Abstract
Pancreatic islet microencapsulation constitutes an attractive therapy for type 1 diabetes mellitus; however, long-term β-cell function remains a major problem. Loss of extracellular matrix interactions during islet isolation dramatically affects β-cell viability. We have previously shown beneficial effects of laminin (LN) in human islet cultures. Herein, we investigated whether LN could improve the outcome of transplantation after islet microencapsulation in Biodritin, an alginate-based material. To test LN-Biodritin stability, microcapsules were subjected to different types of in vitro stress. Focusing on biocompatibility, empty microcapsules were coincubated with the RAW 264.7 macrophage cell line for up to 24 h, and empty beads were implanted IP in mice and retrieved for analyses after 7 and 30 days. Upon culturing for 48 h, mRNA, protein levels, and caspase 3 activity were evaluated in islets microencapsulated with LN-Biodritin. Mice rendered diabetic by streptozotocin injection were transplanted with microencapsulated islets, followed by assessment of body weight, glycemia, and graft function (evaluated by OGTT). Graft efficiency was observed upon microencapsulated islet explantation. The results obtained showed that LN-Biodritin microcapsules were as stable and biocompatible as Biodritin. Modulation of mRNA and protein levels suggested protection against apoptosis and islet stress. Mice transplanted with LN-Biodritin microencapsulated islets presented a better outcome at 198 days postsurgery. Graft explantation led animals to hyperglycemia. In conclusion, LN-Biodritin constitutes a very promising biomaterial for islet transplantation.
Keywords
Introduction
Pancreatic islet transplantation is an attractive alternative treatment for type 1 diabetes mellitus. However, it has not achieved the same standards as pancreas transplantation, largely because of poor quality and low yield of the islet preparations and of inadequate immunosuppressive regimens (41). Ever since the breakthrough caused by the Edmonton protocol (55), there has been great progress in the field, from refinements of protocols to novel strategies for improved islet isolation, implantation, and recipient immunosuppression (54). Irrespective of pancreas or islet transplantation, all patients must be submitted to lifelong immunossupression, which is associated with toxic effects (65). In this context, cell microencapsulation with permeable biocompatible membranes appears a very useful tool, allowing islet transplantation in the absence of immunossupression (4), as observed in experimental (9,19, 31,53) and clinical models (5). Recently, our group has demonstrated the applicability of microencapsulated human pancreatic islets upon transplantation in mice rendered diabetic by streptozotocin injection, using Biodritin, a biocompatible material composed of sodium alginate and chondroitin sulfate (9). Despite the success obtained in this report and in the literature, in general, implanted grafts do not remain functional for long periods of time, which is related to islet stress and the quality and integrity of the microcapsules, leading to immune cell recruitment, inflammation, and transplant failure (15). It is well known that a disrupture of the cell–matrix association during islet isolation leads to a variety of structural and functional abnormalities, including apoptotic β-cell death (60). These alterations may be diminished by restoration, in culture, of the islet natural microenvironment, which includes extracellular matrix (ECM) proteins. Several reports have shown the importance of laminin (LN), the major component of the basal membrane of epithelial and endothelial cells, in enhancing islet insulin secretion in vitro (62) or promoting differentiation of mouse fetal pancreatic cells into β-cells in vitro (25). In this context, LN appears as a promising candidate to be added to the biomaterial used for microencapsulation, in an attempt to counteract part of the deleterious effects associated with the ECM loss. Herein, we investigated whether the addition of LN to Biodritin could lead to a better outcome in islet viability and, ultimately, in islet xenotransplantation.
Materials and Methods
Animals and Diabetes Induction
Male Wistar rats (6–10 weeks old) and male Balb/C mice (6–10 weeks old) were obtained from the animal care facility of the Chemistry Institute (University of São Paulo, São Paulo, Brazil), and all protocols were approved by our Institutional Ethics Committee. All animals employed in our experiments had free access to regular chow and water, unless stated otherwise. Balb/C mice were rendered diabetic by a single intraperitoneal (IP) injection of streptozotocin (250 mg/kg; Sigma-Aldrich, St. Louis, MO, USA) after a 6-h fasting period. In the subsequent days, blood was obtained by puncturing the tail vein, and glycemia was checked with a glucometer (Accu-chek Active, Roche, Basel, Switzerland). The diabetic state was confirmed after three sequential glycemia measurements above 11 mmol/L.
Biomaterials
Biodritin is composed of 80% ultrapure calcium alginate (PRONOVA UP LGV, Drammen, Norway) and 20% chondroitin sulfate (Kin Master, Rio Grande do Sul, Brazil). Alginate and chondroitin sulfate were diluted in 0.15 mol/L NaCl (Sigma-Aldrich) to achieve 1.2% and 0.325% final concentration, respectively. LN-Biodritin formulation was obtained by the addition of laminin 1 (10 μg/ml; Invitrogen, Carlsbad, CA, USA) to the Biodritin solution.
