Abstract
Researchers have accumulated a decade of experience with autologous cell therapy in the treatment of critical limb ischemia (CLI). We conducted a systematic review of clinical trials in the literature to determine the safety and efficacy of cell therapy in CLI. We searched the literature for clinical trials of autologous cell therapy in CLI, including observational series of five or more patients to accrue a large pool of patients for safety analysis. Safety analysis included evaluation of death, cancer, unregulated angiogenesis, and procedural adverse events such as bleeding. Efficacy analysis included the clinical endpoints amputation and death as well as functional and surrogate endpoints. We identified 45 clinical trials, including seven RCTs, and 1,272 patients who received cell therapy. The overall adverse event rate was low (4.2%). Cell therapy patients did not have a higher mortality rate than control patients and demonstrated no increase in cancer incidence when analyzed against population rates. With regard to efficacy, cell therapy patients had a significantly lower amputation rate than control patients (OR 0.36, p = 0.0004). Cell therapy also demonstrated efficacy in a variety of functional and surrogate outcomes. Clinical trials differed in the proportion of patients with risk factors for clinical outcomes, and these influenced rates of amputation and death. Cell therapy presents a favorable safety profile with a low adverse event rate and no increase in severe events such as mortality and cancer and treatment with cell therapy decreases the risk of amputation. Cell therapy has a positive benefit-to-risk ratio in CLI and may be a valuable treatment option, particularly for those challenging patients who cannot undergo arterial reconstruction.
Keywords
Introduction
Critical limb ischemia (CLI) is a complicated health care problem marked by high rates of morbidity, mortality, and resource utilization. The annual incidence is roughly 500–1,000 new cases per million in industrialized countries (53), and disease prevalence is expected to rise with increasing rates of diabetes and the aging of the population. While treatment has been directed at restoring arterial blood flow by either surgical bypass or endovascular procedures, an estimated 25–40% of patients may not be suitable for arterial reconstruction (15,53,64) and still others may fail reconstructive therapy. This subgroup of patients without revascularization options (no option CLI or NO-CLI) is a particularly challenging population with high rates of limb loss and death, and novel therapies are an active area of investigation.
In 1997, Asahara identified a class of bone marrow-derived progenitor cells that contribute to angiogenesis in ischemic tissue (4). The first clinical trial of autologous cell therapy in patients with CLI was published in 2002 by Tateishi-Yuyama et al., who reported that patients treated with mononuclear cells isolated from bone marrow demonstrated improved hemodynamic measurements (66). Since this promising start, researchers have accumulated a decade of experience in the use of autologous cell therapy for CLI with clinical trials progressing from retrospective case series to prospective studies and finally to several recent randomized controlled trials (RCTs).
Data collected from these trials provide insight into the safety, feasibility, and efficacy of cell therapy in the treatment of CLI. We therefore conducted a systematic review of clinical trials in the literature to establish a safety profile of autologous cell therapy as well as to determine the efficacy of cell therapy as a treatment for CLI.
Safety
While most studies of cell therapy have focused on the potential benefits of treatment, the safety profile of cell therapy is rarely addressed in detail because thus far therapy has not presented significant adverse events and the number of patients in individual trials tends to be relatively small (35,45). We sought to use a pooled, large group of patients from the systematic review to identify patterns of adverse events and more precisely establish the safety profile of autologous cell therapy.
In addition to procedure-related events such as bleeding and pain, there are several specific adverse events identified by regulatory bodies such as the FDA as being of potential concern in patients treated with cell therapy. These include death, major adverse cardiovascular events (MACEs), anemia, and unregulated angiogenesis such as retinopathy or arterio-venous malformations, and cancer.
Critical limb ischemia (CLI) patients have significant comorbidities such as coronary artery disease, diabetes, and renal insufficiency, which contribute to a high mortality rate in this population. Given the relatively fragile nature of this patient population, the occurrence of death during a study is not surprising; however, an elevated mortality rate in cell therapy patients compared to control patients would present a clear safety concern.
MACEs include myocardial infarction, heart failure, stroke, and sudden death. Patients with CLI, particularly those with atherosclerosis and diabetes, are at high risk for MACEs due to their disease.
Harvesting bone marrow or peripheral blood for stem cells presents the patient with a hematologic challenge by removing hematopoietic precursors. While this may result in a decreased circulating red cell mass, anemia occurs in only 10% of donors and is usually self-limited, not requiring transfusion (17). Procedure-related anemia usually manifests itself within the first week after harvesting. Stem cell harvesting is remarkably well-tolerated with severe hematological adverse events occurring rarely (63), and many of those in patients receiving granulocyte-colony stimulating factor (G-CSF) administration (related to increased blood viscosity) rather than those undergoing marrow aspiration (23). Nonetheless, anemia remains a potential concern in the CLI population with peripheral and coronary ischemia.
Patients with CLI are at high risk for soft tissue infection, osteomyelitis, and gangrene. This is particularly true for those with tissue lesions (Rutherford 5–6 or Fontaine IV). Progressive infection and gangrene are often indications for amputation, designed to prevent development of life-threatening systemic infection. Insufficient blood flow to a limb decreases the ability to heal tissue lesions (thereby reducing the barrier defenses) and compromises the ability of that patient to mount an adequate response to infectious agents. Injection or infusion of cell therapy is invasive and therefore raises the possibility of infection in a population already at high risk.
Many cell therapy protocols for treating CLI require intramuscular injections of stem cells into the threatened limb. Delivering a volume of cell product to a muscle bed that is compromised by insufficient blood flow raises a concern for muscle damage, specifically rhabdomyolysis and subsequent acute kidney injury. The CLI patient population has a high prevalence of chronic renal insufficiency due to vascular disease and diabetes, which correlates with poor surgical outcomes (54), in addition some patients may have subclinical renal insufficiency following some form of insult to the kidney. Therefore, CLI patients are at high risk of developing acute kidney injury.
