Abstract
Cerebral small vessel disease (CSVD) is composed of several diseases affecting the small arteries, arterioles, venules, and capillaries of the brain, and refers to several pathological processes and etiologies. Neuroimaging features of CSVD include recent small subcortical infarcts, lacunes, white matter hyperintensities, perivascular spaces, microbleeds, and brain atrophy. The main clinical manifestations of CSVD include stroke, cognitive decline, dementia, psychiatric disorders, abnormal gait, and urinary incontinence. Currently, there are no specific preventive or therapeutic measures to improve this condition. In this review, we will discuss the pathophysiology, clinical aspects, neuroimaging, progress of research to treat and prevent CSVD and current treatment of this disease.
Keywords
Introduction
Cerebral small vessels comprise two components. First, the leptomeninges vasoganglion, which is derived from subarachnoid space covering, and the convex surface of brain. Second, perforating arteries are derived from anterior, middle, posterior cerebral arteries that supply the subcortical parenchyma (Fig. 1). The cerebral small vessels are crucial to maintenance of adequate blood flow to the sub-surface brain structure. They include small arteries, arterioles, venules, and capillaries which are commonly sized at 50–400 µm 1,2 .

Cerebral small vessel and its comprised compartments. (1): Posterior cerebral artery (2): Posterior communicating artery (3): Middle cerebral artery (4): Anterior cerebral artery (5): Anterior communicating artery (6): Internal carotid artery (7): Basilar artery. Small vessel atherosclerosis. (A). Eccentric atherosclerotic plaque in a perforating vessel in the putamen, causing significant narrowing of the lumen (asterisk).
Cerebral small vessel disease (CSVD) is a generic term that refers to intracranial vascular disease based on various pathological and neurological processes, as well as a syndrome referring to different clinical manifestations and neuroimaging features caused by the structural changes of vascular and brain parenchyma. Small vessel disease accounts for up to 25% of all ischemic strokes 3 but also put patients at twice the risk for these conditions 4 . In addition, CSVD is a leading cause of functional loss, disability and cognitive decline in the elderly. Neuroimaging development allows increased understanding of CSVD. Thus, a comprehensive knowledge of its pathophysiologic mechanism, neuroimaging, and clinical features is imperative to further study on possible preventive and therapeutic measures.
Pathogenesis and Classification
The pathophysiologic mechanisms of CSVD are not yet clear. The European small brain vascular disease expert group puts forward the classification of CSVD based on cerebrovascular pathologic changes as the following: Arteriolosclerosis, sporadic and hereditary cerebral amyloid angiopathy, inherited or genetic small vessel diseases distinct from cerebral amyloid angiopathy, inflammatory and immunologically-mediated small vessel diseases, venous collagenosis, and other small vessel diseases such as post-radiation angiopathy 1 . These various pathologic changes cited by the European expert group do not only result in damage of brain parenchyma including neuronal apoptosis, diffuse axonal injury, demyelination and loss of oligodendrocytes, but also result in a series of symptoms and unusual neuroimaging findings.
CSVD is thought to result in reduced cerebral blood flow, impaired cerebral autoregulation and increased blood–brain barrier (BBB) permeability. However, the molecular mechanisms underlying CSVD are incompletely understood. Recent studies in monogenic forms of small vessel disease (SVD), such as cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL), and ‘sporadic’ SVD have shed light on possible disease mechanisms in CSVD. Proteomic and biochemical studies in post-mortem monogenic CSVD patients, as well as in animal models of monogenic disease have suggested that disease pathways are shared between different types of monogenic disease, often involving the impairment of extracellular matrix (ECM) function. In addition, genetic studies in ‘sporadic’ CSVD have also shown that the disease is highly heritable, particularly among young-onset stroke patients, and that common variants in monogenic disease genes may contribute to disease processes in some CSVD subtypes 5 .
