Abstract
Purpose
The cardiotoxic effects of anti-cancer agents on cardiomyocytes are a significant challenge for physicians. The current experiment aims to explore the potential cardioprotective effects of cardiovascular agents, i.e., eplerenone, empagliflozin, and colchicine by restoring mitochondrial fusion, reducing fission and mitigating the severity of fibrosis in doxorubicin-treated H9c2 cells.
Materials and Methods
The viability of H9c2 cells are assessed using the MTT assay. Additionally, the underlying mechanisms in H9c2 cells subject to treatment with eplerenone, empagliflozin, colchicine and doxorubicin were explored through a combination of techniques, including fluorescence microscopy, siRNA transfection, flow cytometry, and western blot analysis.
Results
After exposure to doxorubicin, H9c2 cells exhibit a decrease of mitochondrial fusion, an increase in myocardial fibrosis, and perturbed mitochondrial fission. However, treatment with these cardiovascular agents have effectively counteracted these doxorubicin-induced alterations with the restoration of mitochondrial dynamics and the alleviation myocardial fibrosis.
Conclusion
This study suggests that eplerenone, empagliflozin, and colchicine possess direct cytoprotective actions in an in vitro model of H9c2 cells with the potential to serve as therapeutic agents of doxorubicin-induced myocardial fibrosis.
Introduction
Currently, patients with cancer have experienced a substantially prolonged lifespan due to the benefits associated with the rapid advances and extensive application of novel anti-cancer therapies. While the side effects of these anti-cancer agents on the cardiovascular system are becoming increasingly common, such as heart failure, hypertension, coronary heart diseases and arrhythmia. Furthermore, the management of cardiovascular complications in cancer patients has emerged as a major clinical challenge. Myocardial fibrosis is a typical pathological process associated with various cardiac disorders. It can be typically present in conditions such as myocardial infarction, heart failure, hypertrophic cardiomyopathy, dilated cardiomyopathy, and others. As one of the predominant characteristic of which, the excessive accumulation of collagens in the extracellular matrix (ECM) of the myocardium can gradually lead to myocardial stiffness, contractility impairment and electrical disturbances and further result in structural remodeling and impairment of global cardiac function and ultimately contribute to adverse cardiac outcomes.1–3 Importantly, the extent and severity of myocardial fibrosis have been identified as important prognostic factors for adverse outcomes in different cardiac diseases. 1
Doxorubicin is indeed a widely used chemotherapy drug with a significant improvement in the prognosis for various malignant tumors, but its use is associated with the risk of cardiotoxicity, including the development of cardiomyopathy and congestive heart failure (CHF). Cardiotoxicity induce by doxorubicin is dose-dependent, and approximately 10% of patients treated with doxorubicin may develop to cardiomyopathy. 4 Myocardial fibrosis and the subsequent myocardial remodeling have been identified as key factors in the occurrence and progression of doxorubicin-induced cardiomyopathy and impairment of cardiac function. 5 Elucidating the mechanisms involved in doxorubicin-induced fibrosis can facilitate the development of new therapeutic strategies to decrease the possibility or the severity of cardiotoxicity and improve the clinical prognosis of patients under anti-cancer treatment with these agents. 6 Furthermore, doxorubicin can directly affect cardiac fibroblasts, which are the key cells responsible for collagen synthesis and turnover. Understanding the underlying mechanisms of doxorubicin-induced myocardial fibrosis is of great scientific significance in order to develop strategies to prevent or alleviate this adverse effect. Various approaches have been explored, including the use of cardioprotective agents, antioxidants, and targeted therapies aimed at reducing myocardial fibrosis and preserving cardiac function.
Cardiac energy metabolism encompasses an intricate network of interconnected biological pathways, with mitochondria occupying a pivotal position. These organelles are indispensable for the generation of ATP, the primary energy source for cellular activities, contributing to over 95% of ATP production in cardiomyocytes.7,8 The mitochondrial dynamics, including their fusion, fission, and degradation, play a fundamental role in their optimal energy production. Striking a harmonious balance between fusion and fission brings about various benefits to mitochondria, such as efficient trafficking of components, enhanced homogeneity among mitochondrial populations, and optimized oxidative phosphorylation. Mitochondrial fission is the division of a mitochondrion into two smaller mitochondria. The central mediator of mitochondrial fission is dynamin-related protein 1 (DRP1), a GTP-hydrolyzing enzyme. Mitochondrial fusion requires the action of three large GTP-hydrolyzing enzymes belonging to the dynamin superfamily. The mitofusins, Mfn1 and Mfn2, are situated on the mitochondrial outer membrane and are essential for outer membrane fusion. Comprehending the intricate interplay between cardiac energy metabolismvand cardiac dysfunction holds utmost significance in the development of therapeutic strategies for cardiovascular diseases. By targeting mitochondrial and enhancing cardiac energy metabolism, promising avenues can be explored to alleviate cardiac dysfunction and promote overall cardiac health.
