Abstract
Oral diseases, such as cancers, inflammation, loss of bone/tooth/soft tissues, are serious threats to human health since some can cause systemic disease and effective treatments are limited. Thus, discovering promising biomarkers for physiological and pathological processes in oral medicine, and identifying novel targets for therapy have become a most critical issue. Recently, circular RNAs (circRNAs), which were once thought to be a class of non-coding RNAs (ncRNAs), are found to be of coding potential. CircRNAs are highly present in the cytoplasm of eukaryotic cells and are key elements in the physiological and biological processes of various pathological conditions, and are also reflected in oral development and progress. Previous studies have indicated that circRNAs are involved in the initiation and development of different types of diseases and tissues (e.g., cancers, cardiovascular diseases, neural development, growth and development, wood healing, liver regeneration). Moreover, growing evidence demonstrates that circRNAs play vital roles in oral cancers and osteogenic differentiation of periodontal ligament stem cells (PDLSCs). Here, we focus on the biological characteristics of circRNAs, beginning with an overview of previous studies on the functional roles of circRNAs as diagnostic biomarkers and therapeutic targets in oral medicine. We hope this will give us a promising new comprehension of the underlying mechanisms occurring during related biological and pathological progress, and contribute to the development of effective diagnostic biomarkers and therapeutic targets for oral diseases.
Introduction
Oral diseases, such as cancers, inflammation, loss of bone/tooth/soft tissues, have become a severe public health issue worldwide 1 . These conditions not only damage local function, but may also cause other systemic diseases 2,3 . Therefore, there is an urgent need to explore the relevant molecular mechanisms underlying these oral diseases and to identify valuable diagnostic biomarkers and therapeutic targets.
Non-coding RNAs (ncRNAs) are RNA molecules that are generally not translated into proteins but may participate in many biological processes 4 . Functionally abundant and important types of ncRNAs include linear transfer RNAs (tRNAs), ribosomal RNAs (rRNAs), small ncRNAs such as microRNAs (miRNAs), small interfering RNA (siRNAs), small nuclear RNAs (snRNAs), long ncRNAs (lncRNAs), and circular RNAs (circRNAs) 4,5 . The differences and similarities of these ncRNAs are listed in Table 1 6 –12 . Among these different types of ncRNAs, circRNAs are a newly discovered class of functional ncRNAs that exist widely in the cytoplasm of eukaryotic cells 13,10 . Unlike linear RNAs, circRNAs are presumably more stable as they exist in covalently closed continuous RNA loops without 5’ or 3’ ends, and, therefore, are resistant to exonuclease-mediated degradation 14 .
Similarities and Differences of ncRNAs.

ncRNAs: non-coding RNAs; miRNAs: microRNAs; siRNAs: small interfering RNAs; snRNAs: small nuclear RNAs; tRNAs: transfer RNAs; circRNAs: circular RNAs; lncRNAs: long non-coding RNAs.
Recent studies of circRNAs have revealed their functions under physiological conditions and in different types of disease 15,16 . CircRNAs have been demonstrated to play important roles in brain development, neurological disorders, stress handling, and cellular proliferation 10,16 –18 . In addition, circRNAs are shown to be abnormally expressed in multiform types of cancers, including colorectal cancer 19 , hepatocellular carcinoma 20 , pancreatic ductal adenocarcinoma 21 , gastric cancer 22 , lung cancer 23 , osteosarcoma 24 , bladder cancer 25 , and breast cancer 26 . circAmotl1, a circRNA derived from angiomotin-like1 (circAmotl1), which has tumorigenic capacity 27 , is reported to be highly expressed in patient tumor samples and cancer cell lines, and can promote tumor formation by induction of c-myc into the nucleus 27 . In light of these reports, we believe that circRNAs may serve as promising biomarkers, and even provide clues to the discovery of new therapeutic targets to treat cancers and other diseases.
