Abstract
The quality of donor pancreata is important for successful islet isolation. However, in some countries like Japan, the number of donor pancreata is very low; therefore, marginal donors have been used with less restrictive donor criteria. In order to use marginal donor pancreata, we established the Kyoto islet isolation method (KIIM). According to United Network for Organ Sharing (UNOS) in 2005, more than 6,000 pancreata were not clinically used in the US. In this study, we applied the KIIM for brain-dead donors and reevaluated donor usability based on the Japanese islet donor criteria. Islets were isolated with the Ricordi method using pancreata stored in University of Wisconsin (UW) solution (UW group) or by the two-layer method (TLM group) or the TLM combined with ductal injection (DI group). We implemented the KIIM (KIIM group) to confirm the effect of the KIIM on brain-dead donors. Donor charts in Texas from 2005 to 2006 were reviewed. If pancreata were not used clinically, the reason was reviewed and donors were reevaluated based on Japanese criteria. There were no significant differences of islet yield, viability, and purity between the UW and TLM groups. The DI group significantly improved islet yields and isolations were further improved in the KIIM group [UW: 251,663 ± 60,217 islet equivalent (IE); TLM: 243,738 ± 54,170 IE; DI: 498,639 ± 28,853 IE; KIIM: 678,286 ± 55,853]. The KIIM provided high-quality islets in high numbers from islet isolations from brain-dead donors. A total of 236 donor charts were reviewed and 194 pancreata (82%) were not used. Of these, 185 cases identified the reasons that the pancreata were not used. When we applied the Japanese criteria, an additional 82 cases out of 185 (44%) seem to be suitable for islet isolations. With the KIIM, more than 2,500 additional donor pancreata can be used for islet isolation in the US every year when the Japanese criteria are applied.
Keywords
Introduction
Islet transplantation is an option for the treatment of type 1 diabetic patients who maintain hypoglycemic unawareness despite maximal care (20,21). Successful islet isolation is the key to successful islet transplantation; the quality of the donor pancreas is important for successful islet isolation (1–3,5,7,15,22). Highly restricted criteria were made in order to assure high quality of donor pancreata for islet isolation (20). However, in some countries like Japan, the number of donor pancreata is very low; therefore, marginal-quality donor pancreata have been used. In order to use marginal donor pancreata, especially from non-heart-beating donors (NHBDs), we modified the Ricordi islet isolation method and developed the Kyoto Islet Isolation Method (KIIM) (11,14,16,17). Major implementations of this modification are pancreatic ductal preservation, modified two-layer pancreas preservation with Kyoto Solution and density-adjusted continuous density gradient purification (10). Using the KIIM, we successfully isolated islets and transplanted into type 1 diabetic patients in 17 out of 21 cases (81%) (8). This transplantation rate is remarkably high compared with previously published data (4,6,9). In fact, the transplantation rate of the majority of islet centers is less than 50%. Despite this low percentage, the majority of pancreata in the US, even from brain-dead heart-beating donors, are not clinically used. According to UNOS in 2005, only 1,466 pancreata out of 7,593 cases (19%) were clinically used, meaning that more than 6,000 pancreata were not used. However, we postulated that if we implemented the KIIM for islet isolation from brain-dead donors, we should be able to use marginal donor pancreata for islet isolation, meaning that more pancreata could be clinically used.
In this study, we first implemented the KIIM for islet isolation from brain-dead donors. Then we evaluated pancreas donors in our local area by reviewing well-documented donor charts and estimated how many pancreata in the US could be used for islet isolation when we apply the Japanese islet donor criteria in combination with an improved islet isolation technique, such as the KIIM.
Materials and Methods
Pancreas Procurement and Islet Isolation
From January 2005 to August 2007, we performed 27 islet isolations using brain-dead donor pancreata. In all cases, pancreata were stored less than 8 h. All 27 islet preparations were manipulated according to Current Good Manufacturing Practice (cGMP) in the cell processing facility at Baylor Institute for Immunology Research.
