Abstract
Liquid chromatography using a tandem mass spectrometer (LC-MS/MS) is a method of proteomic analysis. A shotgun analysis by LC-MS/MS comprehensively identifies proteins from tissues and cells with high resolution. The hepatic function of mice with acute hepatitis following the intraperitoneal administration of CCL4 was improved by the tail vein administration of the culture conditional medium (CM) of human mesenchymal stem cells from adipose tissue (hMSC-AT). In this study, a secreted protein expression analysis of hMSC-AT was performed using LC-MS/MS; 128 proteins were identified. LC-MS/MS showed that 106 new functional proteins and 22 proteins (FINC, PAI1, POSTN, PGS2, TIMP1, AMPN, CFAH, VIME, PEDF, SPRC, LEG1, ITGBL, ENOA, CSPG2, CLUS, IBP4, IBP7, PGS1, IBP2, STC2, CTHR1, CD9) were previously reported in hMSC-AT-CMs. In addition, various proteins associated with growth (SAP, SEM7A, PTK7); immune system processes (CO1A2, CO1A1, CATB, TSP1, GAS6, PTX3, C1 S, SEM7A, G3P, PXDN, SRCRL, CD248, SPON2, ENPP2, CD109, CFAB, CATL1, MFAP5, MIF, CXCL5, ADAM9, CATK); and reproduction (MMP2, CATB, FBLN1, SAP, MFGM, GDN, CYTC) were identified in hMSC-AT-CMs. These results indicate that a comprehensive expression analysis of proteins by LC-MS/MS is useful for investigating new factors associated with cellular components, biological processes, and molecular functions.
Keywords
Introduction
The clinical application of liver cell therapy using stem cells has great significance. The liver can develop acute hepatitis or chronic liver failure due to the influence of factors such as drugs, xenobiotics, and viruses. Eventually, chronic hepatitis and fibrosis develop and the ability to regenerate hepatocytes is lost 1 . At present, the only effective treatment is liver transplantation; however, liver transplantation is associated with problems such as rejection and limitation of donors. Thus, alternative approaches are necessary, and stem cells are attracting attention as a therapeutic approach. Mesenchymal stem cells (MSCs) represent an outstanding candidate stem cell for clinical treatment. MSCs have been collected from various organs, including the bone marrow (BM) 2 , cord blood 3 , placenta 4 . and adipose tissue (AT) 5,6 . Currently, attention is being given to adipose tissue as a source of MSCs for regenerative medicine 5 –7 . Adipose tissue contains large amounts of MSCs (adipose-derived mesenchymal stem cells (ADSCs)) and is considered to be a useful source of cells for clinical application because of its fast proliferation and high cellular activity.
In recent years, treatment methods using conditional medium of mesenchymal stem cells (MSC-CM) have been reported 8 –11 . Because the culture supernatant does not contain cellular components, there is a high possibility that they will have clinical applications because of the extremely low risk of complications (i.e. pulmonary embolism) associated with the administration of cells in the blood and canceration of the administered cells. Proteins are important components in the regulation of cellular functions such as cell proliferation, cell death, and the immune function, and in the induction of differentiation. Thus, proteomic analyses, which detect the expression of protein, are considered to be a powerful tool for analyzing the system biology and exploring the active factors in MSC-CMs.
Liquid chromatography by tandem mass spectrometry (LC-MS/MS) is an analytical chemistry technique that combines the physical separation capability of liquid chromatography (or high-performance liquid chromatography (HPLC)) and the mass spectrometric ability of mass spectrometry 12 . MS involves a mass separation step; the ionized component is detected as it is. In soft ionization methods such as electrospray ionization (ESI) 13 –15 , molecular weight-related ions are mainly detected (mass spectrum). In tandem mass spectrometry (MS/MS), specific ions are first selected by a mass separator (MS1). In addition, the fragmentation of ions occurs due to the collision of ions with inert gas. The fragment ions obtained are separated and detected by a second mass separator (MS2) (product ion spectrum). Molecular weight-related ions are mainly detected by MS, and precursor ions and product ions are detected by MS/MS. LC-MS/MS allows for the identification of proteins fragmented into peptides by trypsin. Our protocol was based on the bottom-up strategy of a proteomic MS analysis. Enzymatic digestion was carried out using the Filter Aided Sample Preparation (FASP) method with trypsin as protease 16 . The peptide mixture was treated with ZipTip and then on-line coupled nano-liquid chromatography (nano LC) was performed using an Orbitrap Elite Hybrid Mass Spectrometer (Thermo Fisher Scientific, Tokyo, Japan). In addition, an on-line LC-MS/MS system for quantitative proteomics based on data-dependent protein IDs and shotgun-based quantitative proteomics methods was used.
This study was performed to identify functional protein components in the conditional medium of human mesenchymal stem cells from adipose tissue (hMSC-AT-CM) using LC-MS/MS. The identification of the secreted protein components of hMSC-AT and protein components with therapeutic effects is expected to be useful for future cell therapy.
Materials and Methods
Reagents
The MSCGM-CD™ Mesencymal Stem Cell Growth Medium BulletKit™ was obtained from Lonza (Basel, Switzerland). hMSC-ATs (46-year-old Caucasian female) (PromoCell, Heidelberg, Germany) were cultured. Fetal bovine serum (FBS) was obtained from BioWest (Nuaille, France). D-MEM/Ham’s F-12 medium was obtained from Wako (Osaka, Japan). Plastic dishes were obtained from TPP (Trasadingen, Switzerland). All other materials used were of the highest commercial grade.
