Abstract
The effect of epigallocatechin-3-O-gallate (EGCG), a major component of green tea, on platelet preservation was evaluated. Single donor platelets (N = 10) were collected and preserved by the standard method. EGCG was added to the platelet concentrates before preservation and then the functional and biochemical parameters were monitored throughout the storage period. After 6 days of preservation, the aggregability of the platelets was significantly maintained by addition of 50 and 100 μg/ml of EGCG. Platelet prothrombinase activity was also significantly retained by the addition of EGCG. The accumulation of P-selectin and RANTES in the plasma preserved with EGCG was less than those preserved without EGCG, which indicated that EGCG might inhibit platelet activation. Furthermore, EGCG reduced the increase of LDH in plasma during preservation and inhibited the activation of caspase-3 and cleavage of gelsolin, thereby showing that EGCG could inhibit the apoptosis of platelets. These results suggest that EGCG may play an effective role in preserving platelets by inhibiting the activation and apoptosis of platelets.
Introduction
Platelet concentrates (PCs) prepared from the donated blood have been utilized primarily for the treatment or prevention of bleeding (3,23). However, because there is a significant decrease in platelet functional viability as well as an increased risk of pathogen infection during storage of PCs using standard blood banking procedures (4), the duration of storage of PCs is limited to only 5 days internationally or to 3 days in Japan. Therefore, many blood banks are faced with a shortage of PCs and they therefore sometimes cannot meet the demand. During their preparation and preservation, PCs undergo biochemical, structural, and functional changes via a process that is referred to as platelet storage lesions (9,28). Many techniques have been investigated to prolong the preservation period of platelet concentrates, such as improving the preservation bags with high oxygen permeability (36), cryopreservation (8), and the development of platelet storage solutions (17). Unfortunately, none of those studies was able to provide a standard procedure for the preparation and preservation of platelets. Therefore, the current study focused on a method to inhibit the activation of platelets during preservation by the addition of epigallocatechin-3-O-gallate (EGCG), which has a cytoprotective effect (16).
EGCG, one of the green tea polyphenols, known as an antioxidative or antiproliferation agent, has been recently found to protect cells and tissues during preservation (10,13,37). Rat pancreatic islet cells were preserved alive in polyphenol solution for 2 weeks and retained their insulin production activity (13). The successful storage of rat peripheral nerve segments immersed in polyphenol solution for 4 weeks (15) has been reported. Although the exact mechanism of this EGCG action has not yet been elucidated, a previous study demonstrated that EGCG controlled the cell proliferation reversibly and inhibited antigen recognition through the suppression of cell signaling by binding to cell surface molecules (18,22). Therefore, the protective activity might be ascribed to an interaction between EGCG and cell membrane molecules.
This study investigated the potential effects of EGCG on the preservation of human platelets under physiological conditions. To monitor the quality control of PCs, various parameters of in vitro platelet functions and viability were evaluated.
Materials and Methods
Chemicals
The purified EGCG (TEAVIGO™) was purchased from DSM Nutritional Products Ltd. (Basel, Switzerland). The purity of the EGCG was more than 95%. The KBP-PO polyolefin platelet preservation bag was used in this study (Kawasumi Laboratories, Inc., Tokyo, Japan). All other reagents were of chemical grade and used without further purification.
Preparation and Preservation of Platelet Concentrates
Blood samples were collected from 10 healthy volunteers with their informed consent. Freshly drawn venous blood (200 ml) was immediately mixed with one sixth volume of ACD anticoagulant (85 mM sodium citrate, 70 mM citric acid, 110 mM dextrose, pH 6.5) and centrifuged for 10 min at 190 × g. The supernatant platelet-rich plasma was collected.
PCs were mixed with 1 w/w% EGCG saline solution at a final concentration of 0, 10, 50, and 100 μg/ml and stored in KBP-PO bags on a flatbed, horizontal agitator at a controlled, monitored temperature of 22°C for 10 days. Samples were taken from the bags on storage days 0, 3, and 6 for functional analyses and on day 10 for an immunoblotting analysis.
