Abstract
A large number of reports have shown that the two-layer method (TLM), which employs oxygenated perfluorochemical (PFC) and University of Wisconsin (UW) solution, is superior to simple cold storage in UW in islet transplantation. However, two recent large-scale studies showed no beneficial effect of TLM compared with UW storage in human islet transplantation. We reevaluated the effect of TLM by following three groups: group 1: UW simple storage; group 2: TLM performed by multiorgan procurement teams (not specialists of islet isolation); and group 3: TLM performed by specialists of islet isolation (Noguchi and Matsumoto). There were no significant differences between groups 1 and 2, whereas islet yields were significantly higher in group 3 compared with either group 1 or 2. Our data suggest that exact, complete performance of TLM could improve the outcome of islet isolation and transplantation. In this review, we describe the mechanisms of the TLM, the procedure of preoxygenated TLM, and the several possibilities for the reasons of the discrepancy.
Keywords
Introduction
Pancreatic islet transplantation is a promising option for the treatment of type 1 diabetic patients (7–9,30–32,42,44,51,54,55). Donor pancreata are usually preserved with University of Wisconsin (UW) solution. A large number of reports have shown that the two-layer method (TLM), which employs oxygenated perfluorochemical (PFC) and UW solution, is superior to simple cold storage in UW to preserve not only islet but also the whole pancreas transplantation (22,23,26,33,35,47,48,50). The TLM is especially important for preserving pancreata before islet isolation because it helps to preserve the organ, whereas UW preservation results in the deterioration of both islet isolation efficacy and posttransplant islet function (23,33,35). Successful islet transplantation from single donors in some diabetic patients has also been reported by using TLM in a clinical setting (7–9). With TLM, the pancreas is directly oxygenated by PFC during pancreas preservation and maintains a high level of adenosine triphosphate (ATP) in tissues, which maintains cellular integrity and retains parenchymal and nonparenchymal viability (20,56).
However, two recent large-scale studies showed no beneficial effect of TLM, compared with UW storage, on human islet isolation and transplantation (3,10). Kin et al. showed that there were no significant differences in ATP content in pancreatic tissue after preservation, pre- and postpurification islet yields, in vitro insulin secretory function, or utilization ratio of transplantation between the two groups (10). Caballero-Corbalán et al. showed that the ischemic tolerance of human pancreata was not able to be extended by TLM preservation and that TLM did not improve the isolation outcome for pancreata from elderly donors (3). There are several possibilities for these discrepancies compared to previously published results.
There are mainly two type of TLM: one is continuous oxygenation (14); another is oxygen-charged (static) TLM (34), which is widely used worldwide. In this review, we describe the mechanisms of the TLM, the procedure of preoxygenated TLM, and several possible reasons for the recent discrepancies.
Mechanisms of the TLM
PFC in which mice could breathe and survive was reported in the journal Science in 1966 (4). PFC are hydrocarbons in which all or most of the hydrogen atoms have been replaced with fluorine. The most attractive property of PFC is a very high capacity for dissolving respiratory and other nonpolar gases (17). Oxygen solubility in PFC liquids is about 20 to 25 times greater than it is in either water or blood plasma under the same condition, and dissolution capacity for oxygen in PFC liquids is typically 40–50 volume percent. In addition, a negligible oxygen-binding constant of PFC allows them to release oxygen more effectively than hemoglobin into the surrounding tissue. As a result of this unique combination of properties, PFC-based solution has been examined as an oxygen carrier for blood substitutes, myocardial protection, ventilatory support, cell culture (17), and organ preservation before transplantation (14).
The mechanisms of the TLM have been vigorously examined using canine pancreata. During preservation by the TLM, the pancreas is directly oxygenated through PFC and maintains an oxygen tension of about 60% of the normal physiologic level (23,26). During preservation by the TLM, pancreas grafts continuously generate ATP for up to 96 h (5,20,22,56). This ATP is used to drive a sodium-potassium pump to maintain cell integrity; and thus, TLM prevents pancreas swelling more effectively than UW solution (22,56). Furthermore, TLM improves the viability of vascular endothelium and microcirculation (13,25,28). After pancreas transplantation and reperfusion, the level of pancreatic tissue ATP remains high and heat shock proteins are strongly expressed (6). These heat shock proteins may work to provide an antireperfusion or cell repair property. The authors have shown that ATP levels are also enhanced in human pancreata stored by the TLM. This proves the ability of the TLM to oxygenate the human pancreas (12).
