Abstract
Krebs-Henseleit (KH) solution was used to fill the heart chamber of an isolated rat heart before it was immersed in perfluorocarbon (PFC), which is an inert fluid. A gas mixture (PCO2 = 150 hPa and PO2 = 850 hPa) was then aerated at a constant rate into the PFC solution, and the isolated heart was thereafter preserved for 96 h with KH solution perfused continuously at a rate of 0.1 ml/h from the aorta of the isolated heart through a cannula. After preservation, the preserved heart was heterotopically transplanted into the neck of a recipient rat and then it was resuscitated. Using this method for preserving mammalian organs, we attained reproducibility after perfusion preservation for 96 h.
Keywords
Introduction
Clinical transplantation therapy for human lungs, heart, liver, and kidneys has become widely used and has now become a common practice (9). The problem of the annually increasing shortage of organs to be provided to patients who are awaiting transplantation has worsened. Currently, the main method of preservation of human organs for transplantation is cryopreservation, which has a time limit of from 4 to 24 h for preservation (4). Many reasons have been identified as to why organs cannot be preserved for long periods, including such problems as damage to the cell membranes caused by various factors such as the low temperature of 4°C and ischemia (8,17,18). However, it is believed that current systems of supplying organs can be considerably improved if organs can somehow be preserved for long periods in a manner similar to that presently used to preserve blood, and there is therefore an urgent need to establish new and effective techniques for long-term organ preservation (4).
Until now, 4–18 h has been the limit, even in the cryopreservation and resuscitation of isolated rat, rabbit, baboon, and human hearts using the University of Wisconsin Solution (UWS), which has been clinically applied based on the simple method of immersion in a preservative solution (13,26).
Both tissues and cells in organs decompose slightly even at low temperatures, but there have been reports of trial examinations in which the preservation times were extended by supplying oxygen to the preservation solution and perfusion solution (25).
In these attempts, Kuroda et al. filled a heart chamber with UWS in order to improve immersion preservation while supplying a gas mixture (PCO2 = 50 hPa and PO2 = 950 hPa) to perfluorocarbon (PFC) solution, an inert fluid into which a large amount of O2 can be dissolved. They reported that the isolated hearts were transplanted after 24 h (100%) to 48 h (four out of five subjects) into the preservation period and they thus attained a survival time of 6 weeks (10).
Seki et al. (22) focused their study on cryptobiosis, which reduces decomposition by decreasing the amount of water in a living body to facilitate it adaptation to extreme environmental conditions such as dryness and low temperature. They found that tardigrades, which are capable of cryptobiosis, can be regenerated even after exposure to 600 MPa.
This dry cryopreservation method was then applied to living organisms for the preservation of rat organs in an experiment aimed at reducing the amount of water by using a PFC solution before resuscitation, but no significant reproducibility could be attained.
Subsequently, focusing their attention on CO2 gas, which has both anesthetic and decomposition-inhibiting mechanisms for living organisms, Yoshida et al. conducted an experiment in which they preserved hearts in an environment with a reduced amount of water and a high concentration of CO2 (PCO2 = 200 hPa), and they attained a significant reproducibility after 72 h of preservation by resuscitating the hearts (27).
In this study, we also focused our attention on tissue decomposition at low temperatures and attempted a perfusion preservation method from the perspective of eliminating waste products generated through the process. Segel et al. have compared perfusion preservation and nonperfusion preservation using rat hearts (21). The use of perfusion preservation resulted in superior levels regarding the coronary flow, cardiac output, and myocardial oxygen consumption. Linask et al. also evaluated perfusion preservation by generating heart pulsating for 6–9 days at 22°C using a perfusion method in an experiment using rat hearts as well (11).
After immersing a heart isolated from a rat in PFC solution, KH solution was continuously perfused from the aorta at a rate of 0.1 ml/h while highly compressed carbon dioxide gas (PCO2 = 150 hPa) was aerated together with oxygen, and after preservation in a refrigerator at 4°C for 96 h, the heart was then heterotopically transplanted into the neck of a recipient rat and then resuscitated, using electrocardiogram recordings to demonstrate that the transplanted heart continued to function.
Materials and Methods
For this experiment, we used inbred LEW/SsN Slc (male, 6 weeks old) Wister Lewis rats that had been raised for transplantation by Japan SLC Inc. to prevent a rejection response.
