Abstract
Islet transplantation remains the only alternative to daily insulin therapy for control of type 1 diabetes (T1D) in humans. To avoid the drawbacks of intrahepatic islet transplantation, we are developing a scaffolded islet implant to transplant islets into nonhepatic sites. The implant test bed, sized for mice, consists of a limited (2-mm) thickness, large-pore polymeric sponge scaffold perforated with peripheral cavities that contain islets suspended in a collagen hydrogel. A central cavity in the scaffold holds a 2-mm diameter alginate sphere for controlled release of the angiogenic cytokine vascular endothelial growth factor (VEGF). Host microvessels readily penetrate the scaffold and collagen gel to vascularize the islets. Here, we evaluate the performance of the implant in a subcutaneous (SC) graft site. Implants incorporating 500 syngeneic islets reversed streptozotocin-induced diabetes in mice approximately 30 d after SC placement. Controlled release of a modest quantity (20 ng) of VEGF within the implant significantly reduced the time to normoglycemia compared to control implants lacking VEGF. Investigation of underlying causes for this effect revealed that inclusion of 20 ng of VEGF in the implants significantly reduced central necrosis of islets 24 h after grafting and increased implant vascularization (measured 12 d after grafting). Collectively, our results demonstrate (1) that the scaffolded islet implant design can reverse diabetes in SC sites in the absence of prevascularization of the graft site and (2) that relatively low quantities of VEGF, delivered by controlled release within the implant, can be a useful approach to limit islet stress after grafting.
Introduction
Type 1 diabetes (T1D) is a consequence of autoimmune-mediated loss of insulin-secreting pancreatic β cells that results in an inability to maintain blood glucose homeostasis 1,2 . Islet transplantation (utilizing either intact pancreata or islet isolates) remains the only alternative to daily insulin therapy for control of T1D in humans. In the one clinically approved method for transplantation of islet isolates, intraportal islet transplantation (IPIT), islets are injected into the liver via the portal vein, where they lodge in small-caliber hepatic vasculature. Although IPIT is convenient for islet delivery, islet loss can exceed 50% within the first few days of transplantation 3 –5 as a consequence of local necrosis at the graft site (caused by vascular blockage) and by an instant blood-mediated inflammatory reaction (IBMIR) elicited by direct contact of the islets with blood 3 . In addition, islet survival and function are compromised by intrahepatic processes such as detoxification of immunosuppressive agents 6 and glycogenolysis-mediated increase in intrahepatic glucose flux, which incorrectly signals islet α cells to suppress glucagon release during systemic hypoglycemia 7,8 . Collectively, islet loss due to the relatively harsh hepatic environment 9,10 , combined with incomplete control of alloimmune and autoimmune rejection of islets, limits insulin independence of IPIT recipients to less than 5 y 9 .
The problematic nature of IPIT has led to an interest in nonhepatic sites for islet transplantation, such as the kidney, omentum, bone marrow, muscle, and skin 11 –16 . The kidney subcapsular space is optimal in rodents for rapid islet functionality, low marginal islet mass, and glycemic stability; however, the kidney subcapsule in humans is inelastic, with limited space to accommodate large numbers of islets. Moreover, islets implanted in the kidney can die from exocrine contamination 17 , contributing to poor clinical outcomes 18 . Secondary complications of T1D leading to kidney disease 17,19 further limit the kidney as a viable graft site. The omentum requires a relatively low marginal islet mass 14 and has a blood supply and drainage that resembles that of native pancreatic islets 20 but is suboptimal therapeutically with respect to surgical safety, implant monitoring, and capacity for more than one transplantation. Moreover, an omental implant would be susceptible to damage by other surgical procedures performed in the abdominal cavity. Transplantation into bone marrow cavities prohibits monitoring and removal of islets and, for stem-cell therapies, provides direct access of tumorigenic cells to the circulation. Muscle provides easier surgical access compared to sites in the abdominal cavity, but vascularization is relatively poor, resulting in long time periods to achieve functionality and a need for large amounts of islets 14 . Compared to other sites, the subcutaneous (SC) site is ideal in terms of surgical safety, device monitoring and removal, and retransplantation; however, the skin is no better than muscle with respect to blood supply and support of early neovascularization 21 , making a focus on early angiogenesis essential 13,19 . Studies in animal models indicate that prevascularization (PV) of SC sites before islet transplantation improves graft performance 22 –27 , which has led to a phase I/II clinical trial in humans 28 .