Platelet Aggregation
The platelets were washed by a gel filtration method in order to remove the EGCG in the PCs because EGCG has a reversible inhibitory effect on platelet aggregability (7). Sepharose 2B gel (20 ml; GE Healthcare UK Ltd., UK) was washed and equilibrated with HEPES buffer “140 mM NaCl, 3 mM KCl, 1 mM MgCl2, 3 mM NaH2PO4, 5.5 mM glucose, 10 mM HEPES, and 0.1 w/w% bovine serum albumin (BSA), pH 7.4” and packed into a plastic serological syringe (Terumo Co., Tokyo, Japan). Leakage of the gel was prevented by a piece of nylon mesh. Before the start of the separation procedure, the excess buffer was removed from the top of the column and 4 ml of the PCs was layered onto the gel surface. When the whole volume of the applied PCs entered the gel, HEPES buffer was gently layered onto the column. The eluted platelet suspension in clouded drops was collected in plastic tubes before plasma proteins were able to exit the column and the platelet number was counted using a cell counter (Coulter Particle Counter Z-1, Beckman Coulter, Inc., Fullerton, CA, USA). Platelet aggregation was monitored with an aggregometer (HEMA TRACER 1, Nikko Bioscience, Tokyo, Japan) after adjusting the platelet count to approximately 2 × 106/μl by addition of HEPES buffer. The platelet suspension (200 μl) was allowed to sit at 37°C for 60 s and then incubated with thrombin (0.1 U/ml) or collagen (5 μg/ml). HEPES buffer and a platelet suspension without any agonists were used to standardize the aggregometer for 0% and 100% aggregation, respectively. The maximal aggregation rate was recorded. Platelet aggregability in the preserved PCs was defined as the ratio of the maximal aggregation rate of preserved platelets to that of fresh PCs. Triplicate measurements were done for each test.
Platelet Prothrombinase Assay
Platelet prothrombinase assay was performed according to the method of Alberio et al. with modifications (1). PCs were washed by gel filtration and resuspended in HEPES buffer at a concentration of 3.0 × 107/ml. Collagen was diluted in the HEPES buffer to a concentration of 50 μg/ml. The platelet suspension (180 μl) was mixed with 20 μl of the HEPES buffer or collagen solution and incubated 10 min at 25°C without agitation. Factor Xa (20 μl, Enzyme Research Laboratories, UK) was added to a final concentration of 1 nM and the mixture was incubated for 5 min at 25°C. Human prothrombin (Enzyme Research Laboratories, UK) was added to the mixture in a final concentration of 1 μM and then 20 μl aliquots were removed and mixed with 80 ml HEPES buffer containing 10 mM EDTA. Fifty microliters of 1.2 mM chromogenic thrombin substrate [N-(p-Tosyl)-Gly-Pro-Arg-p-nitroanilide acetate salt, Sigma-Aldrich, St. Louis, MO, USA] was added to the mixture and the rate of hydrolysis of the substrate by the generated thrombin was monitored at 405 nm with a microplate reader (VersaMax™, Molecular Devices Co., Sunnyvale, CA, USA).
P-Selectin (CD62P) and RANTES Secretion
To evaluate the spontaneous activation of platelets during preservation, levels of P-selectin (CD62P; Takara Bio Inc., Tokyo, Japan) and regulated upon activation normal T-cell expressed and secreted (RANTES; GE Healthcare UK Ltd.) were measured by the respective enzyme-linked immunosorbent assay kit.
According to the manufacturer's instructions, PCs were added to a 96-well plate coated with mouse monoclonal anti-CD62 antibody and then incubated for 2 h at room temperature. To block nonspecific antibody binding, the wells were blocked with 1% bovine serum albumin in PBS for 30 min at room temperature. After being rinsed three times with PBS, the wells were incubated with peroxidase-conjugated anti-mouse secondary antibody for 1 h at room temperature. Hydrogen peroxide/tetramethylbenzidine was added to the wells for color development and further incubated for 15 min. Afterwards, the enzyme reaction was stopped by the addition of 1 N H2SO4 and the absorbance was determined at 450 nm with a microplate reader.
LDH Release
The LDH concentration in the platelet-rich plasma was measured in order to evaluate the viability of the preserved platelets (2). A high LDH concentration due to platelet rupture indicates low viability. Platelet viability was determined by measurement of the LDH concentration in the supernatant of the platelet suspension, using an enzyme assay (LDH test Wako, Wako Pure Chem. Ind. Ltd., Osaka Japan) following the standard procedure. Briefly, 1 ml of platelet-poor plasma, 1 ml of 2 v/v% Triton X-100/PBS(–) and 1 ml of substrate containing 0.62 mM lithium pyruvate and 0.18 mM NADH in 50 mM phosphoric acid were mixed and the decrease in the absorbance at 340 nm was recorded from 30 to 60 s after mixing (35).
