Abstract
Spontaneous intracerebral hemorrhage (ICH) is one type of the most devastating cerebrovascular diseases worldwide, which causes high morbidity and mortality. However, efficient treatment is still lacking. Stem cell therapy has shown good neuroprotective and neurorestorative effect in ICH and is a promising treatment. In this study, our aim was to review the therapeutic effects, strategies, related mechanisms and safety issues of various types of stem cell for ICH treatment. Numerous studies had demonstrated the therapeutic effects of diverse stem cell types in ICH. The potential mechanisms include tissue repair and replacement, neurotrophy, promotion of neurogenesis and angiogenesis, anti-apoptosis, immunoregulation and anti-inflammation and so forth. The microenvironment of the central nervous system (CNS) can also influence the effects of stem cell therapy. The detailed therapeutic strategies for ICH treatment such as cell type, the number of cells, time window, and the routes of medication delivery, varied greatly among different studies and had not been determined. Moreover, the safety issues of stem cell therapy for ICH should not be ignored. Stem cell therapy showed good therapeutic effect in ICH, making it a promising treatment. However, safety should be carefully evaluated, and more clinical trials are required before stem cell therapy can be extensively applied to clinical use.
Introduction
Spontaneous intracerebral hemorrhage (ICH) is one type of the most devastating cerebrovascular disease worldwide, which accounts for 15% of all strokes 1 . ICH shows high morbidity and mortality. The incidence of ICH is about 0.1–0.2% in the general population and is even higher in elderly people, among which the mortality rate is extremely high, with a death rate of almost 30–50%. Survivors inevitably suffer from long-term and severe neurologic impairment despite multiple treatment approaches 2 . Based on the current data, the prognosis of ICH is extremely poor. There are various risk factors contributing to the onset of ICH, which include coagulation dysfunction, amyloidosis, vasculitis, drug abuse, and genetic factors 3 . However, the most important risk factor inducing ICH is hypertension, constituting about 60% of all ICH cases 4 .
The pathological mechanism of ICH comprises two parts: the primary and the secondary injuries. The first type is the occupying effect and the mechanical damage to adjacent brain tissue resulting from the hematoma. In the meantime, toxic effects of the blood and the decomposed products of blood cells such as enzymes, hemoglobin, and iron ions result in a more severe secondary injury. The secondary injury involves diverse molecular, cellular and biochemical responses induced by the primary injury; typically, inflammation, apoptosis, lipid peroxidation, free radical damage, and glutamate excitotoxicity, to name but a few 5 .
Nowadays, the available treatments for ICH include surgery, the control of intracranial hypertension and blood pressure, the alleviation of cerebral edema, supportive care, and rehabilitation. However, only limited effectiveness of intervention is currently demonstrated 6 . The novel alternatives or efficacious methods for treating ICH are in demand. Stem cell therapy, as a promising approach, has thus aroused considerable interest in researchers worldwide.
Stem cells (SCs) refer to a type of cell that have the potential to proliferate, self-renew, and differentiate into a variety of functional cells in a certain condition 7 . According to the developmental stages, SCs can be divided into two broad types: embryonic SCs (ESCs), which are isolated from the inner cell mass of blastocysts, and adult SCs (ASCs), also known as somatic SCs (SSCs), which can be found in various adult tissues, including neural SCs (NSCs), hematopoietic SCs (HSCs), mesenchymal SCs (MSCs), epidermal SCs, and so forth. According to the differentiative potential, the SCs can be divided into three categories: totipotent SCs, pluripotent SCs (PSCs) and unipotent SCs. The attempts to treat human diseases using SCs have been in existence for several decades. One of the most mature and general applications is the human SC transplantation therapy for multiple malignant or benign hematological diseases, which shows a great clinical value 8 . Furthermore, the treatment of neurological diseases by means of SC therapy, such as ischemic stroke, traumatic brain injury, and subarachnoid hemorrhage, had been developing rapidly in recent years and showed promising results 9 –11 .
Currently, a growing number of studies have been conducted on SC treatment for ICH, not only in animal experiments, but also in clinical trials, which presented favorable curative effects, and potentials in saving the damaged brain tissue and promoting functional recovery. MSCs, NSCs, ESCs, HSCs and induced PSCs (iPSCs) are the most common types of SC in research and having application in ICH treatment.
