Abstract
Adipose tissue-derived stem cells (ASCs) have a self-renewing ability and can be induced to differentiate into various types of mesenchymal tissue. Because of their potential for clinical application, it has become desirable to label the cells for tracing transplanted cells and for in vivo imaging. Quantum dots (QDs) are novel inorganic probes that consist of CdSe/ZnS-core/shell semiconductor nanocrystals and have recently been explored as fluorescent probes for stem cell labeling. In this study, negatively charged QDs655 were applied for ASCs labeling, with the cationic liposome, Lipofectamine. The cytotoxicity of QDs655-Lipofectamine was assessed for ASCs. Although some cytotoxicity was observed in ASCs transfected with more than 2.0 nM of QDs655, none was observed with less than 0.8 nM. To evaluate the time dependency, the fluorescent intensity with QDs655 was observed until 24 h after transfection. The fluorescent intensity gradually increased until 2 h at the concentrations of 0.2 and 0.4 nM, while the intensity increased until 4 h at 0.8 nM. The ASCs were differentiated into both adipogenic and osteogenic cells with red fluorescence after transfection with QDs655, thus suggesting that the cells retain their potential for differentiation even after transfected with QDs655. These data suggest that QDs could be utilized for the labeling of ASCs.
Introduction
Bone marrow mesenchymal stem cells (MSCs) contain a multipotent population of cells capable of differentiating via a number of mesodermal pathways, thus representing an important source of stem cells for cell therapy (12,25). Recently, adipose tissue has been identified as an alternative source of MSCs, because adipose tissue is easier to obtain, carries relatively lower donor site morbidity, and is available in large number of stem cells at harvest (22,30). These cells have been termed adipose tissue-derived stem cells (ASCs), because they are self-renewing ability and can be induced to differentiate into various mesenchymal tissues, including chondrocytes, adipocytes, osteoblasts, myocytes, and endothelial cells (22,30). Because of their potential clinical application, it has become desirable to label the cells for tracing transplanted cells and for in vivo imaging (16,21,32).
Quantum dots (QDs) are novel inorganic probes that consist of CdSe/ZnS-core/shell semiconductor nanocrystals and have recently been explored as fluorescent probes for stem cell labeling (7,8,18,20,23). In comparison to conventional organic probes, QDs have several advantages, such as a high luminance, resistance to photobleaching, a range of excitement wavelengths from ultraviolet to red, and narrower emission bandwidths (6,10,19,28). Because of the stability, permanence, and brightness of QDs, several studies have explored in vivo labeling in live animals for optical imaging of cancer providing greater insight into tumorigenesis (2,3,26,27,29). Furthermore, these properties may allow the observation of long-lasting chemical or biological processes within or around the cell, which includes information on cell communication (31). On the other hand, it is important to understand the potential cytotoxicity of QDs. Cytotoxic mechanisms and the role of QDs in intracellular metabolism are not yet clear. A release of shell components and reactive oxygen species are believed to be a key mechanism of QDs cytotoxicity, whereby apoptosis and necrosis should play a central role in cell death mechanisms (1,9,17).
In this study, negatively charged QDs655 emitting red light of 655 nm wavelength were used with a cationic liposome, Lipofectamine. Seleverstov et al. showed that hMSCs labeled with QDs605 retained their fluorescent signal for the whole period of observation time (52 days), rapid loss of fluorescence signal was observed in QDs525 (24). They concluded that small-sized QDs were removed more rapidly than larger-sized QDs in living cells. A cationic liposome, Lipofectamine, was used for QDs655 transfection. Lipofectamine provides high transfection efficiency and significantly low cytotoxicity.
The purpose of this study was to investigate whether QDs655 could be useful for labeling ASCs without interrupting the ability for their differentiation. In addition, their toxicity and suitability for differentiated ASCs labeling were evaluated.
Materials and Methods
Animals
C57BL/6 mice were purchased from SLC Japan. Mice were housed in a controlled environment (12-h light/dark cycles at 21°C) with free access to water and a standard chow diet before killing. All conditions and handing of animals in this study were conducted with protocols approved by the Nagoya University Committee on Animal Use and Care.
