Abstract
Lactate levels in blood serve as a key physiological indicator of stress and welfare in animals during slaughterhouse processes. Elevated lactate levels lead to an accumulation of lactic acid in the muscle tissue. This rapid lactic acid production lowers the muscle pH, potentially resulting in undesirable meat quality traits such as pale, soft and exudative (PSE) meat. Optical spectroscopic techniques offer a potential solution for measuring blood lactate levels in real-time as a process analytical technology (PAT) tool in slaughterhouses. This article demonstrates the potential of near-infrared (NIR) and Raman spectroscopy for measuring lactate levels in animal blood after exsanguination. All experiments were carried in real-world slaughterhouse conditions. Furthermore, regression modelling was performed to relate spectra with reference lactate values. The results demonstrated that both and NIR and Raman spectroscopy can predict lactate content in blood in slaughterhouse conditions. Furthermore, the relation of lactate values and final meat quality was also evaluated. The findings from the study can be used to develop PAT systems for continuous blood measurement in slaughterhouse conditions.
Introduction
Lactate levels in blood are a key physiological indicator of stress and welfare in pigs during slaughterhouse processes, as lactate is produced through anaerobic metabolism that increases when animals experience stressors such as rough handling, overcrowding, prolonged transportation or inadequate lairage conditions. Elevated lactate reflects the physiological burden of acute or chronic stress and is strongly linked to impaired meat quality, since rapid lactic acid accumulation lowers muscle pH post-mortem and can lead to undesirable traits such as pale, soft and exudative (PSE) meat. Scientific studies1,2 show that higher lactate concentrations correlate with increased cortisol levels, faster pH decline, disrupted protein structure, reduced water-holding capacity and greater drip loss, while stress-induced depletion of muscle glycogen further compromises the extent of post-mortem acidification required for optimal texture and flavour. Monitoring lactate levels at slaughterhouses therefore provides an objective, quantifiable metric for evaluating handling practices and compliance with welfare standards. 3 By minimising stress through improved transport, handling and pre-slaughter care, slaughterhouses can reduce lactate levels, promote a more controlled pH decline, preserve meat quality attributes such as tenderness, colour and shelf life, and ultimately enhance both animal welfare and product value.
Optical spectroscopy provides a promising approach for quantifying lactate levels in animal blood as part of welfare and meat-quality monitoring at slaughterhouses. By analysing how light interacts with biological tissues, spectroscopy can reveal molecular and biochemical information that reflects the chemical composition of a sample, including metabolites such as lactate. Among the most commonly applied techniques are near-infrared (NIR) spectroscopy 4 and Raman spectroscopy, 5 both of which are sensitive to vibrational or overtone frequencies associated with lactate and related metabolic compounds. These spectroscopic methods offer several advantages over conventional biochemical assays, notably the potential for rapid, non-invasive and reagent-free measurements. Because spectra can be collected quickly and processed using chemometric models, spectroscopy enables high-throughput or even inline assessment of physiological markers during slaughter operations. This capability is particularly relevant given the strong relationship between lactate, animal stress and final meat quality: integrating spectroscopic lactate measurement into slaughterhouse workflows would allow continuous monitoring of welfare conditions, early detection of stress-related deviations and real-time optimisation of handling and processing practices. By providing fast and objective information, optical spectroscopy complements and enhances traditional welfare assessment tools, supporting both improved animal treatment and consistent product quality.
The aim of the study was to explore NIR and Raman optical spectroscopy techniques to predict the blood lactate content in real slaughterhouse during animal exsanguination. Furthermore, the relation of blood lactate level with key meat quality parameters were also verified in slaughterhouse conditions.
