Abstract
Pancreas procurement for islet isolation and transplantation is limited by concerns for the detrimental effects of postmortem ischemia. Hypothermic machine perfusion (HMP) preservation technology has had a major impact in circumventing ischemic injury in clinical kidney transplantation and is applied here to the preservation and procurement of viable islets after hypothermic perfusion preservation of porcine pancreata because pigs are now considered the donor species of choice for xenogeneic islet transplantation. Pancreases were surgically removed from young (<6 months) domestic Yorkshire pigs (25–32 kg), either before or after 30 min of warm ischemia time (WIT), and cannulated for perfusion. Each pancreas was assigned to one of six preservation treatment groups: fresh controls—processed immediately (cold ischemia <1 h) (G1, n = 7); static cold storage—flushed with cold UW-Viaspan and stored in UW-Viaspan at 2–4°C for 24 h with no prior WIT (G2, n = 9); HMP perfused on a LifePort® machine at 4–6°C and low pressure (10 mmHg) for 24 h with either KPS1 solution (G3, n = 7) or Unisol-UHK (G4, n = 7). Additional treatment groups to evaluate the effects of prior warm ischemia examined islet isolation after 30 min WIT in situ without (G5, n = 6) or with subsequent 24-h HMP with KPS1 (G6, n = 7). The pancreas was intraductally distended with Liberase PI enzyme and normothermically digested. The isolated islets were purified by a continuous density-gradient centrifugation. Perfusion-induced glandular edema was G3 = 138 ± 19%, G4 = 160 ± 16%, and G6 = 127 ± 22%. Islet yield (IEQ/g of pancreas) varied between the groups: G1 = 1,425 ± 610, G2 = 1,002 ± 262, G3 = 2,242 ± 449 (p < 0.05 vs. G2), G4 = 1,901 ± 420 (p < 0.05 vs. G2), G5 = 1,756 ± 329, and G6 = 1,396 ± 243. Islet stimulation indices were equivalent between the groups and similar to controls (G1). Insulin content (ng/IE) was different between the treatment groups with the highest insulin content in islets harvested from HMP pancreata. Dithizone staining for islets consistently showed more uniform digestion of the perfused organs, with greater separation of the tissue, less entrapped islets, and higher islet yield and purity. The salutary effects of HMP for 24 h were also manifest after 30-min prior warm ischemia. We conclude that 24 h of HMP is well tolerated, leading to moderate edema but no loss of function of the harvested islets. The edema appears to aid in enzymatic digestion, producing a greater yield and purity of islets compared with pancreas subjected to 24 h of static cold storage.
Keywords
Introduction
There is now a worldwide consensus that islet transplantation may be considered a viable option for the treatment of insulin-dependent diabetes mellitus, and clinical trials are under way at many centers around the world (1, 38). As this approach for curing diabetes transitions into routine clinical standard of care, the demand for donor islets will escalate. Moreover, the potential for xenotransplantation to relieve the demand on an inadequate supply of human pancreases will also be dependent upon the efficiency of techniques for isolating islets from the source pancreases (17). Procurement of donor pancreases for islet isolation and transplantation is not yet widely practiced due in part to concerns about postmortem ischemia upon functional islet yields. As we have reviewed recently (43), perfusion/preservation technology has had a major impact in circumventing ischemic injury in kidney transplantation (8, 22, 28, 29, 43).
The choice of a porcine model for this study served two purposes. Firstly, in view of the current resurgence of clinical interest in hypothermic perfusion preservation of organs for transplantation (43), the pig provides the model of choice for large-animal preclinical evaluation of hypothermic machine perfusion (HMP) technology for segmental donor pancreas preservation. More specifically, these studies are designed to provide an initial evaluation of pancreas preservation on the FDA-cleared LifePort® platform (Organ Recovery Systems, Itasca, IL) using a porcine model (44). Secondly, the special case of pancreas preservation prior to islet isolation is of high significance in view of both the worldwide interest in islet xenotransplantation and our preliminary reports that HMP can facilitate improved islet yields without compromising islet function (45, 46). Moreover, the consensus strategic plans recently published by the International Xenotransplantation Association for considering clinical trials of porcine islet products for type 1 diabetes emphasizes the need and importance of sterile, disease-free environment for the source pigs and the products (17). To this end, the established LifePort® system will provide a convenient sterile environment for transport of the source pancreas from the site of procurement to the islet processing facility. Here we applied this approach to the preservation and procurement of viable islets after hypothermic perfusion preservation of porcine pancreata because pigs are now considered the donor species of choice for xenogeneic islet transplantation for a number of compelling reasons (30).
The age of the donor pig has proved to be a significant factor in the islet isolation process with young, so-called market size pigs (<6 months old), proving to be particularly difficult as a source of transplantable islets (7, 10, 50). Nevertheless, despite these challenges young pigs are favored over retired breeders (>2 years old) due to their abundance, lower animal and husbandry costs, and are more suitable to meet regulatory guidelines for donor tissue for xenotransplantation. This study was undertaken to evaluate the efficacy of hypothermic machine perfusion of pancreata from young pigs prior to islet isolation. We also report on the details of our surgical model that was developed in light of special considerations to achieve uniform perfusion of the porcine pancreas during 24 h of hypothermic perfusion at 7°C (44).