Rat Pancreatic Islets Isolation
Rats were anesthetized IP by ketamine (75 mg/kg; Vetnil, São Paulo, Brazil) and xylazine (10 mg/kg; Coopazine, Coopers, São Paulo, Brazil). A midline abdominal incision was made to expose the pancreas and inject Liberase RI (0.17 mg/ml; Roche) via the pancreatic duct. After euthanasia by exsanguination, the pancreatic tissue was surgically removed, cleaned, and incubated at 37°C (20 min) for digestion. The pellet was washed with Roswell Park Memorial Institute (RPMI)-1640 (LGC Biotecnologia, São Paulo, Brazil) supplemented with 10% fetal bovine serum (FBS; Cultilab, São Paulo, Brazil), and islet purification was achieved using a discontinuous four-phase Ficoll density gradient (densities: 1.110, 1.096, 1.069, and 1.037 g/cm3; Sigma-Aldrich) prepared in Eurocollins solution (Renograf; Claris Lifescience, Ahmedabad, India). Islets were handpicked and cultured overnight at 37°C in a 5% CO2 atmosphere.
Culture of Rat Pancreatic Islets and RAW 264.7 Macrophage Cell Line
Islets were kept in RPMI-1640 supplemented with 10% FBS and penicillin (100 U/ml; Affymetrix, Cleveland, OH, USA) at 37°C in 5% CO2 and used in the next 24 h. Prior to transplantation, cells were placed in RPMI-1640 without FBS. For longer culture periods of time, Connaught Medical Research Laboratories (CMRL) 1066 (Mediatech-Cellgro, Manassas, VA, USA) supplemented with 10% FBS and penicillin (100 U/ml) was used. RAW 264.7 cells (adult male mouse, ATCC TIB-71; ATCC, Manassas, VA, USA) were cultured in Dulbecco's modified Eagle's medium (DMEM; LGC Biotecnologia) supplemented with 100 μg/ml streptomycin (Affymetrix), 25 μg/ml ampicillin (Affymetrix), and 10% FBS, at 37°C and 5% CO2. Media were changed every 3 days.
Microencapsulation of Rat Pancreatic Islets and Production of Empty Capsules
Five thousand islets/ml were added to the Biodritin or LN-Biodritin solution, and microcapsules were produced using an air-driven droplet generator (air flow of 2.2 L/min, SP 500; JMS do Brasil, São Paulo, Brazil) and a gelatin solution composed of 20 mmol/L BaCl2 (Sigma-Aldrich) and 20 mmol/L 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES; Sigma-Aldrich). Microcapsules ranged from 700 to 1,000 μm in diameter. After droplet extrusion, the beads were retrieved and washed with 0.15 mol/L NaCl (Sigma-Aldrich). Empty capsules were produced and used in several experiments.
Microcapsule Stability Tests
The resistance of Biodritin and LN-Biodritin empty microcapsules was assessed by four different stability tests. Initially, the diameter of 100 microcapsules was obtained. Microcapsules were submitted to a “temperature challenge” in which they were heated to 37°C and 40°C for 1 h or 24 h. In a “rotational test,” microcapsules were maintained in 0.15 mol/L NaCl (Sigma-Aldrich) inside a polypropylene conical tube (BD Biosciences, San Jose, CA, USA) for 30 days, under constant agitation (150 rpm). In the “culture test,” beads were placed in RPMI-1640 with 10% FBS for 30 days. The “mixed test” involved the three situations mentioned above, where microcapsules were kept in RPMI-1640, at 37°C for 30 days, under constant agitation (150 rpm). Finally, in the “explosive test,” microcapsules were placed in pure deionized water for 15 min or 30 days. At the end of each period, another set of 100 microcapsules was evaluated for broken capsules or swelling under an optical microscope (Nikon Elipse TE300, Nikon Corporation, Tokyo, Japan), and images were acquired.
In Vitro Microcapsule Biocompatibility Study
Microcapsule biocompatibility was analyzed by their ability to activate RAW 264.7 macrophages (ATCC TIB-71), based on interleukin-1β (IL-1β) and tumor necrosis factor-α (Tnfã) mRNA modulation, in a test that closely resembled the one reported by Juste et al. (27). One hundred empty Biodritin and LN-Biodritin micro-capsules were coincubated with 106 macrophages in DMEM containing 10% FBS, 100 μg/ml streptomycin, and 25 μg/ml ampicillin for 3, 9, and 24 h. Total RNA was then extracted with TRIzol (Invitrogen), following the instructions of the manufacturer, quantified, and assessed for quality control by spectrophotometry (ND-1000 Nano-Drop Technologies, Wilmington, DE, USA) and subjected to qRT-PCR as detailed below.