In oral medicine, several studies have suggested that circRNAs play pivotal roles in the progress of oral cancer 28 and the osteogenic differentiation of periodontal ligament stem cells (PDLSCs) 29 . These reports provide us with hints that circRNAs could be important regulatory nova in oral medicine. In this pilot review, we focus primarily on the basic biological characteristics of circRNAs, summarize the roles of circRNAs in oral medicine, and provide insights into the mechanisms underlying the related biological and pathological progresses.
Biogenesis and Categories of CircRNAs
Much effort has been focused on understanding the mechanisms of circRNA formation. Thousands of circRNAs are transcribed from diverse genomic locations in a tissue-, cell-type-, or developmental-stage-specific manner 10 . CircRNAs formed from coding and non-coding exons, introns, intergenic regions, and transcripts antisense to 5’ and 3’ UTRs have been reported 10 . Notably, unlike linear RNAs, many circRNAs are formed by head-to-tail splicing of exons, suggesting a previously unrecognized regulatory potential of coding sequences 10,30 .
According to the different biogenesis patterns from genomic regions, circRNAs can be divided into four subtypes: exonic circRNAs (ecircRNAs), circular intronic RNAs (ciRNAs), exon-intron circRNAs (EIciRNAs), and intergenic circRNAs 15 . Each subtype is produced from a different circularizing mechanism (Fig 1, Table 2).

Biosynthesis of circRNAs. (A) The biogenensis of ecircRNAs and ElciRNAs. (B) The biogenensis of ciRNAs. (C) The biogenensis of intergenic circRNAs. circRNAs: circular RNAs; ecircRNAs: exonic circRNAs; ElciRNAs: exon-intron circRNAs; ciRNAs: circular intronic RNAs; RBPs: RNA-binding proteins.
Differences of Four Categories of circRNAs.

circRNAs: circular RNAs; ecircRNAs: exonic circRNAs; ciRNAs: circular intronic RNAs; EIciRNAs: exon-intron circRNAs.
EcircRNAs
It has been reported that ecircRNAs are generated predominantly from back-splice exons, where downstream 3’ splice donors are covalently linked to upstream 5’ splice acceptors in a reverse order 31 . EcircRNAs account for the majority of identified circRNAs. Their biogenesis process requires RNA-binding proteins (RBPs) to act as trans-factors to promote ecircRNAs formation 32 . Given that ecircRNAs are alternative splicing isoforms, it is suggested that they could function as miRNA or RBP sponges, and regulate transcription of their parental genes 33 .
CiRNAs
CiRNAs are generated from lariat introns that fail in debranching. GU-rich sequences near the 5’ splice site and C-rich sequences near the branch point are sufficient for an intron to escape debranching and become a stable circRNA 34 . CiRNAs are located predominantly in the nucleus and are widely expressed in human cells with little enrichment of miRNA targeting sites. Some ciRNAs have been found to function as positive regulators of RNA Pol II transcriptase to regulate the transcription of their parental genes 34 .
EIciRNAs
EIciRNAs are circRNAs that are circularized with both exons and introns. Internal repeat sequences may play important roles in their generation, and they might be similar to ecircRNAs 35 . EIciRNAs are relatively abundant in the nucleus and can enhance the transcription of their parental genes by interacting with small nuclear ribonucleoproteins 35 .
Intergenic circRNAs
An intergenic circRNA contains two intronic circRNA fragments flanked by GT-AC splicing signals acting as the splice donor and acceptor of the circular junction while forming an integrated circRNA 36 . They were discovered using the CIRI (CircRNA Identifier, a circRNA identification tool) in human transcriptomes by Gao et al 36 . However, at the moment, little is known about the functions of circRNAs.