Materials and Methods
Organ Procurement
Small farm pigs (Domestic Yorkshire, male, 25–32 kg; Hambone Farms, SC) were used as pancreas donors. Following induction of general anesthesia with ketamine (22 mg/kg), acepromazine (0.2 mg/kg), and atropine (0.025 mg/kg), and anesthesia maintenance with isoflurane in oxygen, the animals were intubated and connected to a ventilator. The abdominal cavity was opened through a midline incision from the xiphoid cartilage to just above the pelvis, and the descending aorta was identified and cannulated below the kidneys. The inferior vena cava and aorta were identified, isolated, and close-clamped above the diaphragm. An in situ gravity-driven flushing of the pancreas was initiated using 2 L of cold lactated Ringer's solution while for blood flow the inferior vena cava was cut open above the diaphragm, downstream from the clamp. The pig was euthanized through exsanguinations and a lethal dose of 5% sodium pentobarbital administered intravenously. The latter is an accepted form of euthanasia according to the latest guidelines from the American Veterinary Medical Association Panel on Euthanasia (AVMA). All animal care and handling complied with policies and approval of the Institutional Animal Care and Use Committee (IACUC) at the Medical University of South Carolina, where the organ procurements were carried out.
Organ exposure to warm ischemia was kept below 3 min by using the cold solution vascular flush and by placing ice inside the abdominal cavity during surgical excision of the pancreas. The pancreas was carefully and rapidly exposed and dissected apart from the surrounding tissue and organs. A segment of proximal duodenum starting near the pylorus and inclusive of most of the duodenum's second descending loop was kept intact with the pancreas to protect the superior and inferior pancreaticoduodenal arteries (Fig. 1). The common bile duct and pancreatic duct openings were included as part of the duodenum segment. This considerably facilitated pancreatic duct cannulation, by avoiding the difficulties encountered with retracted duct identification and cannulation, and preserved early duct branches. The latter were necessary to ensure good organ distension for gland digestion and islet isolation. The splenic vein and artery were ligated prior to detachment of the spleen (Fig. 1). A 5–7-cm-long aortic segment was left attached to the pancreas for future organ cannulation. The segment included the openings of both superior mesenteric artery (SMA) and celiac trunk (CT) vessels (Fig. 1). The pancreas was removed from the body, immersed in cold University of Wisconsin solution (UW; Viaspan, Fisher Scientific), and placed on ice for transportation from the operation room to the research laboratory, a trip of less than 30 min. Overall, from the initiation of in situ cold flushing to the beginning of ex vivo hypothermic perfusion, the pancreata exposure to static cold ischemia was kept below 2 h. Upon arrival at the lab all exposed arterial branches on the margin of gastroduodenal and hepatic sides of the pancreas were meticulously identified and ligated to ensure uniform perfusion throughout the gland and allow the effluent to emerge only from the portal vein by avoiding leaks from the many arterial branches.

Schematic diagram showing the pancreas excised with a segment of the descending aorta for cannulation of the celiac trunk (CT) and superior mesenteric artery (SMA). The splenic vein (SV) and artery were ligated. All arterial branches on the margin of the gastroduodenal and hepatic side were ligated to provide uniform perfusion and effluent flow only through the portal vein. IVC, inferior vena cava; LGA, left gastric artery; HA, hepatic artery; GDA, gastroduodenal artery; SMV, superior mesenteric vein; SMA, superior mesenteric artery; PPA, posterior pancreatic artery; SPDA, superior pancreaticoduodenal artery; PDVA, pancreaticoduodenal vascular arcade; IPDA, inferior pancreaticoduodenal artery; IMA, inferior mesenteric artery; IMV, inferior mesenteric vein.
Pancreas Cannulation and Perfusion
During the development of this porcine surgical model the method of cannulation of the pancreas also proved critical for successful uniform perfusion on the selected LifePort® pulsatile perfusion system (Organ Recovery Systems, Itasca, IL). The LifePort® provides closed loop pulsatile perfusion at an imposed systolic pressure selected for control at 10 mmHg in this study, while the pancreas was immersed in cold perfusion solution controlled at 7°C inside the integral organ bath. Due to anatomical configurations and variations of the vasculature in the pancreas from young pigs it proved difficult to achieve a consistent perfusion preparation by using direct cannulation of the SMA and celiac truck individually. This was due to arterial side branches that were easily blocked and impeded by the cannulas as illustrated in Figure 2A and B, and discussed below. This problem was circumvented by employing a seal-ring cannula (10 × 35 mm; Organ Recovery Systems), which has a geometric design that permitted direct access to the openings of the SMA and CT via an aortic patch as illustrated in Figure 2C.

Cannulation of the excised pig pancreas. (A) Excised pig pancreas with attached duodenal section to preserve the superior and inferior pancreaticoduodenal arteries. The openings to the celiac trunk (CT) and superior mesenteric artery (SMA) are identified. (B) Cut-down arterial vessel (CT) illustrating early side branches that may be occluded when straight cannulas such as those illustrated are inserted. This problem was circumvented by using a “seal-ring” cannula (C), which avoids the need to insert cannulas into the individual arteries by allowing perfusion via the openings of the CT and SMA on an aortic patch clamped inside the seal-ring cannula. (D) Pig pancreas immersed in perfusion solution in an organ cassette and perfused via a seal-ring cannula installed on the LifePort? perfusion machine. The scale bar in each panel is 2 cm.