The possible therapeutic mechanisms of SCs in ICH treatment involve multiple factors that have been studied for many years. One of the most important mechanisms is that SC transplantation repairs or replaces the destroyed nerve cells and tissues, including neurons and glial cells, which helps to ensure the integrity of nerve conduction pathways, thus rebuilding neurological functions 12 . Moreover, at the molecular level, incorporated SCs are capable of providing neurotrophic factors through paracrine signaling to develop neurotrophic effects. In addition, SCs can help to reduce ICH-induced secondary injuries including apoptosis, inflammation, and blood–brain barrier (BBB) destruction, and to promote angiogenesis and neurogenesis; therefore, the neuroprotection, together with the neurorestoration, is manifested in SC therapy 13,14 .
Despite the extensive studies of cell replacement in neurodegenerative disorders and ischemic stroke having been established already, studies on ICH have just come into being in the last decade. In this study, we aimed to provide a detailed review on the neuroprotective effects of SCs and the potential mechanisms for treating ICH, which is expected to benefit the application of SC therapy to ICH management in the near future.
Commonly Used Stem Cell Types for Intracerebral Hemorrhage Treatment
Mesenchymal Stem Cells
MSCs are a kind of PSC which originate from early developing mesoderm, having the potency to self-renew and differentiate into many cell types. MSCs were first found in bone marrow; however, they could also be isolated from adipose tissue 15 , umbilical cord blood 16 , peripheral blood 17 , and other tissues. In specific situations, MSCs can differentiate into adipose tissue, bone, cartilage, muscle, tendon, ligament, nerves, liver, myocardium, or endothelial cells, both in vivo and in vitro. Either cultured or cryopreserved MSCs have multidirectional differentiation potential that can be used as ideal seed cells for tissue and/or organ repair caused by aging or disease.
The most commonly applied MSCs in clinical practice are bone marrow (BM) MSCs, human umbilical cord (HUC) MSCs, and adipose-derived (AD) MSCs. BM-MSCs are frequently reported to have therapeutic effects on diverse diseases including stroke, possibly because on the one hand, they can be easily acquired from the host to avoid the transplant rejection 18 ; on the other hand, it has been demonstrated by researchers that the transplanted BM-MSCs are able to pass through the BBB without disrupting the structure 19 . They have displayed the ability to migrate to the injured area, differentiating into neurons or neuron-like cells 20 –22 , thus developing the effects of neurorestoration via secreting various neurotrophic factors 23 . There were lots of studies reporting that BM-MSCs could ameliorate neurological deficits and BBB dysfunction, as well as improve the recovery of neurological function in ICH rats 24 –30 . Yang et al. reported administration of BM-MSCs overexpressing glial-cell-derived neurotrophic factor (GDNF) displays better neuroprotective effects in a rat model of ICH 31 . Cui et al. found that BM-MSCs transplantation could attenuate neurological deficits and promote axonal regeneration through increasing GAP-43 expression in ICH 32 . Moreover, Feng et al. demonstrated the beneficial effects of BM-MSCs in a primate model 33 .
Another kind of widely applied MSC is HUC-MSCs that have been used for the treatment of various neurological diseases including ICH in animal models and patients 34 –36 . HUC-MSCs can be easily acquired in large quantities. Zhang et al. suggested the minimally invasive hematoma aspiration combined with HUC-MSC transplantation might be more effective in reducing neural damage and improving neural functions 37 .
ADMSCs are isolated from adipose tissue in recent years. Adipose tissue has several advantageous characteristics such as being accessible, abundant, and reliable for cell isolation for the purpose of regenerative applications with minimum damage to human body 38 . Studies showed that ADMSCs can stably proliferate and have low apoptotic rate in vitro, which makes them suitable for large-scale cultivation. In addition to the induced differentiation, ADMSCs can also differentiate to specific mature cells by co-culture with the mature somatic cell 39 . The most important application of ADMSCs is the repair of tissue defect and the tissue engineering. However, there is a lot of research focusing on the treatment effects of ADMSCs on diverse diseases including ICH. Chen et al. injected rat ADMSCs into the lateral cerebral ventricle of ICH rats and found them differentiating into neuron-like and astrocyte-like cells around the hematoma; simultaneously, the level of vascular endothelial growth factor (VEGF) and the score of neural function were both increased 40 . Yang et al. injected human ADMSCs into the femoral vein of ICH rat, which also showed significant functional improvement despite the divergence between two species and the different routes of drug administration 41 . Kim et al. also released the similar results. What’s more, ADMSC transplantation in the ICH model could alleviate long-term brain degeneration 42 . Above all, MSC transplantation may become a viable alternative for the treatment of ICH.