Agents i.e. eplerenone, empagliflozin and colchicine have been shown to have various cardioprotective effects in clinical trials and to be able to decrease cardiovascular events and improve the prognosis.9–11 However, whether these agents have a cardioprotective effect against anti-cancer related cardiotoxicity has not fully been elucidated. Therefore, this study aims to explore the potential effects and mechanisms of these cardiovascular protective agents (eplerenone, empagliflozin and colchicine) to alleviate the cardiotoxicity in a doxorubicin-induced cardiotoxicity model of H9c2 cardiomyocytes.
Materials and methods
Reagents
E2,3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyltetrazolium bromide (MTT), Actin-Tracker Red, Mito-Tracker Red CMXRos, enhanced mitochondrial membrane potential assay kit with JC-1 and Liquid Blocker Super PAP Pen were purchased from Beyotime Biotechnology (Shanghai, China). Polyclonal antibodies against dynamin-related protein 1 (DRP1), mitofusin 2 (MFN2), mitochondrial transcription factor A (TFAM) and GAPDH were purchased from Proteintech Group (Chicago, IL, USA). Matrix metalloprotein 9 (MMP9), alpha-smooth muscle actin (α-SMA) were purchased from Abcam Biotechnology (UK). Doxorubicin, eplerenone, empagliflozin, Colchicine and dimethylsulfoxide (DMSO) were purchased from Solarbio Life Sciences (Shanghai, China).
Cell culture
The H9c2 rat cardiomyoblast cell line was obtained from the Cell Bank of the Type Culture Collection of the Chinese Academy of Sciences and used in accordance with institutional biosafety and ethical standards. Cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) medium (Gibico, Grand Island, NY, USA). This medium was supplemented with fetal bovine serum (FBS) (EVER GREEN, Nanijing, China). The cells were incubated at 37°C with 5% CO2 in a humidified atmosphere. To ensure consistent and reliable results, only cells in the logarithmic growth phase were utilized for all experimental procedures. Experiments were performed using cells between passages 10 and 20 Usually, new cultures were re-established from frozen stocks every 3 months.
Cytotoxicity assay
To assess cell viability, H9c2 cells were seeded in 96-well cell culture plates from Corning (NY, USA) at a density of 1.0 × 104 cells/well and allowed to adhere and grow for 24 h. After the specified treatments were applied to the cells for 48 h, they were washed twice with ice-cold PBS. Cell viability was then determined using the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay, following previously reported methods. In brief, MTT was added to each well and incubated with the cells for a specific period of time. After incubation, the formazan crystals formed by viable cells were dissolved in a suitable solvent, and the absorbance was measured at a wavelength of 492 nm using a microplate reader (Thermo Scientific Multiskan MK3, Shanghai, China). The absorbance values obtained from the MTT assay are indicative of cell viability, with higher absorbance values corresponding to higher cell viability. This assay allows for the quantification of the effects of the specified treatments on H9c2 cell viability. Drugs were dissolved in DMSO, keeping the final concentration at or below 0.1% (v/v). In all cytotoxicity experiments, appropriate vehicle controls, which were DMSO solutions at matching concentrations, were included to distinguish drug - specific effects from those of the solvent.
Immunofluorescent staining
Cells were fixed in 4% paraformaldehyde for 20 min, permeabilized with 0.15% Triton X-100 for 10 min, blocked with 10% FBS at room temperature for 30 min, followed by overnight incubation with anti-MNF2 antibody (1:500) or anti-DRP1 antibody (1:500) at 4°C. The coverslips were washed with PBS and incubated with fluorochrome-conjugated secondary antibodies (1:200 dilution) (Proteintech, Chicago, USA) at room temperature for 2 h. The coverslips were washed with PBS three times to remove the excess antibody and stained with DAPI for 10 min. Images were captured using fluorescence microscopy (Beckman Coulter Life Science, California, USA). Images were acquired using identical exposure settings for each fluorescence channel. The exposure duration for each individual fluorescence channel was meticulously maintained at a fixed value throughout the entire image - acquisition process to ensure consistency and comparability across all experimental conditions. Background fluorescence levels were measured in control samples that did not contain the primary antibody (negative controls). The mean fluorescence intensity of these negative control samples was calculated, and a threshold was set at a value that was 3 standard deviations above the mean background fluorescence. For each experimental condition, three fields of view were acquired at random positions and every cell within each field (≈30 cells) was automatically quantified by custom Image-Pro Plus. For each field, a constant threshold was applied to remove background, MFN2/DRP1 integrated density was divided by DAPI density to correct for cell number, then expressed as fold-change versus matched control. Values from n = 3 independent experiments were pooled for statistics.
