Abstract
Autoimmune disease–related critical limb ischemia (AD-CLI) represents a significant portion of no-option critical limb ischemia (NO-CLI). Despite the demonstrated safety and efficacy of cell transplantation for NO-CLI, most studies focused on thromboangiitis obliterans (TAO). There remains a scarcity of reports on cell transplantation for AD-CLI induced by systemic lupus erythematosus (SLE), hypereosinophilic syndrome (HES), and so on. From May 2011 to May 2024, 22 patients with 36 ischemic limbs were enrolled. The primary outcome was amputation, whereas secondary outcomes included toe-brachial index (TBI), Wong-Baker Faces Pain Rating Scale (WBFPS), transcutaneous oxygen pressure (TcPO2), and so on. Among them, 8 had SLE, 11 had HES, and 3 had eosinophilic granulomatous polyangiitis (EGPA). During the follow-up, one EGPA patient died, two patients underwent major amputation, and five underwent minor amputation. The 2-year major and total amputation-free survival rates were 86.4% and 72.7%, respectively. Critical limb ischemia (CLI) relief was observed in 14 patients, with a 1-year cumulative CLI-free survival rate of 61.0%. Significant improvements were noted in postoperative TBI and WBFPS. Autologous cell transplantation showed satisfactory safety and efficacy outcomes for non-TAO AD-CLI patients. Validation of the conclusions awaits more evidence based on the long-term outcomes of a larger number of patients.
Introduction
As the end stage of peripheral arterial disease (PAD), critical limb ischemia (CLI) is often associated with high rates of amputation and mortality 1 . No-option critical limb ischemia (NO-CLI), accounting for approximately 15% to 20% of all CLI cases, refers to CLI that is unsuitable for either surgical or endovascular treatment due to poor anatomical conditions and a high postoperative reocclusion rate 2 . In NO-CLI patients, both the 6-month major limb amputation rate and 1-year mortality rate range from 10% to 40%1,3,4. Autoimmune disease–related chronic limb-threatening ischemia (AD-CLI), defined as CLI caused by autoimmune diseases such as thromboangiitis obliterans (TAO), systemic lupus erythematosus (SLE), hypereosinophilic syndrome (HES), psoriasis, Crohn’s disease, and so on, commonly involves distal or even microvessels, resulting in the ineffectiveness of conventional revascularization5–7. Therefore, AD-CLI constitutes a significant proportion of NO-CLI patients.
Cell transplantation for treating limb ischemia was first reported in 1997 by Asahara 8 . Nowadays, cell transplantation has demonstrated satisfactory safety and efficacy outcomes for AD-CLI patients9–11. However, most studies have primarily focused on patients with TAO. Systemic reports on cell transplantation for treating AD-CLI induced by SLE, HES, and so on are still scarce, given that TAO patients comprise the majority of AD-CLI cases5–7.
In the current study, we retrospectively enrolled all non-TAO AD-CLI patients who underwent cell transplantation at our center over a 14-year period to report their long-term follow-up outcomes.
Materials and methods
This study received approval from the Institutional Review Board of authors’ Hospital and adhered to the ethical principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all enrolled participants.
Patients
Between May 2011 and May 2024, 275 individuals with AD-CLI (Rutherford categories 4 and 5) underwent autologous cell transplantation. The therapeutic regimen comprised purified CD34+ cells (PCC) and peripheral blood mononuclear cells (PBMNC). Detailed selection criteria have been previously published 12 . Key inclusion requirements were age between 18 and 80 years, objectively confirmed CLI via angiography (computed tomography, digital subtraction, or magnetic resonance angiography). Exclusion criteria encompassed: acute major comorbidities (e.g. myocardial infarction, pulmonary embolism, renal/hepatic insufficiency, or cerebrovascular events within 3 months); active or suspected malignancy; anticipated survival ≤6 months. Generally, after the diagnosis of autoimmunological disease was made and other causes of CLI was excluded (atherosclerosis, embolism, thrombosis, etc.) by at least 1 senior vascular surgeon and 1 senior rheumatologist, the diagnosis of AD-CLI was made, and all patients went to the rheumatology and immunology clinic to control the activity of primary disease. However, for patients with rapidly progressing ischemia, diagnosis and activity control of primary disease could only be made by seeking rheumatology consultation during hospitalization. In the current study, we also excluded patients who were diagnosed as TAO.