Materials and methods
The study involves three different trials carried out at different slaughterhouses of Vion Foods Group, The Netherlands. The first trail was from year 2020, where blood samples immediately after slaughtering were measured with lactate analyser (Lactate Scout 4, EFK Diagnostics, United Kingdom) and near-infrared (NIR) spectrometer (Labspec, ASD, Malvern Panalytical, USA). The NIR spectrometer had inbuilt halogen light source for illuminating the blood samples. For NIR measurement the blood were samples in plastic cups and placed below the NIR reflection probe with ~2 cm of distance between probe head and blood. Lactate measurements were performed before spectral measurements. The spectral measurements were performed in reflectance mode as that was the easiest mode of measurements available at the slaughterhouse. During the second trail in 2022, the blood samples were measured with lactate analyser and dip probe Raman spectrometer (Wasatch, Photonics, USA). The Raman spectrometer used a laser source of 785 nm to excite the samples. The blood samples were presented in the plastic cups where the dip probe was immersed in the blood for measurements. After every scan, the dip probe was cleaned with paper to remove remain of blood from earlier samples. The integration time was 5 s. To find the relation in between spectral techniques and lactate values, partial least-squares (PLS) regression was performed. PLS was used as signals were multivariate. To optimise the number of factors for PLS, leave-one-sample-out (LOO) cross-validation analysis was performed. The thirds trial was from May 2023, where meat samples of the animals for which lactate content was measured, went through regular meat quality analysis. Correlation between lactate content and different meat quality parameters were explored.
Results
High blood lactate at slaughter indicates that the pig experienced acute stress shortly before death, which accelerates anaerobic glycolysis and results in rapid lactic acid production in the muscle immediately after slaughter. This lactic acid accumulation lowers muscle pH, meaning that higher blood lactate is associated with a faster and deeper early pH decline. In our slaughterhouse trial (Figure 1), we observed a negative correlation between