The LifePort® pulsatile system was initially designed, and FDA cleared, for kidney hypothermic perfusion/preservation for clinical transplantation (3, 4, 29). Using the kidney system the pancreas was perfused in a closed loop while being completely immersed in the perfusion solution inside the organ cassette, which comfortably accommodated the whole pancreas from these young pigs (Fig. 2D). The cassette also served as a solution reservoir, the perfusate being drawn out by the pump, was passed through the filter, bubble trap, and the infusion port before returning to the pancreas and organ cassette. Pancreas submersion in the temperature-controlled perfusate helped eliminate temperature gradients across the organ surface and ensure high-quality hypothermic preservation. The selected perfusate (1 L) was maintained at 5–7°C. A pulsatile (30 pulses/min) constant low pressure flow regime was imposed with a setting of 10 mmHg for the systolic pressure. The perfusion pressure value of 10 mmHg was selected based on the fact that physiologically the pancreas is a low flow organ and all preliminary experiments performed to optimize the perfusion regime of juvenile pancreata indicated a need for either low pressure, or low flow rate, driven perfusion preservation. The technical features of the already clinically established LifePort? system were able to support these demands (44). Perfusion flow rate and pressure, organ resistance, and perfusate temperature were measured, recorded, and displayed in real time. Organ weight was measured before and after perfusion and used to determine postpreservation fluid accumulation within the organ (edema). More details of the development of the method of perfusion for the pancreas have been published elsewhere (44).
Pancreas Distension and Islet Isolation
Islets were isolated from fresh and hypothermically preserved pancreata. Following either organ recovery or preservation, dissociation enzyme (Liberase PI, Roche, Indianapolis, IN) was delivered to the pancreas via the pancreatic duct by direct syringe infusion. Prior to its use the Liberase (0.5 g) was reconstituted to a final volume of 350 ml with HBSS (Hank's balanced salt solution, VWR, Suwanee, GA) and permanently maintained on ice. Three different batches of Liberase PI were employed throughout the duration of the experiments reported here. The average value of collagenase activity for the three lots was 2192.4 ± 114.6 Wunschs per vial (500 mg) with a standard deviation of 198.6 Wunsch. Following intraductal pancreas distension, all extraneous tissue was removed and the pancreas was cut in seven to nine pieces. The latter were placed in a 1000 ml Ricordi chamber (BioRep Technologies Inc., Miami, FL) containing of nine stainless steel balls and a 500-μm mesh screen. The dissociation chamber, an integral part of the Ricordi islet dissociation system (a 1200-ml pump driven temperature controlled flow circuit), was already primed (RPMI, Invitrogen, Carlsbad, CA) and brought to physiologic temperature (36 ± 1°C). Through combined chamber mechanical agitation and enzymatic digestion the islets were liberated under controlled conditions of temperature and flow rate (400 ml/min). With the aid of dithizone staining, periodic sampling of the tissue digest allowed visualization under the microscope of the progress of tissue digestion and the percentage of free islets, and guided the assessment of digestion end point. When determined, the digestion was stopped and the dilution phase (with 4 L of cold RPMI) was initiated while the tissue digest containing free islets was collected and placed on ice. The tissue digest was washed three times (3 min, at 4°C and 1000 rpm) with cold 10% FCS-HBSS, and the final tissue/islet pellet was consolidated in two 250-ml conical tubes. The packed cell volume was weighed and recorded (always less than 20 g/tube), the islet pellet was resuspended in UW, up to 100 ml per tube, and placed on ice for a cold incubation of at least 30 min. Periodic swirling of tubes was performed to avoid pellet compaction.
Islet Purification
At the completion of cold incubation in UW solution, islet purification based on density gradient centrifugation was performed using the COBE 2991 (Gambro BCT, Lakewood, CO). A continuous Ficoll gradient of 1.108 and 1.069 densities was employed to separate the cells at 2400 rpm for 5 min. The purified Ficoll islet fractions were collected sequentially in six predetermined 250 ml conical tubes (prefilled with 2.5% FCS-M199 media). The remaining content of purification bag was also retrieved in the seventh tube. All tubes were centrifuged at 4°C and 1500 rpm for 3 min. Following appropriate supernatant removal, the fractions were sampled (0.5 ml sample in 2 ml of dithizone solution) to determine under the microscope the purest fractions. Dithizone solution (50 mg diphenylthiocarbazone and 5 ml dimethyl sulfoxide in 45 ml phosphate buffer solution) was used to stain the islets for their identification and quantification. Images of all fractions were recorded for comparison purposes. The fractions containing islets were recombined as found appropriate, properly labeled, and assigned for islet counting and/or viability testing.
Islet Quantification and Assessment
Islet Quantification
Following islet isolation and purification the total number of islets was determined using standard published techniques. A volume of 100 μl of islet fraction was placed in 250 μl of dithizone solution inside a 35 × 10 mm tissue dish with grid. Thus, islets were stained, counted, and converted to islet equivalents (IE) according to standard convention (34). Counts were performed in duplicate by two independent observers. The purity of the islet preparation was also assessed by comparing dithizone-stained tissue to unstained exocrine tissue.