The hearts were isolated under ether anesthesia, and after removing the aorta and the pulmonary artery, each heart was then aerated with a gas mixture (PCO2 = 150 hPa and PO2 = 850 hPa), the blood was removed with a KH solution cooled to 4°C, which was then infused as a preservative solution.
We used a KH solution into which both antibiotics and warfarin were added and four times the normal levels of glucose were dissolved. Each isolated heart was aerated with a gas mixture (PCO2 = 150 hPa and PO2 = 850 hPa) in advance before being immersed in a PFC solution cooled to 4°C. The gas mixture (PCO2 = 150 hPa and PO2 = 850 hPa) was continuously aerated at a rate of 80 ml per minute to the isolated heart, which was being preserved by the KH solution continuously perfused from the aorta at a rate of 0.1 ml/h (Fig. 1).

This figure shows an experiment in which an isolated rat heart is put into a preservation container and preserved in a refrigerator at 4°C. The isolated heart was preserved in PFC (inert gas fluid). During the preservation period, it was kept aerated with PCO2 = 150 hPa and PO2 = 900 hPa and the KH solution was continuously perfused from the aorta at a rate of 0.1 ml/h.
After preservation for 96 h, the PFC solution was removed and the isolated heart was then temporarily immersed in normal saline before being heterotopically transplanted into the right neck, and it was then sutured after the pulsation became stable. The recipient rats were administered drinking water into which antibiotics had been dissolved and postobservation was conducted in the breeding room, where the pulsation of the hearts of the recipient rat and the donor rat was recorded using electrocardiograms (Fig. 2).
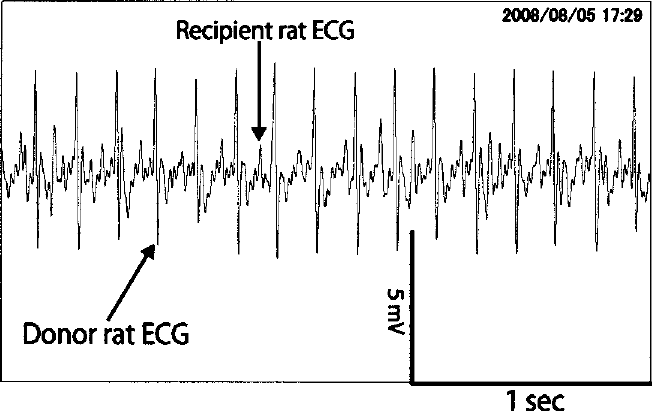
After the isolated rat heart was preserved for 96 h, a heterotopic heart transplantation to the neck was thus resuscitated, and then electrocardiograms of the donor heart and recipient heart were recorded.
Results
For each preserved heart, which was resuscitated after being preserved for 96 h in PFC solution with the KH solution perfused, we inosculated the common carotid artery with the aorta and the external jugular vein with the pulmonary artery from end to end in order to perform five heterotopic heart transplantations. As a result, we obtained four cases in which both the ventricle and the atrium were successfully pulsating and an electrocardiogram could be recorded.
In addition, as a control experiment, the preservation and resuscitation experiment was conducted under the same conditions, except for the fact that one group of samples was not perfused with KH solution, while another was perfused with UW solution. The number of cases preserved for 96 h without being perfused was zero out of five cases. In addition, the number of cases preserved for 96 h with perfusions of UW solution before being resuscitated was zero out of five cases. There were no cases of resuscitation in either group (Table 1, Fig. 3).
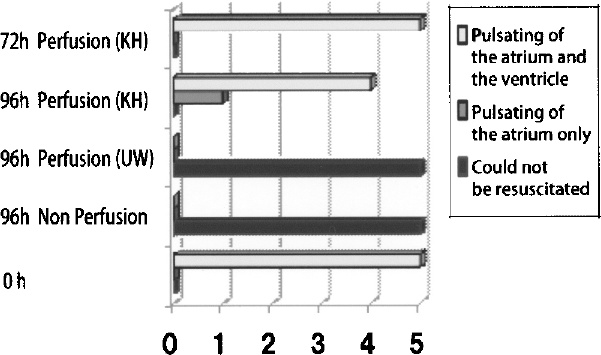
The horizontal axis shows the number of individuals exhibiting the pulsation of the atrium and the ventricle, the number of those with pulsation only in the atrium, and the number of those that could not be resuscitated. The longitudinal axis shows the number of individuals used for each preservation method. Five out of five (100%) of those preserved for 72 h with the perfusion of the KH solution exhibited pulsating of the atrium and the ventricle. Four out of five (80%) of those preserved for 96 h with the perfusion of the KH solution exhibited the pulsation of the atrium and the ventricle, and one out of five (20%) exhibited the pulsation of the atrium alone. None (0/5; 0%) of those preserved for 96 h with the perfusion of UW solution could be resuscitated. In addition, none (0/5, 0%) of those preserved for 96 h without being perfused could be resuscitated.