Real-Time RT-PCR (qRT-PCR)
qRT-PCR Primers

Il-1β, interleukin-1β; Tnfα, tumor necrosis factor-α; Bad, B-cell CLL/lymphoma 2 (BCL2)-associated agonist of cell death; Bax, BCL2-associated X protein; Casp3, caspase 3; Bcl-2, B-cell CLL/lymphoma 2; Bcl-xL, B-cell lymphoma-extra large; Xiap, X-linked inhibitor of apoptosis, Hsp70, heat shock protein 70; Mcp1, monocyte chemoattractant protein 1; InsI, insulin I; InsII, insulin II; Hprt, hypoxanthine phosphoribosyltransferase.
In Vivo Microcapsule Biocompatibility Study
Two hundred empty Biodritin or LN-Biodritin microcapsules were implanted IP in isoflurane (Cristália, São Paulo, Brazil)-anesthetized Balb/C mice through a midline laparotomy. A peritoneal washing with 0.15 mol/L NaCl (Sigma-Aldrich) was performed 7 and 30 days after microcapsule implantation to harvest the beads, and the percentage of retrieved capsules was determined. An optical microscope (Nikon Elipse TE300) was used to measure the capsule diameter and to evaluate cell overgrowth, adopting the following criteria: no detectable cell adhesion (clean capsules), less than 25% of the capsule overgrown by cells (mild overgrowth), between 25% and 75%, and more than 75% cell adhesion.
Effect of Biomaterial Formulation on Pancreatic Islet Culture
After 48-h culture, LN-Biodritin and Biodritin microencapsulated islets were harvested and submitted to RNA extraction with TRIzol (Invitrogen), followed by cDNA production and RT-PCR analysis, using the SYBR Green Dye reagent (Applied Biosystems) to quantify the products. Primers were designed for apoptosis [B-cell CLL/lymphoma 2 (Bcl-2), BCL2-associated agonist of cell death (Bad), BCL2-associated X protein (Bax), caspase 3 (Casp3), B-cell lymphoma-extra large (Bcl-xL), and X-linked inhibitor of apoptosis (Xiap)], stress-related [heat shock protein 70 (Hsp70) and monocyte chemoattractant protein 1 (Mcp1)], and insulin genes [insulin I (InsI) and InsII]. Hprt was the evaluated housekeeping gene. The primer sequences are available in Table 1. In another set of experiments, after a 48-h treatment, the islets were subjected to protein extraction in lysis solution [10 mM Tris pH 7.5; 150 mM NaCl; 5 mM ethylenediaminetetraacetic acid (EDTA); 1 mM ethylene glycol tetraacetic acid (EGTA); 1 mM dithiothreitol (DTT); 1% nonyl phenoxypolyethoxylethanol (NP-40); 0.1% sodium dodecyl sulfate (SDS); 1% sodium deoxycholate; 1 mM sodium orthovanadate (Na3VO4); 25 mM NaF (all chemicals from Sigma Aldrich), and supplemented with phosphatase inhibitor mix (Sigma-Aldrich) with protease inhibitor mix (GE Healthcare, Little Chalfont, Buckinghamshire, UK)], and Western blot analysis was carried out using specific antibodies (Cell Signaling, Danvers, MA, USA) to the following proteins: Bcl-2, Bcl-XL, Bax, and Xiap. For Western blot analysis, 100 μg of protein was separated through electrophoresis under denaturing conditions (sodium dodecyl sulfate-polycarylamide gel electrophoresis; SDS-PAGE) with a molecular weight standard ranging from 5.7 to 170.8 kDa (BenchMark Pre-Stained Protein Ladder; Invitrogen). Protein was transferred to a nitrocellulose membrane (GE Healthcare) by a Bio Rad (Hercules, CA, USA) system. The membranes were blocked overnight with a phosphate-buffered saline (PBS) solution containing 5% bovine serum albumin (Sigma-Aldrich) and 0.1% Tween 20 (Sigma-Aldrich). Proteins were probed overnight with the chosen antibodies according to the instructions of the manufacturer, and, as a control, anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH, 1:2,000; Santa Cruz Biotechnology, Dallas, TX, USA) was used. Next, a horseradish peroxidase (HRP)-conjugated secondary antibody was incubated with the membranes (1:2,000; Vector Laboratories, Burlingame, CA, USA) for 1 h, and protein identification was revealed using the chemiluminescent system ECL Plus (GE Healthcare). Band intensity was quantified by densitometry using the ImageQuant TL Software (GE Healthcare), and the results were compared to the control GAPDH. Caspase 3 activity was also measured in the same protein extract (Caspase 3 Fluorimetric Assay; BioVision, Milpitas, CA, USA).