Embryonic Stem Cells
ESCs are a class of highly undifferentiated cells isolated from early embryos or the original gonads. They have the characteristics including limitless proliferation, self-renewal, and multidirectional differentiation in vitro. The ESCs were firstly isolated and reported by Evans and Kaufman in vivo 43 . Both in vitro and in vivo, ESCs can be induced to differentiate into almost all cell types including neurons and glial cells, which makes them one of the most promising SCs in treating central nervous system diseases 44,45 . Induced by all-trans retinoic acid (ATRA), some of ESCs will express neuron-specific antigens, while the others may have glial-specific antigens. Some neuron-like cells even showed enzyme activity involving acetylcholinesterase or glutamic acid decarboxylase 46 . A few studies have been established, focusing mainly on the treatment effects of ESC-derived neural cells on ICH. Nonaka et al. found that ATRA-treated ESCs injected intracerebroventricularly were transformed into neurons and glial cells around the hematoma cavity in the brain, producing neuroprotective and neurorestorative effects 47 . However, they could not enter the hematoma cavity to remedy the neuronal tissue defects. The related molecular mechanisms of cell migration and cytokine regulation are still unknown, and need further exploration. Leaving aside this incomprehension, the application of ESCs may come into conflict with the ethics, which will impose a limit on the clinical use.
Hematopoietic Stem Cells
HSCs, also known as hematopoietic PSCs (HPSCs), are a group of primitive hematopoietic cells derived from hematopoietic tissues including bone marrow, embryonic liver, peripheral blood, and umbilical cord blood. HSCs are one of the most important components of blood, serving as the initial cells of various blood cells 48 . Till et al. confirmed the existence of HSCs in the spleen of mice in 1961 for the first time 49 . HSCs appear in the yolk sac at the second week of development; after birth, the bone marrow becomes the main source of HSCs. The cell surface markers of human HSCs are CD34, together with CD133 and CD90, excluding CD38 50 . HSC transplantation plays an important role in the treatment of a variety of diseases, especially the hematological ones.
Recently, an increasing number of studies have focused on the therapeutic effect of HSCs on ICH. Sobrino et al. studied the level of circulating HSCs after ICH and found that the higher level of HSCs was associated with the better functional outcome at three months after ICH 51 . Previous studies have demonstrated that the granulocyte-colony stimulating factor (G-CSF) can mobilize HSCs to the ischemic lesion 52 and the transplantation of HSCs could improve the outcome of stroke in a rat model 53 . Based on the above research, England et al. used G-CSF to mobilize HSCs into the circulation and found they could promote functional recovery from not only ischemic stroke but also ICH 54 . They further discovered that the labeled HSCs gathered on the brain injury site to exert neurorestoration effects, which was consistent with the animal experiment 55 . With the convenient acquisition of cells in large numbers, HSCs may as well be broadly used in clinical practice.
Neural Stem Cells
NSCs are a class of SCs that present in the nervous system with the ability to split and self-renew and the potential to differentiate into neurons or glial cells 56 . The concept was first proposed by Reynolds et al. in 1992. He isolated a group of cells from the striatum of adult mouse, which showed the ability to proliferate in vitro and the potency of multiple differentiation 57 . According to the differentiation potential, NSCs can be additionally divided into neuroectodermal cells (neural tube epithelium), neuroblasts (primitive nerve cells) and neural precursors. According to the location, NSCs can be divided into neural crest SCs and CNS SCs 58 .
Some studies have demonstrated that NSC transplantation can promote functional recovery in ICH rats 59 –61 . Transplantation of genetically modified NSCs with some genes overexpressed can even enhance their function of ameliorating the ICH 62 –66 . NSCs were mainly acquired from the embryo or the fetus, which was called exogenous NSCs. For a long time, it had been generally acknowledged that no NSC existed in the adult nervous systems. However, an increasing number of studies have refuted this acknowledgement and further confirmed the fact that NSCs actually exist in the subventricular zone (SVZ) and subgranular zone of the dentate gyrus, which are then named endogenous NSCs 67 . The endogenous NSCs are silent under normal circumstances; however, they can be activated in many pathological conditions, migrate to the injury sites to contribute to neurorestoration. It was reported that endogenous NSCs were activated in the brain of experimental ICH rat and helped the neurons to achieve self-repair 68 . Yu et al. found the up-regulation of hypoxia-inducible factor-1 alpha (HIF-1α) gene could promote the proliferation, migration, and differentiation of endogenous NSCs, thus contributing to neurofunctional recovery from ICH 69 . Even physical exercise was noted to be able to enhance the survival and migration of NSCs after ICH 70 . All of the above implied the neuroplasticity of NSCs, which may be utilized to facilitate the regeneration of NSCs. The NSCs possess low immunogenicity and high histocompatibility with brain tissue, which makes them an available alternative to treating ICH.
