Abstract
The aim of this study was to compare the serum levels of the anti-angiogenic factor endostatin (S-endostatin) as a potential marker of vasculogenesis after autologous cell therapy (ACT) versus percutaneous transluminal angioplasty (PTA) in diabetic patients with critical limb ischemia (CLI). A total of 25 diabetic patients with CLI treated in our foot clinic during the period 2008–2014 with ACT generating potential vasculogenesis were consecutively included in the study; 14 diabetic patients with CLI who underwent PTA during the same period were included in a control group in which no vasculogenesis had occurred. S-endostatin was measured before revascularization and at 1, 3, and 6 months after the procedure. The effect of ACT and PTA on tissue ischemia was confirmed by transcutaneous oxygen pressure (TcPO2) measurement at the same intervals. While S-endostatin levels increased significantly at 1 and 3 months after ACT (both P < 0.001), no significant change of S-endostatin after PTA was observed. Elevation of S-endostatin levels significantly correlated with an increase in TcPO2 at 1 month after ACT (r = 0.557; P < 0.001). Our study showed that endostatin might be a potential marker of vasculogenesis because of its significant increase after ACT in diabetic patients with CLI in contrast to those undergoing PTA. This increase may be a sign of a protective feedback mechanism of this anti-angiogenic factor.
Introduction
Critical limb ischemia (CLI) represents the end stage of peripheral arterial disease (PAD) and portends a high rate of limb loss along with patient mortality rates 1 . The diagnosis and therapy of CLI in patients with diabetes is challenging because they frequently lack typical symptoms due to peripheral neuropathy, and most of the lesions affect infrapopliteal arteries 2 . Traditionally, standard methods in the treatment of CLI have been open bypass surgery or endovascular interventions, such as percutaneous transluminal angioplasty (PTA) or stenting 3 . However, about 30% of CLI patients are not eligible for standard revascularization due to excessive operative risk or unfavorable vascular involvement 4 . These patients with no-option CLI might obtain therapeutic benefit from autologous cell therapy (ACT) as an alternative therapeutic modality for CLI studied in clinical trials 5,6 . Previous studies have shown that therapy using autologous cells injected into the affected lower limb was well tolerated and effective for achievement of better clinical outcomes 7,8 .
ACT is based on cell suspension, which consists of heterogeneous mononuclear cells containing a progenitor cell population harvested from bone marrow (bone marrow-derived mononuclear cells; BMMNCs) or peripheral blood progenitor cells (PBPCs) separated from peripheral blood after stimulation by granulocyte colony-stimulating factor (G-CSF) 9 . According to previous studies 10,11 , the cell population characterized by CD34+ surface marker is probably the most important cell type for vascular regeneration and is mainly composed of hematopoietic stem cells with a subset of endothelial progenitor cells (EPCs) 12,13 . EPCs are capable of migrating from the bone marrow and differentiate into mature endothelium in the areas of ischemia 14 .
While the exact mechanism of action of injected autologous cells at tissue level remains unclear, their involvement in vasculogenesis is assumed. Two possible mechanisms of vasculogenesis – angiogenesis and arteriogenesis – have been suggested 15 . Unlike angiogenesis, which is induced by hypoxia and results in the growth of new capillaries, arteriogenesis is induced by physical forces, mainly increased fluid shear stress that leads to remodeling and maturation of pre-existing collaterals 16,17 . One of the possible explanations for the mechanism of action of ACT is that these injected cells can directly contribute to vasculogenesis by being incorporated into nascent vessels; alternatively, they can release growth factors that affect endogenous vascular cells, or directly instruct host cells to secrete angiogenic growth factors 18 .
The release of pro- and anti-angiogenic factors is a possible modulator of vasculogenesis 19 . The most important pro-angiogenic factors include vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) 20 , angiopoietin-1, platelet-derived growth factors (PDGF-AA, PDGF-BB), or hypoxia-inducible factor (HIF-1α) 21 , whereas the main anti-angiogenic factors are represented by endostatin, angiostatin, arresten, canstatin, tumstatin, or thrombospondins 22 .