The first protocol included the regular Ricordi islet isolation method (19,20) and pancreata were preserved in UW solution (N = 6, UW group) or by the oxygen static charged two-layer method (N = 13, TLM group) (13). For the next protocol, the islet team participated in pancreas procurement and pancreatic ductal injection was introduced at the donor site (N = 3, DI group) (10,11,16). Then pancreata were preserved by the oxygen static charged TLM and islets were isolated by the Ricordi method. For the final protocol, we implemented the KIIM (N = 3, KIIM group) (10,11,17). The key procedures of the KIIM are described as follows. We procured pancreata in conjunction with multiorgan procurement. We removed the duodenum and spleen from the pancreas at the procurement site. A cannula was immediately inserted into the procured pancreas through the main pancreatic duct from the direction of the pancreatic head and M-Kyoto solution was administered intraductally (ductal injection). M-Kyoto solution is 100,000 U/L of ulinastatin (Mochida Pharmaceutical Co. Ltd., Tokyo, Japan) in ET-Kyoto solution (Otsuka Pharmaceutical Co., Naruto, Japan).
For pancreas preservation during transportation, we use an oxygenated perfluorocarbon/M-Kyoto solution-based two-layer method. Before islet purification, we checked the density of the isolation aggregates with a test tube density adjustment method as follows (10). Six discontinuous density test tubes were prepared with 5 ml each of purification solution of different densities (1.085, 1.090, 1.095, 1.100, 1.105, and 1.110 g/cm3). The different densities were achieved by adjusting the ratio of iodixanol (OptiPrep, Axis-Shield PoC AS, Norway) to M-Kyoto solution. Samples were taken from the isolation aggregates and added to the discontinuous density test tubes. The tubes were spun at 1,000 rpm for 5 min and the densities of aggregates were determined by whether they pelleted or floated in the different density solutions. Islets were purified with a continuous density gradient of iodixanol/M-Kyoto solution in an apheresis system (COBE 2991 cell processor, Gambro Laboratories, Denver, CO). The heavy density solution was chosen according to the test tube density adjustment method described above and the gradient was achieved by varying the ratio of iodixanol to M-Kyoto solution.
Islet Evaluation
Islet evaluation was independently judged by two investigators. Islet yield was determined with dithizone staining (2 mg/ml; Sigma Chemical Co., St. Louis, MO) under an optical graticule and converted into a standard number of islet equivalents (IE, diameter standardizing to 150 μm) (12,18). Purity was assessed by comparing the relative quantity of dithizone-stained tissue to unstained exocrine tissue. Islet viability was evaluated using fluorescein diacetate (FDA) and propidium iodide (PI) staining to visualize living and dead cells simultaneously (12,18). The recovery rate after purification was determined by dividing IE before purification by IE after purification. For qualification of transplantation, we used the original Edmonton protocol criteria (20). For a qualified transplantation, islet yield should be more than 5,000 IE/kg patient body weight (we used 60 kg as a default body weight, which means that the total islet yield should be more than 300,000 IE), viability above 70%, purity of more than 30%, and a tissue volume of less than 10 ml (20). In addition, the final product needs to have negative gram staining and have endotoxin levels below 5 EU/kg patient body weight (we used 60 kg as a default so that the total endotoxin level should be less than 300 EU).
Isolated islets from all three cases of the DI group were transplanted into two type 1 diabetic patients. One patient received two islet infusions and the other patients received one. We submitted the data obtained from the KIIM group to Food and Drug Administration (FDA) for the approval of clinical use of the KIIM.