Western blot analysis
Soluble lysates were resolved by SDS-PAGE (11 % gel) and transferred to Millipore Immobilon®-P PVDF membranes. Immediately after transfer, each membrane was placed beside the pre-stained, two-colour molecular-weight marker lane that had been loaded in lane 1 of every gel. The coloured standards provided nine reference bands-250, 150, 100, 70, 50, 35, 25, 15 and 10 kDa-allowing precise visual alignment. Immediately after transfer, the membrane was cut horizontally into strips, each spanning the molecular-weight range of the target protein and processed individually. Following blocking with 5% non-fat milk in Tris-buffered saline containing 0.1% Tween-20 for 2 h at room temperature, the strips were incubated overnight at 4°C with primary antibodies and then with horseradish peroxidase-conjugated secondary antibodies. The membranes were incubated with primary antibody at 4°C overnight and further incubated with the appropriate concentrations of horseradish peroxidase-conjugated secondary antibody. The blots were visualized using the SuperSignal West Pico Chemiluminescent Substrate® purchased from Beyotime Biotechnology (Shanghai, China). Because the membrane was physically segmented prior to antibody incubation, full-length, uncropped blots spanning the entire molecular-weight range do not exist. Brightness and contrast were adjusted uniformly across every western blot images, no further image processing was performed. The protein expression levels were quantified by using ImageJ software. The gray values obtained for each target protein were initially normalized by dividing by the corresponding gray value of GAPDH, which was set as the loading control. Subsequently, the normalized values from the control group were set as 1, and the values from the remaining experimental groups were further normalized by dividing by the control group value. This two-step normalization process ensured the accuracy and consistency of data representation. Finally, the results from three independent experiments were pooled and included into the final statistical analysis to evaluate the significance of the observed differences.
Mitochondrial membrane potential assay
The mitochondrial membrane potential was determined using the JC-1 Assay Kit, and then using fluorescence microscopy (Beckman Coulter Life Science, California, USA). The fluorescence intensity levels were quantified by using Image-Pro Plus software The normalized fluorescence intensity values obtained from three independent experiments were compiled and included into the final statistical analysis.
Transfection of siRNA
siRNA oligonucleotides used in this study.

Measurements of mitochondrial mass content
To stain the mitochondria, cells were incubated with MTG (20 nM) at 37°C for 30 min, followed by the analysis with a FACScan flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA).
Statistical analysis
Data from at least three independent experiments are presented as means ± SD. One-way ANOVA were used for comparisons among groups. ANOVA was used to compare group means. Assumptions of normality and homogeneity of variances were examined for each dataset. Following a significant ANOVA result, pairwise comparisons were performed with Tukey’s honestly significant difference (HSD) post hoc test to control the family-wise error rate. Non-normally distributed data are expressed as median [inter-quartile range] and analyses with Kruskal-Wallis test with Dunn’s multiple-comparison correction. All analyses were conducted by GraphPad Prism 6.0 software. Statistical significance was defined as p < 0.05 and is indicated in figures using the following notation,*p < 0.05, **p < 0.01, ***p < 0.001.