Flow Cytometry
Cell flow cytometry was performed using a NovoCyte® Flow Cytometer (ACEA Biosciences, Inc., San Diego, CA, USA) according to the manufacturer’s instructions. Briefly, hMSC-ATs (1 × 105 cells) were mixed into 0.5 mL of Perfusion Solution (CORNING, Manassas, VA, USA). Each antibody (1/100 of the volume) was added to the cell admixture, which was then incubated on ice for 30 minutes. After washing the cells with Brilliant Stain Buffer (BD Biosciences, Franklin Lakes, NJ, USA), fluorescence activated cell sorting (FACS) measurement was carried out. The following primary antibodies were used: APC Mouse Anti-Human CD29, BV421 Mouse Anti-Human CD44, BV421 Mouse IgG2b κ Isotype Control, APC Mouse IgG1 κ Isotype Control (BD Biosciences, Franklin Lakes, NJ, USA); FITC anti-human CD90 (Thy1) Antibody, FITC Mouse IgG1 κ Isotype Ctrl Antibody, PerCP anti-human CD34 Antibody, PerCP Mouse IgG1 κ Isotype Ctrl Antibody, PE/Cy7 anti-human CD45 Antibody, and PE/Cy7 Mouse IgG1 κ Isotype Ctrl Antibody (BioLegend, Inc., San Diego, CA, USA).
Animal Care
All experimental protocols were in accordance with the guidelines for the care and use of laboratory animals set by Research Laboratory Center, Faculty of Medicine and the Institute for Animal Experiments, Faculty of Medicine, University of the Ryukyus (Okinawa, Japan). The experimental protocol was approved by the Committee on Animal Experiments of University of the Ryukyus (permit number: A2017101). C57BL/6 male mice (8-week-old; Japan SLC, Shizuoka, Japan) were maintained under controlled temperature (23 ± 2°C) and light conditions (lights on from 08:30–20:30). Animals were fed standard rodent chow pellets with ad libitum access to water. All efforts were made to minimize the suffering of the animals.
Preparation of the Mouse Model of Acute Liver Failure
Carbon tetrachloride (CCL4) (Wako 035-01273) diluted with olive oil (Wako 150-00276) was administered intraperitoneally (0.5 mL/kg) to 8-week-old C57BL/6 male mice as a mouse model of acute liver failure. Nine mice each were used for both treated and control animals. At 4 h after the administration of CCL4, 20-fold concentrated culture supernatant was administered via the mouse tail vein (100 μl of PBS and hMSC-AT-CM solution was administered via the mouse tail vein). Blood and liver tissues were sampled at 24 h after the administration of CCL4. Under anesthesia, approximately 500 μL of blood was collected from the descending aorta using a 1 mL syringe (22 G injection needle) passed through heparin, centrifuged (150 g, 30 min, 4°C) after the coagulation, 100 μL of blood was obtained. Four hundred microliters of physiological saline were added to 100 μL of serum and diluted, and the blood components were analyzed (commissioned to SRL). The liver was fixed in formalin after sampling and HE staining was performed after the preparation of tissue sections. Fragmented DNA generated during apoptosis was detected by a TdT-mediated dUTP nick end-labeling (TUNEL) assay to identify apoptotic cells in the liver tissue. TUNEL staining was performed using the In Situ Apoptosis Detection kit (Takara Bio Inc., Shiga, Japan) and visualized using DAB as the chromogen. The Ki67 protein present in the nucleus of cells in G1, S, G2, M cycles (cell growth phase) was detected by using immunostaining in order to identify cells in the growth phase in liver tissue. The reagent Histofine Simple Stain MAX PO (Rubbit) (NICHIRE BIOSCIENCES INC., Tokyo, Japan) and anti-Ki67 antibody (ab 15580) (Abcam, Cambridge, UK) were used.
Preparations of hMSC-AT-CMs for Animal Studies and the Analysis of the Protein Expression by LC-MS/MS
The hMSCs used in this study are limited to three to five passages in order to match the cell nature with clinically used hMSCs. Two milliliters of DMEM/F12 medium was added to hMSC-AT (1 × 106 cells) and cultured for 48 h to prepare hMSC-AT-CMs; this was concentrated to 1/20 of the original volume using a 10 k filter, 100 μL was injected per mouse. The 20-fold concentrated hMSC-AT-CM was serous and successfully passed through a 32 G injection needle (Fig. 1(a)). Two milliliters of clinical Xeno-free medium (MSCGM-CD mesenchymal stem cell BulletKit [Lonza]) was added to hMSC-AT (1 × 106 cells) and cultured for 48 h to prepare hMSC-AT-CMs and then concentrated to 1/20 of the original volume using a 10 k filter, after which the component proteins were analyzed by LC-MS/MS. Twenty-fold concentrated hMSC-AT-CM was subjected to LC-MS/MS. If the medium’s albumin concentration is high, the accuracy of a protein analysis decreases. Thus, after washing these cells with phosphate buffered saline (PBS), they were cultured in albumin-free medium and the resulting culture supernatant was used for this study (Fig. 1(b)). One hundred twenty-eight proteins were identified from the hMSC-AT-CM samples; the identified proteins are listed in Table 1. In this study, DMEM/F12 (containing 0% FBS) was used to prepare hMSC-AT-CMs to be administered to mice, due to the difficulty of accurately observing the therapeutic effect of hMSC-AT-secreted protein when the protein component of clinical Xeno-free medium rich in growth factor proteins is concentrated.

Illustration of the preparation of conditional medium for hMSC-AT. (a) The procedure for administering 100 μL hMSC-AT-CM concentrate to the tail vein of the mouse. (b) The procedure for preparing the hMSC-AT-CM concentrate for the LC-MS/MS analysis.
Details of the hMSC-AT Secreted Protein Identified.

a Protein score is calculated from the score of the peptide attributed to the protein; b pI: (Predicted) isoelectric point.; c Number of matches is spectrum number matched to protein#1; d Number of significant matches is spectrum number that matches protein and exceeds the identification criteria; e Number of sequences is number of peptides matched to protein#2; f Number of significant sequences is number of peptides exceeding the identification criteria matched to proteins; h Sequence coverage is the ratio of the total number of matched peptide residues to the total length of the protein; I Exponentially Modified Protein Abundance Index (http://www.matrixscience.com/help/quant_empai_help.html).