Immunoblotting
Preserved platelets were lysed in a lysis buffer [100 mM Tris-HCl, 10 mM ethylenediaminetetraacetic acid (EDTA), 10 mM ethylene glycol tetraacetic acid (EGTA), 2 mM sodium orthovanadate, 2 v/v % Triton-X 100, pH 7.5] supplemented with protease inhibitors (mammalian protease inhibitor cocktail, Sigma-Aldrich, St. Louis, MO, USA). The proteins were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) on 4–20% gradient gels and transferred to polyvinylidene difluoride membranes, which were then blocked by incubation in 10% bovine serum albumin dissolved in TBS-T (20 mM Tris, 140 mM NaCl, 0.1 v/v % Tween 20, pH 7.6). After blocking, the membranes were incubated overnight with the primary antibodies [anti-caspase-3 (sc-7272; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA, at 1:100 dilution) and anti-gelsolin (Sigma-Aldrich, at 1:1000)]. After incubation with the primary antibodies, horseradish peroxidase-conjugated secondary antibodies (GE Healthcare UK Ltd.) were added (1:1000 dilution) and the mixture was incubated for 1 h at room temperature. Protein bands were enhanced using a chemiluminescence Western blotting determination kit (ECL-Plus, GE Healthcare UK Ltd.). The band intensity was quantified using imaging software (Image J, 1.3 version).
Statistics
All data are presented as the mean ± SD. Statistical analyses were performed using Statistical software (StatView®, SAS Institute Inc., CA, USA). Data among the groups were compared by a one-way ANOVA with post hoc Fisher's protected least significant difference test. Differences were considered statistically significant at a value of p < 0.05.
Results
Platelet Aggregation
PCs were stored with or without EGCG for 6 days and a platelet aggregation study was performed with thrombin or collagen stimulation (Fig. 1). The representative platelet aggregation curves in Figure 1A and B show that the addition of 50 or 100 μg/ml of EGCG to PCs during storage preserved the responsiveness of platelets to thrombin or collagen stimulation, in contrast to PCs stored without EGCG. Figure 1C and D shows the aggregation retaining ratio against fresh platelets induced by thrombin and by collagen after 6 days of storage, respectively. The values of the percent aggregation induced by thrombin were reduced to 87.7% at 3 days and 68.2% at 6 days preservation without EGCG; however, 50 μg/ml of EGCG kept the percent aggregation at 98.7% at 3 days and 78.8% at 6 days (p < 0.01 vs. control). In addition, the values of the percent aggregation by collagen were 10% larger with 50 and 100 μg/ml of EGCG in comparison to without EGCG preservation (p < 0.05 vs. control). Addition of 10 μg/ml of EGCG was not effective and there was no significant difference between 50 and 100 μg/ml of EGCG.

Effects of EGCG on platelet aggregation during preservation. Representative aggregation curves after 6 days of preservation and fresh platelets upon thrombin (A) or collagen (B) stimulation. Changes in the aggregability of the preserved platelets upon thrombin (C) or collagen (D) stimulation. The data among the groups were compared using a one-way ANOVA with post hoc Fisher's protected least significant difference test (*,#p < 0.05 vs. without EGCG, **,##p < 0.01 vs. without EGCG).
Platelet Prothrombinase Assay
Figure 2 shows thrombin generation by the prothrombinase activity of the preserved platelets. Fresh platelets showed the highest prothrombinase activity induced by collagen. After 6 days of preservation, the prothrombinase activity was reduced to 25% but 50 μg/m of EGCG significantly (p < 0.01) maintained the activity at 35% of the activity in fresh platelets.

Collagen-induced prothrombinase activity (thrombin generation) after 6 days of preservation. **The difference between the treated and control samples was significant (p < 0.01).
P-Selectin and RANTES Secretion
An increase of the P-selectin and RANTES released by activated platelets was detected both with and without EGCG during preservation. A statistically significant difference in P-selectin was not seen between platelets stored with or without EGCG, although the mean values in the EGCG were was lower than those in the control group (Fig. 3A). On the other hand, the increase in RANTES was significantly lower (p < 0.001) in the EGCG group (Fig.3B).