lactate and pH₄₅min, consistent with the understanding that a rapid pH drop while the carcass is still warm leads to protein denaturation and myofibrillar shrinkage, thereby reducing water-holding capacity and increasing fluid loss during storage. Accordingly, our results also showed a positive correlation between lactate and drip loss. Because stressed animals often have depleted glycogen reserves and accelerated glycolysis, they continue to exhibit reduced pH in the early post-mortem period, which we confirmed through the negative correlations observed between lactate and pH3h as well as pH6h. For ultimate pH (pH24h), no clear trend was detected in our data, likely reflecting the complex influence of pre-slaughter stress and glycogen depletion. Regarding colour traits, we observed slight positive correlations between lactate and L*, a* and b* values. Higher lactate promotes a rapid early pH drop, resulting in protein denaturation and increased light scattering that produces paler meat, consistent with the elevated L* values characteristic of PSE pork. The decrease in redness (a*) also aligns with light scattering effects, causing meat to appear less red, while b* values may increase slightly for the same reason.
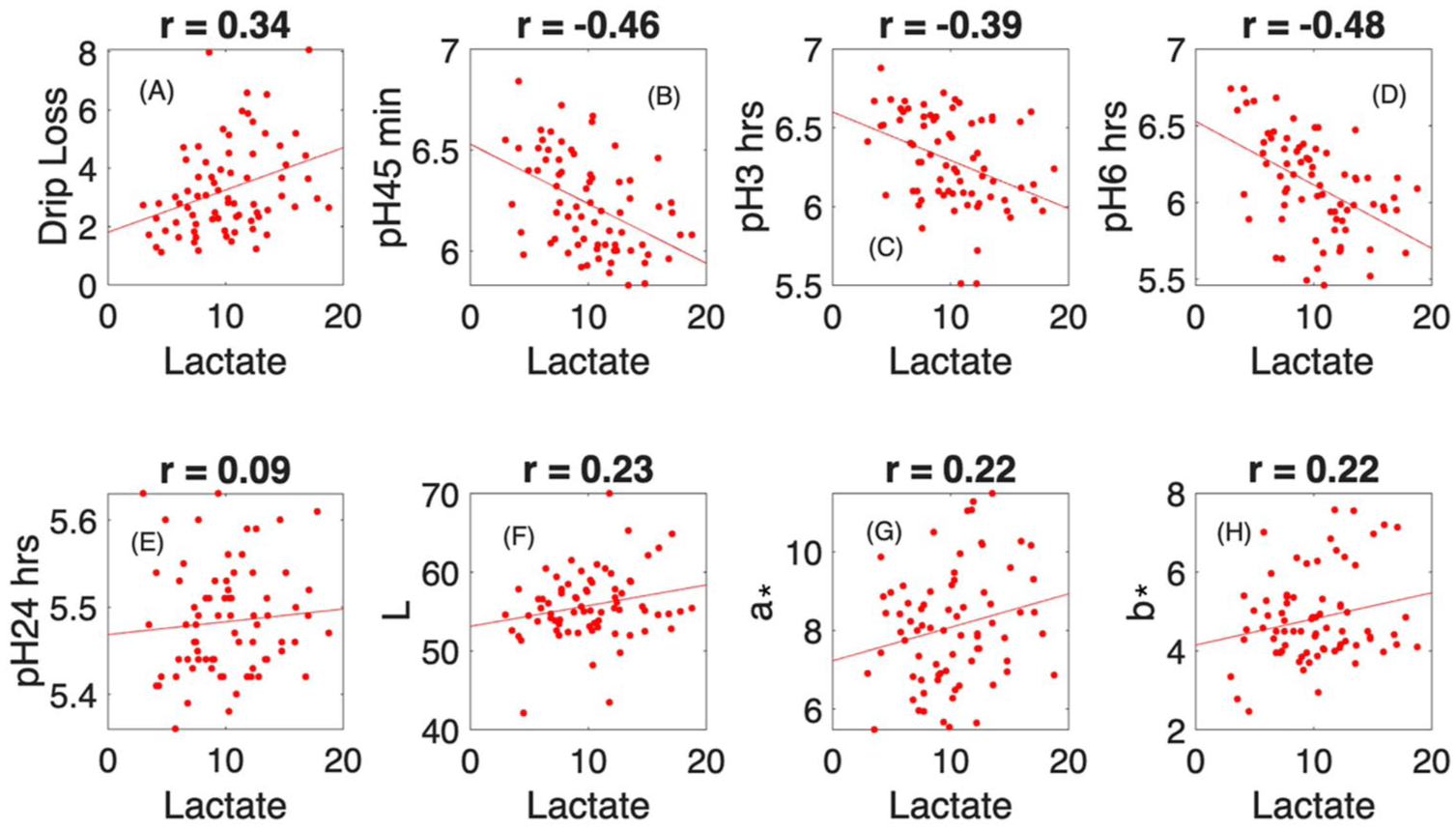
Relation of lactate content in blood with several key quality indicator for meat: (a) drip loss, (b) pH 45 min, (c) pH 3 h, (d) pH 6 h, (e) pH 24 h, (f) L, (g) a* and (h) b*.
The blood lactate ranges from the two trails had a mean ± standard deviation range of 10 ± 3.45 mMol/L, where for NIR modelling, the lactate range was 11.32 ± 3.24 mMol/L (80 blood samples), and for Raman data modelling, the lactate range was 9.92 ± 3.58 mMol/L (57 blood samples). For both the NIR and Raman, the lactate range was similar. The PLS regression cross-validation analysis results are shown in Figure 2. As can be noted, both techniques carried variation related to lactate, but Raman carried higher variation than the NIR. A reason for better explain of lactate variation by Raman could be high sensitivity towards analytes, while NIR signals are usually attenuated by water. For model optimisation, both techniques achieved a clear minimum point for the cross-validation curve depicting that modelling avoided any potential overfitting.