Transplantation of Microencapsulated Islets
A pilot study was performed by transplanting 500, 750, or 1,000 microencapsulated islets to determine the minimum amount of implanted cells that led to reversion of the diabetic state (data not shown). Since 500 islets were not enough to lower mouse glycemia, 750 islets were employed in all of our transplantation experiments.
Rat pancreatic islets microencapsulated in LN-Biodritin and Biodritin materials were transplanted IP into diabetic Balb/C mice anesthetized with isoflurane. Animals received 1 mg/kg meloxican (Vetnil) subcutaneously as a postsurgery analgesic. Mouse glycemia and weight were assessed weekly after transplantation, throughout the experiments, with a glucometer (Accu-chek Active) by drawing blood from the tail vein.
Assessment of Insulin Secretion
Before transplantation, 1,000 rat pancreatic islets were submitted to a static insulin release test upon glucose (Sigma-Aldrich) stimulation. Islets were kept in RPMI-1640 in the presence of 2.8 mM glucose for 1 h. This culture medium was then collected, and the cells were placed in RPMI-1640 containing 16 mM glucose for another 60 min. This culture medium was collected to measure insulin content by radioimmunoassay (40).
Oral glucose tolerance tests (OGTT) were performed 60 and 100 days after microencapsulated islet transplantation. After an 8-h fasting period, mice received 4 g/kg glucose (Sigma-Aldrich) through a gavage needle, and their glycemia was measured for up to 120 min, using a glucometer (Accu-chek Active). The results were compared to those of nondiabetic mice.
Graft Explantation and Animal Follow-Up
Microencapsulated islets were explanted through peritoneal washing with NaCl (Sigma-Aldrich) 0.15 mol/L in six isoflurane-anesthetized normoglycemic Balb/C mice. To assess graft efficiency, the animals were kept alive, and their glycemia was evaluated in the subsequent days.
Statistical Analysis
All statistical analyses were performed with Student's t test, Kaplan–Meyer or one-way ANOVA with Tukey's post hoc test, using the GraphPad Prism Software (San Diego, CA, USA). Differences were considered to be statistically significant when p < 0.05.
Results
LN-Biodritin Microcapsules Are as Stable and Biocompatible as Biodritin Microcapsules
Diameter Ratios of Biodritin and LN-Biodritin Microcapsules After Exposure to Deionized Ultrapure Water

Data are the mean diameter ± SD of three independent experiments with 100 analyzed capsules per experiment. No significant amount of microcapsules was found to be broken. LN, laminin.
Versus Biodritin microcapsules initial measurement, p < 0.01.
Versus LN-Biodritin microcapsules initial measurement, p < 0.05.
Versus LN-Biodritin microcapsules initial measurement, p < 0.01.
In the in vitro biocompatibility assay, Biodritin and LN-Biodritin empty microcapsules were unable to further activate the RAW 264.7 cell line during the time periods tested (Fig. 1). A less purified alginate from Sigma presented a consistent activation of macrophages only after 24 h, while LPS induced Il-1β and Tnfα mRNA upregulation as expected.
Il-1β and Tnfα mRNA expression in macrophages coincubated with a variety of substrates. (A) Interleukin-1β (Il-1β) and (B) tumor necrosis factor-α (Tnfα) mRNA levels in RAW 264.7 macrophages (M) coincubated for 3, 9, and 24 h with empty Biodritin, LN-Biodritin, and “SIGMA” microcapsules or with 2 μg/ml lipopolysaccharide (LPS) as a positive control. Data are displayed as a fold change related to gene expression in nonincubated macrophages (negative control). Results are presented as mean ± SEM of three independent experiments. *p < 0.05 with respect to the negative control baseline.
Biocompatibility in vivo (Table 3, Fig. 2) was similar for both formulations tested with no differences in the percentage of explanted microcapsules between groups, which ranged from 77% to 87%. A significant change in diameter (around 39–52%) was equally observed for both Biodritin and LN-Biodritin after implantation when compared to preimplant measurements.
Photomicrographs of microcapsules retrieved from the mouse peritoneal cavity 7 days after implantation. (A) Clean microcapsule. (B) Microcapsule presenting mild cell adhesion (<25%). (C) Microcapsule presenting a severe cell adhesion (between 25% and 75%). (D) Microcapsule presenting 100% overgrowth. Biocompatibility of Empty Biodritin and LN-Biodritin Microcapsules 7 and 30 Days After Implantation Data are the mean ± SD of three independent experiments. Versus initial Biodritin diameter measurements, p < 0.01. Versus initial LN-Biodritin diameter measurements, p < 0.01.