One of the most attractive effects of the TLM is the ability to resuscitate pancreas grafts that have experienced warm ischemic injury. Examination using canine pancreata showed that the grafts after 90 min WIT lost ATP. However, when the damaged pancreas was preserved/resuscitated by the TLM for 24–48 h at 4°C, the grafts regained the tissue ATP (15). Elevation of ATP levels was shown to be an excellent indicator for successful pancreas preservation by TLM and highly correlated with transplant success (15,38). Importantly, pancreatic ATP levels after TLM also correlated with islet yields after islet isolation (36). We developed oxygen-charged static TLM because the original TLM required continuous oxygenation of PFC, and this procedure is cumbersome for organ transportation (37). With this oxygen-charged TLM, pancreatic tissue ATP levels can be maintained for up to 18 h (37). In addition, warm ischemically damaged human pancreata could be oxygenized with elevation of ATP levels (16). If ATP levels are low after TLM, there should be two main reasons. One is a severely damaged pancreas and the other is inadequate oxygenation—and both yielded poor results. If ATP levels are not elevated after TLM, the results for both pancreas and islet transplantations are the same as using UW storage (10,11).
A recent report showed that improved islet yields from pancreata preserved in TLM is via inhibition of apoptosis mediated by the mitochondrial pathway (53). In this study, gene array analysis was used to analyze the expression of pro- and antiapoptotic genes in isolated islets. Higher expression of the inhibitor of apoptosis (IAP) and higher survival rates were observed in islets isolated from pancreata preserved in TLM compared with UW preservation. This was accompanied by decreased expression of Bad, Bax, and caspases 2, 3, 8, and 9. Therefore, the TLM leads to significant upregulation of antiapoptotic genes and inhibition of proapoptotic genes.
On the other hand, Papas et al. showed that the oxygen penetration depth is about 1 mm and is independent of pancreas diameter, while the oxygenated volume fraction is about 15% for a 2.5-cm-diameter pancreas and about 45% for a 1-cm-diameter pancreas. They concluded that pancreas oxygenation is limited during preservation with the TLM (52). In other words, these data suggest that the percentage of pancreas oxygenation by TLM depends on its thickness.
Procedure of Preoxygenated Static TLM
The procedure of preoxygenated TLM is as follows (34,59):
500 ml of UW solution, 500 ml of PFC, a 1 L Nalgene jar, and a pancreas-locating carriage are prepared. The carriage is made of stainless steel with a perforated plate and three adjustable legs. One leg is hollow with a luer fitting at the top and a small hole at the bottom. This leg is used to deliver respiratory oxygen.
Chilled, sterile PFC is poured into a standard 1-L Nalgene jar, overlaid with UW, and the carriage placed into the solution.
PFC is oxygenated just before pancreas preservation with 100% oxygen at 1–6 L/min for more than 30 min. Respiratory oxygen is supplied through oxygen-impermeable tubing, a 0.22-μm sterile syringe tip filter, and a 150-cm-long sterile extension line that is connected to the luer fitting on the hollow leg. Oxygen flows through the hollow leg and out through the small hole into the PFC. The small hole makes small oxygen bubbles that oxygenate the PFC more efficiently than large bubbles.
After removal of the spleen and excess fat (and the duodenum, if possible), the pancreas is placed under the carriage and the threaded legs adjusted to optimize the pancreas location at the PFC/UW interface. Two thirds of the organ must be covered with PFC (Fig. 1).