A potential concern of using cell therapy to promote angiogenesis is the stimulation of unwanted vessel growth, which manifests either as an arterio-venous malformation or proliferative retinopathy. Diabetic retinopathy develops as functional vessels in the eye are lost, leading to ischemia. This is followed by reactive, aberrant vascular proliferation as a result of hypoxia-mediated cytokines such as vascular endothelial growth factor (VEGF). Stem cells, particularly endothelial progenitor cells (EPCs), play a role in vascular regeneration in the eye, but their differential involvement in normal healing and pathological angiogenesis is complicated (73). While there is a correlation between elevated circulating EPCs and proliferative diabetic retinopathy, it is unclear if these levels rise as a cause or an effect of the disease (7), particularly as most diabetics instead demonstrate decreased levels of circulating EPCs (16,41). On the other hand, researchers are pursuing the use of stem cells to repair retinal degeneration (13), and others see the use of stem cells to stabilize and “mature” aberrant vasculature in the eye (20,34). Nonetheless, administering stem cells to promote revascularization of one ischemic area, such as a threatened limb, and thereby provoking neovascularization in another ischemic area, such as the diabetic eye, remains a conceptual concern.
Finally, with regard to malignancy, there is speculation that bone marrow-derived cells may contribute to cancer development by supporting tumor angiogenesis. Some tumors secrete chemotactic signals to mobilize stem cells from the marrow (40), and these cells have been shown to be incorporated into the vasculature of tumors in mice (43). However, the mere presence of stem cells and tumor together is not sufficient to drive vessel growth; there seems to be an “angiogenic switch” that drives these cells to support neovascularization (21). Still other authors have shown that bone marrow-derived endothelial progenitor cells do not contribute to tumor endothelium in either primary or metastatic tumors (71). Whether or not cell therapy results in new tumor development or contributes to tumor growth in humans is unknown and remains strictly speculative.
Efficacy
The goal of CLI therapy is to promote limb salvage, slow disease progression, and improve symptoms, quality of life, and survival. Clinical trials employ a variety of endpoints to demonstrate the effectiveness of therapy in achieving one or more of these treatment goals. In CLI, the clinical or patient-relevant endpoints are amputation and death, analyzed as amputation rate, mortality rate, time to amputation or death, and amputation free survival. Functional endpoints include clinical classification of disease severity (such as Rutherford or Fontaine class), wound healing, change in pain, and quality of life (QoL). Surrogate endpoints include hemodynamic measures such as ankle-brachial index (ABI) or transcutaneous oxygen pressure (TcPO2), which are markers of improved perfusion and angiographic demonstration of new blood vessel growth (Table 1).
Endpoints in Critical Limb Ischemia (CLI) Trials

Functional and surrogate endpoints—laboratory or other markers of improvement— are often easier to achieve over a short study period and require smaller sample sizes, which make them attractive for early phase trials. While they may contribute to an understanding of a therapy's mechanism of action, these endpoints do not necessarily correlate with patient outcomes (18). This discordance between surrogate and function outcomes and clinical outcomes has been demonstrated in numerous CLI trials (22) and suggests that, while demonstrating improvement in these measures is supportive of the efficacy of cell therapy, they are not sufficient to base treatment decisions.
Functional and surrogate outcome measures are more commonly relied upon in observational trials because they do not require a control group but are instead measured as changes against baseline. Some of these endpoints, like pain or quality of life, have a clear relationship to a patient's clinical status. Others, like angiographic demonstration of vessel growth or increase in the ankle-brachial index, indicate improvements in blood flow but may have no relationship to a patient's clinical status. Each of these endpoints in isolation is of limited value, but taken together they provide evidence of the efficacy of cell therapy
Hard clinical endpoints, on the other hand, are directly related to prognosis and survival—outcomes of clear interest to patients and clinicians. Therapies that demonstrate efficacy in these patient-relevant endpoints are well positioned to change medical practice (61). Furthermore, regulatory bodies such as the US Food and Drug Administration (FDA) will only accept primary clinical endpoints for pivotal trials, and the Centers for Medicare and Medicaid (CMS) place greater emphasis on hard endpoints in the benefit-to-risk analysis prior to approving reimbursement for novel therapies. Therefore, to become a widely accepted treatment, cell therapy needs to demonstrate efficacy in terms of amputation and mortality. We therefore focused our efficacy analysis on these clinical outcome measures.
Analysis of clinical endpoints in CLI patients must take into account the major risk factors for amputation and death which include dialysis dependence, the presence of tissue loss, age > 75 years, and diabetes (3,62). Not only may these affect clinical outcomes, but differences in these variables suggest heterogeneity in study populations, which in turn may influence the validity of results or their applicability to other patient populations. We therefore also examined trials for the presence of these risk factors.
Cell Source
Multipotent mononuclear cells originate in the bone marrow, but trial protocols differ in their method of isolation. Bone marrow mononuclear cells (BMMNCs) are aspirated directly from the bone marrow, whereas peripheral blood mononuclear cells (PBMNCs) are obtained by venipuncture after mobilization of cells from the marrow to the peripheral circulation following administration of G-CSF. We therefore also compared results between the two major sources of autologous cells.
Cell therapy trials also differ with regard to the etiology of CLI with the two major causes being atherosclerosis obliterans (ASO) and Buerger's disease or thromboangiitis obliterans (TAO). We also evaluated procedure-related variables such as cell concentration method and delivery route.
Materials and Methods
Literature Search
We reviewed the literature for clinical trials of autologous cell therapy in CLI by querying PubMed and the Cochrane database of systematic reviews. We used search terms such as “critical limb ischemia” and “cell therapy” or “stem cell” or “bone marrow” or “mononuclear cells” or “granulocyte colony stimulating factor” or “therapeutic angiogenesis.” We included only clinical trials published in English within the last 15 years and excluded case series of fewer than five patients. We examined the references from eligible studies and review articles for additional clinical trials. We only included trials where mononuclear cells were injected or infused into the index limb of patients with CLI. We excluded articles that addressed acute limb ischemia, intermittent claudication, and trials where the CLI portion of the patient population could not be separately evaluated. We excluded trials of G-CSF monotherapy, that is, those trials that only used growth factor to mobilize bone marrow cells to the peripheral circulation without direct delivery of cells to the ischemic limb. We excluded trials that used cell therapy as an adjunct to revascularization procedures. RCTs present the highest quality of data with the least amount of bias and error as well as the greatest opportunity for comparison of safety and efficacy. Therefore, we conducted detailed analysis of the RCTs separately.