Heart Preservation and Resuscitation Rate

Hearts preserved by perfusion of the KH solution are indicated as 1A and 1B, while those perfused with UW solution are indicated as 2, and hearts preserved without being perfused are indicated as 3. In addition, hearts that demonstrated no preservation time are indicated as 4.
Discussion
Kuroda et al. reported that they filled the heart chamber of an isolated heart rat with UW solution and immersed the entire isolated heart in PFC solution, and after preserving the isolated heart for up to 48 h by supplying oxygen through the PFC solution, it was resuscitated to be heterotopically transplanted and thereafter survived for 6 weeks (4/5, 80%) (10).
Modifying the above described method, we immersed an isolated rat heart in PFC solution and preserved the isolated heart while aerating it with carbon dioxide gas PCO2 = 150 hPa in addition to oxygen.
Moreover, we applied a perfusion preservation method, which is known to produce better preservation than simple immersion preservation alone, and as a result, the heart that had been preserved for 96 h could be heterotopically transplanted into the right neck of a recipient rat to be resuscitated.
Changes other than those made to CO2 concentration included the use of KH solution in the place of UW solution in this experiment. Using UW solution, we attempted preservation for longer than 96 h but could not detect any pulsation upon resuscitation. On the other hand, with the KH solution, we found a significant degree of successful resuscitation. We therefore found the KH solution to be a more effective preservation solution than the UW solution. While we will continue to seek the best preservation solution, it appears that the high viscosity of UW solution makes it unsuitable for perfusion preservation.
In addition, Kuroda et al. kept an isolated rat heart submerged with a net to prevent it from floating on the surface of the PFC solution, but we believe that this method may possibly cause partial tissue damage (10). We therefore severed the blood vessels attached to the heart lengthwise and fixed the vessels to the container with thread to prevent floating. As the isolated heart floated in the PFC solution much like a balloon, and therefore we were thus able to eliminate the risk of mechanical tissue injury.
The important factor in this experiment is that the isolated heart was preserved in PFC solution in an environment with highly compressed CO2. As CO2 easily diffuses into tissues, we believe that all of the tissues of the isolated heart were exposed to the environment with highly compressed CO2.
In many biological experiments, CO2 is mixed with O2 and used as PO2 = 950 hPa and PCO2 = 50 hPa (PCO2 = 50 hPa under atmospheric pressure is almost equal to 35 mmHg, the CO2 concentration of living organisms) in combination with a buffer.
On the other hand, PCO2 of 150–200 hPa or more has both anesthetic and decomposition-inhibiting mechanisms (20) and is commercially used as anesthesia for experimental animals (5) as well as for the preservation of grain and the transportation and preservation of fish (15,16).
Theories regarding the mechanisms of these effects of CO2 include theories that it is adsorbed on the surface of a protein biopolymer (14) or theories that the main cause of the effects is that it structures collections of water molecules in living organisms (24), just like inert gases such as Xe that are, just like CO2, used for their anesthetic mechanism (7), their decomposition-inhibiting mechanism (3), or for the preservation of plants (28). In addition, it is believed that decreases in pH that occur with the dissolution of CO2 are also involved, although this has yet to be fully understood.
Seki et al. showed that tardigrades with decreased amounts of water in the body can be resuscitated in PFC solution even under ultra-high pressure (600 MPa) (22).
The phenomenon of living organisms reducing their decomposition by decreasing the amount of water in their bodies in order to adapt to extreme environmental conditions such as dryness or low temperature is called cryptobiosis, and this phenomenon can be found in many forms in the natural world, such as drought dormancy of plants in the Arctic Circle or the phenomena in which many bacteria enter a dormant state when the relative humidity is 60% or less. Such bacteria and tissues are being developed for practical use in preserving dryness (6).
