Abstract
The aim of this work was to determine the effect of nicotine desensitization on dopamine (DA) release in the dorsal striatum and shell of the nucleus accumbens (NAc) from brain slices. In vitro fast-scan cyclic voltammetry analysis was used to evaluate dopamine release in the dorsal striatum and the NAc shell of Sprague–Dawley rats after infusion of nicotine, a nicotinic acetylcholine receptor (nAChR) antagonist mecamylamine (Mec), and an α4β2 cholinergic receptor antagonist (DHβe). DA release related to nicotine desensitization in the striatum and NAc shell was compared. In both structures, tonic release was suppressed by inhibition of the nicotine receptor (via Mec) and the α4β2 receptor (via DHβe). Paired-pulse ratio (PPR) was facilitated in both structures after nicotine and Mec infusion, and this facilitation was suppressed by increasing the stimulation interval. After variable frequency stimulation (simulating phasic burst), nicotine infusion induced significant augmentation of DA release in the striatum that was not seen in the absence of nicotine. In contrast, nicotine reduced phasic DA release in NAc, although frequency augmentation was seen both with and without nicotine. Evaluation of DA release evoked by various trains (high-frequency stimulation (HFS) 100 Hz) of high-frequency stimulation revealed significant enhancement after a train of three or more pulses in the striatum and NAc. The concentration differences between tonic and phasic release related to nicotine desensitization were more pronounced in the NAc shell. Nicotine desensitization is associated with suppression of tonic release of DA in both the striatum and NAc shell that may occur via the α4β2 subtype of nAChR, whereas phasic frequency-dependent augmentation and HFS-related gating release is more pronounced in the striatum than in the NAc shell. Differences between phasic and tonic release associated with nicotine desensitization may underlie processing of reward signals in the NAc shell, and this may have major implications for addictive behavior.
Introduction
Dopamine (DA) has been shown to play an important role in a number of physiological processes, as well as many neurological and psychiatric diseases including Parkinson’s disease (PD) 1,2 as well as schizophrenia and attention-deficit hyperactivity disorder 3,4 . DA has also been indicated as an essential component of the brain reward system 5,6 and is considered central to the mechanism of substance abuse 7 . DA inputs are mediated by both tonic and phasic release from DA neurons. The mechanisms of tonic and phasic DA release are distinct, and it has been proposed that dopaminergic neurons operate in two distinct patterns that depend upon the timescale of its activity 8,9 . During tonic release, dopaminergic neurons fire at low frequencies of 1–5 Hz to maintain a basal steady state concentration of DA in the extracellular compartment. Phasic release is produced by firing in high-frequency bursts (≥20 Hz) that occur in the context of motivationally salient stimuli that predict drug or reward availability 9 . DA may mediate reward behavior by switching activity of dopaminergic neurons from the tonic to phasic state to encode reward-related information 10 .
Nicotine affects release of DA and is related to smoking behavior. It is possible that endogenous cholinergic neurotransmission may be related to smoking habit formation and reward systems associated with nicotine addiction. To further investigate central nervous system DA physiology related to nicotine, we sought to characterize nicotine-augmented DA release in the striatum and nucleus accumbens (NAc) shell and analyze desensitization of the nicotinic acetylcholine receptor (nAChR) and its subtypes. Although the mechanism of nAChR desensitization has not been fully characterized, it has been documented that desensitization of nAChR plays an important role in response to cholinergic agents, and certain AChR-related diseases, to influence synaptic efficacy. Moreover, agent-specific desensitization and recovery from desensitization are important processes for nicotine receptor function that modulate the dopamine network. Therefore, to distinguish the area-specific desensitization in nigrostriatal and mesolimbic systems, in this report we focused on in vitro DA release patterns after infusion of nicotine, a nicotine receptor inhibitor (Mec), and a cholinergic α4β2 receptor inhibitor (DHβe) in the dorsal striatum and NAc shell. These drugs have been classically used to modulate central nicotine cholinergic transmission.
Materials and Methods
Animals
Young adult male Sprague–Dawley rats at 5 weeks old (LASCO Taiwan Co. Ltd., Taipei City, Taiwan) were used in this study. All procedures were reviewed and approved by the National Defense Medical Center Animal Care and Use Committee (IACUC16-258). Animals were provided food and water ad libitum and were housed in a 12-h light–dark cycle room.