Safety Endpoints and Adverse Events
Trials were examined for any reported adverse events. To more fully capture the pattern of adverse events, any reported event was recorded regardless of whether the authors stated it was related, probably related, or possibly related to therapy. We specifically examined trials for the following events: death, MACEs, anemia, bleeding, pain, infection, injection-induced rhabdomyolysis, acute kidney injury, proliferative retinopathy (new or progressive disease), arterio-venous malformation, and cancer.
Efficacy Endpoints
Analysis of efficacy was divided into clinical endpoints (amputation, death, amputation-free survival [AFS]) and surrogate and functional endpoints (clinical classification, pain, QoL, wound healing, ABI, TcPO2, angiography). Major amputation is defined as an ablative procedure performed above the ankle.
Although amputation and death were examined in all trials, determining the influence of cell therapy on these clinical endpoints was limited to comparison of rates between control and treatment groups in RCTs.
We analyzed the results of functional and surrogate outcomes in a binary fashion: If a trial demonstrated a statistically significant improvement in a given endpoint, we defined that as an improvement; any other result was a failure to improve. We then determined the percentage of trials that demonstrated improvement for each outcome measure. Improvement was based on the definition used within each trial rather than a standardized definition. For noncontrolled trials, improvement was based on comparison against baseline.
Results
Overall Clinical Trial Data
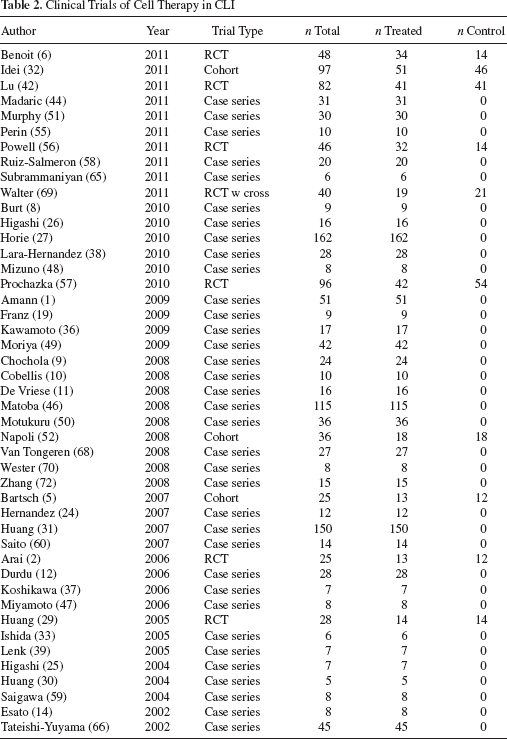
In our systematic review of the literature, we identified 45 clinical trials that evaluated cell therapy in CLI (Fig. 1). These trials enrolled 1,518 patients overall, 1,272 patients who received cell therapy, and 246 who served as controls. There were seven RCTs enrolling 365 patients (195 treatment and 170 control). There were three cohort controlled trials, which enrolled 158 patients (82 treatment and 76 control). The remaining 35 trials were observational case series that evaluated 995 patients, all of whom received cell therapy (Table 2).
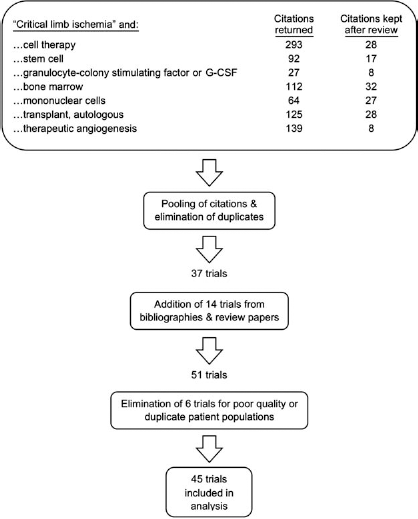
Flow chart of systemic review of cell therapy in no option critical limb ischemia (CLI).
Clinical Trials of Cell Therapy in CLI
The mean age of study subjects (weighted for sample size) was 63.0 years (range: 34–78 years), and the mean follow-up was 16.2 months (range: 1–48 months). The mean number of cells delivered was 32 × 108 (range: 0.39 × 108 to 399 × 108). The most common method of delivering cell therapy was by intramuscular (IM) injection, and this was done in 35 trials. Six trials infused cell via the intra-arterial (IA) route. Three trials used both methods, and a single trial compared IM to IA delivery.
With regard to risk factors, the mean age of the population receiving cell therapy was 63.0 years, and greater than half the patients had diabetes (53.5%) and tissue loss (69.4%). While 13.7% of the overall population required dialysis, this number is largely influenced by a single trial by Horie et al., in which two thirds of the patients (108 of 162) were on dialysis (27). Excluding this trial, the overall dialysis rate was 5.8%.
Etiology
Atherosclerosis was the most common cause of CLI, and 27 trials presented data on ASO patients. Nine trials presented data on TAO patients and five trials did not record the specific etiology of limb ischemia. Twelve trials enrolled a mixed population, but four of these presented their results separately based on etiology and those are included in the numbers of ASO and TAO trials above. ASO patients were older, had significantly higher rates of diabetes and renal failure when compared to TAO patients, although both groups had similar rates of tissue loss (Table 3).
CLI Risk Factors in Cell Therapy Trials

After removing the Horie trial (27) (67% dialysis), the percent dialysis for ASO patients is 9.9%, still significantly greater than for TAO (p < 0.0001).
After removing the Horie trial (67% dialysis), the percent dialysis in PBMNC patients is 11.4%, still significantly greater than for BMMNCs (p = 0.0016). ASO, atherosclerosis; TAO, thromboangiitis obliterans; PBMNC, peripheral blood mononuclear cell; BMMNC, bone marrow mononuclear cell.
Cell Source
The majority of cell therapy trials in the literature used bone marrow-derived cells. In the BMMNC group, there were 35 trials (6 RCTs, 3 cohort trials, 26 case series) that enrolled 1123 patients, 891 who received cell therapy, and 232 who served as controls. In the PBMNC group, 11 PBMNC trials (one RCT and 10 cases series) enrolled 395 patients with 381 treatment patients and 14 controls. One trial by Huang et al., compared BMMNCs and PBMNCs in separate arms; therefore, the sum of BMMNC and PBMNC trials is greater than the total number of published trials (31).
The mean volume of bone marrow aspirate in the BMMNC protocols was 386 ml (50–750 ml). One of the 50-ml protocols used culture expansion to increase cell counts.
