Abstract
Cell therapy has been shown to be a key clinical therapeutic option for central nervous system diseases or damage. Standardization of clinical cell therapy procedures is an important task for professional associations devoted to cell therapy. The Chinese Branch of the International Association of Neurorestoratology (IANR) completed the first set of guidelines governing the clinical application of neurorestoration in 2011. The IANR and the Chinese Association of Neurorestoratology (CANR) collaborated to propose the current version “Clinical Cell Therapy Guidelines for Neurorestoration (IANR/CANR 2017)”. The IANR council board members and CANR committee members approved this proposal on September 1, 2016, and recommend it to clinical practitioners of cellular therapy. These guidelines include items of cell type nomenclature, cell quality control, minimal suggested cell doses, patient-informed consent, indications for undergoing cell therapy, contraindications for undergoing cell therapy, documentation of procedure and therapy, safety evaluation, efficacy evaluation, policy of repeated treatments, do not charge patients for unproven therapies, basic principles of cell therapy, and publishing responsibility.
Introduction
The Chinese Branch of the International Association of Neurorestoratology (IANR) established the first guidelines governing the clinical application of neurorestoration in 2011 (“Chinese Clinical Standard of Neurorestorative Cell Therapy”) 1 . These guidelines were revised in 2012 (“Standard Recommendation for the Application of Chinese Clinical Cell Therapy For Neurorestoration”) 2 , in 2015 (“Chinese Clinical Application Guideline of Neurorestorative Cell Therapy) 3 , and in 2016 (“Clinical Cell Therapy Guidelines for Neurorestoration, China Version 2016”) 4 . The guideline and its revisions have played a significant role in standardizing cell therapy practice in China.
Clinical cell therapies have become increasingly popular around the world. IANR and the Neurorestoratology Professional Committee of the Chinese Medical Doctor Association (Chinese Association of Neurorestoratology [CANR]) collaborated to propose “Clinical Cell Therapy Guidelines for Neurorestoration (IANR/CANR 2017)” based on the Chinese version of the guidelines and approved in principle by the IANR council board members and CANR committee members on September 1, 2016. The document was subsequently edited and literature citations were complemented. The finalized guidelines were then approved by all IANR/CANR members by email communication.
IANR/CANR hopes that these guidelines will be accepted as the applied reference standard for cell therapy of neurological diseases and damage worldwide. In particular, these guidelines may be useful to guide researchers who transplant cells into the brain and spinal cord for therapeutic research purposes. For the detailed protocol and rules of general cell therapies, researchers should first follow the regulations and policies of local governments in their respective countries. Given the rapidly advancing state of the field, the IANR/CANR will amend and update the existing guidelines to reflect the newest results demonstrated in preclinical research, translational studies, and evidence-based clinical studies.
Neurorestoratology is an emerging discipline at the intersection of clinical medicine and neuroscience. Its goal is to restore, promote, and maintain the integrity of impaired or lost neuronal functions and/or structures 5 .
The “Beijing Declaration of IANR” (agreed upon at the IANR 2015 Conference in Tehran) declared as its fundamental tenet that “functional recovery is possible after central nervous system (CNS) injury and neurodegeneration” and noted that “cell therapies may become a key clinical therapeutic option for acute, subacute and/or chronic CNS diseases or damage” 5 . More than 30 types of cells have been identified through preclinical studies as having the capacity for neurorestoration 6 –66 .
The US Food and Drug Administration (Guidance for Industry: Preclinical Assessment of Investigational Cellular and Gene Therapy Products) divided cell therapy products into stem cell–derived cell therapy products and mature/functionally differentiated cell-derived cell therapy products (http://www.fda.gov/BiologicsBloodVaccines/GuidanceComplianceRegulatoryInformation/Guidances/default.htm). Stem cell–derived cell therapy products include embryonic stem cells (ESCs), induced pluripotent stem cells, and adult (multipotent) stem cells. The last one contains neural stem cells (NSCs) and mesenchymal stem cells of different types. Mature/functionally differentiated cell-derived cell therapy products include (1) specialized functional cells such as neural progenitor or precursor cells, olfactory ensheathing cells (OECs), Schwann precursor cells, oligodendroglia precursors, neural-restricted precursors, glial-restricted precursors, neutrophils, neurons, astrocytes, myoblasts, and so on; and (2) nonspecialized functional cells such as bone marrow or umbilical cord blood mononuclear cells, umbilical cord or adipose stromal cells, and fibroblasts and lymphocytes 63,64,67 –71 . Even though there is some disagreement or controversy concerning the nomenclature of MSCs, so far the majority has accepted the MSC standard criteria made by the International Society for Cellular Therapy to identify MSCs 72,73 . While MSCs containing mesenchymal stem cells are able to differentiate into other (adipocytes, chondrocytes, osteocytes, etc.) kinds of cells when cultured in special media for differentiation, this kind of study can be referred to as mesenchymal stem cell research. In those cases, MSCs are cultured for expansion without differentiation; we may refer to this type of study as MSC research. Currently, there remains some misuse of these MSC standard criteria to identify their culturing and expanding MSCs and call them mesenchymal stem cells.