Brain Slice Preparations
The brain slices and carbon fibers (7 μm diameter; Goodfellow Corp., Oakdale, PA, USA) were prepared as described previously 11,12 . Animals were decapitated and the brains were removed and placed in a beaker filled with oxygenated (95% O2/5% CO2) ice-cold cutting solution containing (in mM): sucrose 194, NaCl 30, KCl 4.5, MgCl2 1, NaH2PO4 1.2, glucose 10, and NaHCO3 26. A block of brain tissue containing the NAc and striatum was glued onto the cutting stage of a vibrating tissue slicer (Leica VT1000, Bannockburn, IL, USA) and the tissue block was immediately submersed in ice-cold oxygenated cutting solution. Coronal slices 280 μm in thickness containing the NAc and striatum were transferred to a holding chamber filled with oxygenated artificial cerebrospinal fluid (aCSF; in mM: NaCl 126, KCl 3, MgCl2 1.5, CaCl2 2.4, NaH2PO4 1.2, glucose 11, NaHCO3 26) at 31°C for 20–30 min. The holding chamber was allowed to reach room temperature and maintained at that temperature for the rest of the incubation period. The slices were then transferred to a heated chamber (31–33°C) and perfused with normal aCSF (2 ml/min) for fast-scan cyclic voltammetry (FSCV) recording. Handmade carbon fibers were lowered to a depth of 100 μm into the NAc or striatum under stereoscopic magnification. FSCV recording was performed as described previously 13,14 . A bipolar stimulating electrode was positioned 75–100 μm from the carbon fiber and constant voltage pulses (1–20 volt, 1 ms duration) were delivered between voltammetric scans to elicit DA release. Responses were obtained every 2 min, and only responses that were stable throughout the duration of the recordings were used for analysis. Micropipette assemblies containing the carbon fibers were backfilled with a 4 M potassium acetate/150 mM KCl solution and connected to a standard patch pipette holder/headstage assembly. A potentiostat (Chem-Clamp, Dagan Corp, Minneapolis, MN, USA) was used to apply a potential to the working electrode, relative to an Ag/AgCl reference, and to measure current. Voltammetric scans and electrical stimulus timing protocols were performed using peripheral component interconnect (PCI)-based analog to digital boards (National Instruments, Austin, TX, USA) and LabView-based software (TarHeel CV, University of North Carolina, Chapel Hill, NC, USA). For electrochemical detection, the potential of the carbon fiber was driven from −0.4 to 1.0 V then back to −0.4 V using a triangular waveform (400 V/s, 7 ms duration) applied every 100 ms. A 5-s (50-scan) control period was used to obtain a stable background current that was digitally subtracted from that obtained during the peak of the response following electrical stimulation. Peak oxidation currents were converted to DA concentrations using a calibration performed for each electrode with a 1-μM DA standard solution. All signals used in the statistical analyses matched the expected voltammetric profile for DA 15 .
FSCV Recording
To examine the effects of presynaptic nAChR on dopaminergic axons in the NAc shell or striatum, we measured extracellular dopamine at sub-second resolutions using cyclic voltammetry (CV) in these acutely prepared brain slices. A bipolar stimulating electrode was placed in the NAc shell ∼150 μm from the 5 μm diameter carbon-fiber microelectrode and depolarizing currents were applied at 2 min intervals, providing a stable dopamine response for >1 h. A single pulse stimulus (1p) elicited a maximum dopamine concentration of 0.66 ± 0.11 μM. Using paired pulses at different stimulation intervals (from 10 to 80 μsec), the paired-pulse release ratios (p2/p1) were measured in order to determine the interaction between firing frequency and dynamic release probability. A similar approach was used for striatal recordings.