Real-time PCR and RT-PCR
Five microliters of a cell admixture (concentration, 1 × 107 cells/ml) was collected. RNA was prepared for a qPCR using a SuperPrep Cell Lysis and RT kit according to the manufacturer’s instructions (Toyobo Co., Ltd., Osaka, Japan). Quick Taq HS DyeMix was used according to the manufacturer’s instructions (Toyobo Co., Ltd.). Real-time PCR analyses were performed using a LightCycler 96 Real-Time PCR system (Roche, Basel, Switzerland). The FastStart Essential DAN Green Master (Roche) was used according to the manufacturer’s instructions. An RT-PCR was performed using a GeneAtlas 482 thermal cycler (Astec Co., Ltd., Fukuoka, Japan). Images were recorded using an Aplegen® Omega Lum C (Gel Company, San Francisco, CA, USA), and procedures were performed using the primers listed in Table 2.
Sequences of Primers used for the RT-PCR.

Preparation of hMSC-AT
hMSC-ATs (46-year-old Caucasian female) were cultured (37°C, 5% CO2) on a coated 100-mm culture plate (TPP 93100). The passage of cells was performed every 3 to 4 days after reaching 80% confluence after sowing the cells. The cells were washed with PBS (calcium, magnesium-free), and hMSC-ATs were dissociated using a dissociation solution. Subculturing was carried out by plating on uncoated 100-mm culture plate. An MSCGM-CD mesenchymal stem cell BulletKit (Lonza 00190632) was used for the culture medium. Trypsin/EDTA (Lonza CC-3232) was used for the dissociation solution.
Preparation of Culture Supernatant
hMSC-ATs were cultured on a 100 mm culture plate using an MSCGM-CD mesenchymal stem cell BulletKit (the number of cells was 3 × 106/plate) until reaching 80% confluence. The cells were cultured for 24 h in D-MEM/Ham’s F-12 medium (Wako 4230795) containing 10% FBS, after which the cells were washed with PBS (calcium, magnesium-free); 2 ml of D-MEM/Ham’s F-12 medium was then added to 1 × 106 cells and the cells were cultured for 48 h. After 48 h, the culture supernatant was aspirated with a pipette and centrifuged (1500 g, 30 minutes, 4°C) to remove the cells. After the centrifugation of the medium, the supernatant was concentrated 20 times using Amicon Ultra-15, PLGC Ultracell-PL membrane, 10 kDa (UFC901008) (MERCK, Kenilworth, NJ, USA) and a concentrated solution of culture supernatant was obtained.
Protein Identification by a Nano LC-MS/MS Analysis
A protein solution of 2066 μg/ml was obtained from the concentrated solution of culture supernatant. Finally, 0.4 μg of protein was used for nanoLC-MS/MS. The samples were analyzed via nano LC using an UltiMate 3000 RSLC nano system (Thermo Fisher Scientific, Tokyo, Japan) at the Support Center for Advanced Medical Sciences, Institute of Biomedical Sciences, Tokushima University Graduate School by Ikuko Sagawa. In brief, protein-containing solutions were reduced with 10 mM DTT/8 M urea and Tris buffer containing 2 mM EDTA (pH 8.5), alkylated with 25 mM iodoacetamide/8 M Urea and Tris buffer containing 2 mM EDTA (pH 8.5), subsequently diluted with trypsin (pig-derived trypsin) and digested overnight at 37°C. Peptides were purified and concentrated by solid-phase extraction (SPE) in ZipTip µC18 pipette tips (Merck Millipore, Darmstadt, Germany). Nano LC-MS/MS was carried out using an UltiMate 3000 RSLC nano system. The reconstituted peptides were injected into an Acclaim PepMap C18 trap column (75 μm × 15 cm, 2 μm, C18) (Merck Millipore, Darmstadt, Germany). Solvent A was 0.1% formic acid. Solvent B was 80% acetonitrile/0.08% formic acid. The peptides were eluted in a 229-min gradient of 4% solvent B in solvent A to 90% solvent B in solvent A at 300 nl/min. Orbitrap Elite’s ionization method was set to Nanoflow-LC ESI, positive, and the capillary voltage was set to 1.7 kV. Tandem mass spectrometry was performed using the Proteome Discoverer software program, version 1.4 (Thermo Fisher Scientific, Tokyo, Japan). Charge stated deconvolution and deisotoping were not performed.
Data Analyses
Database Searching
All raw data were searched against the SwissProt 2016-07 database using the Mascot 2.5.1 software program (Matrix Science, London, UK) (unknown version, 551705 entries). The peptide tolerance was set to 10 ppm, and the MS/MS tolerance was set to 0.6 Da. The false discovery rates (FDRs) were calculated for each of the samples using the following formula: FDR = (Ndecoy/Nreal+NDecoy) × 100. This is an indication of the percentage of the random or “false” peptide identifications in the raw data. The relative abundance of the proteins identified by LC-MS/MS was estimated by determining the protein abundance index (PAI) and the exponentially modified protein abundance index (emPAI). Visualized and validated complex LC-MS/MS proteomics experiments were performed using Scaffold (version 4.7.3, Proteome Software Inc., Portland, OR, USA – http://www.proteomesoftware.com/) to compare samples in order to identify biological relevance.
The Criteria for Protein Identification
The Scaffold software program was used to validate the MS/MS-based peptide and protein identifications. Peptide identifications were accepted if they could be established at > 46.0% probability to achieve an FDR of < 1.0% by the Scaffold Local FDR algorithm. Protein identifications were accepted if they could be established at > 5.0% probability to achieve an FDR of < 1.0% and contained at least 2 identified peptides. Protein probabilities were assigned by the Protein Prophet algorithm 17 . Proteins that contained similar peptides and could not be differentiated based on MS/MS alone were grouped to satisfy the principles of parsimony. Proteins that shared significant peptide evidence were grouped into clusters. A protein GO analysis was performed using the GO analysis function of the Scaffold 4 software program with imported data (goa_uniprot_all.gaf [downloaded 2016/10/14]) from the external GO Annotation Source database.