Due to concerns over tumorigenicity and difficulties in controlling differentiation of pluripotent or multipotent stem cells, stem cell–derived cell therapy products require more extensive preclinical and clinical testing. The clinical guidelines presented in this document apply more to mature/functionally differentiated cell therapy. To date, clinical trials of treatments based on those categories of cells have been carried out in over 40 countries with documented safety and functional neurological improvement for patients with CNS diseases and damage 74 –114 .
Recommended Standards for Personnel and Institutions Conducting Cell Therapies
Equipment
Institutions applying cell therapies to patients must have certified laboratory facilities and equipment that comply with the relevant national standards for ensuring cell quality control (Fig. 1).

Recommended standards for personnel and institutions. The standards mainly contain 3 parts: equipment, personal and institutional review board, and ethics committee approval.
Personnel
Clinical personnel
Physicians performing cell transplantation procedures should have documented professional training and certification required to ensure high-level competency in this field. They should have passed all certification examinations recommended by the relevant professional societies or associations.
Laboratory personnel
Directors of cell preparation laboratories should have achieved a high professional rank. Technicians involved in cell preparation should have undergone all relevant professional training and passed all certification examinations in cell preparation recommended by the relevant professional societies or associations.
Oversight personnel
Inspectors assessing cell quality should have undergone all relevant professional training and passed all certification examinations in cell preparation recommended by the relevant professional societies or associations (Fig. 1).
Institutional Review Board and Ethics Committee Approval
All clinical studies or treatment involving cell therapies and human participants must be reviewed and approved by the appropriate institutional review board or ethics committee (Fig. 1).
Provisions
These guidelines include the following provisions: cell type nomenclature, cell quality control, minimal suggested cell doses, patient informed consent, indications for undergoing cell therapy, contraindications for undergoing cell therapy, documentation of procedure and therapy, safety evaluation, efficacy evaluation, policy of repeated treatments, do not charge patients for unproven therapies, basic principles of cell therapy, and publishing responsibility (Fig. 2).

Provisions for cell therapy.
Cell Type Nomenclature
Cells are the basic unit of structure and function of organisms. A stem cell can generate itself in a process known as self-renewal and also produce the other kinds of more differentiated, committed cells with specialized functions. Progenitor or precursor cells are not able to self-renew but instead can multiply rapidly and differentiate into one or more kinds of specialized cells. Mature and functionally differentiated cells usually cannot make additional cells unless they dedifferentiate.
The nomenclature and description of cells used for therapy should specify developmental stage (e.g., embryonic, fetal, neonatal, adult, allogeneic, and autologous), tissue of origin (e.g., blood, bone marrow, umbilical cord blood, placenta, brain, spinal cord, olfactory bulb, subventricular zone, peripheral nerve, adipose, tumor, and cell line), method to isolate or expand the cells (e.g., genetically induced, expanded in culture, laser sorted, minimally manipulated, centrifugation, and type of osmotic gradient), and selection process (e.g., human leukocyte antigen [HLA]- or ABO blood group-matched, CD34+, and aldehyde dehydrogenase expressing). If nonhuman, the species must be specified. If mixtures of cells are used, the type of cells should be specified (e.g., mononuclear) together with the approximate percentages of the different types of cells present in the mixture (Fig. 3).

Provision for cell type nomenclature. The nomenclature and description of the cells used for therapy should specify species, developmental stage, tissue of origin, method to isolate or expand the cells, and selection process. If mixtures of cells are used, the type of cells should be specified.
Cell Quality Control
Quality control is essential for ensuring safety and efficacy of cell therapies. Quality control encompasses cell collection; culturing; identifying; amplifying; detecting composition and relevant cytokines, genetic or other manipulations, and passage number; exogenous factors; cell storage; assessment of biological effects (dynamics proliferation); cell transportation; preparation before clinical use; surgery and cell transplantation; or other administrative approaches. Use of animal serum such as fetal bovine serum (FBS) is discouraged. If FBS is used, it should be washed or otherwise removed before transplantation. If human sera are used, the source and quality of the serum must be documented.