Statistical Analyses
Data are expressed as means ± standard error of the mean. Data for dopamine release input/output curves were analyzed using a two-way analysis of variance (ANOVA) followed by a Bonferroni post-hoc test for multiple comparisons. A one-way ANOVA and a Bonferroni post-hoc test was used to analyze DA concentrations evoked by different stimulation intensities (from 1 to 10 volts), by different stimulation frequencies, by various trains of high-frequency stimulation (HFS), by various stimulation intervals for paired-pulse ratio (PPR) analysis, and for the slope of linear regression under control or nicotine desensitization conditions. All data were subjected to a repeated measure ANOVA (pre-test and post-test) with the alpha level set at 0.05. Post-hoc comparisons were performed using a least significant difference post-hoc test. Mixed effects regression analysis for repeated measures was used to evaluate group differences for evoked DA release in the shell portion of the NAc or striatum. All statistical tests were performed using GraphPad Prism 5.02. A
Results
Nicotine-induced Suppression of Tonic Dopamine Release is Seen in Both the Striatum and NAc Shell
To evaluate the effect of desensitization after nicotine infusion on tonic dopamine release, we analyzed the changes in dopamine release evoked by single pulse (1p/25 Hz) stimulation before, during and after nicotine (0.5 μM) infusion, using FSCV in both the striatum and NAc shell. The time course of DA release (evoked with measurements performed every 30 sec) was markedly suppressed by nicotine desensitization (Fig. 1A, two-way ANOVA,
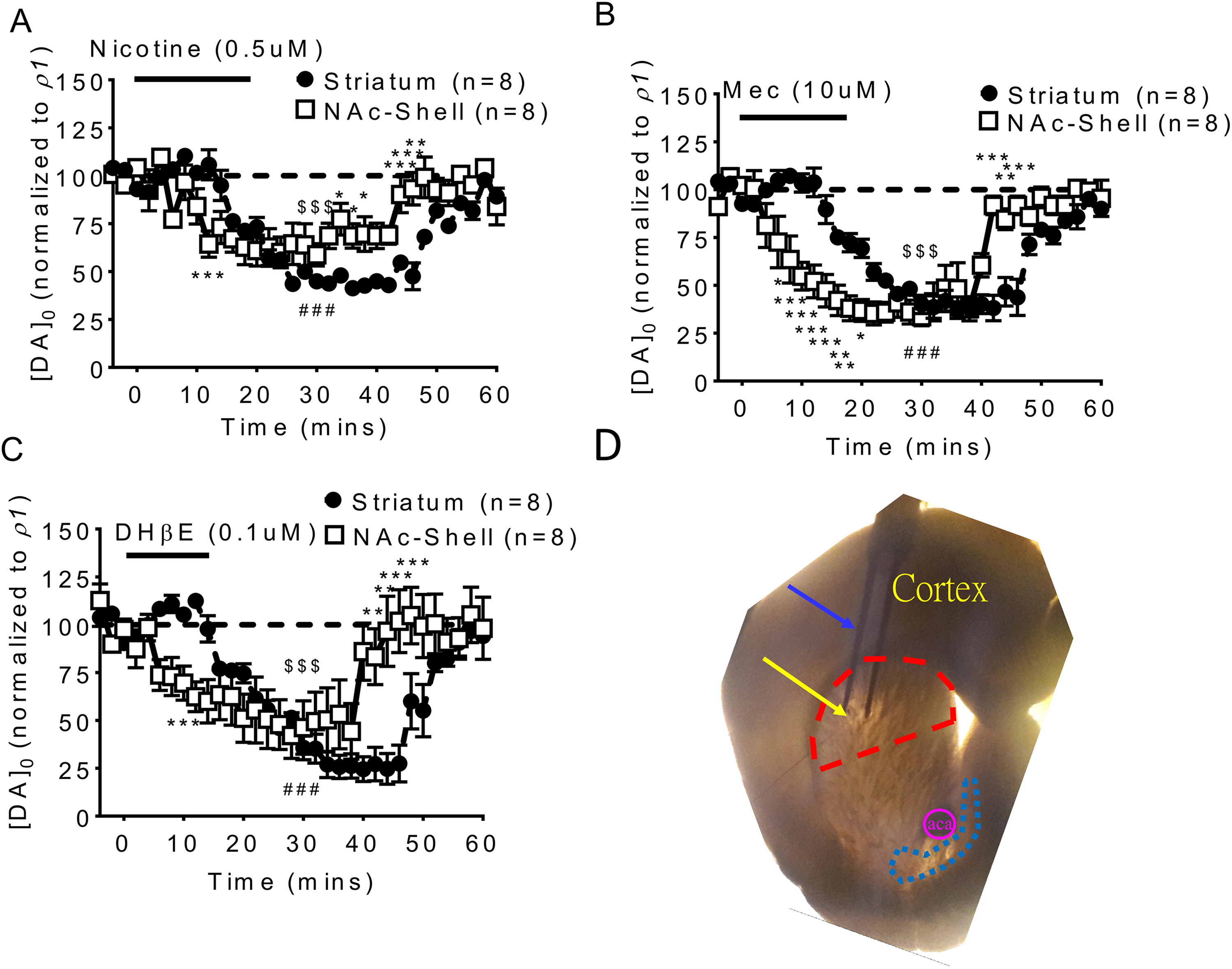
(A) Nicotine infusion induces a desensitizing suppression of tonic DA release (by single pulse 1p/25 Hz) not only in the striatum but also in the NAc shell. The suppression of tonic DA release in the striatum was similar to the NAc shell under 0.5 μM. (B) Suppression effects were also found using the nAChR antagonist mecamylamine (Mec) 1 μM infusion and (C) DHβe 0.1 μM infusion. (
Nicotine-induced Desensitization Suppression of Phasic DA Release is Dose Dependent
We evaluated evoked DA release under different stimulation intensities (input/output; I/O curve, from 1 to 10 volts) under single pulse (tonic) and 10 pulses/25 Hz (phasic) conditions. In the striatum, tonic release was significantly suppressed by nicotine infusion (Fig. 2A, two-way ANOVA,

Nicotine effects on input/output curves for tonic and phasic dopamine release in the striatum and shell portion of the NAc under different stimulation intensities (from 1 to 10 volts) were altered by nicotine infusion. (A) In the striatum, tonic release was significantly suppressed by nicotine infusion. (B) The phasic release in the striatum was enhanced by nicotine infusion. (C) In the NAc shell portion, tonic release was significantly suppressed by nicotine infusion, which was similar to the striatum. (D) The phasic dopamine release in the NAc shell portion was also suppressed by nicotine infusion; this suppression was more significant at higher stimulation intensities. (E) Nicotine desensitization induced a dose-dependent suppression of dopamine tonic release (1p/25 Hz) in the NAc shell. (F) Nicotine desensitization induced suppression of phasic release in the NAc shell but enhanced phasic release in the striatum. *
nAChR-related Augmentation of Frequency Dependence of Dopamine Release in Striatum and NAc Shell were Different