Donor Chart Review
Two hundred and thirty-six donor charts from well-documented multiorgan procurements in Texas (Southwest Transplant Alliance; Dallas, TX and LifeGift Organ Donation Center; Fort Worth, TX, USA) from 2005 to 2006 were reviewed. Twenty-nine pancreata were used for whole pancreas transplantations and 13 pancreata were used for islet isolation. Therefore, 194 (82%) pancreata were not used. In the unused 194 cases, the reasons that they were not used were also reviewed based on donor-specific inclusion and exclusion criteria at Baylor. Then unused donors were reevaluated based on Japanese islet specific donor inclusion and exclusion criteria.
Statistics
Values for the data collected represent means ± SE. Four groups were compared by means of ANOVA followed by Fisher's Protected Least Significant Difference post hoc test. The ratios between the two groups were compared using Fisher's exact test. Values of p < 0.05 were considered significant.
Results
Donor Characteristics
In terms of donor characteristics among the four groups (UW group, TLM group, DI group, and KIIM group), there were no significant differences in donor age, body mass index (BMI), pancreas weight, or cold ischemic time (CIT) (Table 1).
Donor Characteristics

There were no significant differences in all categories among all groups. UW, University of Wisconsin solution; TLM, Two-layer method; DI, TLM + ductal injection; KIIM, Kyoto islet isolation method; BMI, body mass index; CIT, cold ischemic time.
Islet Isolation Outcomes
There was no significant difference in islet yields between the UW group and TLM group (251,663 ± 60,217 IE UW group vs. 243,738 ± 54,170 IE TLM group) (Fig. 1). Islet yields were significantly increased when we introduced ductal injection to the TLM method (498,639 ± 28,853 IE) (Fig. 1). The use of the KIIM further improved islet yields compared to the DI groups (678,286 ± 55,853 IE) (Fig. 1). There were no significant differences of viability or purity of isolated islets among the four groups (Table 2). The recovery rate after purification was significantly higher in the KIIM group compared with the UW and TLM groups (Table 2). Qualifying the transplantation criteria was 2/6 (33%) in the UW group and 4/13 (31%) in the TLM group. After the addition of ductal injection, both DI and KIIM groups had a 100% success rate for qualification of transplantation. The success rate of islet isolation was significantly improved with ductal injection (DI and KIIM groups) [6/19 (32%) without ductal injection (UW, TLM) vs. 6/6 (100%) with ductal injection (DI, KIIM), p < 0.01].

Islet yields of four different groups, including the Ricordi method using UW-stored pancreata (UW), the Ricordi method using TLM-stored pancreata (TLM), the Ricordi method using ductal preservation (DI), and the KIIM (KIIM). Islet yields were significantly higher in the DI group compared with the UW and TLM groups (*p < 0.0001 UW vs. DI and TLM vs. DI). Islet yields were significantly higher in the KIIM group compared with all other groups (*p < 0.0001 UW vs. KIIM and TLM vs. KIIM, **p < 0.01 DI vs. KIIM).
Islet Isolation Outcomes for the Different Groups

p < 0.01 versus TLM and p < 0.05 versus UW.
The clinical results in the DI group are shown in Figure 2. The daily insulin dose was gradually decreased after the first islet transplantation and the patient became insulin independent after the second islet transplantation (Fig. 2, top). Fasting glucose levels became well controlled after the first islet transplantation and further improved after the second islet transplantation (Fig. 2, middle). HbA1C levels gradually decreased after the first islet transplantation and reached normal range (Fig. 2, bottom).