Functions and Roles of CircRNAs
CircRNAs, as a special type of RNA molecules, have an array of functions according to their different origins and structures. Further studies are needed to precisely define the specific roles of circRNAs and to understand how they are regulated. Up to now, circRNAs have been found to function as miRNA and RBP sponges, regulators of protein translation and gene transcription, and competing products during pre-mRNA splicing (Fig 2). Here, we summarize recent studies on the functions of circRNAs and further discuss their regulatory roles under physiological and disease conditions.

Main functions of circRNAs. (A) miRNAs sponge. (B) RBPs sponge. (C) Translate protein. (D) Regulate gene transcription. (E) Splicing regulation. circRNAs: circular RNAs; ecircRNAs: exonic circRNAs; ElciRNAs: exon-intron circRNAs; ciRNAs: circular intronic RNAs; RBPs: RNA-binding proteins.
CircRNAs Can Serve as Efficient miRNA Sponges
Up to now, the most important and most widely studied function of circRNAs is in their role as miRNA sponges. Abundant endogenous circRNA molecules, with multiple complementary binding sites for miRNAs, are inherently resistant to exonucleolytic RNA degradation. Therefore, they form a large class of post-transcriptional regulators acting as potent miRNA sponges carrying out important biological functions 37 . For example, cerebellar degeneration-related protein 1 antisense (CDR1as), is massively bound by miR-7 and miR-671 in human and mouse brains 38 . When the CDR1as locus is removed from the mouse genome, the expression of both miR-7 and miR-671 are mis-regulated in all brain regions. In addition, miR-671 directs cleavage of the circular antisense transcript, the cerebellar degeneration-related protein 1(CDR1), which could produce a highly abundant and circularized species 39 . These results indicate that the interactions between circRNAs and miRNAs are important for normal brain function.
Another study shows that circHIPK2 takes part in regulating astrocyte activation via cooperation of autophagy and endoplasmic reticulum (ER) stress by functioning as an endogenous miR124-2HG sponge to sequester miR124-2HG and inhibit its activity, which in turn results in increased expression of sigma non-opioid intracellular receptor 1. Knocking-down of circHIPK2 expression could significantly inhibit astrocyte activation via the regulation of autophagy and ER stress through the targeting of miR124-2HG and SIGMAR1 40 .
Another circRNA, circITCH, is aligned in a sense orientation to the known protein-coding gene, ITCH—a member of the E3 ubiquitin ligases 23 . circITCH acts as a sponge of miR-7, miR-17, miR-214, and promotes the expression of its parental gene ITCH 23,41 . Interestingly, the upregulation of ITCH could inhibit the Wnt/β-catenin signaling pathway. In light of these reports, it is suggested that circITCH is able to suppress cancer progression by indirectly inhibiting the activation of the Wnt/β-catenin signaling pathway 23,41 .
Furthermore, Huang et al 42 have identified that circHIPK3 binds to multiple miRNAs (9 miRNAs with 18 binding sites), indicating that one circRNA can be associated with a variety of miRNAs. Taken together, circRNAs have been repeatedly demonstrated to function as efficient miRNA sponges to regulate their parental genes and therefore affect their biological functions in health and disease.
CircRNAs Can Act as RBP Sponges
In addition to miRNA sponges, the function of “sponging” other factors, such as RBPs and ribonucleoprotein complexes, has also been proposed for circRNAs 16 . HuR is an RBP that regulates protein expression by interacting with lots of RNAs 43 . circPABPN1, which originates from poly(A)-binding protein nuclear 1 (PABPN1) pre-mRNA, is one of the HuR targeted circRNAs 43 . Besides, it is reported that circPABPN1 inhibits HuR binding to PABPN1 mRNA and reduces PABPN1 production 43 . Furthermore, circAmotl1 is capable of binding to c-myc and STAT3 to contribute to their translocation into the nucleus, thus regulating the expression of their target genes 27,44 .