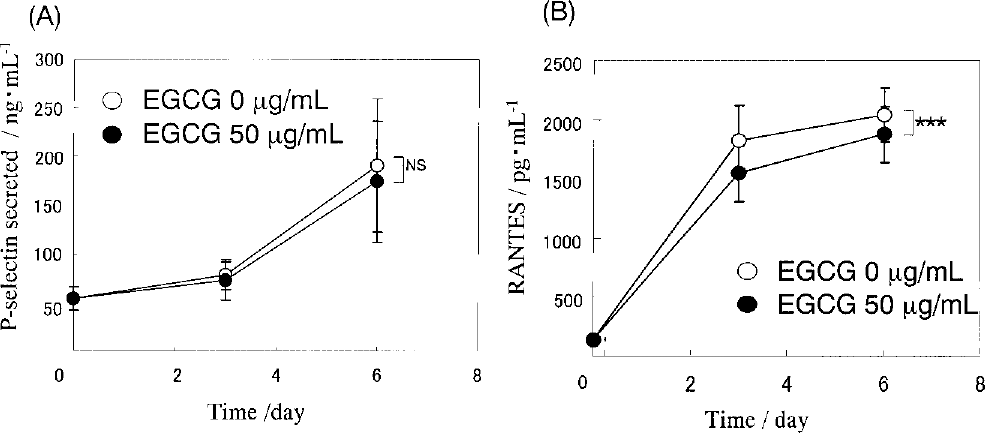
Effects of EGCG on platelet activation. P-selectin secreted (A) and RANTES released (B) into the PCs during preservation. ***The difference between the treated and control samples was significant (p < 0.001).
LDH
Figure 4 shows the level of LDH in PCs was increased with preservation time. However, the addition of EGCG significantly (p < 0.01) reduced the increase of LDH concentration in PCs during preservation. The LDH concentration showed almost no increase for 3 days of preservation and was about 20% lower than that of the control group after 6 days of preservation.

LDH released into the platelet concentrates during preservation. **The difference between the treated and control samples was significant (p < 0.01).
Immunoblotting
To measure the appearance of apoptosis in preserved platelets, caspase-3 activation and gelsolin cleavage were assayed by immunoblotting (Fig. 5). After 10 days of preservation, 35 kDa procaspase-3 was degraded into activated 21-kDa fragment (Fig. 5A) and full length of gelsolin was cleaved to give a 47-kDa fragment (Fig. 5B) in platelets preserved without EGCG. In contrast, activation of caspase-3 and cleavage of gelsolin were significantly both inhibited with EGCG. The ratio of activated caspase-3 to procaspase-3 and cleaved gelsolin to full length of gelsolin are shown in Figure 5C and D, respectively. These data indicate that caspase-3 activation and gelsolin cleavage were both significantly (p < 0.01 and p < 0.05, respectively) inhibited by the addition of EGCG, thus showing that EGCG might inhibit platelet apoptosis.

Expression of apoptosis markers after 10 days of preservation. (A) Representative Western blot images of caspase-3 activation measured as the appearance of the 21-kDa fragment and (B) representative Western blot images of gelsolin cleavage measured as the appearance of the 47-kDa fragment. (C) The quantitative results from the Western blot images of activated caspase-3 (21 kDa) normalized to procaspase-3 (35 kDa). (D) The quantitative results from the Western blot images of cleaved gelsolin (47 kDa) normalized to intact gelsolin (86 kDa). The data among the groups were compared by one-way ANOVA with post hoc Fisher's protected least significant difference test (*p < 0.05, **p < 0.01).
Discussion
The present study demonstrated that EGCG can maintain platelet functions. PCs preserved with EGCG retained the platelet aggregation response, procoagulant activity, and viability, with the inhibition of apoptosis.
The short shelf life of currently available PCs makes it difficult to stock platelets. During preservation, platelets may lose a significant amount of hemostatic activity and exhibit a progressive loss of viability (9,25,28). This is reflected in their loss of membrane integrity, inability to maintain their shape, and a progressive decrease in hemostatic functions such as platelet aggregability and the loss of expression and affinity of surface receptors. Platelets undergo some degree of activation during procurement of blood, shipment, processing, and storage, thus leading to in vivo functional compromise. By using inhibitors of platelet activation and protectants, platelet activation can be minimized through the inhibition of signal transduction (24).