Cell transplantation
The methodology was previously described 12 . Briefly, recombinant human granulocyte colony-stimulating factor (rhG-CSF; Neupogen®; Amgen, USA) was administered subcutaneously at 5–10 μg/kg/day for 4 consecutive days to mobilize bone marrow-derived cells. Concurrent prophylactic enoxaparin (4000 IU daily) was used to prevent hypercoagulability. On day 5, PBMNC were collected through leukapheresis (COM.TEC system; Fresenius Hemocare, Germany). The apheresis product underwent triple-wash centrifugation and was resuspended in 200 ml of ethylenediaminetetraacetic acid-phosphate-buffered saline supplemented with 0.5% human albumin, yielding around 100 ml of PBMNC suspension. PCC (around 40 ml) were isolated from PBMNC using immunomagnetic separation (Miltenyi-Biotec, Bergisch Gladbach, Germany). CD34+ cell quantification combined automated leukocyte counting with flow cytometric analysis. Under general anesthesia, the final cell products were injected intramuscularly at equidistant sites (0.5 ml/injection) throughout the ischemic lower extremities.
Data collection
During hospitalization, demographic characteristics of patients, characteristics of the autoimplants, and so on, were recorded and analyzed. The baseline features of the ischemic limbs, such as numbers, the Rutherford class, and the occlusion level of the arteries, were also recorded.
Outcomes and follow-up
Postoperative surveillance involved regular clinical evaluations at 1, 3, 6, and 12 months post-transplantation with subsequent annual assessments. Primary efficacy endpoints encompassed major amputation (above the elbow/ankle) and minor amputation (below the elbow/ankle), from which major amputation-free survival (MAFS) and total amputation-free survival (TAFS) rates were calculated. Secondary outcomes included improvements in Rutherford classification, toe-brachial index (TBI), transcutaneous oxygen tension (TcPO2), pain severity quantified via Wong-Baker FACES Pain Rating Scale (WBFPS) (0 = no pain, 10 = greatest pain), and disease recurrence. Rest pain alleviation was operationally defined as WBFPS scores became ≤4. During the follow-up, patients were prescribed with antiplatelet drugs and vasodilators. In addition, patients were advised to regularly pay visit to rheumatology clinic to make sure the activity of their primary diseases was well controlled.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation (SD) for normally distributed data or median with interquartile range (IQR) for non-parametric data, with between-group comparisons performed using independent samples t-tests. Categorical data are reported as counts and percentages, analyzed by χ2 or Fisher’s exact tests as appropriate. Time-to-event endpoints including MAFS, TAFS, and cumulative CLI-free rates were evaluated via Kaplan–Meier analysis. All analysis employed a two-sided significance threshold of α = 0.05 and were executed in PASW Statistics version 19 (IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics
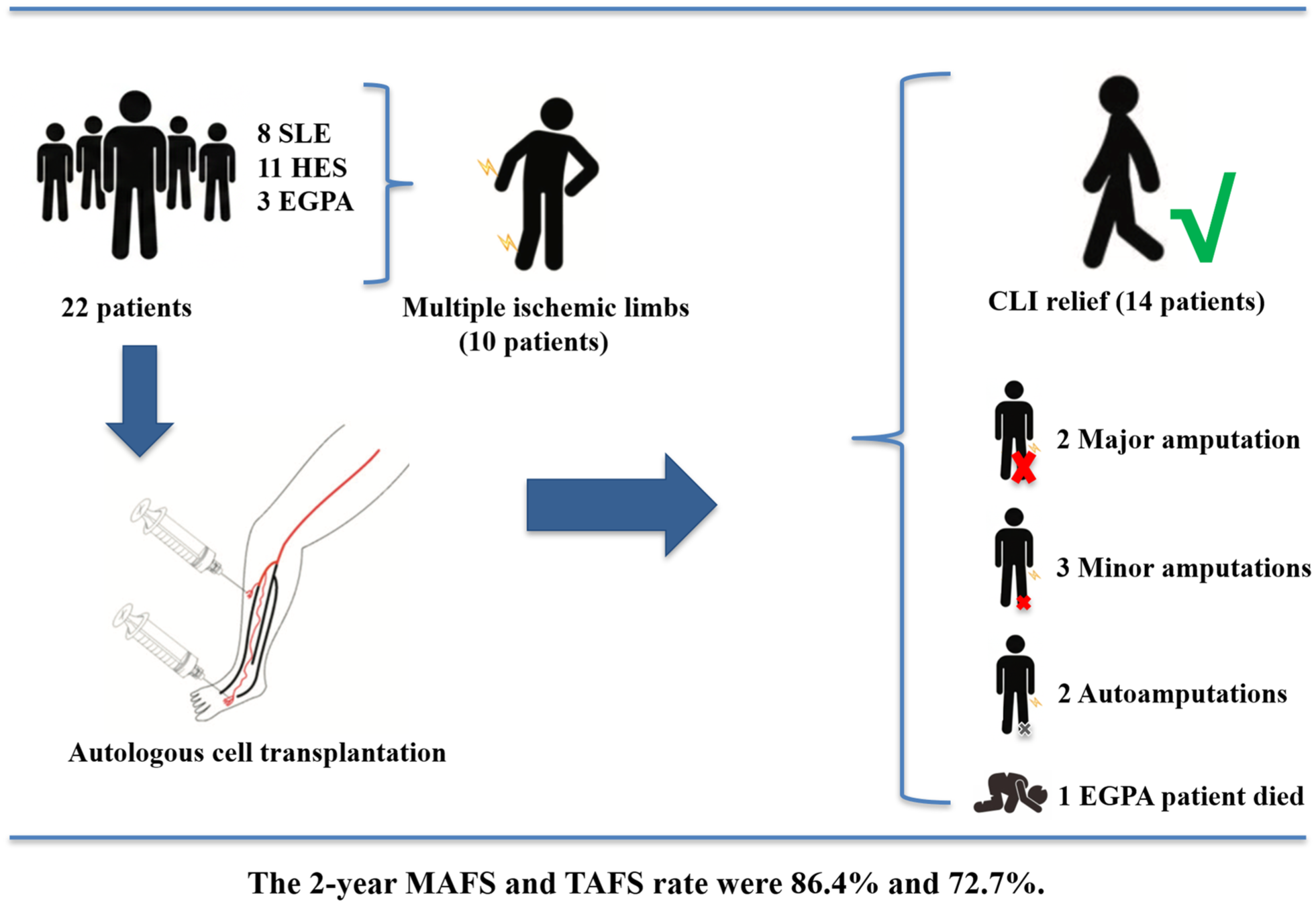
Between May 2011 and May 2024, a total of 281 patients diagnosed with NO-CLI underwent autologous cell transplantation, which included PBMNC and PCC. After excluding 249 patients with TAO, 6 patients with atherosclerotic obliterans, and 4 patients lost to follow-up, a final cohort of 22 non-TAO AD-CLI patients were enrolled (Fig. 1). Among these patients, 81.8% (18/22) were male, with a mean age of 38.9 ± 12.8 years (range, 20–61 years). Five patients (22.7%) had a history of smoking, 2 patients (9.1%) had hypertension, 1 patient (4.5%) had diabetes mellitus, and 1 patient (4.5%) had cardio-cerebrovascular diseases. Among the 22 AD-CLI patients, 8 had SLE, 11 had HES, and 3 had eosinophilic granulomatous polyangiitis (EGPA). Around half of the patients (10/22, 45.4%) had a history of prior open surgery and/or endovascular treatment. Further details of the baseline characteristics are presented in Tables 1 and 2.

Protocol of the current study.
Baseline characteristics.