Isolation and Culture of Mouse Adipose Tissue-Derived Stem Cells (ASCs)
Adipose tissue-derived stem cells (ASCs) were collected from 7-14-month-old female C57BL/6 mice. The mice were killed by cervical dislocation, adipose tissue specimens in the inguinal groove were isolated and washed extensively with Hank's balanced salt solution or phosphate-buffered saline (PBS) to remove blood cells. The isolated adipose tissue specimens were cut finely and digested with 1 ml of 1 mg/ml type I collagenase (Collagenase Type I, 274 U/mg, Koken Co., Ltd., Tokyo, Japan) at 37°C in a shaking water bath for 90 min. Adipose tissue cells were filtrated using 250-μm nylon cell strainers (BD Biosciences) and suspended in Dulbecco's modified Eagle's medium and F12 (DMEM/F12) containing 20% fetal bovine serum (FBS; Trace Scientific Ltd., Melbourne, Australia; Uin: 53141 Lot: B01249-500) and 100 U/ml penicillin/streptomycin. Cells were centrifuged at 1,200 rpm for 5 min at room temperature and ASCs obtained as the sediment. The cells were washed three times by suspension and centrifugation in the culture medium and incubated overnight at 37°C with 5% CO2 in culture medium (DMEM/F12, 20% FBS, and 100 U/ml penicillin/streptomycin). The primary cells were cultured for 4–5 days until they reached confluence and were defined as passage 0. The cells used in experiments were between passage 2 and 5.
Transfection of QDs655 with Lipofectamine Into ASCs
For cytotoxicity, ASCs (1 × 104 cells) were seeded in 96-well plates (BD Biosciences) with 100 μl of culture medium for 2 h. According to the procedures suggested in the user's guide, they were replaced with 100 μl of DMEM/F12 containing 2% FBS. Thereafter, they were transfected with QDs655 (Invitrogen; Qdot ITK Carboxyl Quantum Dots with emission at 655 nm) at the concentrations of 0, 0.08, 0.4, 0.8, 2.0, and 4.0 nM in the presence of Lipofectamine™ LTX (Invitrogen) reagent for 24 h. Viable cells were counted using Cell Counting Kit-8 (CCK-8; Dojindo Laboratories, Kumamoto, Japan). CCK-8 reagent (10 μl) was added to each well and the reaction was allowed to proceed for up to 4 h. The absorbance of the sample at 450 nm was measured against a background control, using a microplate reader.
On the other hand, to measure the proliferation rate, ASCs (1 × 103 cells) were seeded in 96-well plates with 100 μl of culture medium and then transfected with QDs655 at a concentration of 0.08, 0.4, and 0.8 nM in the presence of Lipofectamine™ LTX reagent for 4 h. Thereafter, the medium was replaced with 100 μl of culture medium. After 0, 2, and 6 days, viable cells were counted using Cell Counting Kit-8 in the same way.
Evaluation of Quantum Dots Transfection by Confocal Laser Scannning Microscopy
First, to confirm the mechanism by which QDs655-Lipofectamine penetrate the membrane of ASCs through endocytosis, the effects of the following endocytosis inhibitor was examined at a low temperature (an inhibitor of internalization through endocytosis).
Next, to evaluate intracellular penetration of QDs655-Lipofectamine, the fluorescence intensity inside ASCs was analyzed using confocal laser scanning microscopy (FV1000, Olympus). The nuclei were stained with SYTO16 (λex 488 nm, λem 532 nm) and lysosomes with lysosensor blue DND-167 (λex 443 nm, λem 503 nm). Before imaging, the cells were washed with PBS or serum-free medium.
Adipogenic Differentiation
Adipogenic differentiation was induced by culturing the cells for 3 days in DMEM (high glucose) containing 100 μM indometacin, 1 μM dexamethasone, 1 μM hydrocortisone, 10 μM insulin (Sigma, I-5500), and 10% FBS. The cells were then cultured further in DMEM (high glucose) containing 10% FBS for 2 weeks and the medium was changed every 3 days. Differentiation was confirmed by microscopic observation of intracellular lipid droplets and Oil Red O staining as an indicator of intracellular lipid accumulation. Briefly, the cells were fixed in 10% solution of formaldehyde in PBS for at least 10 min at room temperature and washed with 60% isopropanol. The cells were then stained with 2% (w/v) Oil Red O reagent for 10 min at room temperature followed by repeated washing with distilled water and destaining in 100% isopropanol for 1 min.