In the contexts of convenience, lower treatment costs, and improved patient safety, it would be desirable to eliminate the PV step in SC islet graft protocols; however, work in this area has been limited 21,29 and further investigation is warranted. To this end, the current study evaluates the performance of our scaffolded islet implant in an SC site without PV. The disk-shaped implant, sized for mice, consists of a limited (2-mm) thickness, large-pore polymeric sponge scaffold perforated with columnar cavities that contain collagen hydrogel and suspended islets 30 . Host microvessels readily penetrate the scaffold and collagen gel to vascularize the islets. Implants loaded with 450 to 500 syngeneic islets and implanted on the gut mesentery of streptozotocin (STZ)-treated diabetic mice became vascularized within 2 wk and induced normoglycemia in experiments lasting 54 d 30 to over 200 d. Each implant contains a central cavity in the scaffold into which an alginate sphere can be placed for local, controlled release of bioactive compounds. Here, we show that release of a low level of vascular endothelial growth factor (VEGF) within implants loaded with syngeneic islets and implanted beneath the skin decreases the time to normoglycemia of STZ-treated diabetic mice. Our results suggest that local delivery of VEGF improves implant performance by not only increasing vascular density within the implant but also by reducing islet stress prior to vascularization.
Materials and Methods
Isolation of Islets
Islets were isolated from C57Bl/6 mice (12 to 24 wk of age) by digesting the pancreata with collagenase P and centrifugation of the digests through a culture medium-Histopaque®-1077 (Sigma-Aldrich, St. Louis, MO, USA) interface, as described previously 30 . Immediately after all islets were isolated, the islets were handpicked once, cultured overnight in an incubator (37 °C, 5% CO2,100% humidity), and then handpicked a second time. Yields averaged 125 to 175 islets per mouse. Average diameter of the islets was 136 µm. Total time of islet culture (i.e., from isolation to surgical implantation) was approximately 24 h.
Fabrication of Implant Scaffolds
To fabricate the scaffolds for the implants, 6-mm diameter disks were cut from 2-mm thick sheets of polyvinyl alcohol (PVA) sponge (Type CF90, 500 µm average pore size with no surfactant treatment—a generous gift from Merocel/Medtronic, Inc., USA) with biopsy punches (Sklar Instruments, West Chester, PA, USA). Subsequently, each disk was through-punched with a single central hole (cavity) of 1.5-mm diameter and 8 peripheral cavities of 1-mm diameter, using correspondingly sized biopsy punches (Sklar Instruments). The punched disks were incubated for 10 min in 40 mL of sterile distilled water repeated five times, then air-dried on sterile gauze, transferred to sterile 60-mm dishes, exposed to gamma radiation, and stored until needed for implant assembly.
Preparation of Alginate Spheres Loaded with VEGF
Controlled release of VEGF in implants was accomplished using small alginate spheres. To prepare the spheres, a stock solution of 4% Na alginate (Sigma-Aldrich, Product No. A0682) was dissolved in deionized water and filtered at 0.45 µm, using positive pressure. A stock solution of human recombinant VEGF165 (Peprotech, Rocky Hill, NJ, USA, Product No. 100-20) was prepared at 100 ng/µL in sterile, deionized water with 0.1% normal mouse serum (NMS). For spheres containing VEGF, 32 µL of alginate stock was combined with 28 µL of sterile deionized water and 4 µL of VEGF stock and pipetted in 8 µL volumes (each containing 2% alginate and 50 ng of VEGF) onto a sheet of hydrophobic Parafilm™ “M” (Pechiney Plastic Packaging, Chicago, IL, USA) that was cut into a narrow triangular shape. The Parafilm triangle was held vertically on a clamp positioned 5 cm above a 60-mm petri dish filled with a solution of 100 mM CaCl2. The alginate/VEGF droplets were pulled to the tip of the Parafilm triangle by gravity, where they fell one at a time into the CaCl2 solution and were cross-linked by the free Ca++ ions into spheres approximately 2 mm in diameter. The spheres were cross-linked for 15 min in the CaCl2 solution, then washed twice (2 min each wash) in 10 mL of a saline-HEPES-calcium (SHC) buffer consisting of 0.15 M NaCl–25 mM N-(2-hydroxyethyl)piperazine-N’-2-ethanesulfonic acid (HEPES)–2 mM CaCl2, pH 7.0, transferred to a 35-mm dish filled with SHC buffer, and maintained at room temperature until needed for implant assembly. VEGF-loaded alginate spheres prepared as described above retain approximately 20 ng of VEGF in the sphere (a proportion of VEGF is lost from the alginate during the gelation process) 30 .