Sterility of the cell preparation must be rigorously monitored. Because microbiological cultures may take days or even weeks to complete, the sterility of the cell preparation procedure must be carefully assessed and validated, particularly if the cells are transplanted before sterility data are available. If sterility testing reveals that the cells are contaminated with bacteria, fungus, or virus, sensitivity to antibiotics, antivirals, or fungicides should be determined. If the cells come from donor sources where infections are possible, donors should be screened for human immunodeficiency virus, hepatitis B virus, cytomegalovirus, or other prevalent contaminants. In addition, if tissues are stored at room temperature for any length of time, endotoxin levels should be determined. Before cells are frozen and stored, dimethylsulfoxide should be tested. The standards for sterility depend on the type and source of cells and route of administration. For example, administration of cells to skin or eyes may require criteria as stringent as for their administration to the CNS.
Before transplantation, certain minimal information must be obtained concerning the cells. These include the number of nucleated cells, the number or percentage of therapeutically relevant cells, and cell viability. Some information may be obtained after treatment as long as the preparation process is validated and contaminants can be treated with antibiotics, antivirals, or fungicides. The maximum time between preparation and transplantation of cells should be based on evidence 115 (Fig. 4).

Cell quality control. Quality control is essential for ensuring safety and efficacy of cell therapies from the preparation of cells to cell transplantation. Sterility of the cell preparation must be rigorously monitored.
Minimal Suggested Cell Doses
Cells must be used at an effective dose. Thus, the cell dosage and injection volume must be determined and controlled based on evidence of efficacy and safety. Currently, we recommend that the maximum injection volume of cell suspensions does not exceed 200 μL per injection for brain parenchyma 79,116 –118 , 25 μL per injection into spinal cord parenchyma 74,75 , 10 mL by intrathecal injection into cerebrospinal fluid 119,120 , and 10 to 100 mL by intravenous and intra-arterial routes 120 –124 . The volume or number of cells being transplanted will be reformulated if further trials show stronger evidence or indicate suitable doses in terms of patient body weight.
Current recommendations for a commonly used single dose of cells are as follows (Table 1):
Minimal Suggested Cell Doses.
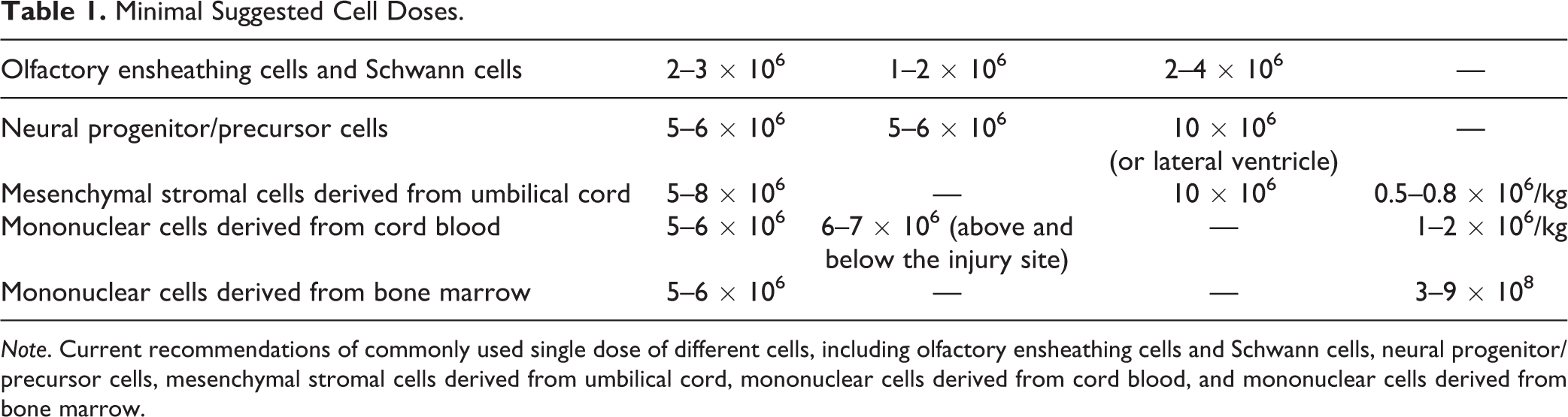
OECs and Schwann cells: 2 to 3 × 106 cells for intrathecal injection, 1 to 2 × 106 cells for intraspinal spinal cord injection, and 2 to 4 × 106 cells for brain parenchymal injection
74,75,79,118,125
. Neural progenitor/ precursor cells: 5 to 6 × 106 cells by intrathecal injection, 5 to 6 × 106 cells for spinal cord injection, and 10 × 106 by brain parenchymal injection
118,123,125
or lateral ventricle
126
. MSCs derived from umbilical cord: 0.5 to 0.8 × 106/kg body weight for intravenous infusion (the dose should be reduced by 1/3 to 1/2 for elderly and frail patients), 5 to 8 × 106 for intrathecal injection, and 10 × 106 for brain parenchymal injection
127
–134
. Mononuclear cells derived from cord blood: 1 to 2 × 106/kg body weight by intravenous infusion (the dose should be reduced by 1/3 to 1/2 for elderly and frail patients), 5 to 6 × 106 by intrathecal injection
81,87,90,94
, and a total of 6 to 7 × 106 cells injected into spinal cord above and below the injury site
135
. Mononuclear cells derived from bone marrow: 3 to 9 × 108 by intravenous infusion and 5 to 6 × 106 by intrathecal injection
84,86,121,122,124,126
.
Patient-Informed Consent