Production of circRNAs Can Regulate Gene Transcription and Translation
Several studies have shown that circRNAs can function as transcriptional and translational regulators. For instance, EIciRNAs, which retain the intronic sequences from their parental genes, could interact with the transcription machinery 45 . As a result, EIciRNAs can regulate the expression of their parental genes in cis via specific RNA-RNA interaction between U1 snRNAs and EIciRNAs 35 .
In addition, ciRNAs, another subclass of circRNAs, are abundant in the nucleus. They play regulatory roles in the Pol II transcription of their parental coding genes, which adds to the growing evidence of circRNAs’ regulatory functions in gene expression 34 .
As an important class of post-transcriptional regulators, circRNAs could directly target (m)RNAs by partial base pairing 10,46 . For example, CDR1 serves as a component in the sense of mRNAs stabilization and plays an important role in regulating RBPs 39 . In addition, the binding of circPABPN1 to HuR can suppress PABPN1 translation 43 . Therefore, circRNAs may take part in the regulation of gene expression.
Certain circRNAs Have Protein-Coding Potential
CircRNAs can serve as translational templates in vitro and in vivo. Two endogenous circRNAs, circMbl and circZNF609, are reported to be translatable 47,48 , suggesting that the peptides and proteins produced by circRNAs could be biologically functional molecules in cells 30 . Recently, Zhang et al. verified that circRNA circFBXW7 encodes a functional protein FBXW7-185aa 49 , which has a negative regulatory role in glioma carcinogenesis, further supporting the coding ability of circRNAs. FBXW7-185aa has a synergistic effect with the parental gene coding protein FBXW7 on inhibiting the expression of c-myc in human cells, which, in turn, decreases glioma cell proliferation and inhibits cell cycle acceleration in vitro and in vivo 49 . In addition, circRNAs can be driven translationally by N6-methyladenosine 50,51 . Some studies indicate that eukaryotic ribosomes can initiate translation of circRNAs with internal ribosome entry site (IRES) elements 52,53 , while other reports show that circRNAs could translate into proteins through a mechanism similar to rolling circle amplification without IRES 54 . Further investigations are needed to determine the mechanisms of how these translatable circRNAs are translated into proteins.
CircRNAs Compete with pre-mRNA Splicing
As the splicing pattern of any pre-mRNA is dictated by competition between alternative pairing of 5’ and 3’ splice sites, the production of circRNAs may play a role in the regulation of pre-mRNA splicing 16,45 . For example, the second exon of the splicing factor muscleblind (MBL) is circularized, which generates a circRNA (circMbl). CircMbl contains conserved MBL binding sites, which are bound by MBL. As a result, modulation of MBL levels can strongly affect circMbl biosynthesis 55 . Taken together, the formation of circRNAs influences the splicing of pre-mRNA to alter gene expression.
Other Possible Functions of circRNAs
Studies have shown that circRNAs can regulate certain cell signaling pathways. For instance, circZNF292 is found to be induced under hypoxic conditions and in gliomas cell lines 56 . The latter report shows that circZNF292 silencing could suppress tumor tube formation through regulating cell cycle by the Wnt/β-catenin signaling pathway 56 .
In addition, circular antisense ncRNAs in the INK4 locus (circANRIL) can regulate rRNA maturation 57 . CircANRIL binds to the C-terminal lysine-rich domain of pescadillo homologue 1, an essential 60S-preribosomal assembly factor, and prevents pre-rRNA binding and exonuclease-mediated rRNA maturation 57,58 . In vascular smooth muscle cells and macrophages, circANRIL impairs ribosome biogenesis, which leads to activation of p53 and a subsequent increase in apoptosis and decrease in proliferation rate 57 .
Moreover, other studies 19,59 have indicated that circRNAs also exist in secreted extracellular vesicles (EV), suggesting that extracellular expulsion is one way that cells can eliminate accumulated circRNAs. Intriguingly, it may also be a way for cells to transfer functional circRNAs as a way of communication, which needs more in-depth investigation.