Results
The Characteristics and Cell Quality of hMSC-ATs
hMSC-ATs were cultured to an 80% confluent state using Clinical Xeno-free medium. We observed the absence of abnormalities in cell size, shape, and culture state with a normal microscope (Fig. 2(a)). Flow cytometry was performed using markers of hMSC-AT (CD 29, CD 44), hematopoietic stem cells (CD 34), and leukocytes (CD 45). Markers of CD29 and CD44 were expressed in hMSC-AT, while the expression of CD34 and CD45 was not detected (Fig. 2(b)). The expression of hMSC-AT markers (CD29, CD44), hematopoietic stem cells (CD34), and leukocytes (CD45) was examined by real-time PCR. CD29 and CD44 were expressed by hMSC-AT, while the expression of CD34 and CD45 was not detected (Fig. 2(c)). The PCR method was used to examine the mRNA expression levels of hepatocyte growth factor (HGF), a suppressor of cancer cell invasion (SCAI) and vascular endothelial growth factor A (VEGFA) expressed in hMSC-AT. (Fig. 2(d)). hMSC-AT-CM was prepared using DMEM/F12 medium. Prior to concentrating hMSC-AT-CM to 1/20 using a 10 k filter, the protein concentration was measured using an ELISA (Fig. 1(a)). The expression of hMSC-AT secreted proteins was examined by an ELISA (R&D Systems, Minneapolis, MN, USA), which revealed that hMSC-AT secreted VEGFA proteins into the culture medium (control group: < 20 [N.D] ± 0.00 pg/ml, n = 3; hMSC group: 886.67 ± 28.93 pg/ml, n = 3) (Fig. 2(e)). hMSC-AT have been reported to secrete HGF9. In our experiments, we could not show the measurement because the detection limit of the ELISA (Otuka, Tokyo, Japan) to detect HGF (0.3 ng/ml) was high (control group: < 0.3 [N.D] ± 0.00 ng/ml, n = 3; hMSC group: < 0.3 ± 0.00 ng/ml, n = 3). We induced differentiation into adipocytes (Fig. 2(f), upper panels) and osteoblasts (Fig. 2(f), lower panel) using hMSC-AT. Mature adipocytes were stained with Oil Red O and mature osteoblasts were stained with alkaline phosphatase (Fig. 2(f), right panel). hMSC-ATs were cultured in three wells of a six-well plate. Adipocytes stained red with Oil Red O staining in all three wells and osteoblasts stained blue with alkaline phosphatase staining in all three wells were confirmed with a normal microscope.

The phenotype and differentiation potential of hMSC-AT in culture. (a) The morphological appearance of hMSC-AT on day 3. (b) The results of flow cytometry of the cell surface markers of hMSC-AT. (c) The results of real-time PCR to detect cell surface markers of hMSC-AT. The expression was calculated using the ΔΔCt method. The expression of the target gene was corrected by the expression of the housekeeping gene. The relative values are indicated. n = 1. (d) The results of an RT-PCR to evaluate the growth factor and cell surface markers mRNA expression of hMSC-AT. (e) The results of an ELISA to evaluate the growth factor protein expression of hMSC-AT-CM. (f) Representative images of adipocyte and osteocyte differentiation of hMSC-AT cultured in growth or differentiation medium.
hMSC-AT-CM Improves the Liver Function of Mice with Acute Liver Failure
CCL4 was intraperitoneally (i.p.) administered to mice to induce hepatic cell damage and model mice were prepared. The upper part of the photo shows the liver histology at 24 h after the administration of CCL4. The hepatocytes of the centrilobular region showed necrotic change. However, when hMSC-AT-CM was injected into the tail vein at 4 h after the administration of CCL4, the number of necrotic cells was reduced. Cells in the growth phase (shown in the panels of Ki67) and apoptotic cells (shown in the panels of the TUNEL assay) of liver tissue sections were detected. In mouse liver administered hMSC-AT-CM, the number of apoptotic cells widely observed in liver tissues was reduced by CCL4 administration. Furthermore, the apoptotic cells were localized to the interlobular vein in liver tissues treated with hMSC-AT-CM. Cells in the growth phase were observed around the cells showing apoptosis due to the administration of CCL4 (Fig. 3(a), left and middle panels). However, cells in the growth phase were uniformly observed in liver tissues treated with hMSC-AT-CM. Ki67 was expressed only in the nucleus, and cells in the proliferation phase had brown-stained nuclei. Mouse hepatocytes in the group treated with hMSC-AT-CM showed more nuclear-stained cells than those in the group treated with PBS, thus indicating that hMSC-AT-CM promoted hepatocyte proliferation (Fig. 3(a), right panel). We also counted the number of positively stained cells in images of TUNNEL-stained sections (× 100). The numbers of positively stained cells in the PBS and CM groups were 15.25 ± 3.96 and 10.00 ± 5.07, respectively (n = 4; P = 0.18) (Fig. 3(a), middle panels). We also counted the number of cells with positively stained nuclei on images of Ki67-stained sections (× 100). The numbers of cells with positively stained nuclei in the PBS and CM groups were 10.25 ± 4.23 and 90.75 ± 38.42, respectively (n = 4; ** P < 0.01) (Fig. 3(a), right panels). These results indicate that hMSC-AT-CM rapidly recovered because of the generation of new viable cells as the older cells died due to CCL4 administration (Fig. 3(a)).