Induced Pluripotent Stem Cells
The iPSCs were first found by two Japanese scholars in 2006. They transferred four transcription factors (Oct4, Sox2, Klf4, and c-Myc) into differentiated somatic cells using viral vectors, so that they could be reprogrammed into a cell type similar to ESCs. By being introduced with exogenous genes, somatic cells can dedifferentiate into PSCs, which are called iPSCs 71 . In September 2014, a Japanese patient with polypoidal choroidal vasculopathy was the first patient in the world to be transplanted with autologous iPSCs 72 .
However, iPSC treatment of ICH remains at the experimental stage. Three consecutive reports from Qin et al. discussed the treatment effects of iPSCs for ICH rats. They observed good therapeutic effects of iPSCs in rat ICH model 73 –75 . The iPSCs could differentiate into neuroepithelium-like/neuroepithelioid SCs and neural cells, which could also secrete neurotrophic factors. Functional improvement may be partially due to neuronal supplementation, anti-inflammation and neurotrophic factors. The iPSC technology is a major breakthrough in the field of SC research. It avoids the ethical issues and solves the problem of immune rejection in SC transplantation, which helps to make it much closer to clinical application. However, the problem of low efficiency of reprogramming still needs to be overcome before the extensive applications. The commonly used SC types for ICH treatment were summarized in Fig. 1 and Table 1.

Commonly used stem cell types for intracerebral hemorrhage (ICH) treatment.
Summary of studies concerning transplantation of commonly used stem cell types for ICH treatment.

* There are no reports on directly-administered HSCs for ICH treatment.
ICH: intracerebral hemorrhage; TSCs: totipotent stem cells; ESCs: embryonic stem cells; PSCs: pluripotent stem cells; MSCs: mesenchymal stem cells; NSCs: neural stem cells; iPSCs: induced pluripotent stem cells; HSCs: hematopoietic stem cells; BM-MSCs: bone marrow mesenchymal stem cells; HUC-MSCs: human umbilical cord-mesenchymal stem cells; ADMSCs: adipose-derived mesenchymal stem cells; IV: intravenous; IC: intracerebral; NA: not available; NSS: Neurological Severity Score; mNSS: modified NSS; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale (mRS); mBI: modified Barthel; MLPT: modified limb-placing test; VTB: video-tracking box; FM: Fugl Meyer; MRC: Medical Research Council.
The Therapeutic Strategies Involving Stem Cells
The Therapeutic Modalities
SC transplantation mainly has two methods. One is the transplantation of proper SCs directly into the body, in which the internal environment and specific signal molecules will guide these SCs towards differentiation into the desired mature cells, thus exerting the necessary functions. The other method is to isolate, cultivate, purify and amplify a certain kind of SC, and induce them to differentiate into the cells with desired functions in vitro so that these mature cells can be implanted into the human body for treatment. The appropriate combination of both the techniques may produce the best effects to patients.
The Routes of Stem Cell Administration
The routes of SC delivery vary greatly in research, and comprise the intracerebral, intraventricular, intranasal, intravenous, intrathecal, intra-subarachnoid space, intra-arterial, and intraperitoneal manners 76 .
Intraventricular/intra-subarachnoid/intrathecal transplantation provides an effective access for the SCs to pass through the BBB, making it possible for the implanted cells to migrate to the injured brain tissue via the cerebrospinal fluid (CSF) flow. However, entrance into the hematoma cavity for the SCs to fill the large deficit is extremely difficult to make. The mechanisms involved in the migration of transplanted cells are still unknown 47 . Intracerebrally delivered SCs may form cell clusters, impeding their further migration towards sites of the damaged brain, which compromises the effects and limits the application 77 . Vaquero et al. reported that the group receiving intracerebral transplantation of BM-MSCs embedded in a platelet-rich plasma scaffold afforded better functional improvement compared with the group receiving BM-MSCs in saline 78 . A cell transplantation pump can also be implanted into the brain to provide SCs continuously, though it has not been put into practice. However, these delivery routes will cause additional damages to the brain tissues of the patients.
Intravenous delivery of cells overcomes the above disadvantages and can afford the transfer of a large number of SCs. The MSCs in the treatment for ICH rats can probably migrate to the areas of the injured brain after the cells are infused into the femoral or tail vein of the rats 28,41 . In addition, G-CSF treatment can mobilize HSCs into the circulation and thus functioning through an intravenous route. However, the ability to pass through the biological barriers or the feasibility of delivering an adequate number of cells to the areas of the injured brain by this route is a controversial issue 79 . Zhang et al. reported that there was no improvement shown in the ICH rats that received intravenous injection of BM-MSCs 80 . The other type of vascular delivery is the intra-arterial route, where cells can be transferred direct to the injury site, and is more effective than the intravenous route 81 . However, occlusion and thrombosis are the biggest problems that affect the applications of these routes.