Relationship of circRNAs with Diseases
According to the functions of circRNAs, it is recognized that circRNAs are essential for the development process of eukaryotes and can influence the pathogenesis and physiology of disease.
For example, in a mouse model of lead-induced neurotoxicity, circRar1 induces the upregulation of apoptosis-associated factors caspase8 and p38 via modulation of their common target miR-671 60 . For another, in tumor cells circFoxo3 can promote MDM2-induced p53 ubiquitination and subsequent degradation, resulting in an overall decrease of p53, which, in turn, prevents MDM2 from inducing Foxo3 ubiquitination and degradation, and results in increased levels of Foxo3 protein 61 . Thereby, according to the previous studies we can conclude that cell apoptosis can be induced by circRNAs directly or indirectly.
On the other hand, circFoxo3 is generated from a member of the forkhead family of transcription factors Foxo3. It is highly expressed in hearts and is correlated with extensive cellular senescence 62 . CircFoxo3 is distributed mainly in the cytoplasm, where it interacts with the anti-senescent protein ID-1 and the transcription factor E2F1, as well as the anti-stress proteins FAK and HIF1a, which results in increased cardiac senescence 62 . Among the differentially expressed senescence-associated circRNAs, circPVT1, which is generated by circularization of an exon of PVT1 gene, is identified as a circRNA displaying markedly reduced levels in senescent fibroblasts 63 . CircPVT1 might selectively inhibit let-7 activity and hence the expression of let-7-regulated mRNA 63 . Silencing circPVT1 can promote cell senescence and reverse the proliferative phenotype after let-7 function is impaired 63 .
CircRNAs are also largely enriched in platelets compared with nucleated cell types, which is a signature of transcriptome degradation 64 . Fusion circRNAs (f-circRNAs) are produced from transcribed exons of distinct genes affected by the chromosomal translocations 65 . During leukemia progression, f-circRNAs could contribute to cellular transformation, promote cell viability and resistance upon therapy, and have tumor-promoting properties in in vivo models 65 .
Beyond that, during the priming phase of rat liver regeneration (LR), the comprehensive circRNAs expression profile has been analyzed by high-throughput RNA sequencing, which revealed their potential roles in rat LR 66 . Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) biological pathway analysis indicates that, during the early phase of rat LR, circRNAs contribute critically to the process of energy metabolism, substance metabolism, and hepatocyte proliferation 66 .
Recently, Yang et al. discovered that circAmotl1 could accelerate healing processes in a mouse excisional wound model 44 . They found that circAmotl1 can increase protein levels of Stat3 and Dnmt3a. The increased Dnmt3a then inhibits the expression of miR-17-5p, which stimulates the expression of fibronectin, Dnmt3a, and Stat3, and leads to increased cell adhesion, migration, proliferation, survival, and wound repair 44 .
Moreover, the investigation of the transcriptomic landscape of human pre-implantation development has identified abundant circRNAs 67 . Quaking (QKI), which functions in the formation of circRNAs, is strongly expressed by Müller glial cells in both the developing and adult retina 68 . It is well accepted that many circRNAs are expressed in tissue- and developmental-stage-specific manners, which reveals a striking regulation of circRNAs during neuronal development. It is plausible that some circRNAs can act as local regulators of gene expression at distal neuronal sites, such as growth cones and synapses, and contribute to aspects of neural development 69 . In addition, emerging evidence also indicates that circRNAs are involved in the development and progression of various neurological disorders 70 .
Taken together, current studies focus mainly on the interactions between RNAs and proteins, while much is unknown about their diverse and complex functions. The use of circRNAs for diagnosis and therapy still needs extensive investigation.
CircRNAs in Oral Medicine
To date, there have been few studies on the roles of circRNAs in oral medicine. However, it is realized that circRNAs are involved in the initiation and development of different types of diseases and tissues, which indicates that circRNAs might be a regulatory nova in this field. In this pilot review, we describe the primary landscape of the correlatively existing literatures on circRNAs in oral medicine (Table 3).