Osteogenic Differentiation
Osteogenic differentiation was induced by culturing the cells for 2 weeks in DMEM containing 200 μM dexamethasone, 50 μM ascorbate-2-phosphate (Wako Pure Chemical Industries Ltd., 013-12061), 10 mM α-glycerophosphate (Sigma, G-9891), and 10% FBS. The differentiation was confirmed by observation of extracellular matrix calcification using Von Kossa's method. The cells were washed twice with PBS and fixed in 10% formalin for 15 min at room temperature. They were washed and incubated with deionized water for 15 min. Thereafter, they were stained with a solution containing naphthol AS MX-PO4, N,N-dimethylformamide, Red Violet LB salt, and Tris-HCl (pH 8.3) for 45 min. Von Kossa staining was carried out with 2.5% silver nitrate solution for 30 min.
Results
Cytotoxicity of QDs655-Lipofectamine in ASCs
To investigate the cytotoxicity of QDs655-Lipofectamine in ASCs, ASCs were transfected with QDs655 at the concentration of 0, 0.08, 0.4, 0.8, 2.0, and 4.0 nM with Lipofectamine and cultured for 48 h at 37°C (Fig. 1A, B). Cytotoxicity was observed in ASCs transfected with more than 2.0 nM of QDs655, especially 4.0 nM of QDs655 induced cell death of most ASCs. However, no remarkable cytotoxicity was observed with less than 0.8 nM. These data suggest that a high concentration of QDs655 induced the death of ASCs.
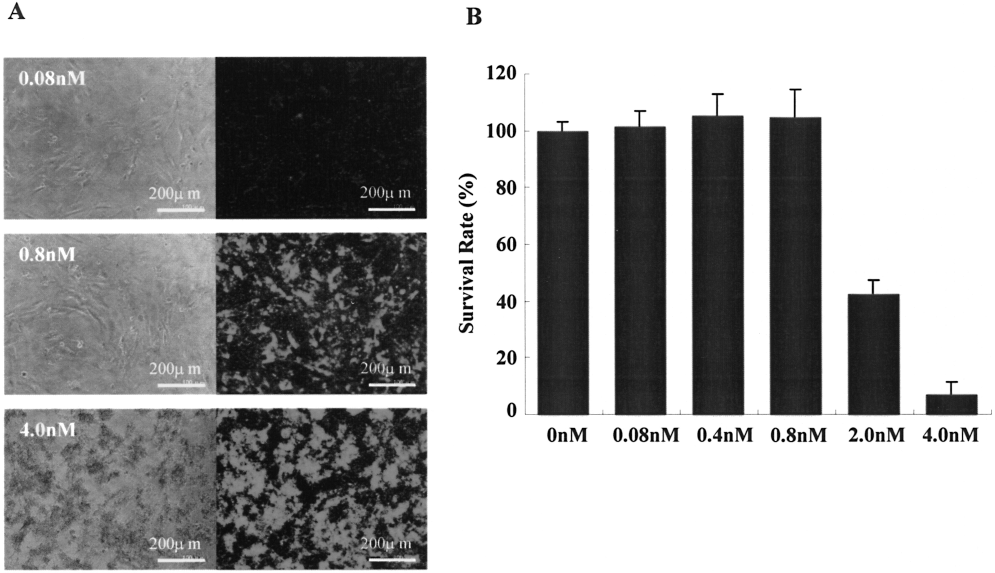
Cytotoxicity of QDs655-Lipofectamine in ASCs. (A) ASCs were transfected with QDs655 by Lipofectamine at concentrations of 0.08, 0.8, and 4.0 nM, as described in the Materials and Methods. The morphology of the cells and fluorescence of the QDs655 were observed by confocal laser scanning microscopy. The bright red granules indicated fluorescence emitted from QDs655. Representative images are shown. Almost all ASCs were dead at 4.0 nM. (B) The survival rate of ASCs transfected with QDs655-Lipofectamine is shown. The cytotoxicity was observed in ASCs transfected with more than 2.0 nM of QDs655.
Proliferation Rate of ASCs Transfected with QDs655
The proliferation rate of ASCs transfected with QDs655 was examined at concentrations of 0.08, 0.4, and 0.8 nM on 0, 3, and 7 days using Cell Counting Kit-8 (Fig. 2). There were no significant differences among three concentrations. The growth of the cells conformed to logarithmic growth phase and it was nearly equal to normal ASCs.