Another alternative route is via intranasal administration, which will not cause secondary injury. Sun et al. reported that intranasally administered hypoxia-preconditioned BM-MSCs could be detected in ICH brain tissues 82 as early as 1 h after intranasal administration 83 . The anatomic characteristics might play an important role in the migration of SCs from olfactory epithelium to the damaged brain. However, the exact mechanisms have not been clarified 84 . In summary, all these routes have been shown as relatively safe with no major complication. The optimal route has not been confirmed. Many aspects, such as the type and number of SCs, the characteristics of the patients, and so forth should be taken into account, and more effective and safe routes need to be investigated.
The Number of Stem Cells Used for Treatment
Similar to the route of SC delivery, the amount of SCs for the treatment of ICH is also indeterminate, which depends on multiple aspects, including the type of SCs, the route of administration, and so forth. Most studies used the million-scale of cells for the treatment of ICH, which showed acceptable results 85 . It is self-evident that insufficient transplanted cells do not exert therapeutic effects, whereas an excess of cells may cause multiple complications including occlusion, thrombosis, and even the increased risk of tumor formation. The most appropriate amount of SCs for treating ICH still needs investigation before clinical use.
The Time Window of Stem Cell Therapy
Regarding the time window of SC therapy for ICH, different studies gave controversial results. On the one hand, most studies supported early treatment. Early delivery of SCs, especially in the first week can effectively reduce the secondary injury after ICH, including inflammation and apoptosis, which contributes to the functional recovery because the secondary injury most often occurs during the first week post ICH 86 . Furthermore, Zhang et al. discovered that MSC transplantation on the third day post ICH showed a better therapeutic effect compared with that on the first, fifth and seventh day post ICH 80 . On the other hand, some studies suggested delayed treatment would be beneficial for ICH. A secondary injury may be so severe during the first week post ICH that the transplanted SCs may be injured and even die. After the acute stage of ICH, SCs can replace the damaged tissues and rebuild neuronal function. Vaquero et al. found that intracerebral transplantation of BM-MSCs embedded in a platelet-rich plasma scaffold 2 months after ICH could also improve neurological function 78 . Otero et al. had similar findings 87 . In addition, the time of transplantation may also determine the fate of cell differentiation. Delivered SCs are prone to differentiate into astrocytes during the acute stage of ICH whereas neurons do so later on 85 . Li et al. found that the therapeutic effects of NSCs depended on the time of transplantation. Animals treated at 7 and 14 days after ICH had a higher percentage of neurons differentiated from NSCs and displayed the most significant functional recovery 88 .
In fact, different cell types with different routes of delivery can affect how the SCs reach the target and work, which will lead to different conclusions, thus there is no comparability with regard to these studies. More specific experiments focusing on the time window of SC delivery for ICH are needed.
Immunosuppressive Therapy
It is still uncertain whether the immunosuppressive therapy necessitates being accompanied by SC therapy. Most studies did not use immunosuppressive therapy after SC therapy, which showed no reject reaction. Only two studies used cyclosporine after allograft SC transplantation for ICH treatment 35,47 . Generally speaking, immunosuppressive therapy is unnecessary for autologous SC transplantation. No study confirmed the necessity of immunosuppressive therapy for allograft SC transplantation, which needs further exploration.
The Neuroprotective Mechanisms of Stem Cells for Intracerebral Hemorrhage
The neuroprotective mechanisms of SCs in ICH involve tissue repair and replacement, neurotrophy, promotion of neurogenesis and angiogenesis, anti-apoptosis, immunoregulation and anti-inflammation and so forth (Fig. 2).