Of the 10 PBMNC trials, nine pretreated patients with G-CSF to promote stem cell mobilization to the peripheral circulation prior to apheresis for collection of cells. The trial by Moriya et al. did not use G-CSF (49). One trial (Lenk et al.) isolated PBMNCs by Ficoll gradient separation of peripheral blood and then culture-expanded cells to a sufficient number (39). The remaining trials used apheresis and cell separators to isolate and concentrate PBMNCs.
While the BMMNC and PBMNC patient populations were fairly similar with regard to risk factors of age and the percentage of patients with diabetes and tissue loss, the peripheral blood group had a higher percentage of patients on dialysis than did the bone marrow group (33.8% vs. 5.1%, p < 0.0001). This discrepancy is largely due to the trial conducted by Horie et al., in which nearly two thirds of the patients were dialysis dependent (27). Even after excluding these patients, however, the PBMNC group still has a significantly higher percentage of patients on dialysis (11.4%). These differences in risk factors complicate comparison of outcomes between these two methods, at least with regard to amputation and mortality (Table 3).
Concentration Techniques
Cell therapy protocols employ a variety of cell concentration techniques. Twenty-seven trials used a cell separator such as the Cobe Spectra (Gabro BCT, CO, USA), Fenwal CS 3000 plus (Baxter, IL, USA), CLINIMACS (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany), or Fresenius AS 104 (Fresenius, Schweinfurt, Germany). Eleven trials used Ficoll gradient centrifugation in a central laboratory. Six trials used point-of-care devices such as Harvest SmartPReP (Harvest Technologies, Plymouth, MA, USA), Genesis CS (EmCyte Corporation, Fort Myers, FL, USA), and MarrowStim (BioMet, Warsaw, IN, USA). Two trials used bone marrow filtered for particulate matter prior to injection. One trial used a plasma extractor. A single trial used a proprietary method for ex vivo culture expansion of mononuclear cells harvested from bone marrow. Three trials used more than one method to prepare cells.
Randomized Controlled Trials
In our review of the literature, we identified seven RCTs of cell therapy in CLI. Five of these used cells isolated from bone marrow aspirate, one used cells isolated from peripheral blood, and one used bone marrow cells expanded by cell culture. Out of the total population of 365 patients, 195 received cell therapy and 170 served as controls. CLI was due to atherosclerosis in 97.8% of patients; thromboangiitis obliterans accounted for the other eight cases (2.2%) (Table 4).
Randomized Controlled Trials of Cell Therapy in CLI

BM, bone marrow; BM-Ex, bone marrow cells ex vivo culture expanded; PB, peripheral blood; ASO, atherosclerosis; TAO, thromboangiitis obliterans; NR, not recorded; POC, point of care centrifugation.
Five trials used mononuclear cells concentrated directly from bone marrow (BMMNCs). One trial by Huang et al. used PBMNCs, and these patients received pretreatment with G-CSF to mobilize cells (29). Of note, these patients remained on an intravenous heparin drip for 5 days during administration of growth factor as prophylaxis against thromboembolic complications associated with G-CSF. Powell et al. aspirated 50 ml bone marrow, which was subsequently expanded in tissue culture to achieve the desired number of mononuclear cells prior to administration (56).
Follow-up in the RCTs ranged from 1 to 6 months. In the trial by Walter et al., control patients crossed over to receive cell therapy at 3 months with the original treatment group receiving a second dose of cells (69). For the purposes of our analysis and to compare events between treatment and control groups, we considered events occurring only during the first 3 months of this trial.
Examining the risk factors between treatment and control groups in the overall RCT population demonstrates similarity between the two groups with regard to age, percentage of tissue loss, and dialysis. However, the control group had a higher rate of diabetes than the treatment group, and this difference was statistically significant (81.2% vs. 71.3%, p = 0.037) (Table 5).
Risk Factors of RCTs

Excludes patients from Powell trial (56), which presented no data on tissue loss. RCTs, randomized controlled trials.
To identify treatment effects due to bone marrow therapy, we focused on the group of five BMMNC trials. This excluded the Powell ex vivo trial (56) (which presented a certain amount of heterogeneity including undocumented tissue loss and higher rates of diabetes in the control patients) and the Huang PBMNC trial (29). This had the added effect of creating a more homogeneous study population in the RCTs; there were no statistically significant differences in risk factors between treatment and control groups, even with regard to diabetes (Table 5).
Safety
In our safety analysis, we defined adverse events due to cell therapy as those documented by a study's authors to be “related,” “probably related,” or “possibly related” to treatment. In our review of the literature, we identified 1,272 patients in 45 trials who received cell therapy, 891 in the BMMNC group, and 381 in the PBMNC group. There were 53 adverse events in the total population (4.2%). There were 39 events in the BMMNC group and 14 in the PBMNC group, which resulted in similar rates (4.4% and 3.7%, respectively).
The most common overall adverse event in the treatment population was pain (20.8% of all adverse events), followed by bleeding (17%) and anemia (15.1%). In the bone marrow group, the most common adverse events were pain (28.2% of events in BMMNC patients), anemia (12.8%), and postprocedural fever (10.3%). In the peripheral blood group, the most common adverse events were bleeding (42.9%), anemia (21.4%), and seizures (14.3%) (Table 6).
Frequency of Specific Adverse Events (AEs) as a Percentage of All Adverse Events

%AE, number of specific AE divided by total number of AEs within group.
Death
In our review of the literature of the 1,272 patients who received cell therapy, there was a single death of a patient following a complication possibly related to treatment. In the trial by Horie et al., one patient developed a gastrointestinal bleed and subsequently died within a month of G-CSF administration and PBMNC therapy (27). Within the same trial, five other patients developed gastrointestinal bleeding (see below). The population in this trial had a high acuity with 67% requiring dialysis and a 24% mortality rate in the patients with atherosclerosis in the first year after enrollment.
While no other deaths in the population from the literature review were defined as related or possibly related to cell therapy, in the interest of completeness we report on the death of a single patient in a trial by Miyamoto et al., which occurred 20 months after BMMNC therapy in a 30-year-old patient who was found dead at home of unknown causes (49).