Two types of informed consent must be obtained. The first is from donors or parents who must give consent for the cells to be used to treat other patients. For example, if cells are obtained from aborted fetuses, the parent must understand what the cells will be used for and give informed consent. The second is informed consent of the recipient of the cells. Patients and their families have the right to know all the possible benefits and potential risks of matters related with the cell transplantation and procedures. Physicians should continue to learn and master the latest cell therapy–related knowledge in order to give objective answers and explanations. All participants must complete and sign a consent form that is approved by the appropriate institutional review board or ethical committee before the clinical study or cell therapy is applied.
Indications for Undergoing Cell Therapy
Animal studies suggest that cell therapies may be beneficial for a variety of neurological diseases and injury including neurotraumatic injury, neurodegeneration, ischemic/hypoxic brain injury, demyelination, sensory motor disorders, neuropathic pain, and nerve damage caused by intoxication, physical/chemical factors, immune and infectious, inflammatory, hereditary, congenital or developmental factors, and so on. However, until formal regulatory approval is obtained for use of cell therapies for specific indications, cell therapy must be administered only under the auspices of clinical trials and studies approved by appropriate institutional review boards, ethical committees, and regulatory agencies.
The relevant committees for each type of neurorestorative treatment should document the special indications for each treatment and the disease categories.
Contraindications for Undergoing Cell Therapy
Patients with poor health or dysfunction of major organs may not tolerate surgery or cell therapy procedures. The presence of infections, pressure sores, bleeding tendency, coagulation disorders, and emotional disturbance likewise may introduce undesirable complications. Patients with active neoplastic diseases, hypersensitivity, or pregnancy likewise should be excluded unless the cell therapy is specifically intended for these conditions. Clinicians should consider the likelihood that a high incidence of complications risks creating unnecessarily negative and undesirable issues for cell therapy or transplantation procedures.
Documentation of the Procedure and Therapy
The operative procedures, cell therapy, and outcomes must be rigorously documented. The documented data include anesthesia methods, cell quality and source, surgical procedures, transplantation method and site, cell preparation and dose, treatment timing at different stages, and both short- and long-term outcomes.
More preclinical and clinical treatment studies are needed to establish the best doses and therapeutic routes for different cell therapies and conditions. Randomized double-blind clinical trials are needed to establish and validate the safest and most effective clinical practices 74,75,79,118,128 –130,136 –139 . Specific sites of transplantation should be compared and assessed. For example, in local brain disorders (trauma or stroke), should the cells be injected into the lesion edge? For nonspecial or diffuse disorders (cerebral palsy, amyotrophic lateral sclerosis), what is the best site to inject cells? Should they be injected into the key points for neural network restoration 75,79 ? Some experience suggests that these key points are located anterior to the lateral ventricle and 23 to 27 mm from the midline, where the frontal corona radiata and pyramidal tract pass through and represent a nexus where numerous projection fibers, association fibers, and commissural fibers converge. Where is the best place to inject into the spinal cord? Some data suggest that cells should be injected into the spinal cord below and above the injury site, at the junction of normal and damaged tissue. For peripheral nerve disorders, should the cells be injected into the damaged site? For brain or spinal cord injury, when is the optimal time window for cell therapy, which mode of cell transplantation is better and what kind of cells should be selected at different stages? All these questions need to be answered in future clinical studies.