Endostatin, one of the most potent inhibitors of angiogenesis, is cleaved from collagen XVIII, which is a component of vascular basement membranes 23 . Endostatin is able to inhibit angiogenesis by restricting endothelial cell proliferation and migration, and by inducing endothelial cell apoptosis 24 . There has been extensive research focused on the potential clinical uses for endostatin, such as an anti-cancer drug or in the treatment of inflammatory diseases, diabetic retinopathy, or obesity. It is also used as a prognostic biomarker or kidney disease marker 23 –26 . On the other hand, the origin of circulating levels of endostatin is still not known and it is unclear what the circulating endostatin levels actually reflect 27 .
The standard technique for non-invasive measurement of limb ischemia is transcutaneous oxygen pressure (TcPO2) as a main parameter for assessing the therapeutic effect in most studies focused on cell therapy of CLI 6,17,28 . In our previous study, we documented the effect of ACT by a significant increase in TcPO2 levels at 12-month follow-up 29 .
Previously, we assessed the serum levels of pro-angiogenic factors as markers of systemic side effects of ACT 19 . We observed no significant increase in the serum levels of pro-angiogenic factors and found no signs of distal systemic vasculogenesis on fundoscopic examination of the eye. Surprisingly, we did document a significant increase in serum endostatin levels after ACT as a finding to suggest lines of further research based on a hypothesis that the anti-angiogenic factor endostatin could potentially reflect vasculogenesis after ACT.
The aim of our present study was to evaluate the serum levels of the anti-angiogenic factor endostatin in diabetic patients with CLI treated by ACT versus PTA, to assess the association of endostatin with the number of injected angiogenic precursor cells (CD34+), and to compare the kinetics of endostatin with changes in TcPO2 potentially reflecting improvement of ischemia after ACT.
Materials and Methods
A total of 25 diabetic patients with no-option CLI treated by cell therapy in our foot clinic during the period 2008–2014 were consecutively included in the study;14 diabetic patients with CLI undergoing PTA in our foot clinic over the same period of time were included in a control group.
The presence of CLI was defined by TcPO2 < 30 mmHg, objectively proven below-the-knee stenosis or obliteration on the angiogram, non-healing ulcer, or ischemic pain consistent with the Trans-Atlantic Inter-Society Consensus II criteria for CLI 30 . Patients meeting the inclusion criteria while ineligible for standard revascularization were treated by ACT; those included in the control group underwent PTA. The severity of angiographic findings was assessed in all patients using the Graziani classification 31 . Patients treated by ACT followed a protocol that included detailed oncological, hematological, and angiographic screening as described in detail in our previous study 29 .
Exclusion criteria for ACT were diagnosed neoplasm of any organ, severe hematological abnormality, deep vein thrombosis in the past 6 months, non-treated proliferative retinopathy, severe limb edema, or severe foot infection with high risk of major amputation. The study was approved by the local ethics committee (Joint Ethics Committee of the Institute for Clinical and Experimental Medicine and Thomayer Hospital), and all patients gave written informed consent.
Using the Jamshidi technique, bone marrow was harvested from both sides of the iliac crest in the operating theater, with BMMNCs subsequently separated using the Smart PReP2 system (Harvest Technologies Corporation, Plymouth, MA, USA) or sedimented using succinated gelatin (Gelofusin; B. Braun, Melsungen, Germany). Final cell suspension of 40–70 ml was injected into the muscles of the affected lower limb in a series of 40–50 punctures of approximately 1–2 ml each, and also to the wound edges.
TcPO2 measurement was performed by standardized method on a TCM400 monitor (Radiometer Medical ApS, Aarhus, Denmark) before revascularization and at 1, 3, and 6 months after the procedure by a foot care specialist.
The serum levels of endostatin (S-endostatin) were measured before and at 1, 3, and 6 months after revascularization either by ACT or PTA, and were compared with changes in TcPO2 at the same intervals.
Endostatin Analysis
Serum samples were stored at –80°C until assayed. These samples were diluted 1:4; endostatin was assessed using the commercial Fluorokine MAP Human Angiogenesis Custom Premix Kit A (R&D Systems, Minnesota, MN, USA) and Luminex device (Luminex Corporation, Austin, TX, USA). The minimum detection level for endostatin was 123.5 pg/ml. Diluted serum samples (50 μl) were mixed with an analyte-specific antibody-coated microparticle and incubated for 3 h at room temperature with gentle, continuous agitation. To this was added a detection antibody for endostatin, then it was incubated again at room temperature for 1 h on a horizontal vortex device. Next, phycoerythrin-conjugated streptavidin (from the Fluorokine Kit) was added and incubated for a further 30 min at room temperature on the vortex. The samples were then immediately measured in the Luminex device. The sample results were calculated by interpolation from particular standard curves.