Recently Published Studies for Detecting circRNAs in Oral Medicine.
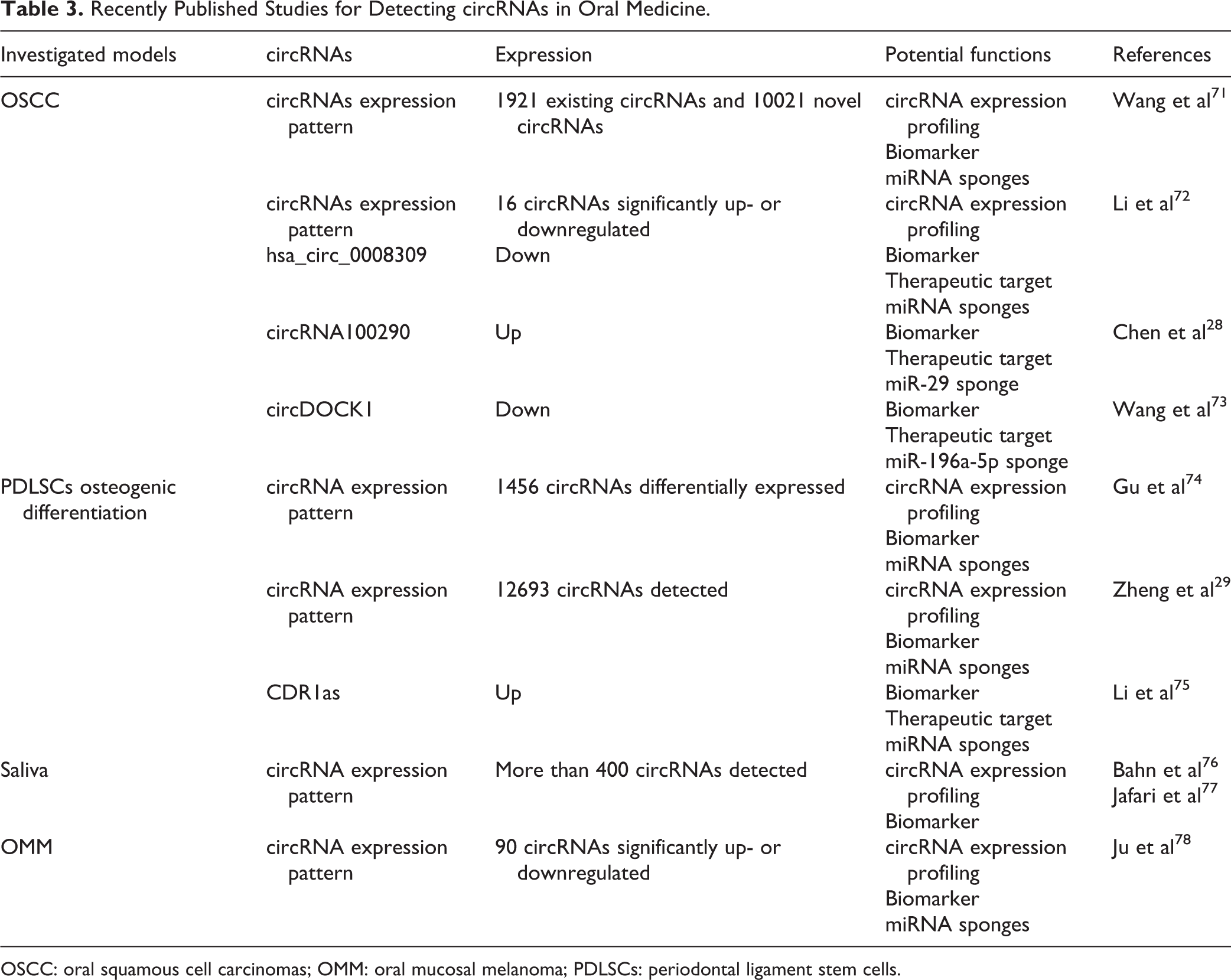
OSCC: oral squamous cell carcinomas; OMM: oral mucosal melanoma; PDLSCs: periodontal ligament stem cells.