To further determine if cell therapy contributes to death in CLI patients, we compared mortality rates between the treatment and control groups in the seven RCTs. The mortality rates were similar: 4.1% in cell therapy patients and 5.9% in controls with a value of p = 0.475. This supports the favorable safety profile of cell therapy with regard to death.
Major Adverse Cardiovascular Events (MACEs)
Three patients (0.2%) experienced MACEs related to cell therapy, two (5.1%) in the BMMNC group, and one (7.1%) in the PBMNC group. Van Tongeren reported two cases of congestive heart failure following aspiration of 750 ml of bone marrow (68). Both patients were treated with diuretics and recovered without sequelae. The authors speculate that protocols for devices that require smaller volumes of marrow aspirate may result in less fluid shifts and may allow the procedure to be performed under local anesthesia, both of which may limit the risks of CHF. In Horie's PBMNC trial, one patient experienced a nonfatal myocardial infarction in the month following G-CSF and cell therapy (27).
In the trial conducted by Arai et al., 14 patients in one arm of the study received G-CSF alone (2). These patients were not included in our overall safety analysis because they did not receive injection of cell therapy; however, it is worth noting that following 10 days of G-CSF one patient developed ventricular tachycardia requiring cardioversion.
Anemia
In our analysis, we defined acute anemia due to cell therapy based on each individual study's criteria. This generally meant a drop in blood counts requiring treatment. We did not rely on laboratory values because most studies did not provide such data. The lack of standard accepted values and the prevalence of anemia of chronic disease in the CLI population further complicate the use of laboratory values to identify anemia.
Eight patients (0.6%) developed anemia after cell therapy. This was one of the more common complications, comprising 15.4% of all adverse events. Anemia developed in 5 of 891 patients (0.6%) in the BMMNC group and 3 of 381 patients (0.8%) in the PBMNC group.
One patient developed anemia after 500-ml bone marrow aspiration leading to ischemic electrocardiography (EKG) changes that resolved after blood transfusion (51). This led the authors to change their protocol and reduce the volume of marrow aspirated to 360 ml with no further instances of anemia. In another trial (1), one patient who became anemic following the aspiration of 450 ml bone marrow required a blood transfusion. Three patients in a single trial (12) developed anemia following aspiration of 650 ml of bone marrow. None of the patients required transfusion but were instead managed by oral iron supplementation.
Three patients in a single PBMNC trial (27) developed anemia (grade 2 or grade 3) in the month following G-CSF treatment, peripheral blood harvesting, and cell therapy. There is no information regarding treatment of these patients.
Bleeding
Nine patients (0.7%) experienced bleeding related to cell therapy, and these complications compromised 17.3% of all adverse events. There were three (0.3%) bleeding events in the BMMNC group and six (1.6%) in the PBMNC group. Bleeding events differed between the two groups in kind and severity. In the BMMNC group, bleeding was associated with bone marrow harvest and was readily controlled and did not lead to anemia or further complications. In the PBMNC group, all six events were gastrointestinal bleeds occurring within 1 month of G-CSF and cell therapy, and one of these patients eventually died as mentioned above. These bleeds occurred in a single trial, and it should be noted that two thirds of the patients in this trial were dialysis dependent.
Pain
Eleven patients (0.9%) experienced significant pain as an adverse event, and this was the most common adverse event overall (20.8% of all events). All of the instances of pain were confined to the BMMNC group (1.2%). Most of these were due to pain at the bone marrow puncture site rather than in the treated limb. Furthermore, these episodes of pain were associated with the procedure itself, all occurring within 48 h of bone marrow harvest and cell therapy injection.
Infection-Related Events
In our population of cell therapy patients from the literature review two patients in the BMMNC group developed soft tissue infections (56) and two developed gangrene (55). These infections were not at the injection site and may have been caused by progression of disease but were deemed possibly related to therapy. There were no instances of systemic infection or sepsis related to cell therapy. In the BMMNC group, four patients experienced postprocedural fever, but these were not related to infections (32). No patients in the PBMNC group developed infectious complications. Of note, while there were no cases of infection caused by contaminated cellular products, there was one instance of bacterial contamination of culture-expanded cells, which prevented treatment (see “Other adverse events” below).
Rhabdomyolysis and Acute Kidney Injury
We found no cases of patients developing significantly elevated creatine phosphokinase (CPK) levels or rhabdomyolysis following intramuscular delivery of cell therapy, we found no cases of acute kidney injury occurring as a result of cell therapy despite the high degree of chronic renal insufficiency in this population.
Unregulated Angiogenesis: Arterio-Venous (A-V) Malformations and Retinopathy
We identified a single A-V malformation and no new cases of retinopathy in the population of patients receiving cell therapy. One TAO patient in the Miyamoto trial developed an arterio-venous shunt in the foot of the index limb 7 months after BMMNC therapy (47). By 1 year, this malformation had spontaneously resolved without sequelae. With regard to retinopathy, we found no new or progression of existing cases of retinopathy following treatment with cell therapy. Although 15 trials excluded patients if they had retinopathy at baseline, in the remaining trials there were 22 patients with retinopathy at enrollment and none of these patients experienced progression of their disease after treatment with cell therapy.
Cancer
We identified 15 documented cases of cancer in 1,272 patients who received cell therapy. This 1.2% incidence over a mean follow-up of 16.2 months equals an annual cancer incidence of 0.89%. The mean age of our study population was 63.0 years. Based on US National Cancer Institute statistics from 2004 to 2008, the age-adjusted cancer incidence for patients 60–64 is 1.3% (28). Because new malignancies are unlikely to appear in the short term following therapy, we focused on trials with follow-up greater than 1 year. In this group there were 14 cancers in 524 patients over a mean follow-up of 25.3 months. This rate of 2.7% corresponds to an annual incidence of 1.3%. By these rough comparisons, patients treated with cell therapy do not have a greater incidence of cancer than the rest of the population. One caveat to this analysis is that trials differ in their screening for preexisting malignancy. Prior to enrollment in one trial patients underwent screening by American Cancer Society recommendations with inclusion conditional on negative results (6). Some trials excluded patients with a history of malignancy. Others (such as the trial in which a patient was diagnosed with metastatic colon cancer 4 weeks after enrollment) did not conduct any cancer screening (69).