Arteriolosclerosis
Among the pathologic changes involved in CSVD, the two most common are arteriolosclerosis and cerebral small vascular atherosclerosis. Arteriolosclerosis, a vascular risk-factor-related SVD, is known to be age-related, and is the most common small vessel alteration in aged brains. The severity of arteriolosclerosis increases with aging and is exacerbated by hypertension and diabetes (Fig. 2) 5 . Thus, arteriolosclerosis is also named hypertensive SVD 6 . Cerebral small vascular atherosclerosis, particularly in arterioles smaller than 50 µm in diameter, is characterized by a loss of smooth muscle cells from the tunica media, degeneration of internal elastic lamina, proliferation of fibroblasts (Fig. 2), deposits of fibro-hyaline material and collagens, thickening of the vessel wall, formation of microatheroma, and narrowing of the lumen 1 . With these changes, the vessels become elongated, tortuous and inflexible (Fig. 2). In addition, wall damage causes distension of its outer portions due to fibrosis, that is microaneurysm, and the stenosis or obstruction of proximal lumen 7 . Ultimately, impaired autoregulation of involved small vessel results in reduced cerebral blood flow (CBF) and chronic cerebral hypoperfusion 6 . The occlusion of arterial lumen leads to acute ischemia, causing lacunar infarction (Figs. 2 and 3). Whereas, critical stenosis and hypoperfusion involving multiple small arterioles, mainly in deep white matter, lead to incomplete ischemia which are visualized as White Matter Hyperintensities (WMH) on neuroimaging 8 . The two pathophysiological pathways above can often overlap, so lacunes and white matter lesions often coexist in the same patient. Kuwabara and colleagues found a 25% decreases in CBF in patients with both Alzheimer’s and Binswanger’s diseases, with the use of positron emission tomography with oxygen-15-labelled water 9 . Conventional risk factors such as hypertension diabetes, smoking, high homocysteine concentrations, obesity, and dyslipidemia have been considered to lead to arteriolosclerosis. In addition, hematological disorders, infection, and hereditary diseases are increasingly recognized in various studies 10,11 .

The relationship between conditions such as diabetes, and hypertension influencing arteriolosclerosis. In addition, the figure shows how cerebral small vessel atherosclerosis is characterized. A series of changes will lead to lacunar infarct.
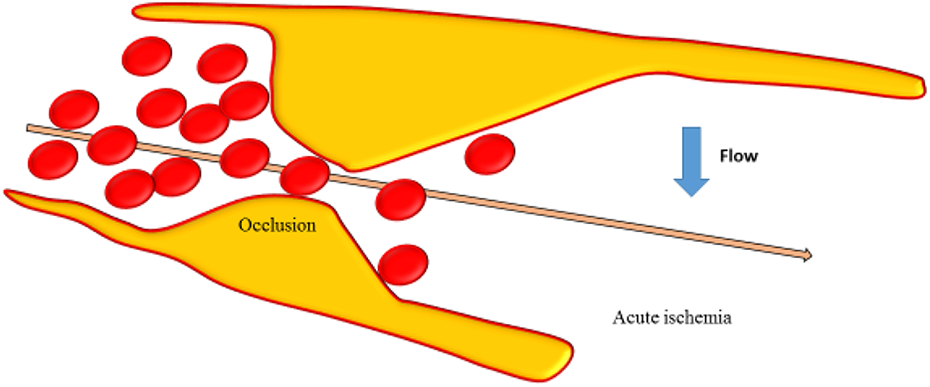
Occlusion of the vessel lumen is represented and acute ischemia due to decreased flow in the vessel occurred.
Cerebral Amyloid Angiopathy
Cerebral amyloid angiopathy (CAA) is the dominant cause of lobar intracerebral hemorrhage. Not only does it result in stroke and cognitive impairment in a significant proportion of older patients, but it is an important component of the senile plaques found in patients with Alzheimer’s disease 12,13 . CAA is characterized by the progressive accumulation of congophilic, immunoreactive, amyloid protein in the walls of small-to-medium-sized arteries and arterioles predominantly located in the leptomeningeal space, cortex, and, to a lesser extent, in the capillaries and veins 1 . A characteristic ‘double-barrel’ lumen of involved vessels is seen under light microscopy in leptomeninges and parenchyma due to the splitting of the internal elastic lamina caused by the deposition of hyaline material in the vessel wall. The thickening vessel walls stained with Congo red, and with thioflavin S and appear green birefringent under polarized light and fluoresce under ultraviolet light, respectively 1,12 .
CAA appears in almost all elderly patients with dementia, and accounts for about 64% to 84.9% of the elderly without dementia 14 –16 . Its distribution in the brain parenchyma is mainly on the region of hippocampus and cortex 17 , which may have a correlation with cognitive impairments caused by CAA, without clear potential causative mechanisms known to date. Many studies indicate that diffuse brain microbleeds, micro-infarcts, hypoperfusion, and white matter hypoxia caused by vessel changes associated with CAA may be responsible for cognitive decline and dementia, independent of Alzheimer’s disease and Lewy body pathology (Fig. 6) 16,18 –21 .