Islet Insulin Content and Stimulated Secretion Assay
Islet insulin release upon exposure to low and high glucose concentrations was determined following an initial recovery of 1 h at 37°C in low (2 mM) glucose (in RPMI-1640) solution. Then consecutive 30-min islet incubation periods (37°C water bath shaker) in 2, 20, and 2 mM glucose solution, respectively, were performed, each followed by careful supernatant removal and freezing (0.5 ml), and islet resuspension in the next glucose concentration solution. The insulin content and glucose-stimulated insulin response assessments were carried out immediately following islet purification and quantification, without prior incubation/culturing. Based on the purest fraction(s) islet yield, small volume aliquots of islet suspension containing 25 IE were distributed to each one of the 12 × 1.5 ml conical tubes containing 1 ml of the corresponding glucose solution. Gravity driven sedimentation of islets within the 1.5-ml conical tubes was used prior to removal of supernatant (0.5 ml per tube) at the end of each glucose stimulation phase. From the purest fraction(s) islet suspension, two samples of 0.5 ml were removed and subsequently frozen to later determine the insulin and amylase content, respectively. The insulin release upon glucose stimulation of the frozen supernatants and the insulin content of the purest fraction(s) samples were quantitatively determined using the Insulin Porcine EIA kits (Alpco Diagnostics, Windham, NH). The latter is a solid phase two-site enzyme immunoassay based on the direct sandwich technique. According to manufacturer's protocol (Mercodia, Uppsala, Sweden), two monoclonal antibodies are directed against separate antigenic determinants on the insulin molecule, the bound enzyme labeled antibody to the insulin molecule is detected by a reaction with the 3,3′,5,5′-tetramethylbenzidine and the end point is read spectrophotometrically (Spectra Max Plus 384; Molecular Devices, 450 nm). The results were normalized to islet equivalent and expressed as ng/IE. The EnzChek Ultra Amylase Assay kit (Molecular Probes, Carlsbad, CA) was used to measure the amylase content, as per manufacturer's specifications.
Islet Viability
Glutathione and ATP were measured as indices of tissue injury and energy status, respectively. For this, from the purest fraction(s) islet suspension samples were removed, 1 and 0.5 ml for glutathione and ATP measurements, respectively, spun and immediately immersed in liquid nitrogen after complete supernatant removal. These two volumes satisfy the analysis requirements for the two assays employed for glutathione and ATP quantification using the Glutathione Fluorimetric Assay kit (Sigma, St. Louis, MO) and Viability-ATP Assay kit (Dojindo Molecular Technologies, Gaithersburg, MD), respectively. The sample analysis was performed in accordance with the manufacturer's assay instructions; glutathione and ATP were determined, normalized to IE and expressed in nM/IE.
Structural Analysis
An optimized protocol for pancreas tissue processing was developed to visualize morphological changes induced by the organ preservation methods. For this all wedge biopsies were fixed overnight in 2% glutaraldehyde/0.1 M Sorenson's buffer solution. Following this, the samples were rinsed (0.1 M Sorenson's buffer) and placed in 2% Osmium/0.1 M Sorenson's buffer solution for 1 h. After another rinsing the samples were dehydrated using a graded series of acetone solution and infiltrated with Araldite 502 resin using initially a 1:1 resin/acetone mixture. After 30 min the samples were moved to a 9:1 resin/acetone mixture, placed on a vertical rotator, and left overnight. The next day, the samples were transferred to 100% resin containing molds ensuring no air bubbles were present and polymerized at 60°C for 24 h. Thick sections (2 μm) were cut using an ultramicrotome, stained with toluidine blue, and viewed with the aid of a light microscope.
Experimental Design
Based on the outcome of our preliminary studies that demonstrated a salutary effect of hypothermic machine perfusion on islet isolation from pig pancreas (43, 46), this study was undertaken to evaluate the effects of both the nature of the perfusate and prior warm ischemia on islet isolation from juvenile pig pancreases. The experimental groups in relation to the conditions of preservation are summarized in Table 1.
Experimental Groups

Preservation Solutions
Three solutions were used for pancreas hypothermic preservation: (i) UW (Viaspan, Barr), for static cold storage; (ii) KPS1 (Organ Recovery Systems), FDA cleared for kidney machine perfusion; and (iii) Unisol-UHK, part of the Unisol™ proprietary family of solutions (Organ Recovery Systems and Cell and Tissue Systems, Charleston, SC) (41). Currently Viaspan, considered the “gold standard” solution for organs hypothermic preservation, is the most commonly used solution in clinical organ transplantation. KPS1, a hybrid “intracellular/extracellular” solution, is the current industry standard for machine perfusion of kidneys (23, 40).
The Unisol™ family of solutions, of which Unisol-UHK is a component, has been designed as a universal solution system for optimum cell, tissue, and organ preservation, and has been granted US patent as a system for organ and tissue preservation and hypothermic blood substitution (41). UHK, the Unisol™ intracellular base solution, was designed for application at profound hypothermic temperatures (<15°C). Table 2 shows the chemical formulations for the solutions used in this study. The UHK solution, prior to its use, was supplemented with fresh reduced glutathione (3 mM), in accordance with its chemical formulation (4, 5, 41, 42, 47). KPS1 solution contains the same amount of glutathione, but was added at the time of solution manufacture.