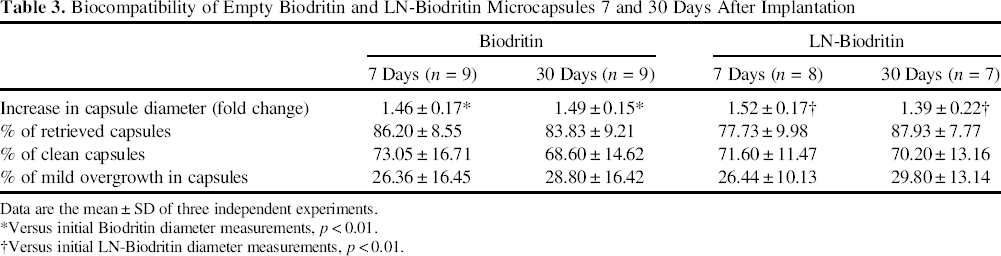
Concerning cell attachment present in the retrieved microcapsules, a mean of 68–73% of the beads from both biomaterials were “clean,” showing no overgrowth whatsoever (Fig. 2A), and most of the remaining beads showed a mild overgrowth (Table 3), accounting for less than 25% of the surface (Fig. 2B). Beads presenting greater cell attachment (Fig. 2C, D) were rarely found.
LN-Biodritin Microcapsules Induce Modulation of Gene Expression at the mRNA and Protein Levels in Pancreatic Islets
After 48 h in culture, LN-Biodritin microencapsulated islets showed an increased expression of antiapoptotic genes (Bcl-2, Bcl-XL) and a decrease in the apoptotic ones (Bax and Casp3) (Fig. 3A). The ratio of apoptotic-related genes is shown in Figure 3B. The expression of stress-related gene Mcp1 diminished, while Hsp70 was not different from the Biodritin group, despite suggesting an upregulation of mRNA levels. InsI gene expression was increased, but no difference was seen for InsII (Fig. 3A). LN-Biodritin induced an increase in Bcl-XL and Xiap antiapoptotic proteins (Fig. 3C), but divergent caspase 3 activity was not found (data not shown).
mRNA and protein modulation in rat pancreatic islets microencapsulated with LN-Biodritin and cultured for 48 h. (A) Expression of genes related to apoptosis [B-cell CLL/lymphoma 2 (Bcl-2), B-cell lymphoma-extra large (Bcl-xL), BCL2-associated agonist of cell death (Bad), BCL2-associated X protein (Bax), caspase 3, and X-linked inhibitor of apoptosis (Xiap)], cell stress [monocyte chemoattractant protein 1/chemokine C-C motif ligand 2 (Mcp1/Ccl2) and heat shock protein 70 (Hsp70)] and islet function [insulin I (InsI) and InsII]. (B) Bcl-2/Bax and Bcl-XL/Bax ratio. (C) Expression of proapoptotic (BAX) and antiapoptotic (Bcl-XL and Xiap) proteins. Results are presented as mean ± SEM of three independent experiments. *p < 0.05; **p < 0.01; ***p < 0.001 with respect to the control Biodritin (dotted baseline).
LN Induces a Better Outcome of Pancreatic Islets Transplantation
Before transplantation, the function of islets was tested a few times with insulin release being at least two times higher at the high-glucose than at the low-glucose concentration (data not shown). At the day of transplantation, mouse weight (g) and nonfasting glycemic levels (mmol/L) were not significantly different among the groups, with their mean values ± SEM being, respectively: 19.40 ± 0.50 and 24.26 ± 0.82 (sham), 21.39 ± 1.25 and 27.97 ± 2.21 (naked islets), 19.63 ± 0.46 and 25.44 ± 0.93 (Biodritin), 19.53 ± 0.74 and 25.42 ± 1.14 (LN-Biodritin). Transplantation of 750 rat microencapsulated pancreatic islets (IP) induced a similar weight gain in animals for both biomaterial formulations (Fig. 4A). However, mice transplanted with islets enclosed in LN-Biodritin were able to maintain euglycemia for longer times (Fig. 4B). After 198 days posttransplantation, we found 60% of mice that received islets microencapsulated in LN-Biodritin to be still normoglycemic, whereas in the Biodritin group, they accounted for only 11% (Fig. 4C). In all experiments, mice transplanted with naked islets failed to reach euglycemia and behaved as expected for sham mice (diabetic controls).
Graft follow-up after transplantation of 750 naked islets (n = 4) or islets encapsulated in Biodritin (n = 11) or LN-Biodritin (n = 12). Sham mice group was used as a control (n = 9). Evolution of (A) body weight and (B) glycemia for 198 days posttransplant. (C) Kaplan–Meyer graph representing the number of normoglycemic mice throughout the experiment. Points are presented as mean ± SEM. *p < 0.05, **p < 0.01 with respect to the control Biodritin. Before transplantation (day 0) mice weight and glycemia were not statistically different between groups.