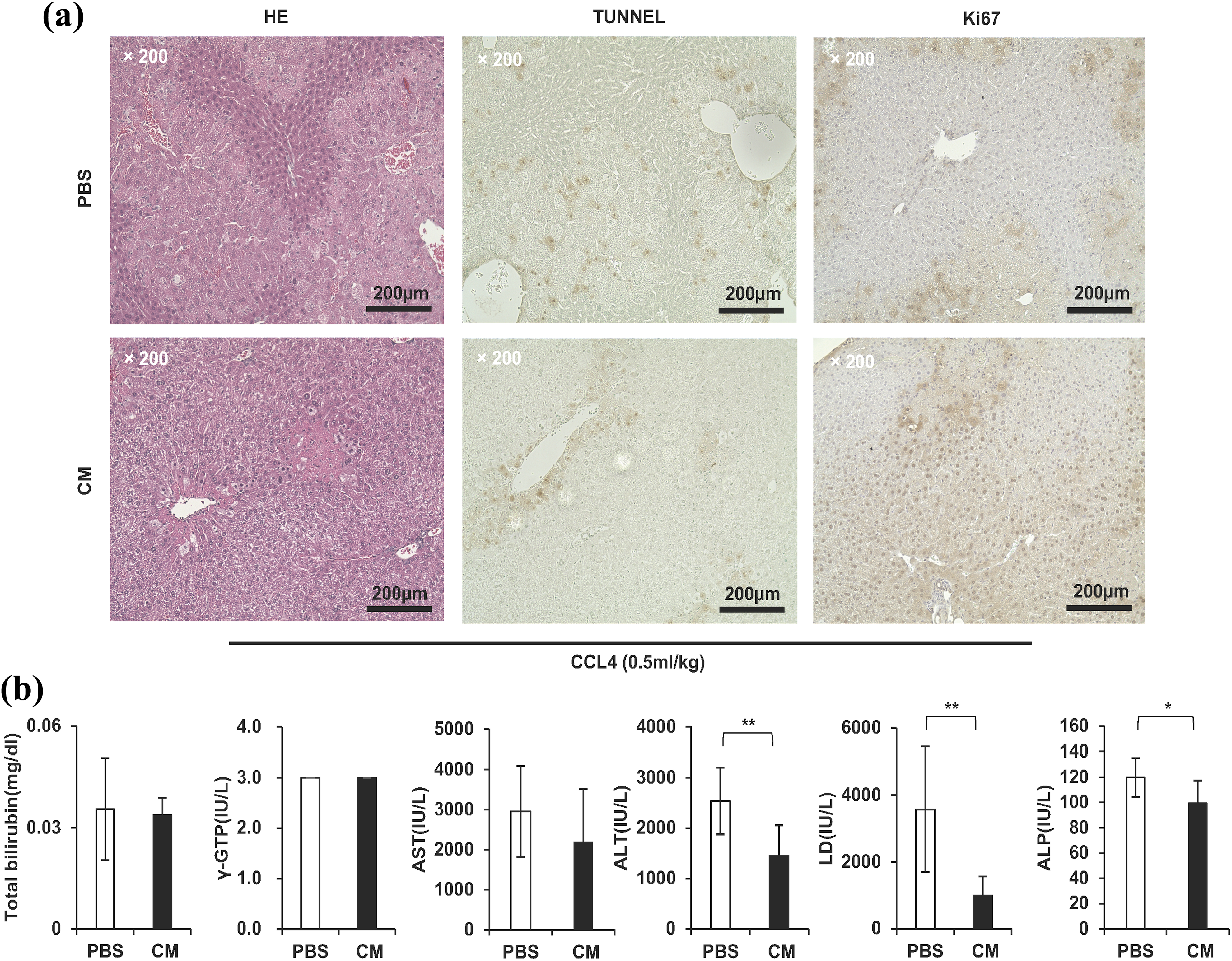
The culture supernatant concentrate significantly improved the symptoms of acute liver failure caused by the administration of CCL4. (a) Micrographic image of H&E staining (left panel), TUNEL assay (middle panels) and tissue immunostaining of Ki67 (right panel) of liver specimens. Scale bar = 200 μm. (b) In the group to which the culture supernatant concentrate was administered, the total bilirubin (95%), AST (74%), ALT (57%), LD (28%), and ALP (83%) decreased in comparison with the group to which PBS was administered. The decrease in the ALT, LD, and ALP values was significant (** P < 0.01, n = 9).
Our experiments show that the administration of MSC-AT-CM from a single vein rapidly promoted the cellular proliferation of mouse hepatocytes. The proteins associated with a growth function (GO analysis), identified by the presence of MSC-AT-CM, were POSTN, SAP, SEM7A, PTK7 (Table 3). Of course, it is not possible to explain the proliferative effect of hepatocytes based on the presence of four proteins. Periostin, which is encoded by the POSTIN gene, has been reported as an extracellular factor that promotes hepatosteatosis 18,19 ; however, many points about proteins with the ability to promote the cellular proliferation of hepatocytes remain unclear. P component (SAP) is a protein that is expressed in hepatocytes and secreted into serum, and is known to be involved in processes associated with immune regulation, such as the action of opsonins 20 . Whether SAP is involved in the cellular proliferation of hepatocytes is unknown. Semaphorin 7A (SEM7A) is known to contribute to TGF-β mediated hepatic fibrosis 21 . It is unknown whether SEM7A promotes hepatocyte cell proliferation. Thus, future studies should investigate whether the growth-associated proteins that are newly identified by GO analyses promote the cellular proliferation of hepatocytes with CCL4-induced impairment. At approximately 10 days of gestation, during the development of the liver, the hematopoietic cells flow from the aorta-gonad-mesonephros region (AGM region) and placenta, and the liver begins to function as a hematopoietic organ 22 . It has been clarified that HGF and various extracellular matrices produced by non-parenchymal cells promote the differentiation of hepatoblasts into hepatocytes during this period 23 . In addition, a recent theory suggests that the biliary tree functions as a source of liver and pancreatic stem cells and progenitor cells. It is known that VEGF is secreted by the biliary tree due as a stress response 24 . From these developmental perspectives, it can be hypothesized that the HGF and VEGF secreted by MSC-AT-CMs have an extremely strong promoting effect on hepatocyte proliferation.
Biological Process.
Serum from the model mice was sampled and biochemically analyzed. The average value of each measurement was s as follows (correction was not made by diluting 100 μL of serum with 400 μL of physiological saline). Total bilirubin (PBS 0.04 ± 0.02, supernatant concentrate 0.03 ± 0.01 (unit mg/ml)), AST (PBS 2956 ± 1133, supernatant concentrate 2195 ± 1319 (unit IU/L)), ALT (PBS 2538 ± 663, supernatant concentrate 1448 ± 608 (unit IU/L)), LD (PBS 3574 ± 1873, supernatant concentrate 997 ± 572 (unit IU/L)), ALP (PBS 120 ± 15, supernatant concentrate 99 ± 18 (unit IU/L)). The serum liver injury markers (ALT, LD and ALP) were significantly reduced at 20 h after the administration of hMSC-AT-CM (Fig. 3(b)).