It is believed that one of the characteristics of this phenomenon is that part of the free water in the cells is lost but the bond water protecting the biopolymer surface remains, and decomposition in the cells is reduced to the regenerable limit as the amount of free water decreases. In the belief that long-term organ preservation is possible, experiments have been conducted on the preservation of isolated mammalian organs, and with a focused attention on CO2 gas having anesthetic decomposition-inhibiting mechanisms on living organisms, we conducted an experiment for the preservation of isolated hearts in an environment with a reduced amount of water and a PCO2 of 150 hPa or more to generate significant resuscitation.
Seki et al. found that after an isolated rat heart was preserved for 24 h in an environment with 400 hPa of partially compressed CO2 at 2 ATA, it could be heterotopically transplanted into a recipient rat and resuscitated, and they also verified the reproducibility (23). It was found that CO2 gas structures free water while maintaining the bond water in a living organism and decreasing the amount of free water.
In addition, Yoshida et al. reported an isolated rat heart that was resuscitated by heterotopic transplantation after being preserved for 72 h using a simple immersion preservation method (27). Regarding this simple immersion preservation as the best method, further extension of the preservation time and improvements in the state of preservation were attempted by maintaining the preservation method for the external solution and devising an internal solution. This method has been reported to lead to a better state of preservation than immersion preservation (11,21). We therefore decided to apply this preservation method.
The perfusion method allows for the elimination of accumulated metabolites, the administering of energetic substrates, and the continuous administering of drugs, and it is believed that a longer preservation time and an improved state of preservation can be obtained. Linask et al. observed pulsating for 6–9 days at 22°C when using the perfusion method in an experiment using a rat heart just like our current study (11). It is believed that this is presently the longest reported preservation period, but the experiment by Linask et al. was an in vitro experiment and transplantation was not actually performed. In that respect, we believe that the perfusion method for isolated rat hearts used in this experiment is an improvement over previous methods because it involves heterotopic heart transplantation into the neck after preservation to allow blood to actually run and pulsate in vivo in a setting much closer to actual clinical conditions.
This experiment was designed to inhibit the decomposition of a heart using a decreased amount of water and CO2 gas in a PFC solution. It is a technique to induce artificial hibernation of an organ. Other concepts for preserving organs include temporarily stopping vital activity completely through low-temperature exposure (1) and inhibiting electron transfer enzymes (2) in a way that allows for resuscitation, and this method is called suspended animation.
Both cases involve the application of the dynamic equilibrium theory that aims to allow for switching between a living state and a material state (12). We would like to propose that this technique be called semibiology.
To select and create the best partially compressed carbon dioxide gas and immersion solution for maintaining a dormant state of cardiomyocytes, as well as to improve the preservative effects of PFC solution, which is an external solution, Salehi et al. are now conducting experiments utilizing a two-layer method (TLM) using a human pancreas (19). Together with basic studies on the effects of CO2, we have used the ATP concentration in the tissues of preserved organs, microscopic histological pictures, and the isolated hearts of large animals and applied them to both preservation and resuscitation experiments. An automobile can be repaired in order for it to be able to be driven again when it breaks down, because there are design drawings, parts, and repair techniques. In the case of humans, design drawings (anatomical drawings) and repair techniques (surgery) have already been completed, but there are unfortunately still no spare parts (organs). These spare parts depend on a supply from brain-dead human donors, but the maximum preservation limit for such organs is 24 h.
Our organ preservation and resuscitation technique has extended the preservation time of these parts (organs) from 24 h to 96 h. If the preservation time of organs can be extended to 1 year or more, then human life could be made semipermanent, much like a semiconductor. It is believed that human life can someday be made semipermanent, much like automobiles, if semibiology techniques can be successfully developed in the future.
The phenomenon of plants and animals in the natural world entering drought dormancy almost every year and awakening in the following spring has already been applied to the cells and tissues of plants and animals, and advances are now being made toward the practical use of these same phenomena. In this experiment, we were able to show that this natural phenomenon, which is repeated in the natural world almost every year, can potentially also be applied to isolated mammalian organs, and moreover, we were able to verify its reproducibility.