In Vivo Insulin-Stimulated Secretion Is Similar in Islets Enclosed in Both Biomaterials
OGTTs performed in transplanted mice (Fig. 5A–C) showed that glucose clearance did not significantly differ between Biodritin and LN-Biodritin at 60 or 100 days posttransplantation. However, only LN-Biodritin induced a faster glucose clearance when compared to the nondiabetic group (Fig. 5A, B). In hyperglycemic animals, glucose clearance was impaired similarly to that obtained for sham mice (Fig. 5C).
Oral glucose tolerance tests (OGTTs) after encapsulated islet transplantation. OGTTs performed 60 (A) or 100 (B and C) days after transplantation of 750 pancreatic islets encapsulated in Biodritin (n = 10 for A and 4 for B) or LN-Biodritin (n = 14 for A and 9 for B). Sham (n = 9 for A and 4 for B/C) and nondiabetic mice (n = 11) were used as controls. Arrows correspond to the times in which the LN-Biodritin group is statistically different from nondiabetic mice (p < 0.05). (C) Curves from hyperglycemic mice that received islets encapsulated in Biodritin (n = 1) and LN-Biodritin (n = 2). Points are presented as mean ± SEM.
Graft Explantation Leads to Hyperglycemia in Transplanted Mice
The retrieval of Biodritin (Fig. 6A) or LN-Biodritin (Fig. 6B) microencapsulated islets from normoglycemic mice was followed by a dramatic increase in glycemia levels in subsequent days.
Mice glycemia before and after the explantation of islets encapsulated in (A) Biodritin (n = 2) and (B) LN-Biodritin (n = 4). Dotted lines represent glycemia measurements after graft retrieval. Points are presented as mean ± SEM.
Discussion
Despite the simplicity of the microencapsulation concept, progress in the field during the past few decades did not meet the high expectations. The low success rates observed in vivo are related to poor biocompatibility, limited immunoprotection, hypoxia, and loss of ECM support (14,44,61,62). The ability to reestablish the critical ECM β-cell signaling may improve current islet culture procedures, as well as the design of an artificial, immunoprotective islet carrier for transplantation. Several reports have demonstrated improved in vitro islet and individual β-cell survival and function when cultured on ECM-derived substrates (8,32,46,48,60–62). Based on that, we proposed the addition of LN to our standard biomaterial Biodritin to generate more suitable microcapsules for cell transplantation. Herein, we discuss the stability, biocompatibility and efficiency of this new formulation.
Mechanical Stability and Biocompatibility of Microcapsules
Biodritin and LN-Biodritin microcapsule stability was probed by five different methods in an attempt to mimic some of the conditions found in vitro and in vivo by microcapsules. In most of these tests, microcapsules presented adequate stability; however, when faced with an osmotic stress, we observed a rapid swelling of beads leading to a dramatic diameter increase (Table 2), but no significant number of broken capsules was found. This result was expected since a major osmotic stress is associated with a sudden hydrating pressure usually inducing capsule breakage (58). We concluded that both biomaterial formulations were equally resistant to the applied stresses.
Polymeric membranes must be highly purified to avoid immune responses in the host organism. Macrophage activation is an important step in the antigraft response, and in our tests, both biomaterials avoided further RAW 264.7 activation (Fig. 1). As expected, less purified alginate from SIGMA and LPS treatment induced an increase in the mRNA levels of the IL-1β and TNF-α cytokines, which are strongly associated with cell/graft apoptosis and necrosis (51). To add a greater challenge to this assay, in future experiments we intend to assess whether the presence of islets in LN-Biodritin beads leads to a less immunogenic profile toward cocultured macrophages than islets enclosed in Biodritin, compared to nonencapsulated islets.