The data presented are the numbers (%) and the means ± standard deviations or medians and the interquartile ranges.
SD: standard deviation; HES: hypereosinophilic syndrome; SLE: systemic lupus erythematosus; EGPA: eosinophilic granulomatous polyangiitis; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; IQR: interquartile range.
Detailed data of all patients.

M: male; F: female; RC: Rutherford class; HES: hypereosinophilic syndrome; SLE: systemic lupus erythematosus; EGPA: eosinophilic granulomatous polyangiitis; PBMNC: peripheral blood mononuclear cells; PCC: purified CD34+ cells; U: upper limb; L: lower limb; CLI: critical limb ischemia; I: iliac artery; F: femoral artery; B: brachial artery; P: popliteal artery; Be: below the knee/elbow; MJ: major amputation; MN: minor amputation; AM: autoamputation.
Ischemic limb
Five patients (22.7%) presented with Rutherford class 4, whereas the remaining (17/22, 77.3%) were categorized as Rutherford class 5. Among the enrolled patients, 6 had 2 ischemic limbs, and 4 had 3 ischemic limbs, totaling 36 ischemic limbs. In addition, 10 ischemic upper limbs were observed in 7 patients (4 with HES, 2 with SLE, and 1 with EGPA). Regarding the highest level of occluded artery, the majority of ischemic limbs were below the knee/elbow (24/36, 66.7%), followed by five (13.9%) in the popliteal artery, six (16.7%) in the femoral/brachial artery, and one (2.8%) in the iliac artery. Further details of the ischemic limbs are provided in Table 2.
Cell product
All 36 ischemic limbs received cell transplantation, 7 (7 patients) with PCC and 29 (16 patients) with PBMNC. The mean volume of cell product was 66.8 ± 31.7 ml (range: 36.6–124 ml) per limb. All patients achieved transplanted CD34+ cells dose of >105 per kg per limb (5.05 × 105 [3.3 × 105–8.2 × 105]) (Table 3).
Characteristics of the cell product.

The data presented are the numbers (%) and the means ± standard deviations or medians and the interquartile ranges.
PBMNC: peripheral blood mononuclear cells; PCC: purified CD34+ cells; IQR: interquartile range.
Outcomes
No perioperative adverse events, such as death, hepatic/renal dysfunction, pathological retinal angiogenesis, cardio-cerebrovascular events, or tumorigenesis, occurred during hospitalization. During the follow-up period, one patient with EGPA died of cardiac disease 3 months after transplantation. Upon admission, five patients presented with rest pain (Rutherford class 4), whereas the remaining patients exhibited ulcers or gangrene (Rutherford class 5). Throughout the follow-up, CLI relief (Rutherford class < 4) was observed in 14 patients, with a mean relief period of 5.0 ± 3.8 months (range: 1–13 months). Most patients (11/14) achieved CLI relief within 6 months post-transplantation. The cumulative CLI-free survival rate at 1 year was 61.0% (95% confidence interval (CI): 41.4%–81.1%) (Fig. 2a). The mean duration of rest pain relief for these 14 patients was 1.8 ± 1.5 months (range: 0.5–6 months). Unrelieved ischemia was observed in seven patients (two SLE, three HES, and two EGPA). Among them, one SLE and one HES patient underwent major amputation in 7 months and 1 month after transplantation, respectively; one HES patient received a second cell transplantation at 18 months; the remaining patients received conservative treatment. Six patients underwent amputation, including two major amputations (one SLE and one HES), three planned minor amputations (one SLE and two HES), and two autoamputations (both HES). The 2-year MAFS and TAFS rates were 86.4% (95% CI: 63.5%–95.4%) (Fig. 2b) and 72.7% (95% CI: 49.1%–86.7%) (Fig. 2c), respectively. Recurrent rest pain was observed in 2 SLE patients (at 72 and 108 months), which was subsequently relieved within 3 months with conservative treatment, including medication and exercise therapy.