All cancers were seen in patients who had atherosclerotic disease. This is not surprising given that the mean age of ASO patients in our study population was 67.5 years compared to 42.7 years in the TAO population, and the incidence of cancer rises with age. Tobacco use is a risk factor for cancer development, and TAO is almost exclusively associated with smoking, yet the lack of cancers in this group following cell therapy is supporting evidence that cell therapy itself may not initiate cancer growth.
We analyzed the occurrence of cancers by cell source. Of the 15 cancers, 11 were seen in the PBMNC group and 4 in the BMMNC group. With 381 patients treated with PBMNCs and 891 patients treated with BMMNCs, this corresponds to rates of 2.9% in the PBMNC group and 0.45% in the BMMNC group. Correcting for the longer mean follow-up in the PBMNC group (18.3 vs. 15.3 months), the annual incidence of cancer was 1.9% in the PBMNC group and 0.35% in the BMMNC group. This difference is statistically significant (p = 0.0102).
The difference in mean ages between the two PBMNC and BMMNC patients (65.3 vs. 62.1) was not great, although it is possible that may have influenced the difference in cancer rates. The biggest difference between the two protocols that may influence cancer rates is the use of the growth factor G-CSF to promote mobilization of stem cells from the bone marrow in the PBMNC group. Although two of the cancers in the PBMNC group occurred in patients who did not receive growth factor, the other nine cases did receive G-CSF. Two of these patients had a previous history of malignancy.
In the bone marrow trial by Walter et al., one patient was discovered to have metastatic colon cancer 4 weeks after treatment (69). The extent of disease led the authors to conclude that malignancy had been present at the time of enrollment. No further tumors were seen in their study population over a mean 30.2 month follow-up. There was one cancer seen in the Idei BMMNC trial as well as one in the cohort control group (32). The Van Tongeren BMMNC trial had two cancers (68). One patient was diagnosed with a locally advanced colon cancer at 41 months. Another patient was diagnosed with laryngeal cancer at 7 months and then lung cancer at 25 months. In the Moriya trial of PBMNC therapy (which did not use G-CSF), two patients were diagnosed with gastric cancer, one at 20 months and the other at 29 months (49). Horie et al. evaluated the long-term outcome of patients treated with G-CSF and PBMNCs (27). They found nine cancers, two of which occurred in patients with a previous history of cancer. These malignancies were located in the colon, pancreas, lung, stomach, and gallbladder. It should be noted that, while the occurrence of gastric cancers documented in cell therapy trials may be higher than expected in a Western population, these cancers occurred in Asian study populations where this malignancy is more common.
Other Adverse Events
We identified 12 additional adverse events that did not fit into any of the above classifications. Two patients in the PBMNC group experienced seizures in the month following G-CSF and cell therapy (27). One patient experienced transient vertigo 1 day after BMMNC therapy (32). There were three adverse events associated with intra-arterial delivery of cells: one hematoma from femoral artery puncture, one thrombus formation in the artery (subsequently removed), and one pseudoaneurysm that resolved without therapy (69). Amann et al. reported a single case of intestinal puncture requiring operative repair during bone marrow harvest and another case of aspiration pneumonia following induction of general anesthesia (1). One patient experienced elevation of liver enzymes during administration of G-CSF, but these returned to baseline after discontinuation of the growth factor (38). In the trial that used ex vivo culture to expand BMMNCs, one patient was unable to receive cells due to contamination of the culture (56). Two BMMNC patients experienced mild edema of the injected limb (8,55).
Bone Marrow Versus Peripheral Blood Protocols and the Timing of Adverse Events
Because of the significant differences between bone marrow and peripheral blood cell protocols, we compared adverse events between the two groups in the literature. While the rates of events were similar in both groups, events differed in their timing after cell therapy, with nearly two thirds of BMMNC-related adverse events occurring in the immediate peri-procedural period. We defined peri-procedural adverse events as those occurring within 48 h and speculated that they were more likely to be related more to the procedure itself rather than the cellular composition. Of the total 53 adverse events, 27 of them (50.9%) were peri-procedural. All peri-procedural adverse events occurred in the BMMNC group, and these events accounted for 64.1% of all adverse events in this group. The events included pain and bleeding at the bone marrow harvest site in 14 patients: five cases of anemia following bone marrow aspiration, four cases of postoperative fever, two cases of congestive heart failure due to fluid shifts after marrow harvest, one aspiration pneumonia after induction of anesthesia, and one case of colon puncture during bone marrow harvest (Table 6).
No peri-procedural adverse events were documented in the PBMNC group, although nearly every trial commented on the frequency of bone pain and arthralgias associated with G-CSF administration. The bleeding and anemia in these patients was likely related to G-CSF and not the delivery of cell therapy itself. G-CSF is associated with thromboembolic events to such a degree that at least one group (Huang et al.) in their three trials kept patients on a heparin drip during the course of G-CSF administration (29).
Adverse Events in Randomized Controlled Trials
Comparing adverse events between treatment and placebo patients in RCTs should provide the best analysis of risks associated with cell therapy. The seven RCTs enrolled 365 patients, 195 of whom received cell therapy and 170 who served as controls (Table 7). There were 16 total adverse events, 14 (7.2%) in the treatment group and 2 (1.2%) in the control group. However, comparison of AEs between groups in RCTs is compromised by differences in protocol between treatment and control patients. For example, 10 of the events in the treatment group occurred within 48 h of procedure and nine of these were due to pain or bleeding associated with bone marrow aspiration. This greatly limits analysis of events between groups. Only a single study conducted a double-blind trial, which included sham bone marrow puncture, and in this trial there were no differences in the number of adverse events related to cell therapy between treatment and placebo groups (6). Of the remaining adverse events in the total RCT population, three adverse events in the treatment group were related to intra-arterial infusion (one hematoma, one thrombus, one pseudoaneursym); one treatment patient had contamination of ex vivo cell culture (but did not receive the contaminated cells); one control patient had edema of the placebo-injected limb; one control patient had pain at the injection site.
Adverse Events in Randomized Controlled Trials

Bleeding and pain at bone marrow puncture site (n = 9) limited to treatment patients complicates comparison of treatment versus control. Only one trial (6) used sham bone marrow puncture in control group. No difference in AEs tx versus control in this trial.
Efficacy
Amputation Rate
All but two of the 45 trials presented data on amputation. Of 1,272 patients who received cell therapy, 203 patients underwent amputation for an overall amputation rate of 16.0%.