Amyloid-beta protein accumulation in capillaries affects BBB integrity, which leads to a loss of tight junction proteins and increased BBB permeability (Fig. 4) 22 . Then, perivascular edema and extravasation of toxic plasma components caused by the disruption of BBB contributes to localized damage to brain parenchyma and enlarged perivascular spaces (Fig. 4) 4,23 . Extensive white matter lesions can be found in patients with CAA, which carry an increased risk of warfarin-related intracerebral hemorrhage after ischemic stroke 24 .
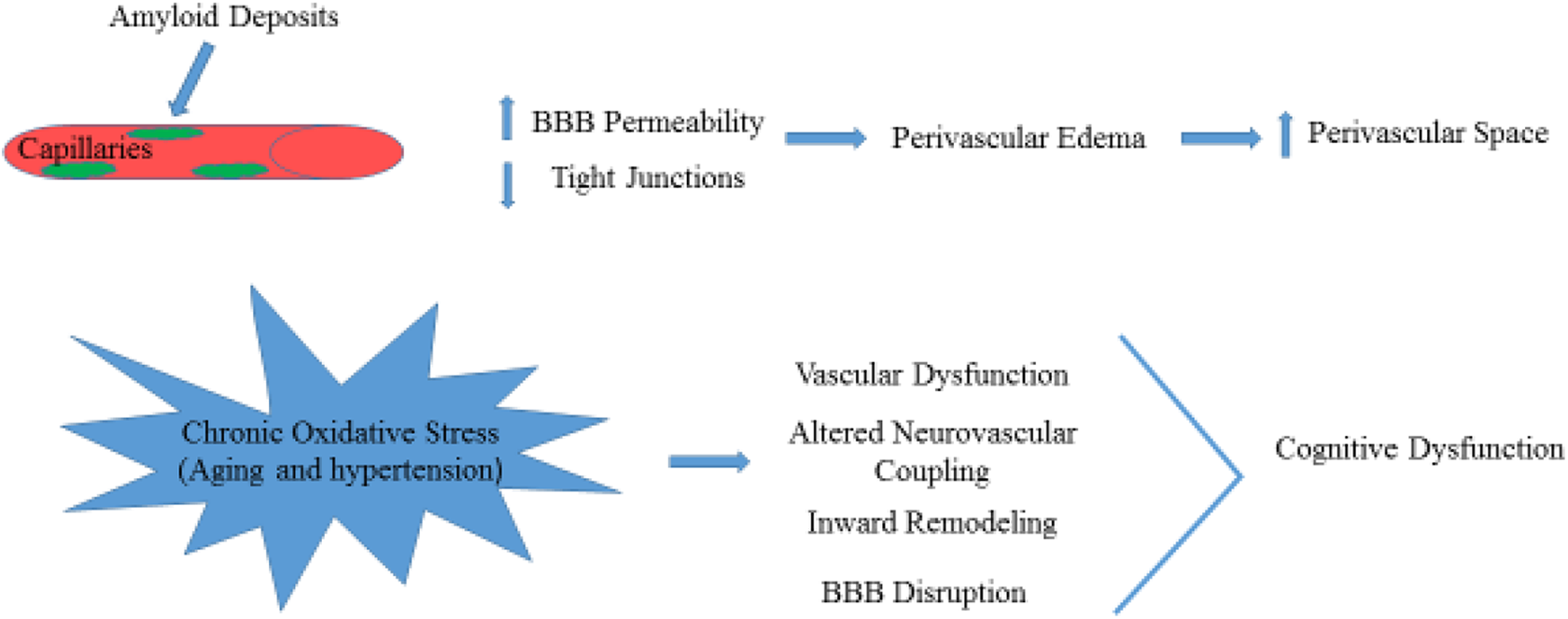
The increase of amyloid deposits in the perivascular space consequently leads to increase in BBB permeability and decrease in tight junctions. The schematic effects of chronic oxidative stress in aging is shown.
CAA is not only a biomarker of cognition impairment, but also found in some rare genetically transmitted diseases 25 such as Down’s syndrome 26 . Probably, CAA is an expression of some systematic amyloid deposition diseases, including hereditary amyloidosis, transthyretin familial amyloid polyneuropathy 27,28 , plasma cell dyscrasia 29 , and myogenic disease 30 .