Preparation of Type I Collagen Solution
To prepare type I collagen solution for the implants, 1 volume of a stock solution of rat tail native type I collagen in dilute acetic acid (Corning, Corning, NY, USA, Product No. 354236) was combined with 1/9 volume of 10 strength NaHCO3-saturated Medium 199 (Gibco/Invitrogen Waltham, MA, USA) and sufficient Dulbecco’s Modified Eagle’s Medium (DMEM) (Gibco/Invitrogen) with NMS to yield a final solution containing 2.5 mg/mL collagen and 10% NMS 31 . The collagen solution was prepared just prior to assembly of the implants and maintained on ice until needed.
Implant Assembly
To assemble the implants, the dry PVA sponge scaffolds were hydrated for 5 min in sterile DMEM, blotted on sterile gauze, and a single, freshly prepared alginate sphere was gently pressed into the 1.5-mm diameter center cavity of each scaffold. The scaffolds were then transferred to 60-mm plastic tissue culture dishes lined with UV-sterilized Parafilm M. As the scaffolds were being prepared, the islets were undergoing the second handpicking and were distributed in equal numbers into 1.5 mL centrifuge tubes containing culture medium—the number of tubes equaling the number of mice to receive islet-loaded implants in a given experiment (i.e., in a given experiment, all mice received the same number of islets). The tubes were then placed briefly in the incubator to allow the islets to settle to the bottom of the tubes. Subsequently, the following 3 operations were performed repetitively to load the islets into each implant: (1) The culture medium was aspirated from the tube and replaced with 60 µL of the type I collagen solution. (2) The collagen solution was gently agitated to uniformly disperse the islets. (3) The collagen solution, with dispersed islets, was pipetted onto the prepared scaffold (over the 1-mm diameter peripheral cavities). The PVA sponge absorbed the collagen solution, which caused the islets to enter the peripheral cavities in an evenly dispersed fashion. The islets did not penetrate into the sponge proper, as the open connections between the pores of the sponge (i.e., the pore “throats”) were too narrow to permit the islets’ passage. Subsequently, the dishes were covered with dish tops (lined with moist filter paper) and incubated for 30 min at 37 °C, 5% CO2, 100% humidity to gel the collagen. The completed implants were transferred to a 24-well tissue culture plate filled with 1 mL/well of pre-equilibrated DMEM–10% NMS–100 µg/mL penicillin–100 U/mL streptomycin and briefly maintained in a tissue culture incubator until implantation into mice.
Induction of Diabetes and SC Placement of Implants into Mice
Three days prior to implant grafting, C57Bl/6 mice were treated with a high dose (200 mg/kg) of STZ 30 . All mice receiving STZ became diabetic within 72 h and had blood glucose levels (BGLs) > 500 mg/dL at the time of surgical implantation. BGLs were measured daily and insulin was given when needed (see following section). Insulin was not administered on the day of surgery.
For the grafting procedure, mice were given buprenorphine (0.05 to 0.1 mg/kg) 15 to 30 min prior to surgery, which was performed under isoflurane. A 1.2 cm vertical incision was made in the dorsal skin near the spine, and a dermal pocket approximately 1.5 cm deep was created using blunt dissection. The scaffolded islet implant was carefully pushed into the pocket, and the skin was closed with 3 to 4 surgical staples. Meloxicam (5 mg/kg) was administered SC immediately after surgery and buprenorphine given at 8 and 18 h after surgery. Removal of implants was done in the same manner as implantation. All work with mice was done in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)-accredited facility and was approved by the Animal Care and Use Committee of the Benaroya Research Institute.
Monitoring of Mice Following Grafting of Implants
Body weights of STZ-treated mice were measured daily. BGLs were measured daily from a saphenous vein blood draw (30-gauge needle) using a Wavesense Presto glucometer (AgaMatrix, Inc., Salem, NH, USA).
To prevent excessive diabetes-mediated weight loss, all diabetic mice received sustained release Levemir® insulin (Novo Nordisk, Bagsvaerd, Denmark) injected SC as needed: 0.12 U of insulin for BGLs of 250 to 350 mg/dL, 0.23 U for BGLs of 350 to 450 mg/dL, or 0.33 U for BGLs < 450 mg/dL 30 , which lowered BGLs to near normal for at least 6 h after administration but did not sustain normal BGLs after 24 h. This response profile allowed us to determine whether a mouse was still diabetic under daily insulin therapy. The frequency of body weight and BGL measurements was reduced for mice that achieved normoglycemia (BGLs < 250 mg/dL) after islet transplantation.
For intraperitoneal glucose tolerance tests (IPGTTs), mice were fasted (water/no food) for 6 h before injection with 1 mg of glucose per gram of body weight 30 . BGLs in saphenous vein blood were measured at 0, 15, 30, 60, and 120 min after injection of glucose.