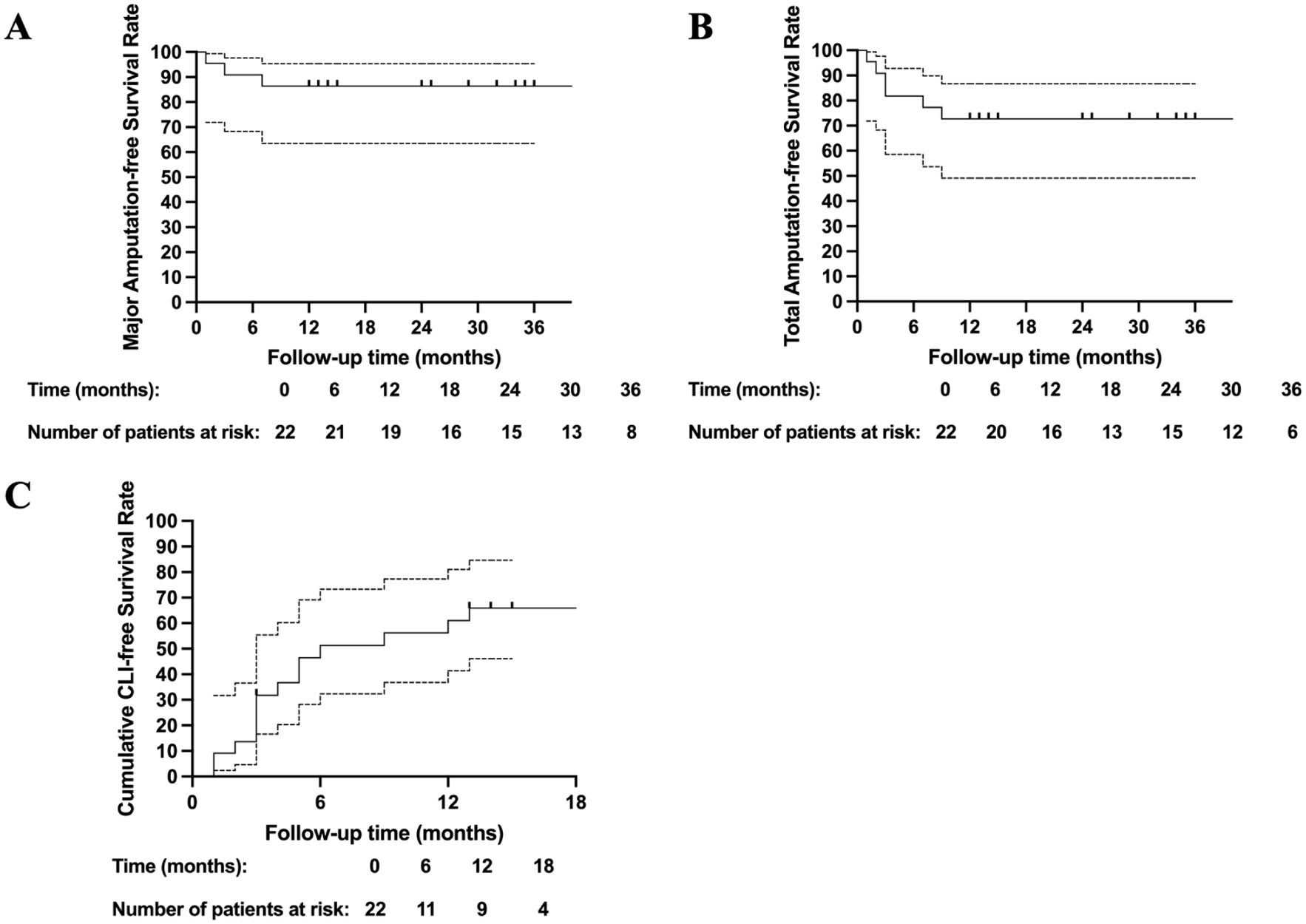
Kaplan–Meier curves showing the probabilities of MAFS (a), TAFS (b), and cumulative CLI-free survival rates (c) of all patients.