CD34+ Cell Analysis
Fluorescence-activated cell sorting (FACS) analysis was done with a FC500 system (Beckman Coulter, Brea, CA, USA), and the number of injected CD34+ cells in the BMMNC suspension calculated based on the detected CD34+ concentration and the volume of injected cell suspension.
All data were expressed as mean ± SD. Wilcoxon pair test, Mann–Whitney test, and Spearman correlation coefficient were used for statistical analysis.
Results
Characteristics of diabetic patients in both groups are summarized in Table 1. We did not observe any significant differences between the study groups except for lower baseline TcPO2 levels and more severe angiographic finding according to the Graziani classification for ACT-treated patients. Patients in the two groups did not differ significantly in baseline endostatin levels.
Basic Characteristics of Patients.

ACT, autologous cell therapy; PTA, percutaneous transluminal angioplasty; SD, standard deviation; TcPO2, transcutaneous oxygen pressure.
While the serum levels of the angiogenic inhibitor endostatin were significantly increased at 1 and 3 months after ACT (from 131.6 ± 88.3 to 159.7 ± 113.1 and 173.1 ± 119.3 ng/ml, respectively; both P < 0.001), no significant changes in S-endostatin were seen after PTA (Figure 1).

Comparison of S-endostatin levels between patients after ACT and PTA. S-endostatin increased significantly at 1 and 3 months after ACT (***P < 0.001) in contrast to no significant changes of S-endostatin after PTA.
The effect of ACT and PTA on CLI was confirmed by a significant increase in TcPO2 levels from baseline values at 1, 3, and 6 months both after ACT (from 18 ± 10.4 to 35.1 ± 14.9, 38.4 ± 11.2, and 41.5 ± 10.4 mmHg, respectively; all P < 0.001), and after PTA (from 27.5 ± 15.2 to 47 ± 13.8, 43.1 ± 20.6, and 48.4 ± 18.8 mmHg, respectively; all P < 0.01; Figure 2). Figure 3 shows an increase in TcPO2 after ACT in comparison to changes in S-endostatin; there was a continuing significant increase in TcPO2 levels at 3 and 6 months after ACT, while the levels of S-endostatin showed a significant increase at 1 and 3 months to return to baseline at 6 months post-ACT. While observing a significant correlation between S-endostatin and TcPO2 only at 1 month after ACT (r = 0.557; P < 0.001; Figure 4), no correlation between S-endostatin and TcPO2 after PTA was seen.
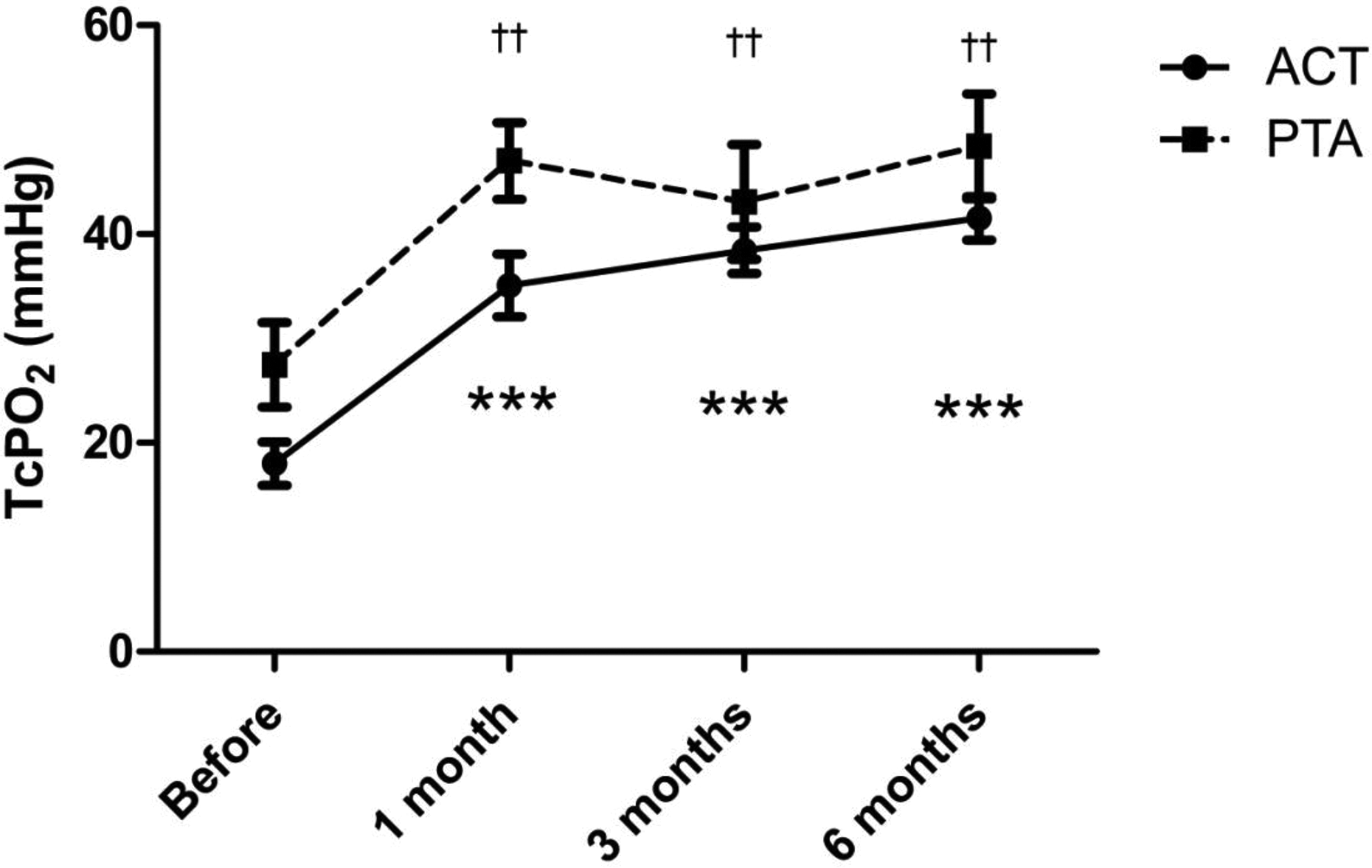