Histological Analyses
To assess islet necrosis and implant vascularization in vivo, implants were surgically removed from mice and fixed in neutral-buffered formalin, dehydrated, and embedded in paraffin. Sections of 8 µm thickness were taken en face across the entire diameter of each implant and stained with hematoxylin and eosin (H&E). Images were recorded with a Leica DM2500 bright field microscope equipped with a SPOT Insight™ digital camera (Diagnostic Instruments, Sterling Heights, MI, USA). Necrotic areas in midplane sections through islets were identified by extensive eosinophilia and the presence of sparse, pyknotic nuclei. To assess vascular density, 8 sections were evaluated for each implant. These consisted of a series of 4 sections, each separated by 50 µm, and a second series of 4 sections taken at a level starting 200 µm deeper in the implant. Collectively, these sections sampled the central 500 µm thickness of each implant. Each entire section was imaged by collecting multiple digital images with a 20× objective (each image covered a 1 mm2 field), and the images were analyzed using ImageJ (http://rsbweb.nih.gov/ij/) to quantify the percentage of vascular area, which was expressed as [total area of vascular profiles divided by the total cellularized area of each implant section] × 100% (the cellularized areas occupied the peripheral cavities, pores, and pore throats of the sponge: the acellular areas occupied by the PVA sponge proper were not included). Data were collected from 3 mice within each VEGF+ and VEGF− group.
Selected sections were stained by immunohistochemistry (IHC) using a polyclonal rabbit antibody against active caspase-3 (Abcam, Cambridge, MA, USA, Product ab2302). Bound antibody was visualized with a Bond™ Polymer Refine Detection Kit (Leica Microsystems, Wetzlar, Germany) using 3,3′-diaminobenzidine as the chromogen.
Statistical Analyses
P values were calculated with Prism® (GraphPad Software, Inc., La Jolla, CA, USA) using a 2-tailed t test. P values for Kaplan–Meier plots were calculated using a log-rank Mantel–Cox test. Intergroup comparison of IPGTT measurements was made using the Mann–Whitney test.
Results
In our previous study, we reported on the capability of scaffolded implants loaded with syngeneic islets and grafted on the mesentery to reverse STZ-induced diabetes 30 . The present study extends that work to demonstrate (1) the biofunctionality of the scaffolded islet implant when placed in a less surgically invasive (SC) site and (2) a reduction in the time interval to normoglycemia through controlled release of VEGF locally within the implant. The primary structural component of the implant is a large-pore PVA sponge scaffold that brings isolated islets (in a supportive type I collagen gel) and a controlled release component (an alginate sphere loaded with the potent angiogenic cytokine VEGF) into close proximity in a unified, compact structure that can be easily assembled, handled, and implanted in a pocket beneath the skin (Fig. 1).

Scaffolded islet implant test bed sized for mice. (a) The implant is comprised of a polyvinyl alcohol sponge scaffold (SS) with a central cavity holding an alginate sphere (asterisk) and 8 peripheral cavities (e.g., arrow) holding collagen gel and islets. (b) Diagram illustrating placement of the implant in a pocket beneath the skin.
In the present study, we incorporated 450 to 500 islets per implant, the same number as in our previous study of implants grafted on the mouse mesentery 30 , in order to compare implant performance in SC versus mesenteric graft sites. Implants containing 500 syngeneic islets reversed STZ-induced diabetes in mice approximately 30 d after placement beneath the skin (Fig. 2). In all mice where diabetes was reversed, we confirmed that the implants were responsible for the reversal by removing them after 116 d, which resulted in a rapid (24 h) return to hyperglycemia (Fig. 2).

Average blood glucose levels (BGLs, black circles) of streptozotocin-treated, diabetic mice following subcutaneous grafting of implants containing 500 islets on day 0 (large open circle; n = 5 mice). Dotted line indicates threshold of hyperglycemia (250 mg/dL). Implants were surgically removed at day 116 (large gray circle), which led to a rapid return to hyperglycemia. Open triangles indicate dose levels of exogenous insulin given to the mice.
Previously, we found that 2-mm spheres of 2% alginate loaded with 20 ng of VEGF released the VEGF in a linear fashion in vitro over a period of 2 wk under physiological conditions of ionic strength, pH, and temperature 30 —a sufficient duration of release that could, in principle, permit VEGF to have a biological effect in vivo when incorporated in scaffolded islet implants. Accordingly, STZ-treated diabetic mice received implants containing 500 islets and 20 ng of VEGF (VEGF+ group) or no VEGF (VEGF− group). Implant-mediated reversal of diabetes in the VEGF+ group was confirmed by rapid (24 to 48 h) return to hyperglycemia following implant removal (Fig. 3a).