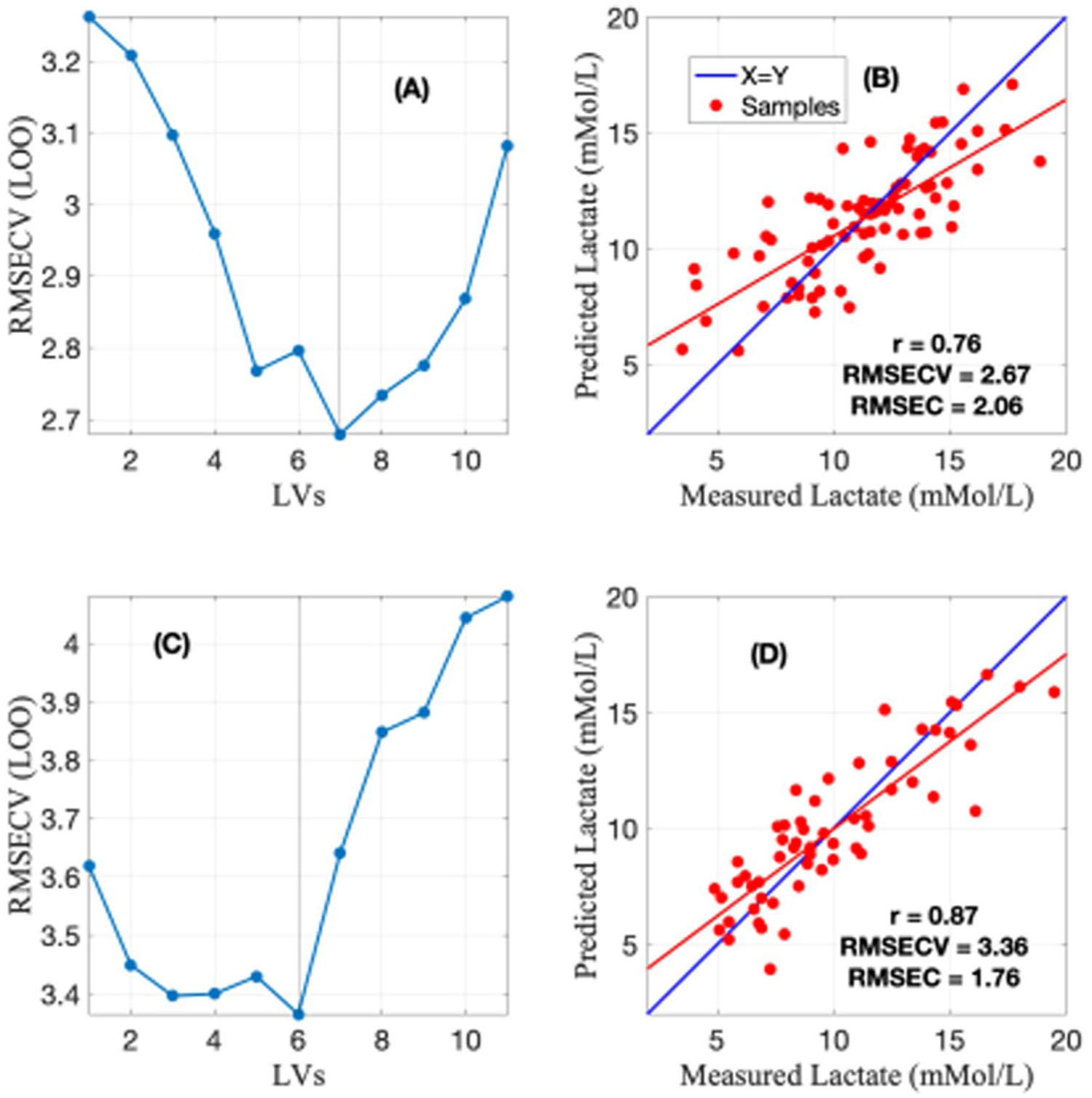
(a) PLS cross-validation for NIR indicating a minimum at seven latent variables, (b) prediction plot for NIR, (c) PLS cross-validation for Raman indicating a minimum at 6 and (d) prediction plot for Raman.
Conclusions and outlook
The observed relationships between lactate, pH decline, drip loss and colour parameters demonstrate the strong impact of pre-slaughter stress on both physiological responses and final meat quality in pigs. The blood lactate concentrations measured across the two trials showed comparability between the two spectroscopic approaches. The PLS cross-validation results further confirmed that both NIR and Raman spectra contained meaningful variation related to lactate levels, with Raman explaining slightly more variance, likely due to its higher molecular specificity and lower susceptibility to water interference. These findings collectively highlight the strong potential of optical spectroscopy as a PAT tool for real-time welfare monitoring in slaughterhouses and control of meat quality. Future work should focus on developing inline-compatible sensor configurations, improving spectral acquisition under industrial conditions and integrating automated chemometric pipelines into slaughter line workflows. With further validation, NIR and Raman spectroscopy could enable continuous, non-invasive and objective monitoring of blood lactate immediately after exsanguination, supporting both improved animal welfare compliance and better control of downstream meat quality.
Footnotes
Acknowledgements
Pork Quality and Safety Assessment Tools (PorQSAT) LWV19176.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