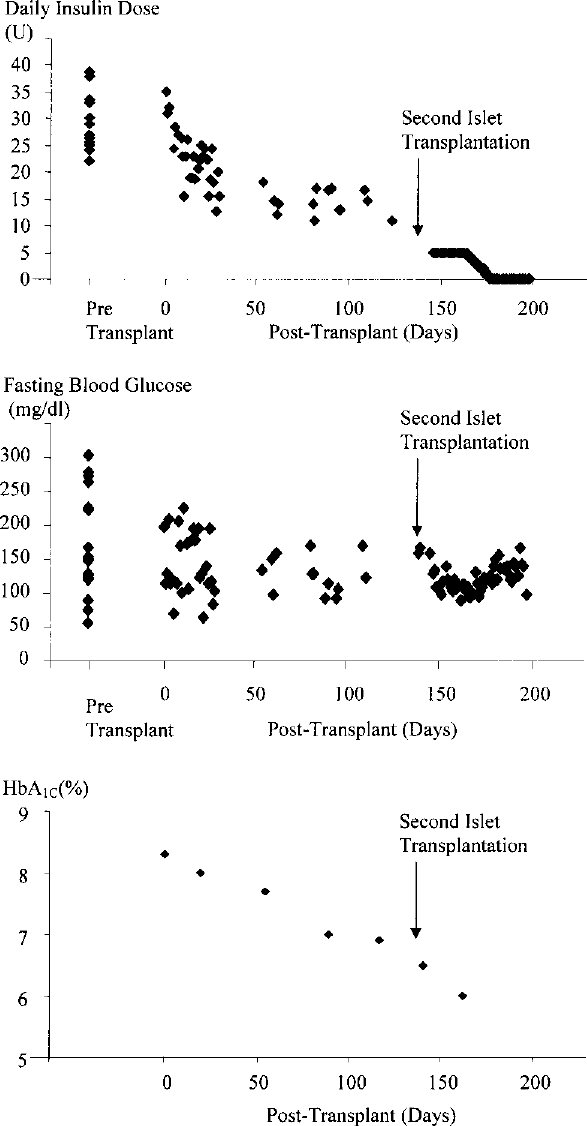
Daily insulin dose (top), fasting blood glucose (middle), and HbA1C (bottom) before and after two islet transplantations with the islets from the DI group. Daily insulin dose decreased after the first islet transplantation and the patient became insulin independent after the second islet transplantation (top). Fasting blood glucose became more stable after the first islet transplantation and became more stable after the second transplantation (middle). HbA1C continuously decreased after the first islet transplantation and achieved normal range after the second transplantation (bottom).
Donor Chart Review
Donor chart review revealed that out of 194 unused cases, the reasons that the pancreata were not used were identified in 185 cases, based on Baylor islet-specific donor inclusion and exclusion criteria (Table 3). Forty cases (21.6%) were aborted during procurement, 37 pancreata (20.0%) were not used due to donor age, 29 pancreata (15.7%) were not recovered due to high glucose, 17 pancreata (9.2%) could not be used due to diabetes, in 15 cases (8.1%) the family did not consent to the procurement, 14 cases (7.6%) had infectious disease, in nine cases (4.9%) no specific reasons were mentioned, eight cases (4.3%) were not attempted due to cardiac arrest events, seven cases (3.8%) were not performed for social reasons, five pancreata (2.7%) were not used due to fatty pancreata, and elevated creatinine levels prevented pancreas procurement in four cases (2.2%) (Fig. 3).

Reasons and number of unused pancreata from brain-dead donors in Texas. Out of 194 unused cases, the reasons that the pancreata were not used were identified in 185 cases, based on Baylor islet-specific donor criteria. The reasons were reevaluated by the Japanese criteria. Eighty-two cases out of 185 (44%) unused pancreata were qualified for islet donation based on the Japanese criteria. Values are number, percentage of total.
Comparison of Baylor and Japan's Donor-Specific Inclusion and Exclusion Criteria

Next, unused donors were reevaluated based on Japanese islet-specific donor inclusion and exclusion criteria (Table 3). Infection, lack of family consent, diabetic pancreata, and social reasons were considered as not qualified cases (63 cases, 34%). Based on the Japanese criteria, high glucose without diabetes, fatty pancreas, or elevated creatinine levels are acceptable for islet donors; therefore, we counted those as qualified pancreata (47 cases, 25%). Ages less than 25 years old and up to 70 years old and cardiac arrest events with less than 30 min of warm ischemia are also acceptable. In some cases where the procedure was aborted, there were no contraindications. Out of 85 of those cases (age, cardiac arrest, aborted cases), 25 cases were qualified for islet donation. A total of 82 cases (44%) were qualified for islet donation.