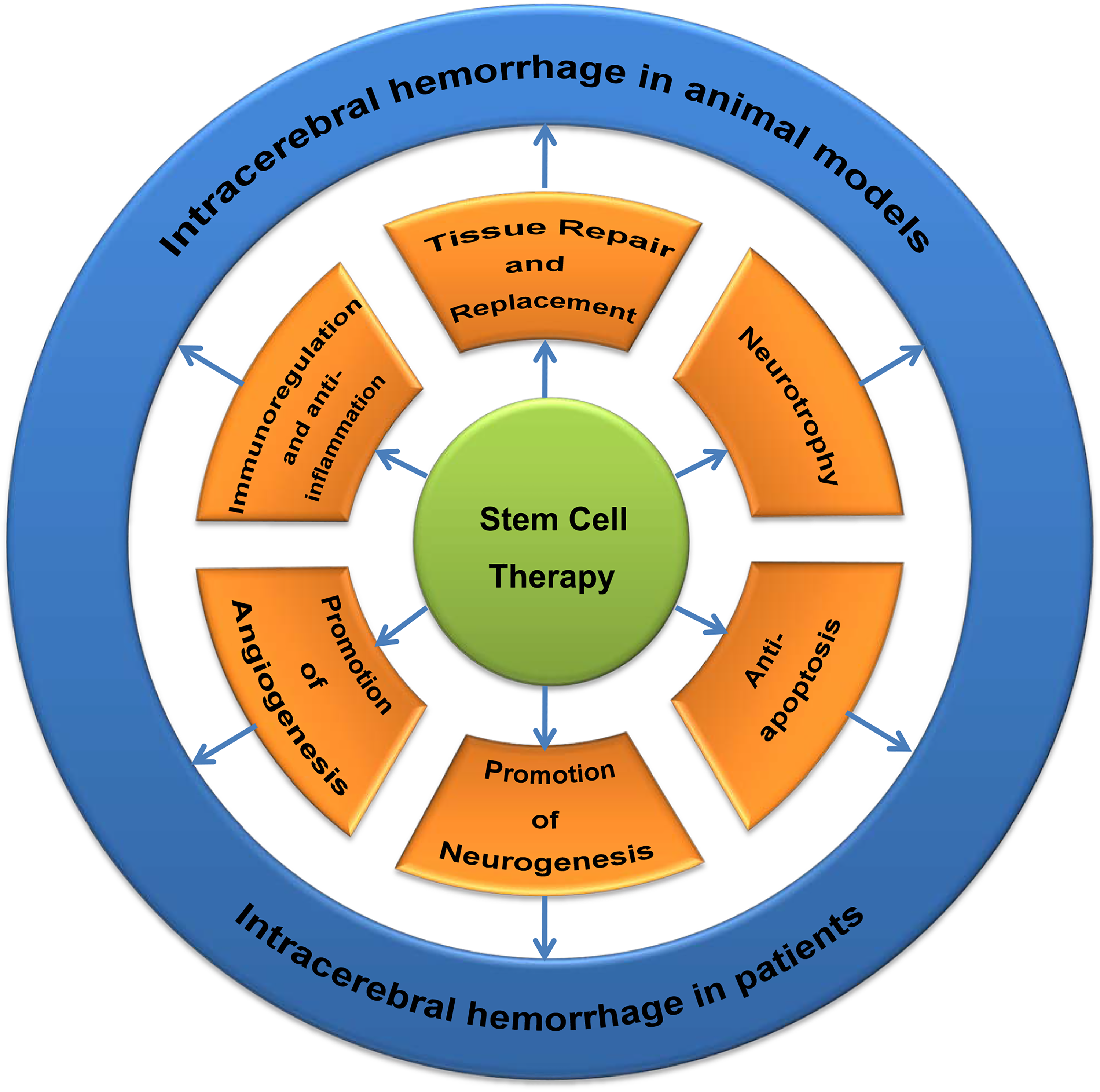
The mechanisms of stem cell therapy for intracerebral hemorrhage (ICH) both in animal models and patients.
Tissue Repair and Replacement
As mentioned above, under the influence of the internal environment and multiple nerve growth factors, many types of transplanted SCs can differentiate into the two most common functional cells after ICH, neurons and glial cells; they can migrate to the injury area to replace the damaged tissues and rebuild nerve conduction pathways. The differentiation directions are related to the time-point of transplantation. However, there are some studies finding the opposite. They found transplanted human amniotic MSCs could just survive at the perihematomal areas but did not differentiate into neurons or astrocytes 13 . These findings suggested two facts: one is that different types of SCs may have diverse capacities of differentiation after being transplanted into the ICH brain; the other is that the beneficial effects must be mediated through other mechanisms, which requires extensive investigation.
Neurotrophic Effects and Promotion of Neurogenesis
Neurogenesis plays an important role in brain injury repair and functional recovery after ICH, which would be beneficial for ICH treatment 89 . Studies showed that BM-MSC transplantation increased the amount of BrdU+, DCX+ and BrdU+/DCX+ cells in the SVZ and perihematomal areas, which suggested enhanced neurogenesis 82 . The reason may be explained by the production of neurotrophic factors. MSCs can secrete various cytokines such as brain-derived neurotrophic factor, GDNF, nerve growth factor, hepatocyte growth factor and G-CSF, which have the ability to promote neurogenesis 34,90 . Furthermore, MSCs can evoke the plasticity of damaged neurons and activate astrocytes to secrete neurotrophic factors 91 . The up-regulation of neurotrophic factors can also provide a favorable microenvironment for the survival of neuronal cells and the inhibition of cell death.