Two-layer method for pancreas preservation. After removal of the spleen and excess fat (and the duodenum, if possible), the pancreas is placed under the carriage and the threaded legs adjusted to optimize the pancreas location at the preoxygenated PFC/UW interface. Two thirds of the organ must be covered with PFC.
The jar is closed. The temperature is maintained at 4–9°C during preservation.
It is very important to oxygenate PFC after constructing the two-layer solution because oxygen easily escapes from the top surface of the PFC layer if it is not covered by the other solution. Sufficient oxygen supply to the PFC is also important and 100% oxygen at the rate of 1–6 L/min for at least 30 min is required to saturate the PFC (37). Moreover, removal of the spleen and excess fat (and the duodenum, when possible) along with adequate contact between the PFC and the pancreas are extremely important for pancreas oxygenation.
Clinical Studies of TLM
A total of 46 publications from several institutes focusing on pancreas preservation with TLM for islet isolation were identified on PubMed. Most of the publications have shown positive effects of utilizing TLM for pancreas preservation. In these publications, some articles reported on clinical studies that had been carried out. We showed the positive effects of TLM for pancreas preservation and human islet isolation, as assessed by the Edmonton isolation protocol (33). We also described an improvement in the transplant rate with the use of TLM (transplant number/isolation number; 3/3; 100%), compared with storage in UW solution alone (2/6; 33%) (37). Ricordi et al. at the University of Miami showed that 2 of 18 (11%) organs preserved with UW simple storage were used for clinical islet transplantation, whereas 8 of 15 (53%) organs preserved with TLM were used (54). Tsujimura et al. at the University of Alberta showed the addition of TLM (additional storage by TLM after UW storage) improved the transplant rate (5/7; 71%) compared with UW storage alone (5/14; 36%) (58). Moreover, the same group showed the evaluation of human islet isolation records from 142 human pancreata between January 2001 and February 2003. The study found that the success rates of islet isolations were at 62% in the TLM group but only 22% in the UW group with 10–16 h of cold storage (57). Hering et al. at the University of Minnesota showed that islets from pancreata preserved by TLM in combination with treatment of anti-CD3 antibody could result in successful single-donor islet transplants, although this study did not compare this approach against UW storage (8).
On the other hand, two negative studies have recently been published. Kin et al. at the University Alberta reported on the evaluation of human islet isolation records from 166 human pancreata between April 2003 and October 2005 (10). They did not find any significant differences between the TLM group and the UW group. However, they also showed that there were no significant differences in ATP content in pancreatic tissue after preservation. In addition, their success rates of islet isolation in both TLM and UW group were low (42.7% and 35.1%, respectively). These data suggest suboptimal performance of TLM. There are several possibilities for the poor performance of TLM in this report, such as incomplete oxygenation of PFC, prevention of O2 delivery to a pancreas due to a high volume of excess fat, insufficient contact between the pancreas and PFC solution (two thirds of the organ must be covered with PFC). In fact, in the evaluation of human islet isolation records from 142 human pancreata between January 2001 and February 2003, their findings actually suggest a positive effect of TLM (57). Caballero-Corbalán et al. at Uppsala University Hospital showed the analysis of the outcome of 200 human islet isolations (3). They found no beneficial effect of TLM compared with UW storage on human islet transplantation. Again, their success rate of islet isolation in both the TLM and UW groups were low (45% and 47%, respectively), suggesting problems with the way that the TLM was performed. Interestingly, the same group clearly showed the PFC-based one-layer method improved islet yield (1) and the isolation index (fragmentation rate of islets, which is calculated as the ratio between islet equivalents and islet number), compared with TLM (2). The data clearly suggest the beneficial effects of pancreas oxygenation by PFC. Although they did not show the concentration of ATP in the clinical study, the reason for no beneficial effect of TLM may be similar to that of Edmonton group.