With regard to cell source, there was no difference in amputation rate between BMMNC and PBMNC trials (15.8% vs. 17.1%, p = 0.504). Amputation rates differed significantly between patients with atherosclerosis and those with thromboangiitis obliterans (20.5% vs. 5.4%, p < 0.0001), which is to be expected given the overall worse prognosis of patients with atherosclerotic disease.
Analyzing the effect of risk factors on amputation rates is limited by the lack of individual patient data; however. it is possible to make conclusions based on the proportions of patients with a given risk factor in each of the trials. Trials in which greater than 50% of patients had diabetes demonstrated a significantly higher amputation rate than those with a lesser proportion of diabetics (19.1% vs. 10.2%, p < 0.0001). Trials that enrolled patients on dialysis had a significantly higher amputation rate than those that had no dialysis patients (27.0% vs. 12.2%, p < 0.0001). Trials in which greater than 50% of patients had tissue loss had a significantly higher amputation rate than those with a lesser proportion of tissue loss (16.8% vs. 7.7%, p = 0.0096). In patients with atherosclerosis, trials with a mean age of greater than 67 years (the mean age of all ASO patients) had a significantly higher amputation rate than trials with a younger proportion of patients (25.0% vs. 17.3%, p = 0.0074). This influence of age on amputation rate was not seen in patients with thromboangiitis obliterans.
To best estimate the efficacy of cell therapy on amputation rate, we focused our detailed analysis to RCTs. The duration of follow-up in the RCTs was similar (mean 4.1 months, range 1–6 months), which facilitated comparison of amputation rates among these trials.
In the seven RCTs, patients receiving cell therapy had a significantly lower amputation rate than control patients (14.4% vs. 27.6%, p = 0.0019). Focusing on the five trials using bone marrow protocols alone, the positive effect of cell therapy persists with an amputation rate of 14.8% in BMMNC-treated patients and 25.4% in controls (p = 0.0278) (Table 8).
Amputation Rates From RCTs: All Trials Versus BMMNC Trials Alone

Value of p is calculated using Fischer's exact test.
To further estimate the impact of cell therapy on amputation, we performed a meta-analysis on the seven RCTs. The odds ratio was 0.36, indicating that patients treated with cell therapy had a 36% chance of amputation when compared with control patients (p = 0.0004) (Fig. 2).

Meta-analysis of cell therapy on amputation rate. All RCTs and bone marrow trials only.
Time to Amputation
Although published trials do not present individual patient data on time to amputation, evaluation of amputation rates based on trial follow-up offers some insight into the timing of amputation. As trial duration increases from 3 to 6 to greater than 12 months, the amputation rate increases from 8.0% to 24.5% (Table 9). Longer trials provide more time in which amputations may occur. Looking at the percentage of total amputations based on trial duration suggests that amputations do not occur at a steady rate but are grouped largely in the first 6 months. While 12.3% of amputations occur in trials under 3 months and 41.9% occur in trials under 6 months, 49.8% of amputations occur in trials under 12 months. From this, we can infer that most amputations may take place within 6 months of enrollment with a prolonged “tail” of amputations occurring as follow-up progresses. This suggests that cell therapy trials with duration of 6 months may capture sufficient data with regard to amputation.
Relationship Between Trial Duration and Amputation Rate

Death
There were 119 deaths in the group of 1,272 patients who received cell therapy, resulting in an overall mortality rate of 9.4%.
Initial comparison of mortality rates between trials with different cell sources suggests that BMMNC-treated patients had a lower mortality than those receiving PBMNCs (5.5% vs. 18.4%, p < 0.0001). However, these results are heavily influenced by the trial by Horie et al., which had a long follow-up (median 26.4 months) and a 66.7% dialysis rate, both of which increase the mortality rate (27). Excluding this trial, the mortality rate in PBMNC trials was 9.1%, which, although higher than the rate in bone marrow trials, is not significantly different (p = 0.059).
Mortality rates differed by etiology. Patients with atherosclerosis had a 12.7% mortality rate while only 0.5% of patients with thromboangiitis obliterans died (p < 0.0001).
As with amputation, evaluating the effect of risk factors on mortality is limited to analyzing outcomes in trials with different proportions of a given risk factor. This analysis suggests that diabetes, dialysis, tissue loss, and age contribute to higher mortality rates. Trials in which greater than 50% of patients had diabetes had a significantly higher mortality rate than those with a lesser proportion of diabetics (12.5% vs. 3.4%, p < 0.0001). Trials that enrolled patients with dialysis had a significantly higher mortality rate than those that had no dialysis patients (24.0% vs. 4.0%, p < 0.0001). Trials in which greater than 50% of patients had tissue loss had a significantly higher mortality rate than those with a lesser proportion of tissue loss (10.1% vs. 4.0%, p = 0.050). In patients with atherosclerosis, trials with a mean age greater than 67 years (the mean age of all ASO patients) had a significantly higher mortality rate than trials with a younger proportion of patients (18.8% vs. 8.4%, p < 0.0001). This influence of age on mortality rate was not seen in patients with thromboangiitis obliterans.
Again, as with amputation, to estimate the effect of cell therapy on mortality, we limited our analysis to RCTs. In the seven RCTs, there was no significant difference in mortality rates between patients who received cell therapy and controls (4.1% vs. 5.9%, p = 0.475). This lack of difference in mortality rates was also true of the BMMNC trials alone (Table 10). While this does not demonstrate efficacy of cell therapy with regard to improving patient survival, it supports the fact that cell therapy is a safe procedure.
Mortality Rates From RCTs: All Trials Versus BMMNC Trials Alone

Value of p is calculated using Fischer's exact test.
Functional and Surrogate Outcomes
Functional and surrogate outcome measures are more commonly relied upon in observational trials because they do not require a control group but are instead measured as changes against baseline. The most commonly used measures were ABI and pain, and the least common were QoL and clinical classification. For each measure, at least 60% of trials demonstrated improvement. Cell therapy demonstrated the most improvement in wound healing (93.5% of trials) and reduction of pain (89.2%). This rough analysis suggests that cell therapy improves a variety of surrogate and functional outcomes in CLI patients (Table 11).