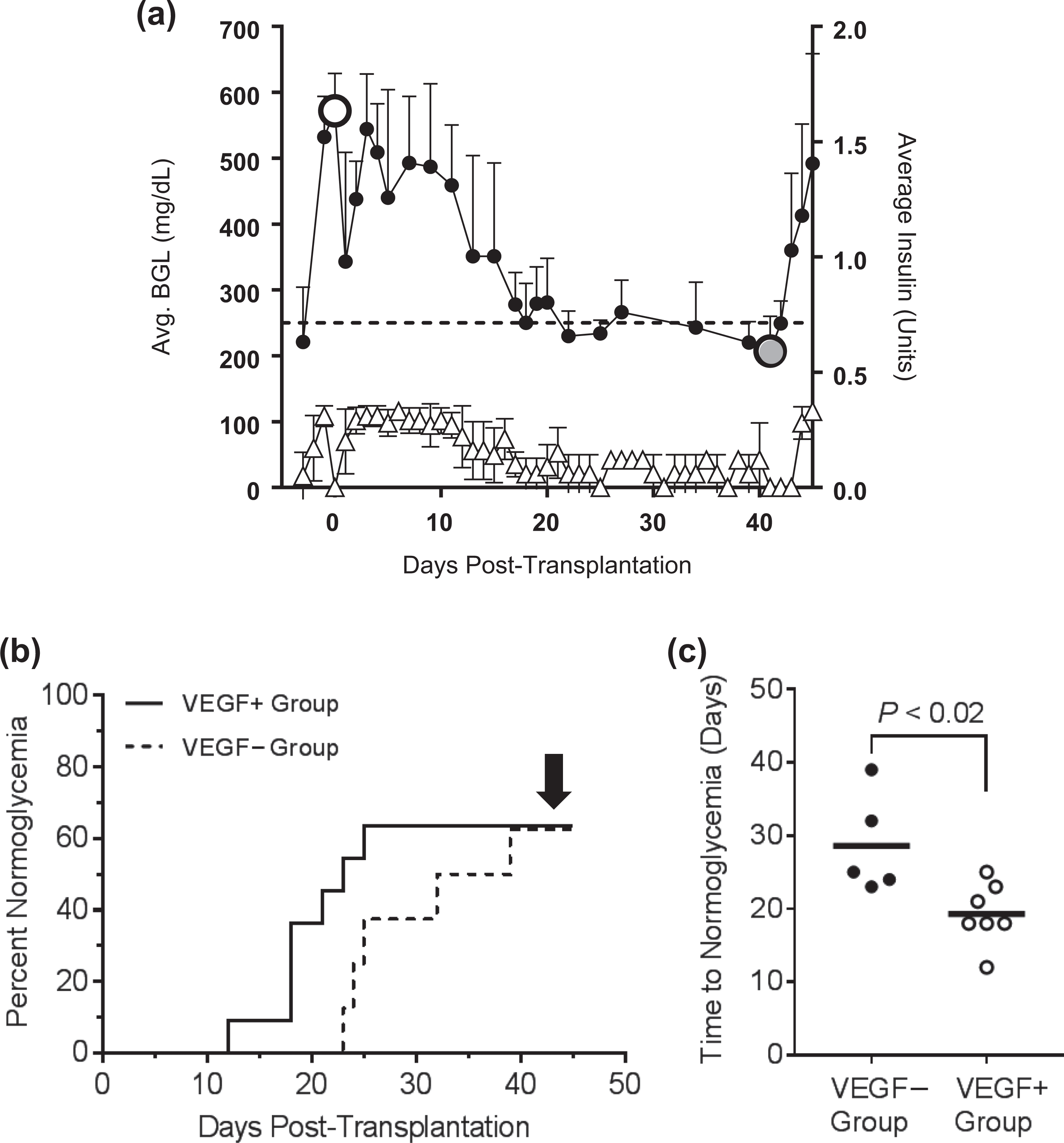
Streptozotocin-treated, diabetic mice received subcutaneous implants containing 500 islets loaded with 20 ng of vascular endothelial growth factor (VEGF; VEGF+ group) or no VEGF (VEGF− group). (a) Implant-mediated reversal of diabetes in the VEGF+ group (n = 7 mice) was confirmed by rapid (24 to 48 h) return to hyperglycemia following implant removal at day 42 (large gray circle). Blood glucose levels and exogenous insulin doses are indicated by black circles and open triangles, respectively. (b) The fraction of mice that ultimately achieved normoglycemia (arrow) was not significantly different between the VEGF+ and VEGF− groups. (c) Of the mice that achieved normoglycemia in the VEGF+ and VEGF− groups, the VEGF+ group had a shorter average time to normoglycemia than the VEGF− group.