A nAChR-related augmentation of frequency dependence on DA release was seen in the striatum but was less prominent in the NAc shell. In the striatum, DA release was suppressed after low-frequency stimulation and enhanced after HFS as well as with nAChR desensitization and blockade; this was not seen in the absence of nicotine (Fig. 3A, non-treatment versus treatment, two-way ANOVA, [

nAChR desensitization-related frequency-dependent augmentation of dopamine release is different in striatum and NAc shell. (All of the signals in each plot were compared with the control p1 signal.) (A) The suppression in tonic release reversed to an increment with high-frequency stimulation in the striatum under nicotine infusion, while this increment was not significant under control conditions (control: red solid, nicotine infusion: red open circles). In the NAc shell, the release of DA in the shell portion was suppressed significantly under nicotine infusion compared with the control group. The increment in DA release along with stimulation frequency was found under both control and nicotine infusion conditions (blue solid and open squares). (B)With infusion of mecamylamine, suppression of DA tonic release was seen in both the striatum and NAc shell, and DA release increased after high-frequency stimulation only in the striatum. These increments with increased frequency were found in the NAc shell with or without Mec infusion, but the DA level was significant lower only in the Mec infusion group. (C) Infusion of the α4β2 receptor inhibitor, DHβe, induced similar effects in NAc, which suggests that most receptors associated with the nicotine effects on DA release in NAc shell were of the α4β2 receptor subtype. (D) The difference between tonic and phasic release concentrations in the striatum significantly increased with increasing stimulation frequency (from 1 to 100 Hz) after nicotine infusion. In the NAc shell, these differences were seen both in controls and after nicotine infusion. The frequency-dependent augmentation after nicotine infusion was similar to the control. Similar effects were found with (E) Mec and (F) DHβe infusion. (G) The slope of the difference in the concentration equation indicates the release probability of DA in each group; the highest release probability was found in the striatum with nicotine infusion, and the slope was similar between control and nicotine infusion groups in the NAc shell portion. (H) Representative DA signals (IT) in the striatum under different stimulation frequencies with significant frequency augmentation are shown in nicotine, Mec, and DHβe infusion groups, compared with the control slices. (I) In the NAc shell, the frequency augmentation of DA release was found in the control slices and also after nicotine, Mec and DHβe infusion. The signals were reduced after nicotine, Mec and DHβe infusion compared with control signals. **
Nicotine Desensitization-induced Phasic DA Release, Induced by HFS (100 Hz), is Different in the NAc Shell and Striatum
In the striatum, the increase in phasic DA release evoked by HFS (100 Hz) along with the number of trains of HFS occurred with nicotine (Fig. 4A,