The Biological Processes, Cellular Components and Molecular Function of Proteins Identified from hMSC-AT-CM
The biological processes of proteins were analyzed using the Mascot software program with the SwissProt 2016 database.
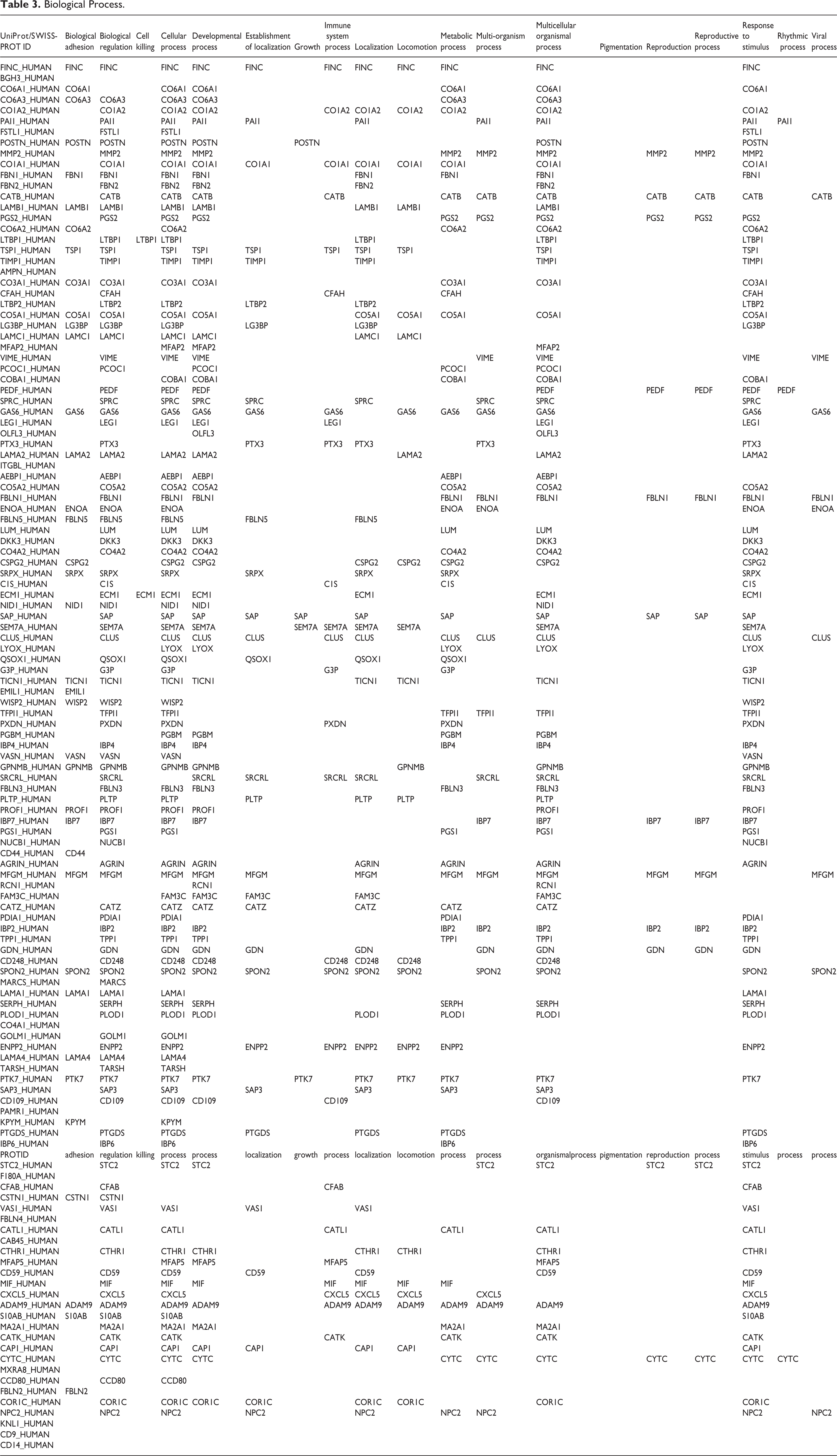
In this study, a secreted protein expression analysis of hMSC-AT was performed using LC-MS/MS and 128 proteins were identified (Table 1). LC-MS/MS showed that 106 new functional proteins and 22 proteins (FINC, PAI1, POSTN, PGS2, TIMP1, AMPN, CFAH, VIME, PEDF, SPRC, LEG1, ITGBL, ENOA, CSPG2, CLUS, IBP4, IBP7, PGS1, IBP2, STC2, CTHR1, CD9) were previously reported in hMSC-AT-CMs. In addition, various proteins associated with growth (SAP, SEM7A, PTK7); immune system processes (CO1A2, CO1A1, CATB, TSP1, GAS6, PTX3, C1 S, SEM7A, G3P, PXDN, SRCRL, CD248, SPON2, ENPP2, CD109, CFAB, CATL1, MFAP5, MIF, CXCL5, ADAM9, CATK); and reproduction (MMP2, CATB, FBLN1, SAP, MFGM, GDN, CYTC) were identified in hMSC-AT-CMs.
Biological processes
FINC, CATB, TSP1, GAS6, SAP, SEM7A, SRCRL, MFGM, GDN, SPON2, PTK7, ADAM9 and CYTC all seemed to be widely involved in the function of hMSC-AT-CM under the classification of ‘biological processes’ (Table 3). FINC was distributed in sites such as those associated with the response to biological adhesion, biological regulation, cellular processes, the developmental process, the establishment of localization, the immune system process, localization, locomotion, the metabolic process, the multicellular organismal process, and response to stimulus. Collagen types I, V, VI and XII, and fibronectin (ECM components) were detected in hMSC-AT-CM by MALDI-TOF/TOF mass spectrometry 25 . Fibronectin is a major ECM component that supports cell adhesion by presenting an integrin binding domain 26 . FINC in plasma is taken up by the fibrin clot during tissue injury, contributing to the platelet function and hemostasis. The cell’s FINC is then synthesized by the cells to reconstitute the damaged tissue 27 . CATB, TSP1, GAS6, SAP, SEM7A, SRCRL, MFGM, GDN, SPON2, PTK7, ADAM9 and CYTC were newly detected in hMSC-AT-CM. MFGM was distributed in sites such as those associated with the response to biological adhesion, biological regulation, cellular processes, the developmental process, the establishment of localization, localization, the metabolic process, multi-organism processes, multicellular organismal processes, reproduction, the reproductive process, and the viral process. Jang et al. presented a pathology model showing that MFGM inhibits hepatic fibrosis via the signal of transforming growth factor (TGF)-β 28 (Fig. 4a).