CircRNAs and Oral Cancers
Oral cancer is a multifactorial pathological disease thought to be the result of genetic modifications (including activation of oncogenes and silence of tumor suppressor genes). However, there is a lack of accurate diagnosis and efficient treatment 64 . Therefore, reliable biomarkers for the timely detection of the disease and patient stratification are required.
So far, miRNAs are the most well studied ncRNAs in the physiological and pathological processes of all types of oral cancers 79 . A great number of miRNAs have been discovered as key participants in tumorigenesis, acting either as tumor suppressors or as oncogenes (oncomiRs) 80,81 . Moreover, emerging lines of evidence predict that circRNAs, as sponges of miRNAs, are associated with the progression and induction of oral cancers 82 .
There have been studies on circRNAs in oral squamous cell carcinoma (OSCC) 28,71 –73 . Using circRNA and mRNA microarrays, Chen et al. identified circRNAs that are differentially expressed between OSCC tissue and paired non-cancerous matched tissue 28 . They discovered that circRNA100290 is upregulated in OSCC tissues, and its knockdown decreases the expression of CDK6—a direct target of miR-29b—and inhibits proliferation of OSCC cells in vitro and in vivo 28 . Their results reveal that circRNA100290 may function as a competing endogenous RNAs (ceRNAs) to regulate CDK6 expression by sponging of miR-29b family members in OSCC 28 . Meanwhile, it is reported that circDOCK1 can suppress cell apoptosis via inhibition of miR-196a-5p by targeting BIRC3 in OSCC 73 .
In addition, Ju et al. found that the expression pattern of circRNAs is altered in oral mucosal melanoma (OMM), suggesting that circRNAs might play vital roles in metastasis of OMM and in regulating OMM progression 78 .
In conclusion, these reports indicate that circRNAs exert regulatory functions and might be potential biomarkers in oral medicine. Based on the functions of circRNAs in various cancers, we believe that they have tremendous potential to serve as biomarkers for the diagnosis and treatment of oral tumors, even as new therapeutic targets for the treatment of oral cancers.
CircRNAs and Oral Inflammation
Although there is still no study on the roles of circRNAs in common inflammation of oral diseases such as periodontitis, there have been studies of circRNAs involved in rheumatoid arthritis (RA) 83 and osteoarthritis 84 . It is reported that circRNAs are expressed differentially in RA patients compared with healthy controls, indicating that circRNAs play crucial roles in the regulation of expression of symbol genes that control the occurrence and development of RA 83 . Interestingly, periodontitis shares a similar occurrence and developmental process with RA 85 , which suggests that circRNAs may play certain roles in oral inflammation.
It is known that inflammation in oral medicine is manifested mainly via periodontal diseases—the most common form of oral disease 76 . Intriguingly, it is reported that more than 400 putative circRNAs that may function in intercellular signaling and inflammatory response exist in human cell-free saliva 76 . Consequently, salivary circRNAs, as endogenous RNAs, may be novel clues for detecting a wide range of local or systemic diseases, including inflammation and be used routinely in personalized medicine 76,77 .
CircRNAs and Cell Differentiation and Tissue Regeneration
Some reports have clearly established the participation of circRNAs in the maintenance of pluripotency. For example, circBIRC6 acts as a sponge for miR-34a and miR-145 to regulate the molecular circuitry in human embryonic stem cells 86 . And during human epidermal stem cell differentiation, circRNAs are abundantly expressed and upregulated in a coordinated manner 87 , further suggesting that they may play important roles during the process.