Dopamine release related to HSF (100 Hz). (A) DA release did not increase with the number of HFS trains in the striatum but DA release increased with the number of HFS trains in the striatum after infusion with nicotine. DA release in the nicotine infusion group was lower than in control group in the NAc shell, but both groups showed an increase with trains of HFS. (B) Mec infusion suppressed DA release initially and the release increased along with the number of trains in the striatum (red open circles). In the NAc shell, DA release was suppressed with Mec infusion. In both the control and Mec infusion groups, DA release increased along with the number of trains. (C) DA release was suppressed initially with DHβe infusion in the NAc shell, and an increment in release with train number was found similar to the control group. (D) Linear regression for the difference between large trains of HFS phasic and tonic concentrations (normalized to control p1 concentration); the slope, referred to as the release probability of DA in the NAc shell, was steeper than in the striatum. (E) A similar situation was found with Mec infusion and (F) with DHβe infusion. (G) The slopes in the NAc shell portion, related to nicotine desensitization, were higher than in the control group and in the striatum. (H) Representative IT and CV curves in the striatum and (I) NAc shell with different numbers of HFS trains are shown. *
Nicotine-Related Short-Term Facilitation of DA Release was Similar in the NAc Shell and Striatum
PPRs (p2/p1) induced by paired pulses at varying inter-pulse intervals reflect the interaction between firing frequency and dynamic release probability. The important characteristics of PPR in the control slices were different in the striatum and the NAc shell. Short-term depression was seen only in the control striatum at all inter-pulse intervals but potentiation could be found in control NAc shells with inter-pulse intervals shorter than 50 msec, whereas the PPR showed depression with longer intervals. In contrast, facilitation could be found under nicotine-induced desensitization conditions in both the striatum and NAc shell (Fig. 5A, non-treatment versus treatment, two-way ANOVA,
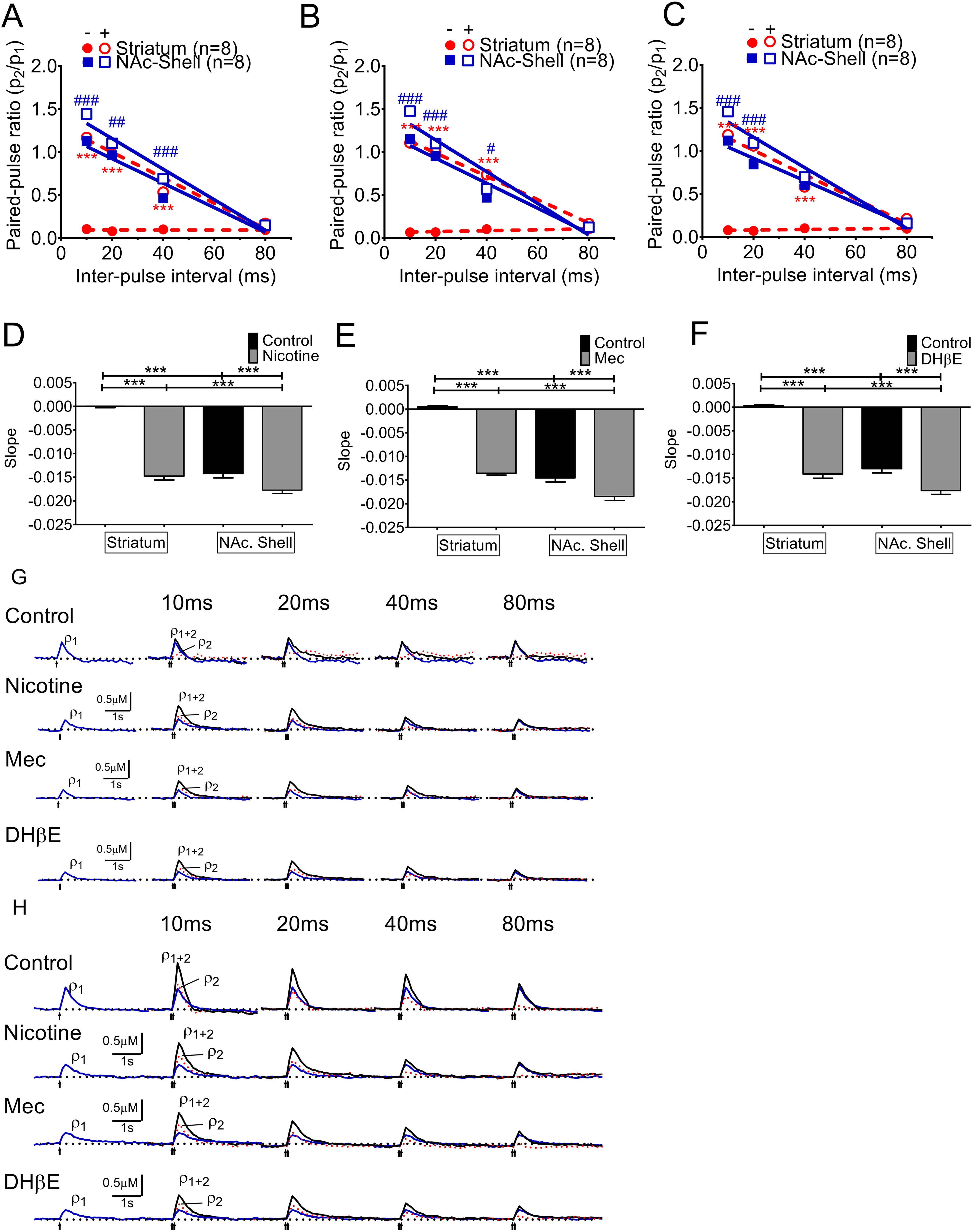
Nicotine-related short-term facilitation of DA release. PPRs (p2/p1) showed a characteristic short-term depression in the controls at all inter-pulse intervals at the striatum whereas the Paired pulse facilitation (PPF) could be found in the short interval (<50 msec) in the NAc shell portion and then the PPF became Paired pulse depression (PPD) while the interval was getting longer. (A) Both the striatum and the NAc shell area showed nicotine desensitization-related facilitation of DA release. This facilitation was dependent on frequency, with the largest at short stimulation intervals and suppressed at longer intervals. (B) Mec infusion induced a similar facilitation of DA release with short stimulation intervals and suppression at longer intervals. The suppression effects were more profound in the shell portion. (C) In the NAc shell, DHβe produced facilitation effects similar to those in the striatum. (D) The slopes of linear regression of the PPR related to the stimulation interval, and the slopes of the nicotine desensitization-related decline in the NAc shell portion, were higher than those in the striatum. A similar relationship could be found with (E) Mec and (F) DHβe infusion. (G) The voltammetric signals in the striatum with paired pulses at 10 ms intervals indicate synaptic facilitation by nicotine (blue line for the first response p1, red dotted line for the subtracted second response p2, and black line for the summation of p1+p2). This facilitation is suppressed by nicotine and Mec infusion with increased stimulation intervals, whereas there was no facilitation in the control groups. (H) The voltammetric responses in the NAc shell portion were similar to the striatum. ***