The biological processes, cellular components and molecular function of the hMSC-AT-CM proteins (as determined by GO). The PCA of proteome dynamics based on the protein information generated by high-resolution mass spectrometry. (a) The ordinate shows each protein’s biological function, and the abscissa indicates the proteins that were identified. The names of the proteins classified in Table 3 are listed by their abbreviated names. (b) The ordinate shows the name of each organelle, and the abscissa indicates the number of proteins identified. The names of the proteins classified in Table 4 are listed by their abbreviated names. (c) The ordinate shows each protein’s molecular function, and the abscissa indicates the proteins identified. The names of the proteins classified in Table 5 are listed by their abbreviated names.
Cellular components
Proteins synthesized in the rough endoplasmic reticulum are transported to the lumen of the rough endoplasmic reticulum and transported or secreted to the cell membrane via the Golgi apparatus. In hMSC-AT-CM, GAS6, CLUS, NUCB1, CATZ, PTGDS, STC2, CSTN1, and CD59 also seem to be widely involved in the function of hMSC-AT-CM under the classifications of endoplasmic reticulum, Golgi apparatus, membrane, and extracellular region of ‘cellular component’ (Table 4). STC2 suppresses the oxidative stress-induced cell damage of MSC. In the clinical application of MSC, it was suggested that STC2 promotes the long-term therapeutic effects of therapeutic cells 29 . GAS6, CLUS, NUCB1, CATZ, CSTN1 and CD59 were newly detected in hMSC-AT-CM (Fig. 4b).
Cellular Component.

Molecular function
In hMSC-AT-CM, LTBP1, AMPN, GAS6, FBLN1, PXDN, FBLN3, PGS1, ENPP2, PTK7 and MIF also seem to be widely involved in the function of hMSC-AT-CM under the classification of ‘molecular function’ (Table 5).
Molecular Function.

POSTN, PGS2, TIMP1, PEDF, LEG1 and IBP7, the protein function of which was not especially wide was related to the biological processes, cellular components and molecular function of hMSCs (Table 1). POSTN has previously been reported as a factor that promotes the in vivo proliferation activity of cancer in association with hACS transplantation 30 . POSTN has been reported to promote the cell migration of MSC-BM via PI3K/Akt signaling through receptor integrin αvβ3 31 . The simultaneous administration of MSC-BM and PGS2 was reported to significantly improve thioacetamide-induced the rat model of hepatic fibrosis in comparison with the administration of MSC-BM alone 32 . The TIMP1 contained in the culture supernatant of the immortalized MSC line RCB2157 was reported to inhibit the migration and invasion of breast cancer cells 33 . MSC-BM in aged mice show the increased expression of PEDF. PEDF was reported to promote or inhibit the growth of cells affected by myocardial infarction 34 . PEDF has also been reported to promote the expression of bone formation genes and mineral deposition genes of human MSC-BM 35 . It has been reported that IBP7 has an important function in the action of MSC-BM in preparation for immune regulation in a mouse model of colitis 36 . LTBP1, AMPN, GAS6, FBLN1, PXDN, FBLN3, PGS1, ENPP2, PTK7 and MIF were newly detected in hMSC-AT-CM (Fig. 4c).
Discussion
In recent years, genome sequencing and epigenetic analysis techniques have provided important information to help clarify the causes of diseases. The application of cell therapy in regenerative medicine is expected to be useful for the treatment of many types of diseases. Genetic, epigenetic, and proteomic analysis techniques play an important role in inducing the differentiation of cells used for cell therapy. Several papers focusing on the MSCs involved in the treatment of liver diseases have been published and the functions of the factors identified in the latest analysis have been explained.
A proteomic analysis using LC-MS/MS provides evidence to support the possible application of cell therapy using MSCs and information regarding the potential application of MSCs in the treatment of liver disease. This information provides important clues for investigating the function. However, MSCs are distributed throughout the body, and there are different types of MSCs, such as mesenchymal stem cells from adipose tissue (MSC-ATs), bone marrow (MSC-BMs), umbilical cord blood (MSC-UCs), and dental pulp (MSC-DPs). In previous reports, to identify the proteins expressed in MSCs, MSC-BMs, components contained in the culture supernatant of MSC-DPs and MSC-ATs were examined 8,10,11 . Banas et al. showed that hMSC-AT secreted interleukin (IL)-1 receptor antagonist (IL-1RA), IL-6, IL-8, and granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), monocyte chemotactic protein 1 (MCP-1), nerve growth factor (NGF), and HGF using a protein-array analysis 8 . The authors explained that these factors were effective in improving the mouse liver function. Poll et al. showed that the analysis of the serum levels of pro-inflammatory cytokines, which are known to be upregulated during liver injury, revealed a nonsignificant decrease in the levels of IL-1 and significantly lower levels of TNF-α and IL-6 after MSC-CM treatment. On the other hand, the levels of IL-10 (an anti-inflammatory cytokine) were increased four-fold in MSC-CM–treated animals. These data suggest that the infusion of MSC-CM alters the systemic cytokine profile associated with acute liver failure to a more anti-inflammatory state 11 . Yukawa et al. reported that the administration of mouse MSC-ATs into the blood resulted in an improvement of the liver function and a reduction in the blood concentrations of TNF-α, IL-1β and IL-6 in mice 37 . The authors cited a paper that reported that IL-6 is effective for improving the liver function of mice among these factors and explained the improvement of the liver function of MSC-CM 38 . Parekkadan et al. reported that the majority (69/174 [30%]) of proteins contained in MSC-BM-CM (according to a protein array) are chemokines and are widely involved in immune regulation and liver regeneration 11 . Similarly to the abovementioned studies, hMSC-AT-CM was also shown to improve mouse liver function (Fig. 3(a) and (b)). This study showed that hMSC-AT-CM was administered to mice to ameliorate the symptoms of acute liver failure induced by the administration of CCL4. Our findings indicate that hMSC-AT-CM is likely to have the effect of ameliorating symptoms of human liver disease. Therefore, the MSCGM-CD mesenchymal stem cell BulletKit (Lonza) was used to create hMSC-AT-CM. This medium was a clinical grade medium approved by the Japanese Ministry of Health, Labor and Welfare for use in human clinical treatment research. However, we must bear in mind that the components and amounts of hMSC-AT-CM secreted by hMSC-ATs will likely change depending on the composition of the culture medium.