Recent findings also demonstrate that expression profiles of circRNAs are significantly altered during osteogenic differentiation of PDLSCs, which provides clues leading to further insight into the roles of circRNAs in osteoblast differentiation in an oral setting 29 . Moreover, Gu et al. found a total of 1456 circRNAs differently expressed during PDLSCs osteogenic differentiation 74 . What’s more, it is suggested that specific circRNAs could serve as ceRNAs to promote PDLSCs osteogenic differentiation and periodontal regeneration. For instance, CDR1as was proved to promote osteoblastic differentiation of PDLSCs via the miR-7/GDF5/SMAD and p38 MAPK signaling pathway 75 .
However, up to now, there have been only a few reports about circRNAs’ functions during orthodontic tooth movement (OTM), which involves alveolar bone remodeling. It is recognized that PDLSCs are key seed cells in OTM and alveolar bone remodeling 88,89 . Previous studies by our research team have found that circRNAs expression changed in mechanical force-induced PDLSCs 90 . Besides, it is known that circRNAs can serve as regulators of cellular stress both positively and negatively: to maintain homeostasis, combat acute infection, and regulate cell growth and death, or dysregulation of others to enhance stress response to adapt chronic stress 91 . All this evidence indicates that circRNAs may participate in the regulation of OTM and bone remodeling.
Although there is currently not enough understanding of tissue regeneration and bone remodeling in oral medicine, we believe that there will be more reports on the functions of circRNAs in this field in the near future.
Conclusions
Current research in oral medicine usually focus on tissue regeneration, tooth development, oral cancer, tooth movement, and inflammatory disease. To explore the relevant potential mechanisms of each of these processes has extreme clinical significance.
CircRNAs are shown to play important roles in various physiological and pathological processes. It is generally accepted that circRNAs are involved in regulating cell proliferation, apoptosis, and multiple signaling pathways. Currently, circRNAs have been found to play pivotal roles in different types of diseases and tissues. We have also learnt that circRNAs play a vital part in OSCC 28,71 –73 , metastatic OMM 78 , saliva 76,77 and the osteogenic differentiation of PDLSCs 29,74,75 . Based on these studies, we suggest that circRNAs participate in the regulation of oral biological processes, and might be a regulatory nova in the development and outcome of oral medicine. There is no doubt that a great number of studies will be carried out to further explore the functions of circRNAs and the associated mechanisms.
Undeniably, findings on the roles of circRNAs in oral medicine are also confronted by many challenges. On the one hand, strategies and technical limitations might affect the detection of circRNAs in experiments 92 . For example, some smaller circRNAs may be missed in length selection, and the use of some artificial primers may affect the enrichment of circRNAs 92 . On the other hand, there is still no uniform verdict on the predictions of the detection algorithms of different circRNAs 33,93 . In addition, low expressed-circRNAs also tend to be overlooked by bioinformatics analysis 94,95 . All these limitations restrict functional studies of circRNAs in oral medicine and other fields. Therefore, establishing a gold standard with which to assess the accuracy of predictions from algorithms and choose appropriate thresholds are needed to overcome these challenges, in addition to reducing operational errors during experiments 92,94 . The continued development of such a standard will provide a statistical test that could be used to evaluate the false discovery rate, and defining appropriate thresholds will be beneficial for the detection of high-confidence circRNAs 92 . Besides, normalization procedures to assess the enrichment or depletion of circRNAs are necessary to improve the accuracy of the results during the experiments 92 .
Motivated mainly by the finding that they function as miRNA sponges, circRNAs are stepping forward into the light of broader comprehension and clinical application of their physiological and pathological phenomena in oral medicine. We hope that this pilot review can help us tap into this new field and lay the groundwork for a more in-depth understanding of the diagnosis, prognosis, and treatment in oral diseases, and offer a primary glimpse into the functions of circRNAs involved in oral health.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work is supported by the National Natural Science Foundation of China (81771030;81470709) and the Construction Engineering Special Fund of “Taishan Scholars” (tsqn20161068).