In a Kaplan–Meier plot of the results (Fig. 3b), the fraction of mice that ultimately achieved normoglycemia was not significantly different (by log-rank Mantel–Cox test) between the 2 groups (VEGF+ group = 63.6% [7/11] vs. VEGF− group = 62.5% [5/8]).
However, of the mice that achieved normoglycemia, the VEGF+ group had a significantly reduced time to normoglycemia compared to the VEGF− group (2-tailed t test, P < 0.02). Specifically, the time to reach normoglycemia in the VEGF+ group (n = 7) averaged 19.7 ± 4.2 d versus an average of 28.6 ± 6.8 d in the VEGF− group (n = 5; Fig. 3c).
Three weeks after the implanted mice registered BGLs below 250 mg/dL, we did not observe a statistical difference in basal BGLs between the VEGF+ and VEGF− groups. The capacity of these normoglycemic mice to respond to glucose challenge was evaluated by IPGTTs (Fig. 4). At 10 to 30 min following glucose challenge, the VEGF+ and VEGF− groups had similar BGLs, which were significantly higher than healthy, nondiabetic mice that had not received STZ or implanted islets (controls). However, within 60 to 120 min of glucose challenge, BGLs of the control and transplanted groups had returned to normal and were not significantly different from one another, indicating that the scaffolded islet implants, placed beneath the skin, could effectively respond to major changes in BGL. At all time points of the IPGTTs, BGLs were not significantly different between the VEGF+ and VEGF− groups.

Intraperitoneal glucose tolerance test performed on streptozotocin-treated, diabetic mice receiving subcutaneous implants. Assays were performed on mice that had achieved normoglycemia for at least 4 wk and were off insulin (n = 4 per group). Vascular endothelial growth factor minus (VEGF−) group (black circles); VEGF+ group (open circles); healthy, nondiabetic control mice (black squares).
Previously, we found that inclusion of 20 ng of VEGF in the implants prevented transitory hypoglycemia that occurred 24 h after placement of the implants in gut mesenteric pockets 30 . As this effect occurred well before host vasculature could enter the graft site, we concluded that local release of VEGF was reducing postoperative islet stress by acting directly on the islets. Although, in the present study, we found that transitory hypoglycemia did not occur in mice that received SC implants (with or without inclusion of VEGF), it was possible that early postoperative mitigation of islet stress might contribute to the shortened interval to normoglycemia we observed in the VEGF+ group. Accordingly, implants (450 islets, ±20 ng of VEGF) were placed beneath the skin in STZ-treated diabetic mice and removed 24 h later. Levels of islet central necrosis (identified by extensive eosinophilia and the presence of sparse, pyknotic nuclei) were measured from H&E-stained sections of the implants (Fig. 5a vs. 5b). There was little evidence of apoptosis, as determined by IHC for activated caspase-3 (Fig. 5c, d). Measurements of necrosis were expressed per islet as the fraction (×100%) of necrotic area/total islet area of sections taken through the midplane of each islet. Notably, the implanted islets of the VEGF+ group had significantly less central necrosis than the islets of the VEGF− group (14.1% ± 2.8% for the VEGF+ group vs. 43.4% ± 4.3% for the VEGF− group, P < 0.0001; Fig. 5e). Average midplane areas of the islets that were sampled were not significantly different (P > 0.2) between the VEGF+ and VEGF− groups (Fig. 5f), indicating that the difference in necrotic area between the 2 groups was not influenced by differences in islet size.

Controlled release of vascular endothelial growth factor (VEGF) in subcutaneous implants inhibits early (24 h) posttransplantation islet necrosis. Streptozotocin-treated diabetic mice received implants containing 450 islets with 20 ng of VEGF (VEGF+ group) or without VEGF (VEGF− group; n = 3 mice per group). Implants were removed at 24 h, sectioned, and stained with hematoxylin and eosin to assess necrosis. (a, b) Examples of midplane sections of islets without (a) and with (b) central necrosis (b, asterisk). Of the islets that had central necrosis (c, d, asterisks), only a few (c) showed immunohistochemical staining for activated caspase-3 (brown 3,3′-diaminobenzidine stain, arrows), whereas most islets were negative (d) for this apoptotic marker. (e) Islets of the VEGF+ group had significantly less necrotic area, on average, than islets of the VEGF− group (red bars). (f) Average midplane areas (red bars) of islets of the VEGF+ and VEGF− groups were not significantly different. Scale bars in (a), (b), (d) = 50 µm; (c) = 100 µm.