Promotion of Angiogenesis
The neurological function recovery lies on not only the neuronal cells, but also the microenvironment around them, which includes vascularity and extracellular matrix (ECM). Angiogenesis refers to the new vessel formation, which can help to repair tissues. Angiogenesis occurs during many brain insults including ICH, which becomes a critical therapeutic target 92 . Transplanted MSCs can secrete VEGF to promote vascular stabilization as well as decrease VEGF-induced BBB breakdown after ICH. Another mechanism suggested is that MSCs merge into the cerebral vasculature and significantly increase vascular density in the perihematomal areas 93 . The ECM works as the supporting component of neuronal tissues. The ECM component, including fibronectin, which is derived from MSCs, and the cell adhesion molecules such as integrin, cadherin, and selectin, can all promote neurorestoration and axonal regeneration 18 . Above all, various trophic factors and molecular components participate in the reconstruction of neurovascular unit, which promote functional improvement in combination.
Anti-Apoptosis Effects
Another widely studied mechanism of SC therapy for ICH is the anti-apoptosis. Apoptosis is involved in almost all neurological diseases including ICH, which is responsible for neurological functional recovery as well. Apoptotic cells are often detected by the techniques such as terminal deoxynucleotidyl transferase-mediated 2’-deoxyuridine 5’-triphosphate (dUTP) nick-end labeling staining (TUNEL), flow cytometry, immunohistochemistry and electron microscope. Yang et al. confirmed that TUNEL-positive cells were reduced after intravenous injection of human umbilical-tissue-derived cells, which was consistent with the improvement of motor function 94 . The changes of the expression of apoptosis-related proteins can also influence the apoptosis. Western blotting showed that the pro-apoptotic proteins such as p53, caspase-9, caspase-3, and Bax greatly decreased, while anti-apoptotic proteins such as Bcl-2 and the signaling molecules of cell survival like Akt1 and ERK-MAPK significantly increased in the ICH brains receiving the transplantation of SC compared with those without SC transplantation, which explained the functional recovery in ICH mice 64,95 .
Immunoregulative and Anti-Inflammatory Effects
At last, SCs also exert immunoregulative and anti-inflammatory effects on ICH. MSCs could develop immunomodulatory features in vitro, which was shown in animal model of cerebral ischemia 96 . In ischemic brain, MSCs reduced the numbers of Iba-1+ and ED1+ inflammatory cells 97 . In ICH animal models, transplanted neural SCs increased the regulatory T cells in the brain and peripheral blood but decreased the gamma–delta (γδ)T cells. Accordingly, the anti-inflammatory cytokines such as interleukin 4 (IL-4), IL-10, and transforming growth factor beta (TGF-β) were increased, and the pro-inflammatory cytokines such as IL-6 and interferon gamma (IFN-γ) were decreased 98 . Bao et al. transplanted Flk-1+ BM-MSCs into ICH brain, which resulted in the decreased activation of inflammatory cells in the perihematomal areas, as well as the reduction of inflammatory factors such as IL-1β, IL-2, IL-4, IL-6, and tumor necrosis factor alpha (TNF-α) 93 . Lee et al. reported early intravenous NSC injection could reduce inflammatory infiltrations in hemorrhagic stroke 99 . All of the above suggested SCs exert significant immunoregulative and anti-inflammatory effects for ICH treatment.
The Influence of the Central Nervous System Microenvironment on the Stem Cell Therapy
The microenvironment of the CNS plays an important role in the tissue repair. The neurovascular unit (NVU) refers to a complicated system that comprises neurons, glial cells, blood vessels and ECM which form the microenvironment of the CNS and is closely inter-related to the maintenance of its homeostasis 100 . The NVU functions to regulate the blood flow and the metabolism, modulate the exchange of substances across the BBB, provide trophic support and repair the injured neurons; it also contributes to the immune surveillance 100 . The brain tissue damage caused by ICH is thought to involve all cell and matrix components of the brain. The transplanted SCs seated in the microenvironment may receive multiple complex signals from the components of NVU. Damaged tissue and microenvironment can affect the survival, migration and differentiation of the transplanted SCs, thus influencing the effectiveness of SC therapy 101,102 .