We evaluated the effect of TLM by following three groups: group 1 (n = 9): UW simple storage; group 2 (n = 16): TLM performed by multiorgan procurement teams (not specialists of islet isolation); and group 3 (n = 9): TLM performed by specialists of islet isolation (Noguchi and Matsumoto). As shown in Figure 2, there were no significant differences between groups 1 and 2, whereas islet yields were significantly higher in group 3 compared with either group 1 or 2. In group 3, all cases (9/9) met the transplantation criteria, which means that the success rate of islet isolation was 100%. It may be the reason why the reports are inconsistent. Our data suggest that exact, complete performance of TLM could improve the outcome of islet isolation and transplantation.
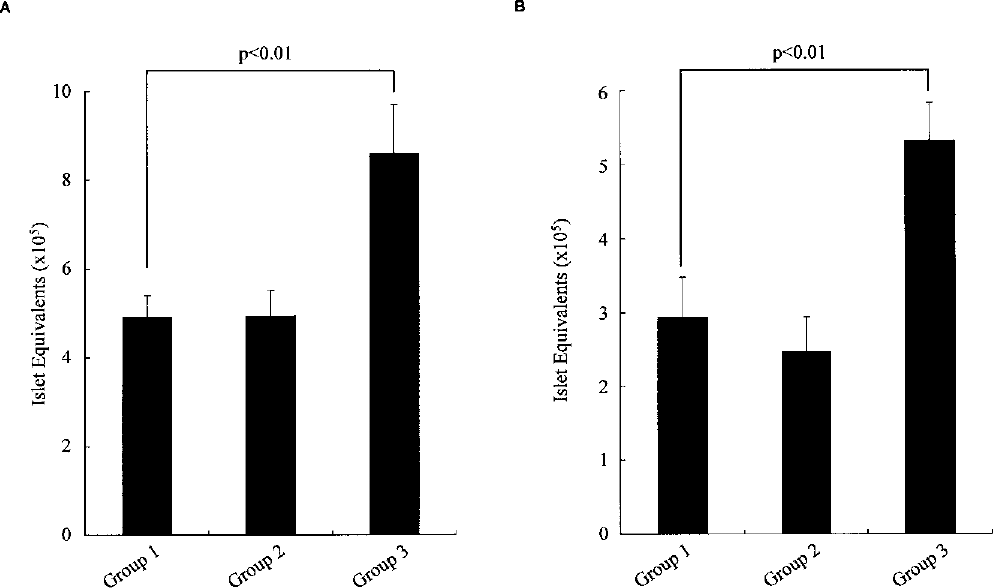
Islet yields before purification (A) and after purification (B). Group 1 (n = 9): UW simple storage; group 2 (n = 16): TLM performed by multiorgan procurement teams (not specialists of islet isolation); and group 3 (n = 9): TLM performed by specialists of islet isolation. There was no significant difference between groups 1 and 2, whereas islet yields were significantly higher in group 3 compared with either group 1 or group 2. Data are expressed as the mean ± SE.
Conclusion
Because there is still no reliable method to generate β-cells from stem/progenitor cells (40,41,43,49), improving the efficacy of islet transplantation seems the most realistic and prudent method to cure diabetes. Recent advances in islet transplantation, including the utilization of donors after cardiac death (DCD) (18,31,42), single donor islet transplantation (7–9), and living-donor islet transplantation (30,32) were based on advanced pancreas transport systems (9,19–27,34–37,56), revised immunosuppressant protocols (42,44,45), improved islet isolation methods (29,44), and enhanced islet engraftment (39,46). The improved viable islet yield resulting from the innovations, such as the procurement and the preservation of pancreata, means that transplantable islets can be recovered from marginal pancreata, including DCD donor pancreata. TLM has been used for not only islet transplantation from DCD donors but also pancreas transplantation from both DCD and donors after brain death in Japan because most pancreata from the donors in Japan are considered marginal. This advanced technology and strategy will be useful to expand the donor pool using marginal donors and to improve the outcome of islet transplantation.
Footnotes
Acknowledgments
The authors thank Dr. Carson Harrod for his careful reading and editing of this manuscript. This work was supported in part by the Juvenile Diabetes Research Foundation International (JDRFI); the Ministry of Education, Science and Culture, the Ministry of Health, Labour and Welfare; and Baylor All Saints Health Foundation.