Changes of TcPO2 after revascularization. TcPO2 increases significantly from baseline values both after ACT (***P < 0.001) and PTA (††P < 0.01).

Comparison of kinetics of S-endostatin and TcPO2 after ACT. Levels of TcPO2 significantly increase from baseline values and remain elevated at 6 months after ACT (***P < 0.001), while S-endostatin levels increase at 1 and 3 months after ACT (†††P < 0.001) to decrease to baseline values at 6 months.

Correlation between S-endostatin and TcPO2 at 1 month after ACT (r = 0.557; P < 0.001).
The mean number of CD34+ cells in the suspension injected to the lower limb was 12.9 ± 10.8 × 106; there was no significant association between the number of injected CD34+ cells and S-endostatin levels.
Discussion
Our study showed a significant increase in the serum levels of the endogenous angiogenesis inhibitor endostatin at 1 and 3 months after ACT, in contrast to statistically non-significant changes in this factor after PTA. These results may support our hypothesis that the increase in endostatin could be a compensatory mechanism during transient vasculogenesis after ACT through a negative feedback loop, in contrast to PTA whereby no vasculogenesis occurs. PTA re-establishes the patency of an artery by transluminal or subintimal procedures without a direct impact on new vessel formation.
The mechanism of the anti-angiogenic activity of endostatin was described previously. Endostatin shows potent inhibition of endothelial cell migration and proliferation, and induction of their apoptosis 22 ; it is able to inhibit the VEGF-induced mobilization of EPCs into the circulation, thereby suppressing angiogenesis 32 . Another proposed mechanism of the anti-angiogenic action of endostatin is inhibition of matrix metalloproteinases that facilitate endothelial cell migration and invasion during angiogenesis by proteolytic extracellular matrix degradation 23 . These effects of endostatin are most likely due to its binding to cell surface receptors and influencing a series of intracellular signaling cascades through the interaction with a number of proteins such as integrins, tropomyosin, glypicans, or laminin that mediate the anti-endothelial functions of endostatin 33 . Folkmann et al. 34 suggested that only “pathogenic-derived” angiogenesis usually involves a signal pathway through integrins, which are directly affected by endostatin in contrast to “physiological” angiogenesis that occurs during the wound-healing process.