Discussion
Two patterns of dopamine release have been indicated in previous in vivo studies: (1) tonic, which refers to DA neurons firing at low frequencies of 1–5 Hz for basal DA tone on high-affinity dopamine D2 receptors in the mesolimbic DA system, including the NAc; and (2) phasic, which refers to high-frequency DA neuron bursts (≥20 Hz) to produce transient high DA levels sufficient to occupy low-affinity DA D1 receptors in the NAc 16 –18 . Thus, different stimulation frequencies used here mimic different physiological firing activities from tonic to phasic bursting that typically accompany reinforcement or reward prediction 10,19 .
There is evidence that drug addiction involves multiple functional processes including reward by the mesolimbic dopaminergic pathways, emotion by the amygdala, learning, memory by the hippocampus, and executive functions by the prefrontal cortex. Each of these processes are governed by the cortico-limbic network 20 . The role of the NAc and striatum in the development of drug addiction is controversial but is thought to involve distinct dopaminergic innervations in each of these areas. The dorsal striatum receives projections from the substantia nigra pars compacta (SNc), while the ventral striatum, including the NAc, receives dopaminergic input primarily from the ventral tegmental area (VTA) 21,22 . The ventral striatum, believed to underlie the reinforcing effects of abused drugs, is innervated by excitatory inputs from the prefrontal cortex (PFC), amygdala and hippocampus 23,24 . On the other hand, the dorsal striatum gates sensorimotor functions thought to underlie habitual drug seeking via efferent projections from the motor cortex 25,26 .
Upregulation of nAChR subtypes may be induced by long-term exposure to nicotine, which is reflected by an increase in high-affinity nicotine binding sites in multiple regions of brain 27 –29 . On the other hand, a higher dose of nicotine exposure may cause rapid nAChR desensitization, associated with a loss of receptor function. After long-term desensitization, upregulation may be triggered to compensate for the reduced signal from inactivated receptors 30,31 . These changes may induce a higher sensitivity to nicotine and have been related to nicotine addiction 32 .
Desensitization has also been postulated to play a role in nicotine tolerance and dependence 33 –36 . When desensitized α4β2 nAChRs become unoccupied and recover to a responsive state during periods of abstinence, such as sleeping, craving and withdrawal, symptoms are seen in chronic smokers that are alleviated by nicotine binding and desensitization of these receptors during smoking 37 –39 . Therefore, to prevent withdrawal symptoms, smokers may need to smoke continuously to maintain plasma nicotine levels and search for environmental contexts that reinforce smoking behavior in order to derive rewarding effects 40,41 . Although nicotine activates DA release and has been indicated to be involved in addiction mechanisms, high-dose applications or fully occupied nAChRs may lead to desensitization and a prolonged depression of evoked dopamine release 33,42,43 .