Biodritin and LN-Biodritin microcapsules behaved similarly during in vivo biocompatibility assays. The efficiency of capsule retrieval observed in our tests was around 82%. In the literature, there are biocompatibility assays with recovery rates ranging from 90% to 100% (16,19,20,59), but most of them are based on the volume of the implanted microcapsules before and after bead harvesting. Because beads may swell in vivo, and their retrieval also depends on the implant site (19) and animal species/lineage (29), we decided to count the exact number of implanted/retrieved microcapsules. This was particularly important since we observed an increase in capsule diameter after transplantation (Table 3). Microcapsule swelling could lead to fewer interactions between alginate chains and a greater inefficiency in cutting off unwanted antibodies, proteins, or immune cells. The concentration of alginate used in our experiments (1.2%) might be the reason for the pronounced swelling observed in vivo, and tests with different alginate concentrations are necessary to avoid this problem in the future. Around 18% of the implanted beads could not be retrieved by peritoneal washing, and they were found immersed in peritoneal fat and in the omentum, rarely presenting a severe cell overgrowth. Despite observing that the great majority (around 70%) of microcapsules retrieved were clean (Fig. 2), we still could not achieve the percentage reported by some groups (more than 85%) after implantation of biomaterials for different time periods (16,19,50). Differences found in those reports are related to the alginate employed, presenting a high mannuronic acid content, while ours has 60% guluronic acid and the gelating ion used for capsule production (Ca2+ or Ba2+). Since high-guluronic alginates, gelated in a Ba2+ solution, generate more resistant microcapsules (67) and material purity is considerably more important than alginate guluronic/manuronic content (42), we believe other factors might be affecting our results. The impact of surgery itself (15), the implant of microcapsules in the IP site (19), and also a possible loss of the barium ion (68) are factors related to an increase in local inflammation and capsule overgrowth. In previous in vitro experiments, we found that the loss of Ba2+ from Biodritin microcapsules in culture occurs mostly in the first 24 h (data not shown), when beads are still in culture, but since we observed a 50% increase in capsule diameter, we wonder if that influenced cell attachment. Assessment of microcapsule surface properties aimed at better understanding the mechanisms by which the employed biomaterials mediate the interaction between enclosed islets and the external environment is under way. Overall, our data reveal that the LN-Biodritin material is as biocompatible as Biodritin.
Gene Expression Profile in Biodritin and LN-Biodritin Microencapsulated Islets After 48 h Culturing
Upon isolation of pancreatic islets, vascular and nervous connections are interrupted as well as the islet–ECM interaction, which leads to a diminished integrin expression and a decrease in phenotypic characteristics (13,48,49). Massive cell apoptosis follows due to various stimuli including anoikis (57), stress-activated signal transduction phosphokinases [e.g., extracellular signal-regulated kinase (ERK), c-Jun-amino-terminal kinase (JNK), and p38 mitogen-activated protein kinase (MAPK)] (45) and proinflammatory markers [i.e., nitric oxide (NO), cytokines, prostaglandins, free radicals, etc.] (3), dramatically affecting the outcome of islet transplantation. Since LN is an important basement membrane protein and binds integrins, it has been vastly studied by different groups (8,13,25,60) and by our own (32), being an excellent candidate to improve islet viability in biomaterials (56).
The Bcl-2 family of proteins constitutes a critical intracellular checkpoint of apoptosis, including pro- (e.g., Bax and Bad) and antiapoptotic (Bcl-2 and Bcl-XL) factors (10). When apoptosis is triggered, effector caspases are activated. Microencapsulated islets suffer a great mass loss (around 40%) mostly in the first month after transplantation as shown by De Vos et al. (17). To improve the transplantation success rate, efforts have been made to diminish the levels of islet proapoptotic factors and/or to increase the antiapoptotic ones (22). In our experiments (Fig. 3A, B), we observed mRNA expression of apoptosisrelated genes and found that LN-Biodritin is associated with a protective gene modulation pattern. In agreement, the differences found in Western blot experiments (Fig. 3C) also support this view. Caspase 3 activity assays presented no significant difference between groups (data not shown), but an increase in the expression of Xiap, a natural inhibitor of caspases 3, 7, and 9 was seen (Fig. 3C). Reports in the literature show that the inhibition of the intrinsic pathway of apoptosis might (12,30) or might not (66) result in cell protection since the extrinsic pathway is also important in cell death. However, both pathways end in caspase activation, and an increase of Xiap has been reported as efficient in avoiding islet apoptosis (21). It remains to be elucidated whether the presence of LN actually increases islet cell viability and function per se; however, the data presented here suggest an increase in islet survival.
The MCP1 proinflammatory chemokine is highly associated with graft failure in islet transplantation due to its prominent capacity of macrophage recruitment and activation (7,11,38,47). Blockage of MCP1 was reported to lead to the survival of islet allografts (34). The downregulation of Mcp1 gene expression, which we observed in islets microencapsulated in LN-Biodritin (Fig. 3A), has the potential to contribute to a less stressed cell culture. Despite not finding a statistical difference between mRNA levels of Hsp70 between groups (Fig. 3A), we observed a tendency toward an increase in gene expression that could be verified by performing more experiments. HSP70 is a molecular chaperone, which is activated during cellular stresses acting as a potent cytoprotector (33,52), and its local increase has been efficient in decreasing cytokineinduced islet impairment, ischemic damage, or death (23,37,39).
Finally, LN modulated insulin I and II mRNA levels (Fig. 3A): Insulin impairment is a major event in β-cell dysfunction, which is associated with the characteristic hyperglycemia in patients with diabetes (24). It has been shown that insulin I levels are markedly increased in stimulated islets when compared to insulin II (28,36). Also, insulin II may be depleted after prolonged insulin release stimuli, since its production does not increase as fast as its release (36). Therefore, our data suggest LN might be stimulating insulin synthesis and release in microencapsulated islets.