Proliferation rate of ASCs transfected with QDs655. The graph shows the proliferation rate of ASCs transfected with QDs655-Lipofectamine at concentrations of 0, 0.08, 0.4, and 0.8 nM. The number of cells was evaluated by Cell Counting Kit-8 after 0, 3, and 7 days.
Transfection of QDs655 with Lipofectamine by Endocytosis
The internalization of QDs655 at 4°C was compared to that at 37°C incubation. After incubation at 4°C, QDs655 was observed on the surface of ASCs (Fig. 3A, B). On the other hand, after incubation at 37°C, QDs655 was incorporated into the cytoplasm of ASCs (Fig. 3C, D). These results suggested QDs655 penetrates the cell membrane. Next, nuclei and lysosomes of ASCs were stained with SYTO16 and lysosensor blue DND-167 (pseudocolored light green), respectively (Fig. 3C–F). The signal of QDs655 was not colocalized with the nuclear fluorescence. On the other hand, it was colocalized with the lysosomal signal, suggesting QDs655 was internalized by endocytosis and then absorbed by lysosomes (15).

Intracellular distribution of QDs655 in ASCs. (A, B) ASCs were transfected with QDs655-Lipofectamine at the concentration of 0.4 nM for 24 h at 4°C and (C, D) at 37°C. Internalized QDs655 is shown by the red fluorescence. (E) The green represents nuclear staining by SYTO16. The signal of QDs655 is not colocalized with the nuclear fluorescence. (F) The blue fluorescence (pseudocolored light green) represents lysosomes using by lysosensor blue DND-167. The yellow fluorescence, which colocalized the two types of fluorescence, was detected in the lysosome. (G) The image shows a merged image of (E) and (F).
Relationship of Transfection Time and Fluorescence Intensity
The relationship between fluorescence intensity and transfection time was investigated. As shown in Figure 4, the fluorescence intensity gradually increased until 2 h at the concentrations of 0.2 and 0.4 nM, while, the intensity increased until 4 h at 0.8 nM and then the signals were observed without degradation until 24 h. These data suggest that the fluorescence intensity of QDs655 was increased within 2–4 h and thereafter was maintained until 24 h.
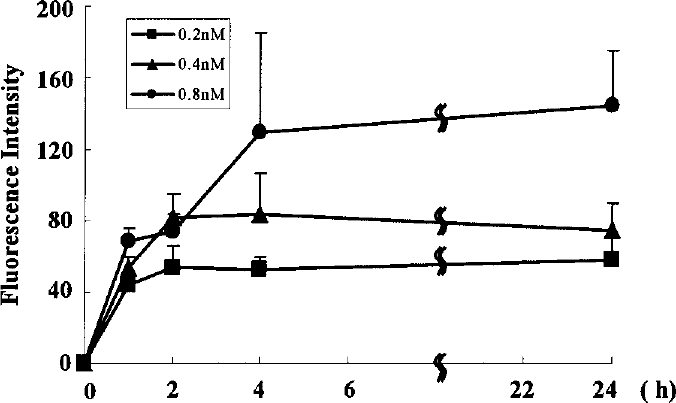
Relationship between transfection time and fluorescence intensity. ASCs were transfected with QDs655 conjugates at concentrations of 0.2, 0.4, and 0.8 nM. The fluorescent intensity in the cytoplasm was examined for 24 h. The intensities at 0.2 and 0.4 nM almost reach these peaks at 2 h. The intensity at 0.8 nM almost reaches its peak at 4 h. Peaks of their fluorescence intensity increased with increasing concentrations of QDs655.
Differentiation of ASCs Transfected with QDs655 Into Adipogenic and Osteogenic Cells
ASCs were transfected with QDs655 and cultured with adipogenic and osteogenic medium for 2 weeks. Red fluorescence of QDs655 in ASCs was observed 1 day after transfection (Fig. 5A) and remained in the ASCs for more than 2 weeks when the cells were cultured with control medium (Fig. 5B). After induction by adipogenic medium, intracellular lipid droplets in ASCs transfected with QDs655 were observed (Fig. 5C). Moreover, cuboidal shapes and tightly packed arrangements in ASCs transfected with QDs655 were observed after induction by osteogenic medium (Fig. 5D). These cells showed the red fluorescence, thus suggesting that the cells after transfection with QDs655 have the potential to differentiate into adipogenic and osteogenic cells.