Preservation Solution Formulations

Results
Islet Retrieval Data
It was observed throughout the study that the method of preservation had a significant impact on the extent of digestion time and the amount of free islets released from the pancreatic digest. Data are summarized in Table 3 and illustrated in Figure 3. Microscopic examination of the different preparations using dithizone staining for islets showed a consistently more uniform digestion of the pancreata from G3 and G4 compared with G1 and G2, with greater separation of the tissue and less entrapped islets (Fig. 4). Tissue digest from both fresh (G1) and SCS control group (G2) pancreata showed more mantled (incompletely cleaved islets with adherent exocrine tissue) and entrapped islets (Fig. 4A–D) in comparison to perfused organs (Fig. 4E and F). Islet sampling during the process of digestion revealed early free islets and a more homogenous digest, without fragments of exocrine tissue, for the machine perfused pancreata (Fig. 4E).
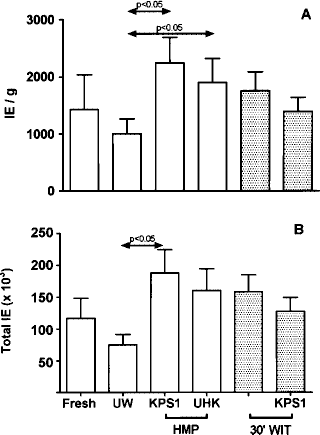
Islet yields expressed as both islet equivalents (IE) per gram of pancreas (A) and total IEQ (B). Data for each group are expressed as the mean (±SEM).

Light micrographs (100x magnification) showing the relative purity of the respective islet preparations at the end of the digestion phase (A, C, E) and after density gradient purification (B, D, F). Islets are identified by dithizone staining and appear purple-red in contrast to the unstained exocrine tissue, which appears gray-brown. (see text for details). All panels are shown at the same magnification and represented by the 100 μm scale bar shown in the top left hand corner.
Pancreas Preservation and Islet Isolation Indices

N = 4.
N = 6.
p < 0.05 versus G1, G2 (ANOVA, Tukey's posttest).
p < 0.01 versus G1, G2, G3, G4 (ANOVA, Tukey's posttest).
p < 0.01 versus G1, G2, G3 (ANOVA, Tukey's posttest).
p < 0.05 versus G2.
p < 0.01 versus G2–G6.
The islet retrieval data are summarized in Figure 3, which shows that pancreas perfusion, resulted in a high yield of islets that was statistically significantly (p < 0.05) when compared to the experimental control cold flush group (G2). Machine perfusion allowed the remnant blood to be washed out and also, based on the amount of water accumulation (edema), provided a disrupted extracellular space without a negative impact on the ductal distension. These ultimately helped rapidly free more islets and a correlation between edema and digestion time seems to exist with shorter digestion times in pancreases with higher edema (Table 3). The slightly negative edema observed in the cold flush group (-2.8 ± 0.7%) was due to the hypertonicity of the UW-Viaspan solution.
Following purification, for all the experimental groups, the purified islets were found in two fractions; frequently one fraction had more islets than the other one, which often contained bigger islets and less exocrine tissue. The fractions were labeled chronologically, in the order of collection, from 1 to 7. Most of the free pure islets in the preps from perfused pancreata, with or without 30 min of warm ischemia exposure (G3, G4, G6), were found in fractions 3 and 4. The purest islets of static stored pancreata (G2) and nonpumped warm ischemic (30 min) organs (G5) were usually seen in fractions 2 and 3. However, in the case of fresh and Viaspan control group pancreata, free islets were also contained by fractions 5 and 6, for these group pancreata the prep revealed entrapped islets within exocrine tissue fragments that were unable to migrate in the density gradient. In the HMP group, where significantly shorter digestion times were needed and more uniform preps of separated islets and exocrine tissue were seen, a density gradient separation was more efficient, with a higher yield and purity of islets (see Fig. 4). Fraction 7, which contained the remnants from the density gradient bag, had no islets for perfused pancreata while it contained entrapped islets from control and fresh organs (Fig. 4B, right). The purity of the islet preparations after density gradient purification, measured as the ratio of insulin (from islets) to amylase (from exocrine cells), was also increased in the perfused groups compared with both fresh and static cold storage (Table 3). The size distribution of the islets harvested in the different groups is summarized in Table 4, which shows that a very high percentage (>90%) of the islets harvested from preserved pancreases was in the range 50–100 μm irrespective of the mode of preservation. This was not significantly different (ANOVA) to the size distribution obtained from control untreated pancreases obtained from these young pigs. The islet counts given in Table 4 represent the yield expressed as absolute islet numbers irrespective of their size (>50 μm) and is distinct from the yields shown in Figure 3, which are expressed in terms of “islet equivalents” using the standard convention (34). The size distribution of islets obtained from these juvenile pigs concurs with previous reports in the literature comparing adult and young porcine donors (10, 20, 51, 52).
Islet Size Distribution

Total islet count represents the number of individual islets with a diameter greater than 50 μm.
The effect of prior warm ischemia on islet yield is also shown in Figure 3. Islet retrieval from young porcine pancreases was not compromised after 30 min of warm ischemia (G5) and was further maintained after an additional 24 h of hypothermic machine perfusion (G6).
Islet Integrity
Table 3 summarizes the data for islet function in terms of insulin content and the ability to respond to a secretory glucose challenge. The latter is expressed as the Stimulation Index determined by comparing the insulin released during sequential exposure to a low (non-stimulatory, 2 mM) and high (stimulatory, 20 mM) concentration of glucose. The mean insulin content of islets isolated from perfused pancreata was significantly higher than that of the UW-Viaspan cold stored control group and was not significantly different to the mean values from fresh tissue. Moreover, the stimulation indices showed that the insulin secretory function of the islets isolated from perfused pancreata was not compromised when compared with the control groups even after 30 min prior warm ischemia. Ischemia alone, without subsequent perfusion, produced greater variability in the secretory function (G5) compared with all of the other groups as reflected in the standard error that was an order of magnitude greater. However, the imposition of HMP after 30 min WIT (G6) appeared to stabilize this response and insulin secretory function was not significantly different to controls. The energy status of the isolated islets, in terms of ATP content, was also preserved during the 24-h perfusion technique.