Long-term and chronic oxidative stress in aging, and hypertension lead to cerebral vascular dysfunction including impaired neurovascular coupling, inward remodeling, rarefaction, and BBB disruption, which result in brain injury and cognitive dysfunction (Fig. 4) 31 . In the inherited or genetic SVDs such as Fabry’s disease and CADASIL 32,33 , there is still a controversy about the mechanisms of brain injury. Moore and colleagues reported that the deposition of globotriaosylceramide (Gb3) leads to altered vascular reactivity, resulting in increased blood flow and metabolic vulnerability in the deep white matter. These findings are contrary to chronic cerebral hypoperfusion found in other studies 34 –36 .
Neuroimaging Features of CSVD
Due to mild clinical symptoms or lower mortality than common stroke, the onset of CSVD was frequently neglected. Thus, neuroimaging has become an important tool in diagnosing CSVD and the silent neurovascular disease, especially at early stage. Neuroimaging of CSVD primarily involves visualizing recent small subcortical infarcts, lacunar infarct, WMH, microbleeds, enlarged perivascular spaces, and brain atrophy 1 (Fig. 6).
Recent Small Subcortical Infarcts
Recent small subcortical infarcts are considered to result from acute severe ischemia of a single perforating artery, and are lesions occurring in the territory of one perforating arteriole within the previous few weeks, with neuroimaging evidence or clinical symptoms of recent infarction (Fig. 5) 37 . Recent infarcts are regarded as round or ovoid lesions less than 20 mm in maximal diameter in the white matter, basal ganglia, or brainstem. They present hyperintense on diffusion-weighted imaging (DWI), hypointense on the apparent diffusion coefficient map, and either normal or hyperintense to a normal brain on fluid-attenuated inversion recovery (FLAIR)/T2 imaging, and with less hyperintensity than cerebrospinal fluid on T2 38,39 . However, a number that can reach 50% of patients with acute stroke may appear with no responsible lesions on computed tomography (CT) and magnetic resonance imaging (MRI). DWI is the most sensitive sequence for acute ischemic lesions, allowing detection of acute ischemia within the first few hours after stroke onset. With the increasing recognition of recent small subcortical infarcts, Gattringer and colleagues recommended the new term ‘recent small subcortical infarct’ instead of lacunar infarct 40 . From recent small subcortical infarct to cavity formation, there are a number of morphological changes, including volume and diameter reductions, occurring within the first 90 days of onset 39,41 . In the development of recent small subcortical infarcts, more than one-third (39%) of the lesions recovered with no residual cavities, and 40% of the infarct lesions were located adjacent or fused in preexisting white matter lesions 39 . It is reported that the incidence of cavity formation after recent small subcortical infarcts is in the broad range of 28–94% 41 –44 . There are three outcomes from recent small subcortical infarcts: (1) A common lacuna and/or (2) white matter hyperintensity without apparent cavitation on T2-weighted sequences, and (3) less common fates that disappearing with little visible consequence on conventional MRI.

Subcortical infarcts resulting from acute severe ischemia. It also shows microbleeds occurring in the cerebral cortex and gray/white matter junction due to deposition of amyloid. Cavitation is also shown as a result of acute severe ischemia.

Gradient echo magnetic resonance imaging (left image) in a patient with small vessel disease and lacunar stroke showing microbleeds (arrowed) in the subcortical region not seen on fluid-attenuated inversion recovery (right image).
Lacunar Infarct
Lacunar stroke accounts for up to a quarter of all acute ischemic strokes. it is a small fluid-filled cavity that was thought to mark the healed stage of a small deep brain infarct. In neuroimaging, a lacuna is a round or ovoid, subcortical, fluid-filled cavity with similar signal to cerebrospinal fluid (CSF) (Fig. 5). It measures between 3–15 mm in diameter, which is consistent with a previous acute small deep brain infarct or hemorrhage in the territory of one perforating arteriole 37 . Lacunar infarcts are typically located in the basal ganglia, internal capsule, thalamus, corona radiata, centrum semiovale (CSO), and brainstem. Poirier and colleagues divided the lacunas into three subtypes based on the formation: Subtype I lacunas are secondary to old lacunar infarction; subtype II lacunas secondary to old hemorrhagic lesions; subtype Ⅲ lacunas are secondary to enlarged perivascular spaces. Herve and colleagues classified the lacunar lesions by three-dimensional MRI reconstruction, according to their shapes into four types: Slab, stick, multiple components, or ovoid/spheroid, then proposing that most of lacunar infarcts (83%) were ovoid or spheroid. Infarct lesions manifest isolated, adjacent to or fused into white matter hyperintensity 38 . Moreau and colleagues found that lacunas almost always present at 90 days after acute lacunar infarction and appear as a central CSF-like hypointensity with or without a surrounding border of hyperintensity on FLAIR sequence but only CSF-like hypointensity and hyperintensity on T1-weighted and T2-weighted, respectively. Moreover, the sensitivity of FLAIR for cavitation was greatly lower than for T1-weighted sequences 41 .