Differentiation of ASCs transfected with QDs655 into adipogenic and osteogenic cells. (A) ASCs transfected with QDs655 at a concentration of 0.4 nM after 1 day. The red fluorescence from internalized QDs655 was observed by confocal laser scanning microscopy. (B) No differentiation of ASCs transfected with QDs655 at a concentration of 0.4 nM after 2 weeks was observed by confocal laser scanning microscopy. The red fluorescence from internalized QDs655 was slightly observed. (C) Adipogenic differentiation of ASCs transfected with QDs655 at the concentration of 0.4 nM was confirmed by the presence of intracellular lipid vesicles. (D) Osteogenic differentiation of ASCs transfected with QDs655 at the concentration of 0.4 nM was confirmed by extracellular matrix calcification. After both adipogenic and osteogenic differentiation, the red fluorescence from internalized QDs655 was observed.
Discussion
Because of the characteristics and high photostability exhibited by CdSe/ZnS QDs, the use of the QDs has been increased in biomedical research and extensive studies have been directed to its potential application in labeling cells. The transfection method based on Lipofectamine has successfully introduced QDs655 into ASCs without further modification of QDs655, as shown in Figures 1A and 3. These data indicate that the liposome-mediated transfection effectively introduced QDs into the ASCs and was consistent with the finding of previous reports regarding the labeling of HeLa cells with QDs (14).
The cytotoxicity of QDs is a major concern when QDs are used as markers for cells (31). Although the cytotoxicity of cadmium has long been recognized, previous studies on the CdSe/ZnS QDs have revealed that labeling of cells with these nanocrystals does not interfere with normal cell morphology and physiology or cause any negative effects on cells. In the present study, the ASCs were incubated with QDs655-Lipofectamine at concentrations of 0.08–4.0 nM. The cytotoxicity was observed in ASCs transfected with more than 2.0 nM of QDs655 and it was primarily due to the Lipofectamine. However, no remarkable cytotoxicity was observed at 0.4 nM. Besides, the labeling of ASCs with QDs655 did not affect their proliferation.
No fluorescent signal of QDs655 was observed in the nucleus, which was revealed by staining with the green fluorescence-emitting agent SYTO16. The QDs were localized in the lysosome, cytoplasm, or perinuclear regions and appeared to be restricted in a vesicle-like structure, as observed in previous papers (5).
When QDs are utilized clinically, the transfection time is important. The current study confirmed the relationship between fluorescence intensity and transfection time. The line graph indicates the intensity reached its peak between 2 and 4 h.
Moreover, the differentiation of ASCs transfected with QDs confirmed that they maintained their multilineage potential after transfection with QDs655. When ASCs transfected with QDs655 were cultured with adipogenic and osteogenic medium for 2 weeks, the cells were differentiated into adipogenic and osteogenic cells, respectively. The ASCs with QDs655 cultured with the control medium maintained their stem cell potency. Consistent with these findings, it has been reported that the CdSe/ZnS QDs labeling does not affect the differentiation of other labeled stem cells (24).
In undifferentiated ASCs transfected with QDs655, the fluorescence intensity of QDs655 was attenuated after 2 weeks of culture. On the other hand, in differentiated ASCs transfected with QDs655, the fluorescence intensity was relatively higher than in undifferentiated ASCs (Fig. 5). The differences may be due to their proliferative activity (11). Undifferentiated ASCs have a high rate of proliferation, whereas differentiated ASCs reduce their proliferation. However, even in undifferentiated ASCs 2 weeks after transfection with QDs655, obvious signal of QDs655 was observed, thus suggesting that QDs could be useful for labeling of ASCs for 2 weeks.
Recently, Hsieh et al. reported that although proliferation of hBMSCs was not affected by the internalized QDs, the induced osteogenesis, including expression of osteocyte specific marker genes, osteopontin and osteocalcin, as well as alkaline phosphatase activity, was significantly suppressed (4,13). It will be necessary to verify not only the osteogenic differentiation of ASCs transfected QDs but also other types of differentiation in further studies.
Footnotes
Acknowledgment
We thank the Kobayashi Pharmaceutical Co., Ltd and Rina Yokota (Nagoya University) for their assistance.