Histological integrity of the pancreases was evaluated from wedge biopsies taken at the end of the preservation interval and examples from the control and experimental groups are shown in Figure 5. Figure 5A shows the typical morphology of fresh tissue with an intact islet, which stains more lightly with toluidine blue than the surrounding acini that are characterized by the abundance of zymogen granules. In marked contrast, tissue stored for 24 h in UW-Viaspan shows some degenerative changes characteristic of ischemic injury (Fig. 5B). These include budding, rounding, and vacuolated cells. Breakdown of the acini is also apparent with separation of cells and degranulation. Comparable micrographs prepared from pancreases perfused for 24 h with KPS1 or Unisol-UHK are shown in Figure 5C and D, respectively. Figure 5C shows an intact islet surrounded by acinar tissue that clearly shows changes in the acini compared with fresh tissue (Fig. 5A). The exocrine cells of the acini appear to have a looser structure consistent with the moderate edema that developed during perfusion. Figure 5D shows an islet in pancreas perfused with Unisol-UHK having intact morphology surrounded by acinar tissue. In this specimen the exocrine tissue appears better preserved with less disruption than that detected in either the 24-h cold storage group (Fig. 5B) or the group perfused with KPS1 (Fig. 5C). Figure 5E and F illustrates the morphology of islets and acinar tissue in sections of pancreas perfused for 24 h with KPS1 after 30 min prior warm ischemia. Again some acinar disruption is apparent consistent with both tissue edema and warm ischemic changes, but the islets have an intact morphology comparable with those from islets subjected to hypothermic perfusion without prior warm ischemia.

Histology of pancreatic biopsies sampled for each of the treatment groups: (A) Fresh control pancreas; (B) 24 h cold storage in UW-Viaspan; (C) 24 h HMP perfused with KPS1 (WIT = 0); (D) 24 h HMP perfused with Unisol-UHK (WIT = 0); (E) 24 h perfused with KPS1 (WIT = 30 min); (F) 24 h HMP perfused with KPS1 (WIT = 30 min). Scale bars: 10 μm.
Discussion
Today, there is a considerable worldwide effort to further develop the concept for treating type 1 diabetes by transplanting islets, but clinical application of the techniques developed in animal models is fraught with many challenges. The source of the islets remains a primary concern, and isolation from donor pancreases demands resolution of questions concerning the source, supply, and condition of the donor organs. Reliance upon an adequate supply of human organs for this purpose is considered futile, such that alternative sources are actively been sought (6, 18, 19).
Pigs are considered the donor species of choice for xenogeneic islet transplantation for a number of compelling reasons. Pigs share many physiological similarities to humans and porcine insulin has demonstrated clinical efficacy for many years. Pigs are raised as a food source and provide an ethical source of donor islets by being housed in a controlled environment to ensure safety for porcine islet xenotransplantation (9). However, experiences in many laboratories over the past 10 years show that isolation of porcine islets appears to be more difficult (13, 15, 27, 30, 50), compared with the isolation of human (21), bovine (12), or rodent islets (37). Porcine islets are less compact and tend to fragment during the isolation procedure and during prolonged periods of in vitro culture (33). Moreover, the age, and even the strain, of the donor pig has been documented by several groups to markedly influence the islet isolation process, with young, so-called market size pigs (<6 months old) proving to be particularly difficult as a source of transplantable islets (7, 10, 50). Islets from adult pigs (>2 years old) offered not only higher yields, but retained the ability to preserve intact morphology during the isolation process and culture, in association with higher functional properties after transplantation. Despite the challenge encountered by many groups attempting to isolate islets from young pigs, donor pigs of market weight (<80 kg = <12 months old) are preferred to retired breeders (>200 kg = >2 years old) due to their abundance, lower animal and husbandry costs, and they are more suitable to meet regulatory guidelines for donor tissue for xenotransplantation. This study was undertaken to evaluate the efficacy of hypothermic machine perfusion (HMP) of pancreata from young pigs prior to islet isolation in light of recent developments in the efficacy of HMP for clinical organ preservation (29, 31, 32, 43).
Perfusion Technique
Earlier studies have demonstrated that pancreas hypothermic preservation by machine perfusion is feasible and can be safely extended to 24 and 48 h (2, 14, 26, 48, 49). Dedicated renal perfusion systems have been employed mostly (14, 26, 48, 49) after appropriate modifications required to accommodate the physiologic low flow and pressure needs of the pancreas (2). The latter helps avoid excessive organ edema that postsegmental transplantation and reperfusion has been documented to result in subcapsular bleeding, hemorrhagic necrosis, venous congestion, and hemorrhagic pancreaticoduodenal secretions (48). Transplanted islets isolated from 24-h perfused dog pancreata have been reported to result in 60% recipient survival post transplantation, providing similar outcome to fresh islets implantation (49). Islets isolated from human pancreas after 13 h of cold static storage and 4 h of hypothermic pulsatile perfusion on a Waters RM3 system were characterized by higher viable yield and stimulation index relative to cells isolated from organs that sustained more than 8 h of static storage alone (16). In light of this brief history of pancreas perfusion preservation and current resurgence of clinical interest in HMP of donor organs (43), we embarked upon the development of a perfusion technique for pancreas preservation prior to islet isolation (46). Considerable attention to detail was necessary in the development of a successful technique for perfusion of pig pancreata on the LifePort? perfusion machine. Both the surgical dissection, including the mode of cannulation, and configuration in the organ cassette on the machine proved to be important in the development of a technique that guaranteed 24-h continuous perfusion.