White Matter Hyperintensities
The white matter is the most vulnerable region to suffer hypoxia/hypoperfusion due to the watershed effect. WMH are supposed to be comprehensive expressions including disturbances of small blood vessels, breakdown of the BBB, small infarcts in the white matter, glial activation, loss of oligodendrocytes, and demyelination caused by chronic diffuse hypoperfusion or reduced cerebral blood flow 1,31 . Bilateral, mostly symmetrical hyperintensities on T2-weighted and FLAIR MRI are characteristic features of white matter lesions accompanied with some hypointensity different from CSF on T1-weighted MRI and low density on cerebral CT in most older individuals with or without cognition decline. In addition to white matter, the hyperintensity lesions are also located in subcortical gray matter structures, such as basal ganglia, and brainstem 37,45 . However, whether the hyperintensities of gray matter and brainstem should be adopted into WMH is controversial 37,46 . To differ WMH from other lesions, such as lacuna and atrophy, and then research their specific role on dementia, diffusion tensor MRI (DT-MRI), and magnetization transfer MRI (MT-MRI) were increasingly used to give a quantitative information on the state of the brain’s white matter.
Microbleeds
Cerebral microbleeds (CMBs) are magnetic resonance (MR)-visible small (generally 2–5 mm in diameter, but up to 10 mm) areas of signal void caused by perivascular collections of hemosiderin deposits that are foci of past hemorrhages resulted from small vessels involved in CAA or arteriolosclerosis (Fig. 5) 18,37 . Small hypointense lesions appear on paramagnetic-sensitive MR sequences such as T2-weighted gradient-recalled echo (GRE) or susceptibility-weighted imaging sequences with a ‘blooming effect’ (larger or more conspicuous on GRE than on spin-echo MRI) 18 , and are generally not seen on CT, or on FLAIR, T1-weighted, or T2-weighted MR sequences. Microbleeds lesions, either round or oval in shape, are most commonly located in the cortico-subcortical junction, and deep gray or white matter in the cerebral hemispheres, brainstem, and cerebellum. Lesions were classified as three types according to location: lobar, deep and subtentorial CMBs 47 . Lobar microbleeds are especially associated with a decline in executive functions, information processing and memory function relative to a decline in motor speed associated with deep or subtentorial microbleeds 48 .
Enlarged Perivascular Spaces
Perivascular spaces, as well as Virchow–Robin spaces, are extensions of the extracerebral fluid-filled spaces that follow the typical course of a vessel as it goes through gray or white matter 49 . These spaces follow the path of perforating arteries including arteries, arterioles, veins, and venules. Given the different imaging planes, the lesions appear linear, round or ovoid CSF-like intensity on all MRI sequences, with the diameter generally smaller than 3 mm 50 . Enlarged perivascular spaces are predominantly located in the basal ganglia and CSO, and with increased signal intensity equal to cerebrospinal fluid on T2-weighted images, hypointensity on T1-weighted and occasionally hypointensity on FLAIR images without hyperintense rim to distinguish from old lacunar infarcts 49 .
Brain Atrophy
Brain atrophy indicates a lower brain volume on neuroimaging that is not related to definitive macroscopic focal injury such as trauma or infarction. Characteristic manifestations of atrophy are symmetrical or asymmetrical decreased total volume, increased ventricular volumes, enlarged superficial sulci, and decreased specific gray or white matter volumes. The hippocampus is an example on imaging examinations that include cranial CT or MRI that has characteristic manifestations of atrophy 51,52 . Brain atrophy frequently occurs together with WMH in elderly, and is greatly associated with cognitive decline, and dementia. Some investigators studied the association between brain atrophy and WMH and indicated that increased hyperintensities would accelerate brain atrophy. This was especially true by visualizing the loss of deep tissue demonstrated by an increase in ventricular size 52,53 .