Hyon and Kim first observed that EGCG might help to preserve tissues and cells, particularly rat pancreatic islets (13) and, since then, several tissues have been preserved with EGCG (10,14,15,37). It must be noted that some of the activity of EGCG is related to its surface binding ability. Kim et al. demonstrated that EGCG can prevent allostimulation in murine allografts by blocking cell surface molecules, resulting in weak or incomplete stimulation of alloreactive T cells (18). In fact, the inhibition of platelet aggregation is reversible. This study confirmed that gel-filtered washed platelets had their aggregation response restored. A previous study showed the inhibition of cell proliferation by EGCG to also be restored by washing (22). Cetin et al. suggested that a reversible inhibition of the platelet function could protect against activation during storage (6). Furthermore, EGCG is known to have a strong antioxidative activity, which protects cells from oxidative damage (16,29).
The formation of platelet-fibrin thrombi at sites of vascular injury is the principal mechanism involved in the arrest of bleeding and the formation of occlusive intravascular thrombi. The mechanisms of platelet adhesion to the subendothelium has been attributed to the interaction and activation with platelet agonists such as collagen, thrombin, and adenosine diphosphate (ADP) to promote the formation of platelet aggregates at injury sites (32,33). The initiation process of aggregation is the formation of intracellular bridges by fibrinogen, von Willebrand factor, and platelet membrane glycoprotein (GP) IIb-IIIa (26). Activated platelets also contribute to coagulation by providing a catalytic membrane surface for assembling prothrombinase complexes. In this reaction, factor X is converted to factor Xa by a complex of factor VIIIa and factor IXa and the prothrombinase reaction catalyzes the conversion of prothrombin to thrombin by a complex of factor Va and factor Xa. The aberration of these two principal functions (aggregation and coagulation) of platelets is related to the loss of membrane lipids and proteins. The current results indicated that EGCG maintained the aggregation (Fig. 1) and coagulation (Fig. 2) response after 6 days at a significantly higher level than in the control because EGCG might attached with platelet membrane molecules to protect and to maintain the integrity via its antioxidative ability. It has been reported that lipid peroxidation and microvesiculation are the major mechanisms of lipid loss (20). EGCG may also inhibit the lipid peroxidation with its high antioxidation ability by adsorption to the membrane. There was no significant difference between 50 and 100 mg/ml EGCGC groups in aggregability (Fig. 1C and D). Therefore, other functional and biochemical properties were evaluated between the groups without EGCG and with 50 μg/ml of EGCG.
The addition of EGCG significantly inhibited the increase in the secretion of P-selectin (Fig. 3A) and the release of RANTES (Fig. 3B) during the 6 days of preservation. P-selectin, an alpha granule membrane protein, is a useful marker for in vitro platelet activation, because its expression on the surface correlates with a loss of viability upon transfusion of the preserved PCs (12). Platelet activation also leads to degranulation and release of platelet-derived cytokines. RANTES may be implicated in nonhemolytic transfusion reactions (19). The current results suggest that EGCG inhibited the activation of platelet functions and protected the surface proteins and lipids from oxidation during preservation by adsorption onto surface molecules and then, functions were restored by the maintained surface molecules with the detachment of EGCG by washing.
Apoptosis refers to any cell death event that is mediated by constitutively expressed proteins, activated in a proteolytic cascade by different stimuli. Vanags et al. first suggested the presence of apoptotic mechanisms in platelets (31). During in vitro preservation, platelets exhibit increased expression of proapoptotic proteins associated with morphological and biochemical features similar to those found in nucleated cellular systems (5). Platelets express many components of nucleated cell apoptosis, including caspase-3, -9, and Apaf-1 (5,27,31). Caspase-3 has been implicated in a specific subset of platelet activation responses and strong agonists can induce apoptosis including phosphatidylserine exposure, cell shrinkage, plasma membrane microvesciculation, and gelsolin cleavage (27,34). Gelsolin is an actin-binding protein that is cleaved during apoptosis in other cell systems with the formation of a fragment of 47 kDa and this proteolytic event is another marker frequently used for detecting the occurrence of apoptosis (21,30). Immunoblotting demonstrated that caspase-3 activation (Fig. 5A) and gelsolin cleavage (Fig. 5B) were significantly inhibited in platelets preserved in EGCG. The antiapoptotic property of EGCG has been reported in several cells and tissues (11,37). EGCG may lead to the inhibition of platelet apoptosis, which is consistent with the maintenance of lower LDH level (Fig. 4), indicating lower cell death in preservation with EGCG.
The addition of EGCG to the PCs during preservation may therefore be a potentially novel and useful method to prolong the platelet storage period. Although further investigation is necessary with respect to the mechanisms behind the preservation and regarding the improvement of its efficacy, the addition of EGCG to PCs may potentially be able to successfully extend the preservation period for platelets.