Surgical Model
Until recently, the anatomy of the pig pancreas was not well documented (11, 36, 39). Physiologically and topographically the pig and human pancreata are considered similar. The pancreas is an elongated retroperitoneal gland as shown in Figure 1. In both pigs and humans, the pancreas head is closely related to the proximal duodenum, but for pigs the pancreatic duct opening is found on the duodenum distal and separate from the common bile duct (39). There are a variable number of vessels originating from the splenic, hepatic, gastroduodenal, superior mesenteric, and celiac arteries that on an individual basis have irregular configuration of blood supply to the pancreas. Commonly, blood to the head is supplied by the posterior and anterior arcades arising from the gastroduodenal and superior mesenteric arteries (Fig. 1). In pig, the head does not surround the pancreaticoduodenal arteries and veins—the latter lie between the head and duodenum with the branches to the pancreas easily identifiable (39). The neck and the body of the pancreas are usually vascularized by the dorsal and inferior pancreatic arteries. The former can originate from the either the splenic, hepatic, or directly from the celiac arteries. The inferior pancreatic artery may begin from the superior mesenteric artery (SMA) under the neck of the pancreas and course toward the tail along the posterior inferior margin of the pancreatic surface in intimate contact with the gland. It can communicate with a varying number of splenic artery branches. The neck of the pancreas is also the site of the portal vein at the confluence of the splenic and superior mesenteric veins. The pancreas tail receives its blood supply mainly from the splenic artery.
In preparation for attaching the pancreas to the LifePort® perfusion machine, all exposed arterial branches on the margin of gastroduodenal and hepatic sides of the pancreas were meticulously identified and ligated to ensure uniform perfusion throughout the gland and allow the effluent to emerge only from the portal vein. This surgical approach proved optimal for pancreas perfusion/preservation for islet isolation as we have described elsewhere (44). The inclusion of the duodenum segment along with the pancreas head allowed for consistent perfusion. The leaks from the small vessels diverging from the pancreaticoduodenal arteries (the two loops around the head in Fig. 1, which in pig are between the head and duodenum) were eliminated by maintaining vessels integrity and therefore allowing for a uniform perfusion of the pancreas head and neck. Moreover, the opening of the pancreatic duct into the duodenum was preserved. This considerably facilitated pancreatic duct cannulation, by avoiding the difficulties encountered with retracted duct identification and cannulation, and preserved early duct branches. The latter were necessary to ensure good organ distension for gland digestion and islet isolation. The identification and tight ligation of all exposed vessels on the hepatic and gastroduodenal side of the pancreas was of high importance. Usually 12–14 vessels were tied up on the back table prior to perfusion. This eliminated the possibility for a pathway of “least resistance” for the flow throughout the organ that would have resulted in inconsistent organ perfusion, pressure, and temperature gradients across organ surface and suboptimal pancreas preservation.
Pancreas Cannulation for Perfusion
The LifePort® perfusion machine provided a controlled closed loop pulsatile perfusion at a set systolic pressure of 10 mmHg. In order to hook up the pancreas to this machine for consistent 24-h uninterrupted perfusion, several methods of cannulation needed to be evaluated. Initially, a 5–7-cm-long aortic segment, inclusive of both the superior mesenteric artery (SMA) and celiac trunk (CT) arterial openings, was used by ligating one end of the aortic segment and inserting a straight cannula (6.25 mm OD connector) into the other end. The cannula was attached to the infusion port of the LifePort® pump. However, this arrangement proved to be problematic due to the configuration of the clinical LifePort® machine, which was unable to reach and maintain the target perfusion pressure. Without going into detail, it was presumed that the inherent compliance in the aortic segment relative to the vascular resistance of the pancreas contributed to this phenomenon. While it was conceivable that the LifePort® could be modified to eradicate this problem we preferred to maintain the original LifePort® specification, which has FDA clearance for clinical use. To this end, alternative modes of cannulation were evaluated. The next attempt involved direct straight cannulation of the SMA and celiac trunk individually using two 4 mm OD luer-to-barb connectors joined together with a coupler attached to the pump infusion port (Fig. 2). The success of this arrangement proved to be dependent on anatomical differences from one pig pancreas to another. Specifically, we often encountered increased flow resistance and eventual pump stalling with incomplete perfusion. Upon investigation this proved to be due to occlusion of arterial side branches by the cannulas inserted into the SMA and CT as illustrated in Figure 2, which occurred in about one third of the cases. These flow problems were alleviated by using a proprietary seal ring cannula (10 × 35 mm; Organ Recovery Systems) illustrated in Figure 2C. These cannulas are designed to enclose the openings of the SMA and CT by clamping an aortic patch as shown in Figure 2C. In this way it provided a sealed flow link between the pancreas and the perfusion system without compromising the normal physiological flow even if early side branches were present. These constraints may be peculiar to the anatomy of juvenile pigs but the use of the seal ring cannula permitted consistent, trouble-free perfusion for 24 h (present study), and even 48 h (unpublished observations).