Similar to our study, Xue et al. 35 investigated early changes in the expression of endostatin and VEGF in patients after acute ischemic stroke. They found that serum endostatin levels tended to increase significantly later than those of VEGF, and remained higher until the end of the observation. This asynchronous expression of VEGF and endostatin indicates that VEGF may possibly play an important role in the early stages of cerebral ischemia through triggering endogenous angiogenesis and increasing vascular permeability, whereas endostatin may represent protective feedback mechanisms. These results are in agreement with possible endostatin feedback regulation of vasculogenesis after ACT of CLI in diabetic patients.
We did not observe any correlation between the number of injected CD34+ cells and serum levels of endostatin. A supposed clinical effect of ACT is probably mediated by the interaction of cells in cell suspension and influenced by the viability of these cells rather than by the total number of injected CD34+ cells. Similar to our finding, no relation between the number of injected CD34+ cells and clinical effect has been confirmed in previous trials: Kawamoto et al., using three different doses of CD34+ cells per kilogram in patients with CLI, observed no significant dose–response effect in terms of the clinical effect as assessed by TcPO2 36 . Likewise, Dong et al., using purified CD34+ cells in CLI patients, did not report a correlation between improvement of clinical outcomes and low, medium, or high doses of injected CD34+ cells 37 . On the other hand, Madaric et al. investigated factors associated with clinical benefits of ACT in patients with no-option CLI and discovered that the number of injected CD34+ cells was an independent predictor of limb salvage and wound healing 38 .
The clinical effect both of ACT and PTA was confirmed by a significant increase in TcPO2 levels from baseline at all intervals during follow up 29 . A significant correlation between endostatin and levels of TcPO2 was observed only at 1 month after ACT because of the different kinetics of endostatin and changes of TcPO2. A temporary significant increase in endostatin at 1 and 3 months post-ACT may reflect vasculogenesis with a subsequent decrease in endostatin after completion of the new-vessel-formation process. Kinnaird et al. published that the process of remodeling of a completely patent three-layered collateral artery restores blood flow within 4–6 weeks 21 . Unlike the temporary increase in endostatin, increased TcPO2 levels persist because the ACT-mediated improvement of microcirculation increases tissue oxygenation for a longer time as documented in our previous study 19 .
The increase in endostatin as a potential marker of new vasculogenesis in our present study seems to be in contrast to the results of several earlier studies in which elevated endostatin levels were associated with ischemia, not with revascularization. For example, Qin et al. 39 reported that S-endostatin was significantly higher in patients with severe coronary artery disease and poor collateral circulation compared with patients with normal collaterals. The discrepancy of these results with our findings of a transient endostatin increase as a marker of vasculogenesis could be explained by different physiological pathways: In our study, the increase in endostatin was likely stimulated as a feedback reaction to ACT-induced new vasculogenesis in contrast to the increase in the levels of endostatin as a marker of poor collateral formation in Qin’s study. In another study, Golledge et al. reported that S-endostatin was elevated in men with symptoms of intermittent claudication compared to those without ischemic leg pain 40 . The discrepancy between the above results and ours could again be explained by the different mechanisms causing the changes in endostatin levels.
The advantages of our study include its prospective follow up, well-defined inclusion criteria and methods used for assessment, and comparability of both groups of patients in terms of their demographic characteristics. The main limitation of our study is our inability to verify increased vasculogenesis after ACT by appropriate immunohistochemical techniques at tissue level due to the absence of such a method or unavailability of the necessary technologies 41 . Another limitation of this study is its pilot design, with a limited number of patients in both groups.
Conclusion
In conclusion, our study showed that the anti-angiogenic factor endostatin, temporarily elevated after ACT, could reflect new vasculogenesis induced by this therapy in diabetic patients with CLI. A potentially protective feedback role of endostatin in ACT-mediated vasculogenesis poses a future research challenge.
Footnotes
Ethical Approval
This study was approved by the Joint Ethics Committee of the Institute for Clinical and Experimental Medicine and Thomayer Hospital, Prague, Czech Republic.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Ethics Committee of the Institute for Clinical and Experimental Medicine and institutional review board’s approved protocols.
Statement of Informed Consent
All patients gave informed consent.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Charles University in Prague, project GAUK 16415 and by the Ministry of Health of the Czech Republic, grant no. 16-27262A and by the project for Development of Research Organization 00023001 (IKEM, Prague, Czech Republic) – Institutional support.