Upon admission, the TBI could not be measured for the digit ulcer or gangrene in eight ischemic limbs of four patients. Among the remaining 28 limbs, the baseline TBI was 0.41 ± 0.17 (range: 0.1–0.72). All patients presented with unbearable rest pain, with a baseline WBFPS score of 9.0 ± 1.8 (range: 8–10). TcPO2 measurements were performed for all ischemic limbs, yielding a baseline value of 25.1 ± 8.0 mm Hg (range: 10–42 mm Hg). During the follow-up period, significant and persistent improvements in TBI (Fig. 3a) and WBFPS (Fig. 3b) were observed from the sixth month post-transplantation onward. However, there were no significant changes in TcPO2 levels observed during the follow-up (Fig. 3c).

Longitudinal changes in TBI (a), WBFPS (b), and TcPO2 (c). The values are presented in linear graphs that show the means and SDs.
Discussion
Cell therapy has emerged as a promising approach for the treatment of NO-CLI. Autoimmune diseases are commonly associated with coagulation abnormalities, platelet activation, inflammation, and deposition of autoantibodies, contributing to pathological thrombosis and tissue ischemia. AD-CLI typically involves distal or even microvessels, rendering conventional revascularization ineffective and thus constituting a significant proportion of NO-CLI cases. However, due to the rarity of many AD-CLI, such as SLE and HES, cell transplantation has predominantly been reported in the treatment of TAO5–7. As one of the largest centers for cell transplantation in China, we have treated over 290 patients with AD-CLI to date. Therefore, the current study aims to report the long-term outcomes of all non-TAO AD-CLI patients who underwent cell transplantation at our center over a 14-year period.
In the current study, 22 non-TAO AD-CLI patients were enrolled. Most patients were with a surgical history, including thrombectomy, thrombolysis, percutaneous transluminal angioplasty, and so on, but derived little benefit from these interventions, further confirming their classification as NO-CLI cases. SLE and HES constituted the majority of the cohort. It has been reported that the prevalence of vasculitis in SLE patients ranges from 10% to 56%, characterized by excessive fibrosis and microvascular abnormalities 8 . Consequently, many SLE patients presented with finger/toe ischemia. In a cohort of 485 SLE patients, among the 7 presenting with CLI (2 had rest pain, 3 had toe/finger gangrene, and 2 had hand/foot gangrene), most experienced only digit loss 9 . Similarly, three out of four SLE patients in Rutherford class 5 in our cohort exhibited digit ulceration/gangrene, suggesting that the ischemic progression in most SLE-related CLI patients might be limited, making major amputation an unsuitable efficacy indicator. Hence, we also employed rest pain relief as an efficacy indicator for cell therapy. In our study, the majority of SLE patients (6/8) achieved rest pain relief within 3 months, indicating the satisfactory efficacy of cell therapy (Fig. 4). Likewise, reports of HES-related limb ischemia are rare. A review encompassing four HES-related CLI cases noted that all cases were male, with both small and medium-sized vessels of the limbs affected 10 . The HES subgroup in our study was also predominantly male (81.82%, 9/11) and exhibited a high proportion of patients with multiple ischemic limbs (54.55%, 6/11). More than a half of HES patients (7/11, 63.64%) achieved CLI relief within 1 year after transplantation. Eosinophils play a crucial role in the pathogenesis of EGPA, a type of antineutrophil cytoplasmic antibody-associated small vessel vasculitis characterized by necrotizing granulomatous vasculitis11–13. While EGPA can affect various organ systems, such as the lungs, skin, and nervous system, limb ischemia is a rare complication, described only in a few case reports14,15. Our study enrolled only three EGPA patients, one of whom died of cardiac disease and others experienced unrelieved ischemia, suggesting the uncertainty surrounding cell therapy in such cases.

Ischemia relief progress in a 49-year-old male (patient 7) with SLE. The patient was enrolled with gangrene in the left index finger (a). After PBMNC transplantation, his gangrene generally healed in 1 month (b) and 2 months (c). At 3 months after transplantation (d), his gangrene basically healed while cyanosis still can be observed. In 6 months, 1 year, and 2 years, his finger was in good condition and cyanosis disappeared.