Islet Retrieval and Integrity
The primary purpose of this study was to evaluate the relative merits of hypothermic machine perfusion (HMP) of donor pancreases as a prelude to islet isolation. The general aim was to hopefully capitalize upon the benefits of HMP demonstrated for other organs (principally the kidney) as a means of better preservation during extended periods of storage, especially for suboptimum organs (43). To this end, these studies revealed an unexpected salutary effect of machine perfusion applied to the application of islet harvesting. The progressive development of edema during extended machine perfusion of organs is a phenomenon that is generally regarded as undesirable. Steps are usually taken to minimize the problem by adjusting the mechanical perfusion parameters such as flow and pressure, as well as the composition of the perfusate, to minimize the development of interstitial edema. Having resolved a technical problem with respect to cannulation of the pancreas that affects the efficiency of perfusion as described above, we determined that 24 h of HMP resulted in moderate edema in the gland compared to the controls that were simply flushed with and immersed in cold UW-Viaspan solution. Contrary to expectations, development of edema (up to 150%) did not prove deleterious, but was shown to be of considerable benefit by correlating with a more efficient disruption of the pancreas during enzymatic digestion to yield a significantly greater number of islets. Our hypothesis is that the edema causes sufficient disruption to the extracellular matrix and architecture of the pancreatic gland that the subsequent distension and digestion of the gland proceeds more effectively. This is evidenced by significantly shorter digestion times (Table 3), a more homogeneous digestion product (Fig. 4), and better gradient purification resulting in higher yields and purity of the final islet preparation. The structure and function of the islets per se did not appear to be compromised by the level of tissue edema encountered in these studies. Concerns that a change in the hydration of the isolated islets due to HMP might alter the buoyant density of the islets and thereby critically alter their ability to be separated from exocrine tissue on a density gradient did not appear to be a problem. This is presumably due to the fact that any inherent edema in the islets is counteracted by the pregradient incubation in UW solution, which is a hypertonic medium that would dehydrate the islets during the 30-min cold incubation prior to loading on the density gradient for purification. The latter is a routine step used in islet isolation protocols (24, 25).
The morphological integrity of the islets in situ in the preserved pancreata was evaluated by taking wedge biopsies at the end of the preservation interval. Changes associated with ischemia and the mode of preservation are illustrated and discussed in Figure 5. Dithizone staining of both the digest samples and purification fractions was used to evaluate the gross structure, purity and numbers of islets in the respective samples. Figure 4F shows the typical appearance of the highest purity preparations obtained from the HMP-treated pancreases. The islets have an irregular cluster shape that has been described by others as “grape-like” (35) and this appearance is characteristic of islets isolated from young pigs, reflecting the irregular shape observed in the endogenous pancreas prior to isolation (7). This characteristic irregular, fragmented appearance of islets from young pigs contrasts sharply with the more normal regular rounded shape of islets from adult pigs (7) and is not a reflection of the method of preservation. Figure 4B shows that islets from the fresh control pancreases have the same morphology.
The unanticipated mechanical benefit of HMP described above was achieved without compromising the quality of the harvested islets. The functional ability, in terms of their insulin secretory index, of the islets isolated from the perfused pancreases was equivalent to that of the controls including fresh pancreas. Moreover, the insulin content was significantly higher than the control group comprising pancreases stored statically in cold UW-Viaspan solution, which is currently the standard method employed clinically. It was further demonstrated that these effects and standards of preservation were achieved irrespective of the nature of the perfusate because equivalence was demonstrated using the two proprietary solutions, KPS1 and Unisol-UHK. Further improvements and benefits to this technique are anticipated by optimizing the composition of these baseline perfusates by adding cytoprotective agents design to minimize preservation and reperfusion injury (42).
Summary and Conclusion
Ever since the first experimental attempts to ameliorate type 1 diabetes by transplantation of allograft donor islets the field has been challenged by the need for improved methods of retrieving islets from donor pancreata. During the course of these studies we discovered that the technique of hypothermic machine perfusion preservation resulted in a greater yield of islets than either fresh or static cold stored pancreases. In fact, the increased yield over the fresh group of pancreases was 1.6–1.8 times greater and approximately two- to threefold greater than for pancreases preserved in UW-Viap-san. If this unanticipated finding is validated in further studies the implication would be very significant for the field of islet transplantation. Despite many efforts to improve the technique of islet isolation the field remains constrained by the limitations and vagaries of enzymatic digestion of a gland that comprises less than 5% endocrine tissue. Consequently, harvesting islets from a single donor pancreas often yields insufficient islet mass to reverse diabetes in a recipient, such that multiple donors often have to be considered for treating a single recipient. Our initial results using HMP strongly suggest that the development of a moderate degree of interstitial edema while preserving the integrity of the islets greatly facilitates islet isolation to the extent that approximately twice as many islets may be retrieved compared with nonperfused pancreases. Moreover, the salutary effects of HMP were also manifest after prior warm ischemia.
Footnotes
Acknowledgments
This work was funded in part by grants from the NIH (R44DK065508 and R44DK076326). The authors are presently or were previously employed by either Cell and Tissue Systems, Inc. or Organ Recovery Systems, Itasca, Inc. as indicated.