Safety Evaluation
Detailed records must be kept for cell therapy–related adverse events by using standardized terminology such as fever, headache, nausea, vomiting, anorexia, infection, rash, poor wound healing, dyspnea, increased/decreased blood pressure, increased/decreased heart rate, neurological deterioration, cerebrospinal fluid leakage, twitch, and so on. In the case of mortality, autopsies should be carried out to determine the cause of death and disposition of the transplanted cells.
Efficacy Evaluation
Efficacy of cell therapies should be evaluated by validated and established standards or scales currently used in the international community to assess the patients’ functions for different diseases (referred to as neurorestoratology 138 or CNS neurorestoratology 139 ). Many organizations, including IANR, regularly hold training courses for physicians to learn specific standards and assessments of patients, to test and evaluate proficiency and consistency, such as volitional control which is evaluated by a motor task involving single and multijoint movement performed repetitively at the same rate and amplitude and provide professional certification. Independent third-party examiners are recommended for clinical trials. Randomized double-blind controlled trials are preferred and required for regulatory approval. Whenever possible, detailed imaging information should be obtained before and after therapy, including magnetic resonance imaging (MRI) scans, such as functional MRI, and diffusion tensor MRI and electrophysiological examination such as transcranial magnetic stimulation (TMS), evoked potential (EP), and electromyogram (EMG) to document the presence of the cells or therapeutic effects. Patients should be scheduled for long-term follow-up examinations to determine whether beneficial effects are lasting. Functional and quality-of-life scales should be used to assess the impact of treatment effects. Natural recovery can occur over a period of time before it stops. This time may differ in different pathological conditions. It is therefore important to record the time elapsed between the pathological event and the intervention and to document other previous treatments (surgical, medical, or pharmacological) and the time prior to the cellular intervention.
Policy of Repeated Treatments
Repeated cell therapies should be based on convincing evidence of efficacy. In the absence of such evidence, patients should not be told that repeated transplants are more effective. If such evidence is not available, clinicians should do randomized trials to obtain such evidence. For example, one possible approach is to use a randomized crossover trial design, where patients are randomized to early or late repeated therapy. To rule out potential placebo effects, some trials should involve sham transplant procedures.
Do Not Charge Patients for Unproven Therapies
There should be agreement within the treatment community that if clinical trials do not show convincing evidence of benefit, practice of the therapy should be discontinued. Patients should not pay for experimental therapies. While there may be differing standards for proof of safety and efficacy, charging patients for unproven cell therapies without regulatory approval is not only illegal but may give cell therapy a bad reputation with regulatory agencies and delay acceptance of cell therapy by the mainstream clinical community. Approved medical treatments including cell therapies will not be fit for this agreement. Many countries and areas such as the United States, India, and Europe 140 , however, are adopting the practice of allowing compassionate use of therapies shown to be safe and with some evidence of efficacy to treat disorders for which there is no known effective therapy. Regulatory agencies will often negotiate with the clinician or company for the patient to pay the cost of the therapy.
Basic Principles of Cell Therapy
A number of clinical studies concerning cell therapies with a positive outcome have led to the suggestion that combination cell therapies and/or certain transplantation approaches with rehabilitation can be more effective 107,123,128 –130,136,138 –200 . IANR/CANR will evaluate such data and actively organize multicenter studies to test these practices for different cells and diseases. Randomized, double-blind, and controlled clinical studies should be carried out, if at all possible.
Publishing Responsibility
All groups that practice clinical cell therapies must promptly analyze and publish their data in peer-reviewed journals, so that other physicians can have access to the information.
Summary
Clinical cell therapies for neurological diseases and damage have shown promise for functional neurorestoration. These guidelines will help to promote the development of clinical cell therapies. Although currently multiple cell types have being used or have continued use for neurorestoration 201,202 , additional studies are necessary to determine the best type, doses, route, and timing window for administration. Further investigations in human patients will enhance our comprehension of this up-and-coming treatment.
Footnotes
Authors’ Note
This manuscript was approved by the International Association of Neurorestoratology and Chinese Association of Neurorestoratology.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paul R. Sanberg is Co-Editor-in-Chief of Cell Transplantation. Neither Paul R. Sanberg nor any of his colleagues were involved in the peer review or decision making processes for this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