Clinical Manifestation of CSVD
The clinical manifestations of CSVD vary depending on the specific cause of the disease, as well as the brain regions affected. Individuals may present sudden onset stroke symptoms, progressive cognitive deterioration, dementia, gait disorder, sphincter dysfunctions, and psychiatric disorders, etc. 54 –56 .
Cognitive Decline and Dementia
SVD is thought to be among the main causes of vascular cognitive impairment, and is thought to account for about 45% of dementia cases, which is also associated with so-called silent lacunar infarcts which are asymptomatic infarcts with definite lesions on neuroimaging 57 –60 . Cognitive decline caused by CSVD presents with executive dysfunctions, attention and memory decline, set-shifting disabilities, slower speed of information processing, decline of verbal fluency, and delayed recall. On the behavior area, symptoms showed apathic, mood disorder, depression and daily living disability 1,54,56 . Among others, some clinical features include sleep disorders, vertigo, tinnitus, and hearing disorder.
Neuropsychiatric Symptoms
Neuropsychiatric symptoms resulting from SVD mainly include hallucination, agitation, depression, anxiety, disinhibition, apathy, irritability, sleep disturbance, and appetite changes 61 . It is found that the presence of multiple cerebral microbleeds, particularly multiple lobar microbleeds, is associated with higher global neuropsychiatric burden, particularly with depression and disinhibition. Tang and colleagues reported that the emotional disinhibition was associated with CMBs, while the behavioral disinhibition symptoms are commonly found among patients with Alzheimer’s disease 62 .
Urinary disturbances are common in cerebral vascular pathology, which mainly include nocturia, incontinence, urinary frequency, and urgency. In the LADIS (Leukoaraiosis And Disability) study 62 , Poggesi and colleagues researched 639 individuals with age-related white matter changes (ARWMC) ranging from mild to severe, and reported that 70% of the participants complained of at least one urinary symptom. In addition, 60% suffered from nocturia, and approximately 20% reported urinary frequency, incontinence, and urgency. Urinary frequency and nocturia are more prevalent in men, whereas incontinence is more frequent in women. Urinary urgency is associated with the severity of ARWMC, while urinary frequency is only associated with the stroke history. In patients with Alzheimer’s disease, larger ARWMCs in volume were found to be associated with urinary incontinence.
Gait disturbance (GD), characterized by impairment of locomotion, equilibrium and gait ignition, is another common manifestation of CSVD 63 . Both White Matter Lesions (WMLs) and lacunar infarcts are independently associated with several gait parameters including a lower gait velocity, a shorter stride length and a reduced cadence 64 . De Laat and colleagues offered the first indication that microbleeds may be associated with GDs, independent of other coexisting markers of CSVD including white matter lesions and lacunar infarcts. They suggested that a higher number of microbleeds was associated with a shorter stride length, a lower gait velocity and a longer double-support time 65 .
Preventive and Therapeutic Measures of CSVD
The precise diagnosis of CSVD depends on neuropathological examination, which is difficult in clinical practice. Therefore, the clinical diagnosis bases on clinical features, neuroimaging of the brain parenchyma, and the ancillary use of other investigative techniques, such as ultrasonography (carotid and cardiac), combining with the risk factors. The prognosis is better than other strokes in the short term after onset, because the primary lesion is small, and the rate of recovery is often rapid.
Risk factors for lacunar infarcts include non-modifiable risk factors (age, regional distribution, sex) and acquired risk factors (hypertension, cigarette smoking, diabetes, atrial fibrillation, hyperhomocysteinemia, chronic kidney disease, high circulatory phosphate level and obesity) 66 –69 . Controlling or removing risk factors is important for the prevention of CSVD. Hypertension is the most prevalent and important risk factor for stroke in general, as well as the most treatable factor. Many studies showed that lowering blood pressure reduces stroke 70 , and dementia or cognitive decline in patients who have had a stroke and in those without cerebrovascular disease. In a recent study, researchers drew a conclusion that targeting a systolic blood pressure of <120 mmHg, as compared with <140 mmHg, had a non-significant 11% lower incidence of stroke in patients at high risk for cardiovascular events but without diabetes 71 . Similarly, the patients whose intensive systolic blood pressure goal was <130 mmHg in the Secondary Prevention of Small Subcortical Strokes trial, and <120 mmHg in the ACCORD trial had a non-significant 19% and a significant 41% lower incidence of stroke, respectively, than the incidence with higher targets 70 . Statins, primarily used to lower low density lipoprotein cholesterol levels, have been reported as having cardiovascular benefits in recent years. Statins may improve vasomotor reserve capacity 72 and cerebral endothelial function by inhibiting cerebral vascular superoxide production 73 , enhancing endothelial-derived Nitric Oxide bioavailability and decreasing oxidative stress, thus protecting against cerebral ischemia. In the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) study, patients with stroke or transient ischemic attack were treated by atorvastatin, and therefore had a significant reduction in stroke 74 while high-dose atorvastatin (80 mg/d) may increase the incidence of hemorrhagic stroke 75 .