Development of reward behavior is associated with conditioned stimuli and reinforcement 44 , involving signals conveyed by phasic DA release in the mesostriatal and mesocorticolimbic systems 45 . The mesocorticolimbic system mediates the sense of pleasure 46 . Since it is densely innervated by tonically active cholinergic interneurons and dopaminergic neurons that originate from the VTA, the NAc shell is thought to be the principle region associated with nicotine reinforcement 47 . These neurons possess both cell body and presynaptic nAChRs 48 . Microdialysis has shown that nicotine shifts VTA neurons from tonic to burst-firing 49 and greatly enhances extracellular dopamine levels 50 . In addition, serum nicotine levels in chronic smokers (∼250–300 nM) are likely to rapidly desensitize nAChR to block tonic acetylcholine activation and inhibit evoked DA release 51 . However, the relationship between nicotine-induced elevations in extracellular DA and depression of evoked DA release in both the striatum and NAc shell remains controversial 52 .
DA release encoded by nicotine may play an important role in nicotine addiction, so mechanisms and responses to nicotine-encoded DA release are important. In this study, we measured the effect of nicotine desensitization on DA release. We found that DA release in both the striatum and the NAc shell is suppressed by nicotinic acetylcholine receptor desensitization. The I/O curve data in the NAc shell revealed that tonic DA release may be affected by nicotine infusion but phasic release is affected only at higher doses (Fig. 2). Frequency-dependent augmentation of DA release was seen in the striatum only with nicotine infusion, whereas this type of augmentation in the NAc shell could be found with or without nicotine (Fig. 3).
Enhancement of bursting DA release related to trains of HFS and nicotine desensitization was prominent in the NAc shell with or without nicotine, but was not seen in the striatum without nicotine. With nicotine desensitization and nAChr blockade (Mec infusion), DA release with lower trains of bursts was seen in the NAc shell, while this suppression subsided gradually with increasing the trains of bursting stimulation trains (Fig. 4). Furthermore, when the DA concentration was normalized to the control p1, we found that the incremental proportion related to nicotine desensitization in the NAc shell was significantly higher than in the striatum (Fig. 4).
In Fig. 3, the different frequency-specific effects of nicotine in the striatum and NAc shell portion are shown. We then explored the frequency-specific effects of nicotine by using paired pulses at varying inter-pulse intervals to determine the interaction between firing frequency and dynamic release probabilities 47,53 . The frequency-specific effects of nicotine were different in the striatum and the NAc shell. PPRs (p2/p1) showed characteristic short-term depression in controls at all inter-pulse intervals in the striatum whereas the PPF could be found at short intervals (< 50 msec) in the NAc shell portion. The PPF became PPD when the intervals increased (Fig. 5A–C). These phenomena are referred as frequency filtering 47 . The data we shown here are similar to previous reports showing that nicotine-induced desensitization appears to set a high-frequency pass filter for impulse-dependent DA release 10,54 . Through nAChR desensitization, DA release would be suppressed during non-reward, low firing frequencies; whereas release would be selectively enhanced during reward-related event due to relieving short-term depression at higher burst-like frequencies 53,55,56 . Supporting this concept, the decline of PPR was faster in the NAc shell compared with the striatum (Fig. 5D–F).
The patterns of DA release we observed related to nicotine desensitization in the striatum and NAc shell are summarized in Table 1. The similarities between the striatum and NAc include: (1) tonic release is suppressed by Mec, a nicotine receptor antagonist and the α4β2 receptor inhibitor (DHβe) in the striatum and NAc shell; (2) PPR is facilitated by nicotine and Mec infusion in both the striatum and the NAc shell; and (3) facilitation is suppressed by increasing the stimulation interval.
The Relationship Between Nicotine Desensitization and DA Release in the Striatum and NAc Shell.

DA: dopamine; Dhβe: α4β2 cholinergic receptor antagonist; HFS: high-frequency stimulation; Mec: mecamylamine; NAc: nucleus accumbens; PPD: Paired pulse facilitation; PPF: Paired pulse depression.
The differences between NAc and the striatum include: (1) input/output curve analysis indicates that nicotine desensitization induces suppression of tonic DA release and enhanced phasic release in the striatum, whereas in the NAc shell both tonic and phasic release are suppressed by nicotine infusion; (2) phasic burst stimulation data indicates that nicotine infusion induces significant frequency augmentation in the striatum, whereas phasic DA release in the NAc shell is lowered by nicotine and frequency augmentation, both with nicotine infusion and under control states; and (3) DA evoked by various trains of HFS (100 Hz) reveal that nicotine gating of DA release is significantly enhanced with three or more trains of HFS in the striatum, whereas HFS stimulation-induced DA release enhancement is seen with both the presence and absence of nicotine in the NAc shell. The concentration difference between tonic and phasic release related to nicotine desensitization was more prominent in the NAc shell.
