCs in major pelvic ganglion (Figure S7) may suggest that a local injection of hAFSCs could promote pelvic nerve regeneration and improve bladder dysfunction. The amelioration of the decrease in neurofilament density after hAFSCs transplantation is possible due to both the improved reinnervation of bladder by pelvic afferents and the prevention of the degeneration of bladder afferent terminals that have been cut off from their cell bodies.
The present study has some limitations. First, a single concentration of hAFSCs was used in our study. It is possible that higher concentration of hAFSCs could have better effects on PNT-induced bladder dysfunction. However, previous studies 25,26 have shown that timing and cell dose of transplantation may determine the survival of stem cell grafts and affect the outcomes and neuroinflammatory response after ischemic stroke. Wang et al. 26 demonstrated that increasing the number of implanted bone marrow stem cells from 1 × 106 to 1 × 107 did not cause a significantly greater number of surviving cells in an animal model of ischemic stroke. It is postulated that exceeding the optimal threshold of cells being transplanted may saturate the damaged striatum, result in insufficient amounts of nutrients to reach the grafted cells, and thereby progressively decrease the survival rate. Second, we examined the functional and morphological alterations of bladders at 10 and 28 days after PNT, but there could be better results if cystometries were examined at later time points after hAFSCs transplantation. Third, we did not directly examine the cellular differentiation after hAFSCs transplantation into pelvic lesion. Fourth, behavioral tests were not examined to demonstrate the return of sensory or motor function. Fifth, we did not adopt immune-deficient rodents for our experiment to prevent rejection against hAFSCs. However, hAFSCs have been reported to possess immunomodulatory properties, making these cells a good candidate for allo- and xenotransplantation 27 .
Conclusion
Bladder dysfunction induced by PNT can be improved after hAFSCs transplantation, and PGP9.5, GAP-43, neurotrophins, and encephalin could be involved in the mechanisms of nerve regeneration in PNT bladder.
Supplemental Material
Supplementary_Fig._S1.-PGP9.5 - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S1.-PGP9.5 for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Fig._S2.-GAP_43 - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S2.-GAP_43 for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Fig._S3.-NGF - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S3.-NGF for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Fig._S4.-P75 - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S4.-P75 for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Fig._S5.-_IF-PNT-MPG_-SDF1(CXCL12).tif.ijg6k9j - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S5.-_IF-PNT-MPG_-SDF1(CXCL12).tif.ijg6k9j for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Fig._S6.-_IF-PNT-MPG-CCL7 - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S6.-_IF-PNT-MPG-CCL7 for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Fig._S7.-_IF-PNT-MPG-ENK_190725 - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S7.-_IF-PNT-MPG-ENK_190725 for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Fig._S8._-EdU_-20cm - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Fig._S8._-EdU_-20cm for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Supplemental Material
Supplementary_Table - Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection
Supplementary_Table for Amniotic Fluid Stem Cells Improve Rat Bladder Dysfunction After Pelvic Nerve Transection by Ching-Chung Liang, Sheng-Wen Steven Shaw, Hung-Hsueh Chou, Yung-Hsin Huang and Tsong-Hai Lee in Cell Transplantation
Footnotes
Author Contribution
Liang CC, Lee TH, Huang YH, conception and design, financial support, provision of study materials, collection and assembly of data, data analysis and interpretation, manuscript writing, and final approval of the manuscript;
Shaw SW and Chou HH, conception and design, provision of study materials, data analysis and interpretation, manuscript writing, and final approval of the manuscript.
Ethical Approval
The hAFSCs isolation and culture for transplantation was approved by the institutional review board of Linkou Chang Gung Memorial Hospital, Taoyuan, Taiwan (No. 201601690A3).
Statement of Human and Animal Rights
This article contains animal studies that were approved by Institutional Animal Care and Use Committee of Linkou Chang Gung Memorial Hospital, Taoyuan, Taiwan (No. 2016121515).
Statement of Informed Consent
There were no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Chang Gung Memorial Hospital grants: CMRPG3H1051 (CC Liang) and CMRPG3H1041 (TH Lee) and Ministry of Science and Technology Taiwan grants: MOST 106-2314-B-182A-131, MOST 107-2314-B-182A-101 (CC Liang), and MOST 106-2314-B-182A-034 -MY3 (TH Lee).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