Since the data in the present study were obtained from the hMSC-AT-CM from one donor, we must consider the reliability of the data. In addition, the proteins were detected by a label-free method. Protein quantification was determined from the peptide ion data obtained by mass spectrometry using the number of peptide fragments identified by the database analysis as an index. This principle is based on the PAI 39 method, which states that, “quantitatively more proteins can detect more peptide fragments in the same protein.” This method was used to determine the emPAI 40 , which estimates the protein abundance based on the peptides calculated and theoretically observed tryptic peptides for each protein using the Scaffold software program. This program identifies and quantitatively displays proteins using proprietary algorithms (Peptide/Protein Prophet, Protein grouping). Thus, the quantification of the amount of protein in this paper is a theoretical value estimated based on the emPAI 40 function of the Scaffold software program. The ratio of the number of measured peptides to the number of theoretical peptides is linearly related to the logarithm of the protein concentration, and the number obtained by subtracting 1 from the index of the peptide number ratio was defined as the emPAI 40 . The larger the emPAI 40 value, the greater the amount of protein. Proteins quantified using emPAI were listed from the top in the tables showing the GO analysis results (Tables 1, 3, and 4) in descending order of concentration.
Conclusions
In this study, which used an LC-MS/MS measuring system, we focused on the quantified amount of protein and components contained in hMSC-AT-CM that improve the liver function, with a focus on the function of proteins classified by a GO analysis. These analyses revealed a number of new candidates associated with growth (SAP, SEM7A, PTK7); the immune system processes (CO1A2, CO1A1, CATB, TSP1, GAS6, PTX3, C1 S, SEM7A, G3P, PXDN, SRCRL, CD248, SPON2, ENPP2, CD109, CFAB, CATL1, MFAP5, MIF, CXCL5, ADAM9, CATK); and reproduction (MMP2, CATB, FBLN1, SAP, MFGM, GDN, CYTC). MSC-CM contains proteins secreted by MSCs and the proteins that were initially added to the culture medium. In Table 6, the proteins identified in hMSC-AT-CM are listed in the far-left column, with the medium component proteins using culture medium for hMSC-ATs listed in the next column. Proteins secreted by hMSC are predicted by the following formula: hMSC-AT-CM containing protein – medium containing protein = hMSC-AT secreted protein. Table 6 also lists eight articles that can be searched using the keywords MSC, ADSC, mesenchymal stem cell, LC/MS/MS, CM, conditional medium, protein, and secretion on the PubMed database (https://www.ncbi.nlm.nih.gov/pubmed/). Secreted proteins of MSCs are listed in Table 6. This research method differs from a protein array and enables a comprehensive analysis of the protein expression. We succeeded in identifying 106 types of novel proteins contained in MSC-CM. The newly identified protein components contained in hMSC-AT-CM provide valuable information to support the clinical application of hMSC-AT-CM.
Previous Reports; hMSC Secreted Protein Identified.

a human Mesenchymal Stem Cells from adipose tissue;b human Mesenchymal Stem Cells from Bone marrow; c human Mesenchymal Stem Cells from dental pulp; d conditional medium.
Supplemental Material
Supplemental Material, Nakashima(SupFig1)2018-6-11(3) - A Liquid Chromatography with Tandem Mass Spectrometry-Based Proteomic Analysis of the Proteins Secreted by Human Adipose-Derived Mesenchymal Stem Cells
Supplemental Material, Nakashima(SupFig1)2018-6-11(3) for A Liquid Chromatography with Tandem Mass Spectrometry-Based Proteomic Analysis of the Proteins Secreted by Human Adipose-Derived Mesenchymal Stem Cells by Yoshiki Nakashima, Saifun Nahar, Chika Miyagi-Shiohira, Takao Kinjo, Zensei Toyoda, Naoya Kobayashi, Issei Saitoh, Masami Watanabe, Jiro Fujita, and Hirofumi Noguchi in Cell Transplantation
Footnotes
Acknowledgements
We thank Naomi Kakazu (University of the Ryukyus) for clerical assistance and Saki Uema, Yuka Onishi, Maki Higa, Youichi Toyokawa, Yuki Kawahira and Saori Adaniya (University of the Ryukyus) for providing technical support. We thank Masayoshi Tsukahara (Kyowa Hakko Kirin Co., Ltd.) for his expert technical advice on cell culture methods, which was provided under a cooperative research contract with Kyowa Hakko Kirin Co., Ltd.
Ethical Approval
Ethical Approval is not applicable for the article. (In this paper, we did not conduct clinical studies that required Institutional review).
Statement of Human and Animal Rights
All experimental protocols were performed according to the guidelines for the care and use of laboratory animals set by Research Laboratory Center, Faculty of Medicine, and the Institute of Animal Experiments, Faculty of Medicine, University of the Ryukyus (Okinawa, Japan). The experimental protocol was approved by the Committee on Animal Experiments of University of the Ryukyus (permit number: A2017101).
Statement of Informed Consent
Statement of Informed Consent is not applicable for the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Japan Society for the Promotion of Science (JSPS; KAKENHI Grant Number 16H07094), Japan Agency for Medical Research and Development, The Naito Foundation, and Okinawa Science and Technology Promotion Center (OSTC). This work was supported by the Research Laboratory Center, Faculty of Medicine, and the Institute for Animal Experiments, Faculty of Medicine, University of the Ryukyus.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