Overall, these first results showed modulation of gene expression associated with a less stressful environment in LN-Biodritin microcapsules.
Diabetes Reversal by Xenotransplantation of Microencapsulated Rat Pancreatic Islets in Immunocompetent Mice
Ever since the first attempts to treat diabetes with microencapsulated islets (35), scientists have pursued a better biomaterial, to allow greater graft survival in the absence of immunosuppression and employing a marginal islet mass (22). Based on our previous results, we hoped to reach a better outcome of transplanted islets enclosed in LN-Biodritin. We implanted 750 rat islets per immunocompetent Balb/C mice, an amount that is in the range of those used in the literature (64).
In xenotransplantation experiments (Fig. 4), the advantage of microencapsulating islets in the LN-Biodritin biomaterial became clear 198 days postsurgery, with most of the transplanted animals remaining normoglycemic. This result directly shows that the addition of LN to Biodritin can effectively protect the islets, possibly restoring interactions between this ECM protein and the microencapsulated cells, which could improve survival during the most critical period of engraftment, thereby improving the transplantation outcome. In accordance, Wang and Rosenberg reported that the expression of integrins immediately following islet isolation and during in vitro culturing was progressively diminished, which led to a decrease in β-cell function (downregulation in the proinsulin gene expression and insulin content/release) and cell apoptosis, effects that could be prevented or delayed by the exposure of islets to ECM proteins (60). Indeed, islets were reported to secrete LN during in vitro culturing and also after exposure to inflammatory cytokines (1), an effect believed to be compensatory. Beneficial effects of LNs on β-cells have been demonstrated by different groups, including our own (32), accounting for improved insulin secretion after glucose stimulation (8,26,32,61–63), increased cell viability (62,63), and replication (2). These actions shed some light into the protective pattern associated with the biomaterial containing LN and the importance of restoring islet microenvironment after isolation. Also, LN could be acting in a completely different manner, since it is an 850-kDa protein that could contribute to reduce microcapsule permeability and, therefore, limit the access of immune factors to the encapsulated islets. This would be similar to what a polylysine layer confers to some microcapsules (43). In any case, addition of LN is likely to protect the graft, but future experiments are necessary to unravel its specific mechanisms of action.
OGTT analyses (Fig. 5) allow the evaluation of islet function with greater precision and reliability than in vitro tests (6). The greater insulin release elicited by islets enclosed in LN-Biodritin, when compared to nondiabetic animals (Fig. 5A, B), could reflect an increase in insulin secretion by the transplanted β-cells associated with a delay in islet response, since the location of the graft (IP site) does not allow the same strict control as in the pancreas (18). Hyperglycemic animals presented an impairment in glucose clearance as expected (Fig. 5C). Diabetes reversal was completely dependent on graft function (Fig. 6). Unfortunately, we were not able to evaluate the explanted encapsulated grafts to assess islet structure and viability, since the majority of the beads were damaged upon capsule retrieval and cleansing.
In conclusion, our results indicate that addition of a single component (LN) to Biodritin does not interfere with microcapsule stability/biocompatibility, provides a better environment for microencapsulated rat pancreatic islets, and leads to an improved outcome in islet xenotransplantation.
Footnotes
Acknowledgments
The authors acknowledge the excellent technical support of Zizi de Mendonça, Marluce da Cunha Mantovani, Debora Cristina da Costa, Ricardo Krett de Oliveira, and Sandra Regina de Souza (Chemistry Institute, University of São Paulo). This work was supported by Fundação de Apoio à Pesquisa do Estado de São Paulo (FAPESP), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), FINEP (Financiadora de Estudos e Projetos), BNDES (Banco Nacional de Desenvolvimento Econômico e Social), and the Brazilian Ministries of Science, Technology and Innovation (MCTI) and Health (MS-DECIT). This work has been published in abstract form in the Annals of the International Pancreas and Islet Transplant Association (IPITA) and of the Brazilian Society of Biochemistry and Molecular Biology (SBBq), in 2011. Ana Lucia Campanha-Rodrigues performed most of the experiments, researched data, and wrote the manuscript. Gisella Grazioli performed some of the experiments, researched data, and contributed to discussion. Talita C. Oliveira performed some of the experiments and researched data. Ana Carolina V. Campos-Lisbôa researched data and contributed to discussion. Thiago R. dos Mares-Guia researched data and contributed to discussion. Mari Cleide Sogayar reviewed/edited the manuscript and contributed to discussion. Mari C. Sogayar is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. This work was carried out in collaboration with CellProtect Biotechnology Ltda. The authors declare no conflict of interest.