When we extrapolated the 44% qualification rate that we identified here, but of the currently unused 6,000-plus pancreata, more than 2,500 pancreata could be used for islet isolation in 1 year in the US.
Discussion
Currently, we have established the KIIM for islet isolation with marginal donor pancreata (8,10,11,14,17). With the KIIM, we successfully isolated transplantable islets from NHBDs in 17cases out of 21 isolations (81%) (8). All transplanted islets secreted insulin and all of the patients improved glycemic control without hypoglycemic unawareness. Three out of five multiple transplantation cases (60%) became insulin independent (8).
In this study, we first compared UW and TLM storage. We then tested ductal injection (DI group) and KIIM (KIIM group) to confirm the effect of DI and KIIM on islet isolation from brain-dead donors. We did not see any significant differences between the UW group and the TLM group. Previously, we demonstrated that TLM improved islet yields and the effect was more apparent when pancreata were stored for longer periods (12). In this study, we restricted CIT to less than 8 h. Therefore, the effect of the TLM became less apparent. In addition, in the previous study, members of the islet team procured pancreata for the TLM. In this study, a multiorgan procurement team procured pancreata and stored them by TLM. The University of Alberta group also demonstrated that the TLM had no significant impact on islet transplantation (6). In their study, the multiple organ procurement team but not islet team procured pancreata. These facts suggest that the type of procurement team has a significant impact on islet isolation. This is because the TLM requires expertise to perform properly (e.g., immersing at least two thirds of pancreas into PFC, removing fat surrounding pancreas before storage).
Even though the number is small, introduction of ductal injection and procurement by the islet team significantly improved islet yield; and introduction of the KIIM further improved islet yields. One of the major advantages of the KIIM is the density adjusted continuous density gradient with Kyoto solution plus iodixanol (10). This method enabled us to maximize the recovery rate after purification; and this study showed the significant improvement in recovery rate after purification. Therefore, the KIIM seems beneficial for islet isolation from brain-dead donors. The fact that transplanted islets from the DI group resulted in excellent glycemic control and insulin independence by the recipient further supported this concept. In order to confirm the benefit of the KIIM for isolating islets from brain-dead donors, further research is necessary to increase the case number. This is our current ongoing research.
In this study, we reevaluated pancreas donors in the Texas area with the Japanese criteria. Previously, we demonstrated that islet isolation by KIIM using NHBDs that had elevated blood creatinine levels and/or transaminase levels, or who had experienced cardiac arrest events, which are current contraindications in the US for donor eligibility, did not have a significant impact on the isolation results (8). Therefore, those factors could be eliminated from the list of contraindications and, in fact, they are not contraindications under the Japanese criteria. Under our current criteria in the US, ages of less than 25 years old are contraindication for islet donors. However, it was recently shown that younger donors could provide high-quality islets even though isolation of islets from young donors is difficult (5). In fact, we isolated islets with the KIIM from a 14-year-old donor pancreas. From this isolation, we obtained approximately 500,000 IE islets that resulted in successful islet transplantation. Therefore, we propose that donor ages of less than 25 years should not be a contraindication for islet donors in the US. The Japanese criteria do not impose such a limitation.
We showed that more than 40% of the unused pancreata were actually suitable for islet isolation under the Japanese criteria with the KIIM. We conclude that with the KIIM and the Japanese criteria, more than 2,500 additional donor pancreata might be used for islet isolations annually in the US.
Footnotes
Acknowledgment
We thank Dr. Carson Harrod and Yoshiko Tamura for their careful review of this manuscript. This research is partly supported by All Saints Health Foundation and Otsuka Pharmaceutical Company. This research was partially presented at the 34th annual meeting of The Japan Society for Organ Preservation and Medical Biology.