Intravenous tissue plasminogen activator (t-PA) is the gold standard of treatment of acute ischemic stroke, but the role of t-PA in patients with lacunar infarction has been debated due to the different pathomechanisms compared with common stroke related to large-vessel changes and increasing risks of hemorrhage in patients with WMH or CMB. Some investigators have found that the increased risk of symptomatic Intra Cerebral Hemorrhage (ICH) attributable to CMB is small and unlikely to exceed the benefits of thrombolytic therapy 76,77 while there is significantly higher risk of ICH in patients with multiple CMBs 78,79 . A higher number of microbleeds (>10 CMBs) is associated with a higher risk for symptomatic ICH after intravenous thrombolysis (IVT) when compared with patients with 0 to 10 CMBs or 1 to 10 CMBs on pretreatment MRI. Thus, high microbleed burden may be included in individual risk stratification scores predicting ICH risk following IVT for AIS 80 .
Anti-platelets are generally used in ischemia stroke. The Guidelines for management of ischemic stroke and transient ischemic attack in 2008 by the European Stroke Organization Executive Committee 81 . However, Lundström and colleagues reported that non-responsiveness to clopidogrel or clopidogrel resistance presented in the patients with CSVD, not carotid atherosclerosis, after minor ischemia stoke and transient ischemic attack, possibly because of glucose intolerance and insulin resistance 82 . In the Secondary Prevention of Small Subcortical Strokes (SPS3) trial, the results showed no benefit of dual antiplatelet treatment on the prevention of recurrent stroke, and there was no significant different rate of recurrence between the patients on dual therapy (2.5%) and those taking aspirin (2.7%) 83 . Pearce and colleagues reported that neither dual antiplatelet treatment nor the lower blood pressure target was associated with the difference in the change of cognitive function during up to 5 years of follow up 83 .
There are some other therapeutic measures except for the usual methods above. Homocysteine lowering using B-vitamins was proved to reduce WMH volume increment in those with severe baseline SVD 84 . In addition, vitamin E tocotrienols were recently found to attenuate the progression of WMH among healthy subjects with WMH 85 . In the study of spontaneously hypertensive stroke-prone rats, which is the best model to simulate human cerebrovascular disease, it had been reported that chronic spironolactone treatment increased tone and reactivity of cerebral vascular and alters vascular structure of the middle cerebral artery (MCA) by increasing the lumen and outer diameter, without any change in blood pressure. Thus, these changes may enhance the autoregulatory behavior of the MCA, therefore helping to protect the brain in the event of an ischemic stroke 7 . In a randomized double-blind trial in CADASIL, Dichgans and colleagues reported that donepezil does not result in cognition impairment at week 18; however, improvements were detected in executive function and processing speed, but there was no additional benefit of donepezil at week 24 compared with week 18 85 .
CSVD is a relatively homogeneous disease process and an important cause of stroke, cognitive decline, and age-related disability. Although there have been a large number of researches on CSVD, the mechanism of vascular pathology and brain injury is still not clear, and there are numerous controversies on the prevention and management. More attention and targeted efforts are needed to better define the clinical consequences of these diseases. The main difficulty of investigating CSVD is the frequent coexistence of different forms including white matter lesions, lacunar infarcts, and microbleeds. Furthermore, more clinic trails should be investigated to study the diagnostic criteria of CSVD and the preventive and therapeutic measures to reduce the burden of disability or dementia caused by CSVD, and animal models should be established to study the specific pathogenesis of different forms of CSVD.
Footnotes
Author Contribution
Qian Li and Yang Yang contributed equally to this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