VEGF-mediated reduction in islet necrosis was observed within 24 h of grafting—well before entry of host neovessels into the graft—suggesting that VEGF could mitigate islet stress by acting directly on the islets. Of course, VEGF is a potent stimulator of angiogenesis and, therefore, might also stimulate later-stage growth of host vasculature into the implant. Accordingly, we assessed whether controlled release of VEGF in SC implants could enhance vascularization by the host. As transplanted islets themselves are a potential source of both vasculature and VEGF 32 –35 , islets were omitted from the implants in these experiments. Twelve days after grafting (near the estimated 2-wk time span of VEGF release), vascularization of the implants was measured from multiple sections as a percentage of the vascular area relative to total cellularized area of each implant. We found that implants containing 20 ng of VEGF had a significantly (1.85-fold) higher vascular density than implants lacking VEGF (1.39% ± 0.25% for the VEGF+ group vs. 0.75% ± 0.14% for the VEGF− group, P < 0.04; Fig. 6).

Controlled release of vascular endothelial growth factor (VEGF) enhances growth of host vasculature into subcutaneous implants. (a) Implants lacking islets but containing 20 ng of VEGF (black bar) or no VEGF (gray bar) were removed after 12 d, sectioned, stained with hematoxylin and eosin, and assessed for degree of vascularization. The presence of VEGF significantly increased vascular growth into the implants (n = 3 mice per group). (b, c) Representative 8 µm sections of cellularized areas of implants with (b) or without (c) VEGF. Vascular profiles of various calibers (a few are indicated by arrows) are filled with red blood cells. In (b) and (c), scale bars = 50 µm.
Discussion
Major impediments to greater use of islet transplantation in the treatment of T1D are poor short-term islet survival 9,10 and failure to achieve durable immune tolerance to the graft 36,37 . Although development of better tolerogenic strategies is crucial, it is also important to consider how the “early” postgraft environment can be optimized to promote islet survival and function. Stem cell sources of islets are unavailable at present and the quantity of cadaveric islets is limited (multiple donors are often required for each recipient 38,39 ); therefore, improvements in early postgraft islet survival could increase the number of T1D patients served. A major step in this direction is to identify a suitable nonhepatic graft site; however, candidate sites have specific drawbacks, particularly the kidney subcapsular space which, despite a high degree of effectiveness in rodent models, is not translatable to human recipients. Other nonhepatic graft sites (e.g., muscle, omentum, and skin) are translationally feasible but exhibit delayed normoglycemia and require larger numbers of islets 14 .
To improve islet graft performance in nonrenal sites, 2-step PV approaches have been evaluated, in which vascular growth is initiated in the graft site by introduction of proangiogenic agents, followed by a second surgery to introduce islets. Approaches to PV (in rodents) include delivery of the proangiogenic cytokine basic fibroblast growth factor (bFGF) via PVA hydrogel (1 wk PV) 40 , agarose rods (1 wk PV) 25 , gelatin microspheres (2 wk PV) 41 , or Matrigel™ (3 wk PV) 23 or, alternatively, induction of neovessels by foreign-body responses to removable nylon catheters (4 wk PV) 26,27 or indwelling stainless steel mesh tubes 42 (40 d PV).
As mentioned earlier, a single-step graft procedure omitting PV would be desirable from the standpoint of convenience, patient safety, and lower treatment costs. To this end, the scaffolded islet implant described here serves as a useful test bed to evaluate single-step strategies to accelerate vascularization in SC islet grafts. The limited (2-mm) thickness, large pore (0.5 mm) scaffold, and supportive collagen gel facilitate vascularization of the implant, which is augmented by local release of a proangiogenic cytokine. We selected VEGF because it specifically targets endothelial cells, as opposed to bFGF, which is a mitogen to a broader spectrum of cell types, such as fibroblasts, keratinocytes, and melanocytes of the skin 43 . Islets transfected to overexpress VEGF have been shown to enhance graft vascularization 44,45 , but this approach poses problems therapeutically. Consequently, we used an acellular approach, delivering purified VEGF from a biocompatible alginate hydrogel.