Efficacy of Cell Therapy in CLI Trials: Improvement in Functional and Surrogate Outcomes

Improvement is based on the definition used within each trial, not a standardized definition. For noncontrolled trials, improvement is based on comparison against baseline.
Number of trials demonstrating improvement divided by number of trials evaluating that outcome measure.
Discussion
In our systematic review of the literature of autologous cell therapy for CLI, we identified 45 clinical trials that included 1,272 patients who have received cell therapy in a research setting.
We found cell therapy to be safe, without increased risks of mortality or cancer associated with therapy and a low overall adverse event rate of 4.2%. Cell therapy also demonstrated efficacy, most notably in reducing amputation rates in treated patients compared with controls (odds ratio [OR] 0.36), as well as in improving a variety of functional and surrogate outcome measures.
The field of autologous cell therapy for CLI has demonstrated significant growth in recent years; in 2010 Fadini et al. published a comprehensive review in which he identified a single RCT (15). In our search of the literature conducted less than 2 years later, we identified seven published RCTs and an additional 500 patients receiving cell therapy.
While large RCTs provide the highest quality of data with the least amount of bias, we included smaller noncontrolled case series and cohort controlled trials specifically to expand our collection of safety data and thereby identify any patterns of adverse events in a larger pool of patients. The overall adverse event rate was low, and we observed few serious adverse events and no fatal adverse events related to the cell therapy procedures.
We did observe a difference in the pattern of adverse events between BMMNC and PBMNC protocols, with bone marrow protocols having more events related to the bone marrow puncture and harvest, such as pain, bleeding, and the sequelae following withdrawal of large volumes (>400 ml) of bone marrow. Several of the peri-procedural adverse events in the BMMNC group may be associated with the “learning curve” as clinicians become more familiar with harvesting bone marrow from the CLI patient population. Techniques that limit exposure to general anesthesia and limit the volume of marrow aspirate, such as point-of-care cell concentrators, may lessen the risks of cardiopulmonary complications such as anemia, heart failure, or aspiration pneumonia. In the PBMNC group, on the other hand, nearly every trial commented on the frequency of bone pain and arthralgias associated with G-CSF administration, and bleeding and anemia in these patients was likely related to G-CSF and not the delivery of cell therapy itself. G-CSF is associated with thromboembolic events to such a degree that at least one group (Huang et al.) in their three trials kept patients on a heparin drip during the course of G-CSF administration (29).
With regard to efficacy, RCTs are the only way to measure treatment effect in clinical outcomes such as amputation and death where comparison against historical controls is inaccurate.
In the absence of contemporaneous matched controls, many case series compared their amputation rates to historical controls, but such comparisons are of questionable value due to patient heterogeneity as well as the observation that event rates like amputation may be decreasing over time in the CLI population. While cohort controlled trials avoid these problems to some degree they are vulnerable to selection bias, which also may compromise their results. Therefore to estimate the efficacy of cell therapy on amputation rate we limited our detailed analysis to RCTs. Meta-analysis of amputation rates in the seven RCTs demonstrated a beneficial treatment effect with an odds ratio of 0.36, indicating that patients receiving cell therapy had a 36% chance of amputation compared to control patients. These are promising results for this challenging population of patients.
Mortality rates did not differ between patients receiving cell therapy and those treated as controls. Failure to demonstrate efficacy with regard to decreasing mortality is not a surprise given that traditional treatment of CLI is largely directed at limb salvage, and therefore would not be expected to have a significant impact on patient mortality. Although some authors have suggested that treatment of a threatened limb may remove an inflammatory or infectious focus and thereby improve patients' survival, detection of a treatment effect on mortality is difficult in the CLI population. CLI patients have high mortality rates related to both their vascular disease and comorbidities such as diabetes and renal insufficiency. In trials with longer follow-up, the influence of novel therapy on mortality may also be obscured by deaths from other causes because mortality rates necessarily rise with time, and this factor complicates comparison of mortality rates between groups with different lengths of follow-up. Evaluation of all-cause mortality in the setting of clinical trials is nonetheless important to identify any increased risks posed by novel therapy. Our observation that patients treated with autologous cell therapy did not have an increased mortality rate supports this as a safe procedure in the fragile CLI patient population.
Our review also highlights the influence of risk factors such as diabetes, tissue loss, and dialysis on clinical outcomes in CLI. Trials with higher proportion of patients with these risk factors had higher rates of amputation and mortality. While other reports have made this point with regard to patients undergoing arterial reconstruction (62,67) and we have previously published on the impact of tissue loss on amputation in the NO-CLI population specifically (6), our findings in this review emphasize the importance of risk factor stratification in patient enrollment in cell therapy trials in CLI.
Study Limitations
Lacking access to patient-specific data, our findings were dependent on events as described in each article. Given the high rate of morbidity associated with CLI, one might expect higher rates of adverse events than reported, the latter best assessed by independent monitoring and adjudication. Additionally, the evaluation of certain outcomes, such as retinopathy, is likely to have varied between studies. However, despite this intrinsic drawback to analyses such as ours, it is likely that published data accurately reflect serious events such as death, amputation, and malignancy.
Conclusions
Our systematic review of the literature identified 45 clinical trials of autologous cell therapy in CLI, which included seven RCTs. Overall 1,272 patients received cell therapy. Cell therapy in this population has a favorable safety profile with a 4.2% overall adverse event rate, no increase in mortality compared to control patients, and no increase in cases of malignancy when analyzed against population incidence rates. Protocols that isolate mononuclear cells directly from bone marrow were associated with more procedure-related adverse events such as pain, whereas peripheral blood protocols had adverse events related to G-CSF therapy such as bleeding. With regard to efficacy, cell therapy reduces amputation rates when compared to controls as well as improving a variety of functional and surrogate outcome measures. Our findings demonstrate that cell therapy in CLI has a positive benefit-to-risk ratio, which is of critical importance in this challenging patient population with limited therapeutic options.
Footnotes
Acknowledgments
The authors wish to acknowledge the statistical support of Dr. Eugenia Henry of inVentiv Clinical Solutions, LLC (Hunt Valley, Maryland), and Francisco Silva in helping edit the manuscript. Drs. Benoit and O'Donnell have served as medical consultants for Harvest Technologies, Inc. (Plymouth, Massachusetts). The authors declare no conflict of interest.