ICH can cause the release of a variety of bioactive substances such as thrombin, erythrocyte lysate, excitatory amino acid, free radicals and nitric oxide due to ischemia and hypoxia, as well as the coagulation, dissolution and absorption of the hematoma 103 , which consequently leads to inactivation or even death of the endogenous or exogenous SCs. However, ischemia and hypoxia can induce both angiogenesis and the increase of related cytokines and their receptors in the brain, such as VEGF and basic fibroblast growth factor, which promote the angiogenesis 104 . New blood vessels can eliminate necrotic cell debris and toxic substances, as well as transport neurotrophic factors that induce SC proliferation, differentiation, migration and the neuronal regeneration 105 .
Glial cells are also important components of the NVU, which provide structural support for neurons, control neuronal activity through synapse formation, and may participate in the formation of local capillaries. Once activated, astrocytes can form protuberances and release a series of neurotrophic factors and growth factors to integrate the transplanted SCs and the host cells, thus exerting repairing effects 106 . Activated and proliferated microglia can help to remove the extracellular oxidative proteins and devour cell fragments, which provide a favorable microenvironment for the SCs to promote tissue repair 107 . Oligodendroglia forms myelin sheath in the CNS. Injured oligodendroglia is able to express myelin-associated inhibitor factor, which will inhibit the neuroregenerating effects of SCs 108 .
ECM is another important component of NVU, which is derived from both host cells and transplanted SCs and forms the supporting structure in the neuronal tissues. ECM in the microenvironment suck as fibronectin, integrin, cadherin and selectin can guide the proliferation, differentiation and migration of transplanted SCs and promote neurorestoration of SCs 18 . After ICH, the content of specific cytokines such as brain-derived growth factor, platelet-derived growth factor, insulin-like growth factor in the brain are significantly increased, which can improve the survival of the transplanted SCs. The combined application of these cytokines will greatly improve the therapeutic effect of SC transplantation.
Moreover, the diversity of the microenvironment could determine the multiple differentiations of the SCs 109,110 . As described before, the time of transplantation also determines the fate of cell differentiation, which may also be due to the different microenvironments.
Conversely, exogenous SCs may also alter the local environment to be more conducive to neuronal regeneration 101 . Otero-Ortega et al. reported that the exosomes derived from MSCs might work as paracrine effectors which were responsible for promoting neurovascular remodeling and functional recovery in an animal model of ICH 111 . Suda et al. found that autologous bone-marrow-derived mononuclear cells (MNCs) could protect the integrity of the NVU and alleviate inflammation and neurological deficits after ICH 112 .
The Safety of Stem Cell Therapy
As previously described, a growing number of experimental and clinical studies have suggested good curative effects of SC treatment for ICH. However, the safety and reliability of SC therapy should not be neglected. The administration of SCs may cause severe adverse reactions and/or complications, which impose the limitation to its applications. Some studies reported excess SCs and/or excessive infusion rate might cause occlusion of the blood vessels by thrombosis or embolism 113,114 . A few studies deemed that transplanted SCs could lead to some level of immune rejection and thus suggested concurrent immunosuppressive therapy 35,47,115 .
The most severe side effect of SC transplantation is the tumorigenicity and instability 75 . It had been reported that having NSCs directly implanted into adult rodents could form cell clusters in their brains 47 . Miura et al. found that murine BM-MSCs could spontaneously transform into malignant cells and form fibrosarcoma in vivo. The possible mechanisms may lie with the cumulative chromosomal abnormalities, gradual increase in telomerase activity, and the elevated expression of c-Myc 116 .
In addition, the SC transplantation can cause other side effects such as seizure, infection, hyperpyrexia and even death 75,117 . Nakamura et al. reported a rare case where the transplanted MSCs after ICH caused the formation of arteriovenous malformation a few years later 118 . The safety issues associated with SC therapy should be carefully evaluated and investigated thoroughly. Reducing the adverse effects may be more important than increasing the efficacy of SC treatment.
Conclusions and Perspective
Above all, SC therapy shows good therapeutic effects on ICH, making it a promising treatment for ICH. However, it is still in its infancy and there are some studies challenging this conclusion. Issues of concern include ethical conflicts, therapeutic effects, adverse reaction, complications, immune rejection, cell purification and the most important problem, tumorigenicity, which brings huge safety risks 47 . The detailed therapeutic strategies and mechanisms, together with the challenge of controlling its proliferation and differentiation properly are still undetermined and need further exploration. In addition, most studies have been conducted in animal models due to lack of clinical studies. A small number of clinical trials on SC transplantation in ICH patients have been conducted that give us optimistic results 36,119 –124 . However, before application in clinical practice, SC therapy for ICH needs more animal experiments to assess the abovementioned challenges. Large-scaled and multicenter clinical trials are also required. It is believed that with the continuous evolution of SC technology, SC therapy will certainly achieve a great breakthrough for the clinical treatment of ICH.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Foundation of China (No. 81601003).