The 1-year cumulative CLI-free survival rate and 2-year MAFS were 61.0% and 86.4%, respectively, mirroring results reported in studies focusing on TAO5,16–18. These findings underscore the satisfactory efficacy of cell therapy in preserving limbs and alleviating ischemia for non-TAO AD-CLI patients. In our previous study, the total transplanted CD34+ cell count was identified as an independent risk factor for persistent ischemia within 6 months after therapy 19 . Similarly, in the current study, the majority of patients with persistent ischemia presented with multiple ischemic limbs (5/6), a situation comparable to those who did not achieve ischemia relief within 6 months (9/11). The presence of multiple ischemic limbs necessitates a higher number of cells, potentially leading to an insufficient relative cell count. Moreover, controlling the activity of the primary disease in AD-CLI patients before cell therapy is crucial for optimal outcomes. However, some patients present to our clinic with rapidly progressing ischemia before an accurate diagnosis of the primary disease is made, necessitating urgent intervention to alleviate ischemia. These patients received concurrent treatment for their primary disease. During the follow-up, increased disease activity, as evidenced by elevated erythrocyte sedimentation rate and serum complement levels, was observed in two SLE patients with recurrent rest pain, underscoring the importance of controlling the primary disease to achieve favorable outcomes.
The present study has several limitations. First, being a single-center retrospective study, there is a risk of selection bias inherent in the study design. Second, the relatively small number of enrolled patients is attributable to the low incidence of AD-CLI. Third, there is a paucity of long-term clinical outcome data regarding limb salvage and ischemia relief following conventional treatments in AD-CLI patients, making it challenging to compare clinical outcomes. Finally, while PCC and PBMNC demonstrated comparable therapeutic efficacy in TAO patients, their differential effects remain uncharacterized in non-TAO ones. In the present cohort, robust subgroup analysis was precluded by two methodological constraints: (1) insufficient sample size across treatment subgroups and (2) significant baseline heterogeneity (SLE comprising >80% of PCC patients vs <15% in the PBMNC group). These limitations should be considered when interpreting the findings of the study.
Conclusion
In conclusion, results of the current study indicated that autologous cell transplantation showed satisfactory safety outcomes and efficacy outcomes in terms of ischemia relief for non-TAO AD-CLI patients with a 1-year cumulative CLI-free rate of 61.0% and 2-year MAFS of 86.4%. Strict and regular follow-up is needed to monitor not only the condition of limb but also the activity of primary disease. Validation of the conclusions is pending more evidence on the basis of the long-term outcomes of a larger number of patients.
Footnotes
Acknowledgements
None.
Ethical Considerations
The study design was approved by the Ethics Committee of Zhongshan Hospital, Fudan University, Shanghai, China (Approval No. B2022-498, Date 9/1/2024).
Consent for Participation
All included patients were informed about the nature of the study and gave their written informed consent.
Consent for Publication
All included patients were informed about the nature of the study and gave their written informed consent.
Author Contributions
Hao Liu drafted the manuscript. Hao Liu, Lingwei Zou, Tianyue Pan, and Zhihui Dong contributed to the conception and design of the study. Hao Liu, Yuan Fang, Gang Fang, Xiaolang Jiang, and Yifan Liu contributed to data acquisition, analysis, and interpretation. Bin Chen, Zheng Wei, Shiyang Gu, Peng Liu, and Weiguo Fu provided research supervision. Tianyue Pan and Zhihui Dong critically revised the manuscript. All authors have full access to all study data and are responsible for ensuring data integrity and accuracy of the analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by China National Natural Science Funds (grant no. 82400568; 82270507; 82370499), Noncommunicable Chronic Diseases-National Science and Technology Major Project (2023ZD0504300), Outstanding Resident Clinical Postdoctoral Program of Zhongshan Hospital Affiliated to Fudan University (2024), and Clinical Research Special Fund of Zhongshan Hospital Affiliated to Fudan University (2023ZSLC59).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The deidentified participant data will be shared on a request basis. Please directly contact the corresponding author to request data sharing.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