We found that release of a relatively small quantity (20 ng) of VEGF within the implant significantly decreased (by 31%) the time required for diabetic mice to achieve normoglycemia relative to controls lacking VEGF. It is likely that blood vessel growth in response to VEGF played a role in this result; as 2 wk after grafting, the VEGF+ group had nearly twice the vascular density of the VEGF− group (although this difference was observed in implants without islets and may or may not be valid for implants that contain islets). Interestingly, we did not see a VEGF-mediated reduction in time to normoglycemia when implants were grafted on the gut mesentery 30 . The reason for this discrepancy is unclear but may relate to (1) the high endogenous levels of VEGF in mesenteric structures relative to other tissues 46 , which may make mesenteric vasculature less responsive to exogenous VEGF than SC vasculature and/or (2) the relatively lower baseline vascularity of dermis versus mesentery, which might result in dermis having a larger difference in vascularity between VEGF-stimulated and unstimulated conditions than mesentery would. Although addition of VEGF to the implants accelerated the time to normoglycemia, the fraction of mice that ultimately achieved normoglycemia was not significantly different between VEGF+ and VEGF− groups. This may be due to the fact that a “subtherapeutic” number of islets were implanted in order to prolong the time to normoglycemia to demonstrate the effects of VEGF with the sample size we used.
Central necrosis is a factor in islet transplantation and has been observed in vivo 47 and in vitro 48 . It is noteworthy, therefore, that we observed VEGF to markedly reduce islet central necrosis 24 h after grafting. As this effect occurs well before penetration of the graft by host blood vessels, we conclude that the exogenous VEGF acts directly on the islets, probably by stimulating endogenous endothelial cells within the islet (intra-islet endothelial cells). In this context, it has been reported that intra-islet endothelial cells of transplanted islets can contribute to revascularization of the graft, integrating with vessels of the host 32 –34 . Although it has been shown that endothelial cells of isolated mouse islets are lost after a few days of culture (e.g., approximately 8% are present after 3 d 34 ), we cultured our islets for only 24 h—at which time approximately 45% of the endothelial cells are still present in the islets 34 . Of note, islets from Tie-2 LacZ mice cultured 24 h and then transplanted under the kidney capsules of wild-type recipient mice had LacZ-expressing, donor islet endothelial cells present in the graft site at 5, 10, and even 21 d after transplantation 32 . Collectively, these observations suggest that intra-islet endothelial cells would be present in donor islets in our implants 24 h after grafting where they could respond to locally released VEGF in the implant.
It is somewhat surprising that locally released VEGF can bring about a substantial reduction in islet necrosis in such a short time (24 h). Nonetheless, this observation is consistent with our earlier finding that VEGF rapidly reduced islet stress in mesenteric islet grafts, as indicated by prevention of acute insulin release (leading to transitory hypoglycemia) 24 h after grafting 30 . Moreover, we have observed a similar rapid effect of VEGF in vitro—cultured mouse islets exposed to 10 ng/mL of VEGF for only 2 h released significantly less insulin in response to a subsequent 30 min exposure to elevated (16.6 mM) glucose than did control islets not exposed to VEGF 30 . It is known that macro- and microvascular endothelial cell responses to VEGF stimulation can be very rapid—for example, changes in barrier function in vitro are maximal on the order of 30 to 40 min after exposure to 10 ng/mL of VEGF 49 . Therefore, it is not unreasonable that elements of a rapid response to VEGF by intra-islet endothelial cells might include pro-survival signals to islet endocrine cells.
Whatever mechanism may underlie the pro-survival effects of VEGF on islets, our past and present results in mice, in combination with an earlier finding that VEGF preserved β-cell mass in rat islet cultures 50 , support the proposition that VEGF would be a useful agent to incorporate in islet transplantation devices to limit early postgraft islet stress.
In conclusion, the current study demonstrates that scaffolded islet implants grafted in SC sites in the absence of PV can reverse diabetes and that controlled release of a relatively small quantity of VEGF within the implant reduces the time to normoglycemia. Contributing factors to this latter effect appear to be (1) a VEGF-mediated reduction in islet stress/necrosis immediately after grafting and (2) increased graft vascularity. In subsequent studies, we will use scaffolded test beds that hold multiple alginate spheres to determine whether multiple cytokines, released concomitantly within a single SC implant, can interact synergistically to improve the survival and function of transplanted islets.
Footnotes
Authors’ Note
John A. Gebe and Robert B. Vernon contributed to research design; John A. Gebe, Anton Preisinger, Michel D. Gooden, Leonard A. D’Amico, and Robert B. Vernon contributed to research performance; John A. Gebe and Michel D. Gooden contributed to data analysis; John A. Gebe and Robert B. Vernon contributed to manuscript writing.
Acknowledgments
We would like to thank Dr. Rebecca L. Hull for her valuable help in optimizing the islet isolation procedures and Dr. Virginia M. Green for her editorial assistance.
Ethical Approval
Animal protocols in this study were approved by the Animal Care and Use Committee of the Benaroya Research Institute.
Statement of Human and Animal Rights
Treatment of research animals was performed in accordance with the USA Public Health Service Policy on Humane Care and Use of Laboratory Animals.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by a major grant from the Klorfine Foundation (to R.B.V.).