The desensitization of nAChRs reduces tonic DA release and allows a rapid rise in DA release after phasic firing patterns associated with incentive/salience paradigms. Thus, nicotine may enhance the ‘signal to noise ratio’ of DA signals associated with behavioral cues, and nicotine’s effects mainly depend on the firing pattern of DA neurons 47 . These phenomena have been observed in both the striatum and NAc shell. Our data support that nicotine’s effects depend on the firing pattern of DA neurons, so desensitization of nAChRs limits DA release with tonic firing and allows release enhancement under phasic firing patterns associated with incentive/salience paradigms 10,47 .
In the striatum, nicotine desensitization increases the concentration difference between tonic and phasic DA release and therefore the strength of the reward signal related to reinforcing and learning behaviors 10 . The mechanism of action of nicotine in the striatum may involve the dual action of nicotine on DA release, which has been previously shown to suppress initial tonic release and enhance higher frequency stimulation evoked (phasic) release. Our data reveal similar effects of nicotine on the dynamic release probability of DA (relative release per pulse) after a phasic burst (100 Hz) in the striatum 10 .
In the NAc shell, transition from tonic to phasic release for frequency-dependent augmentation was inhibited by nicotine desensitization, and there was an influence of stimulus frequency on release with or without nicotine. There was less of a difference in absolute concentration between tonic and phasic release in the NAc shell related to the frequency of stimulation. However, if the concentration is normalized using all evoked DA signals compared with the control P1 concentration, nicotine desensitization expands tonic and phasic release, and DA release increases along with frequency (from 1–100 Hz) (Fig. 3). DA release is significantly correlated with the HFS train number (Fig. 4) in the NAc shell but not in the striatum; after normalization the NAc shell also exhibited a correlation even greater than in the striatum.
Currently, both nAChR activation and desensitization have been postulated to be important in drug reinforcement and affective behavior 57 . Through nicotine desensitization, the reward system might suppress the low-frequency DA release associated with non-reward-related stimuli, but at a higher frequency, bursting DA release selectively might enhance the reward-related signal by relieving short-term depression 10 . Our data suggest that not only does phasic release play an important role in addictive behavior, but the difference between phasic and tonic release concentration relates to a reward signal. This effect in the NAc shell and mesocorticolimbic system is prominent and may have important implications for addiction.
Conclusion
The tonic release of DA in the striatum and NAc shell are both suppressed by nicotine desensitization that may occur via the α4β2 subtype nAChR. After nicotine desensitization, frequency-dependent augmentation and HFS-related gated release are more significant in the striatum compared with the NAc shell. However, absolute phasic release may play a less important role in addictive behavior than the difference between phasic and tonic release concentrations. This effect is most prominent in the NAc shell within the mesocorticolimbic system. These findings may have relevance for the pathophysiology of addictive behavior.
Supplemental Material
Supplemental Material, Nicotine_1_Encoding_Dopamine_TopCop_4-13-18R4 - Differences in Nicotine Encoding Dopamine Release between the Striatum and Shell Portion of the Nucleus Accumbens
Supplemental Material, Nicotine_1_Encoding_Dopamine_TopCop_4-13-18R4 for Differences in Nicotine Encoding Dopamine Release between the Striatum and Shell Portion of the Nucleus Accumbens by Yuan-Hao Chen, Bon-Jour Lin, Tsung-Hsun Hsieh, Tung-Tai Kuo, Jonathan Miller, Yu-Ching Chou, Eagle Yi-Kung Huang, and Barry J. Hoffer in Cell Transplantation
Footnotes
Acknowledgments
Yuan-Hao Chen and Bon-Jour Lin contributed equally to this work.
Ethical Approval
All procedures were reviewed and approved by the National Defense Medical Center Animal Care and Use Committee (IACUC16-258).
Statement of Human and Animal Rights
All procedures involving laboratory animals were reviewed and approved by the National Defense Medical Center Animal Care and Use Committee (IACUC16-258).
Statement of Informed Consent
Statement of Informed Consent is not applicable for this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Council of Taiwan under grant: MOST105-2314-B-016-001-MY3, and by Medical Research Project grants: TSGH-C107-068, TSGH-C107-071 and TSGH-C107-070 from the Tri-Service General Hospital of Taiwan and MAB-107-022 from the National Defense Medical Center. This project was also supported in part by philanthropic support from the George R. and Constance P. Lincoln family.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