Results
Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) effectively alleviate doxorubicin-induced fibrosis levels in H9c2 cells
Doxorubicin-induced cardiomyocytes damage is partly manifested as myocardial fibrosis,12–14 therefore we examine the potential impact of eplerenone, empagliflozin and colchicine on doxorubicin-induced myocardial fibrosis in an in vitro model of H9c2 cells. We compared the cytotoxicity of doxorubicin on H9c2 cells in the absence or presence of empagliflozin, eplerenone or colchicine. Based on previous reports,15–18 the commonly used concentrations of doxorubicin, eplerenone, empagliflozin, and colchicine are 1 μM, 10 μM, 10 μM and 10 μM respectively. To investigate the cytotoxic effects of these drugs on H9c2 cells, a series of experiments were conducted. These results reveal that the maximum concentration levels of eplerenone, empagliflozin and colchicine were 20 μM, 20 μM and 10 μM, respectively, at which these agents did not significantly affect the survival of H9c2 cells. Doxorubicin inhibited H9c2 cell growth in a dose-dependent manner with an IC50 of 10 μM (Figure 1(a)). These concentration levels of doxorubicin, eplerenone, empagliflozin, and colchicine were further chosen into subsequent tests. H9c2 cells were pre-treated 2 h with eplerenone, empagliflozin or colchicine, exposed to doxorubicin for 48 h. The MTT reduction assay showed that Empagliflozin, eplerenone or colchicine significantly inhibited the cytotoxic effects of doxorubicin on H9c2 cells (Figure 1(b)). Fluorescence microscopy observations reveal that treatment with eplerenone, empagliflozin or colchicine effectively alleviated the doxorubicin-induced loss of cell polarity and disorganization of stress fibers (Figure 1(c)). These findings are further supported by data demonstrating that doxorubicin treatment led to an upregulation in the expression of fibrosis-related proteins, MMP-9 and α-SMA. Notably, the unregulated expression those proteins was partially reversed by the administration of eplerenone, empagliflozin and colchicine (Figure 1(d)). These findings suggest that eplerenone empagliflozin and colchicine may offer a protection effect to H9c2 cells by alleviating the fibrosis induced by doxorubicin. Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) effectively alleviate doxorubicin-induced fibrosis levels in H9c2 cells. (a) Dose-response of doxorubicin, eplerenone, empagliflozin, or colchicine in H9c2 cells. Cell viability was determined by an MTT assay. Data are mean ± SD of n = 3 independent experiments (each run in triplicate). Doxorubicin versus control, **p = 0.008, **p = 0.006, ***p = 0.0007, ***p = 0.001; Eplerenone versus control, p = 0.1531, p = 0.0693, *p = 0.027, **p = 0.0011; Empagliflozin versus control, p = 0.3279, p = 0.0568, **p = 0.0017, ***p = 0.0002; colchicine versus control, p = 0.0595, *p = 0.0111, **p = 0.0025, ***p = 0.0008. (b) H9c2 cells were exposed to doxorubicin, either as a monotherapy or in combination with eplerenone, empagliflozin or colchicine for a duration of 48 h. Subsequently, cell viability was quantified using an MTT assay. n = 6, Data are means ± SD. Doxorubicin versus control, ***p = 0.0007; eplerenone versus doxorubicin, ##p = 0.0046; empagliflozin versus doxorubicin, #p = 0.0498; colchicine versus doxorubicin, ###p = 0.0001. (c) Immunofluorescence staining of F-actin was carried out using Actin-Tracker Red-594, followed by visualization under fluorescence microscopy. (d) Western blot analysis was performed to assess the expression levels of fibrosis-related proteins, including MMP-9 and α-SMA. Fold change of the band densities of specific protein to control is expressed. Data are means ± SD, n = 3. Doxorubicin versus control, *p = 0.0193 (α-SMA), *p = 0.0236 (MMP-9); Doxorubicin + eplerenone versus doxorubicin, #p = 0.0361 (α-SMA), #p = 0.0116 (MMP-9); Doxorubicin + empagliflozin versus doxorubicin, #p = 0.0206 (α-SMA), #p = 0.0178 (MMP-9); Doxorubicin + colchicine versus doxorubicin, #p = 0.0175 (α-SMA), #p = 0.0152 (MMP-9).
Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) significantly ameliorate doxorubicin-induced mitochondrial fusion in H9c2 cells
The mitochondrial dynamics, encompassing the opposing processes of fusion and fission, along with biogenesis and degradation, are visualized through the MitoTracker Deep Red (MTR) dye. Our fluorescence microscopy analysis unveils that doxorubicin exposure led to a truncated and fragmented appearance of mitochondria (Figure 2(a)). As shown in Figure 2(b), the expression of the mitochondrial fusion-related protein MFN2 notably decreased in the mitochondria of H9c2 cells exposes to doxorubicin. However, treatment with eplerenone, empagliflozin or colchicine significantly reverses this decreased expression. Additionally, the immunostaining results further confirmed that these cardiovascular agents could significantly upregulate the MFN2 expression (Figure 2(c)). To investigate the role of MFN2 in doxorubicin-induced myocardial fibrosis, MFN2 is knocked down using siRNA in H9c2 cells. The efficiency of siRNA transfection is subsequently confirmed by Western blot analysis (Figure 2(d)). Consistent with previous findings, the knocking down of MFN2 notably attenuated the fibrosis-ameliorating effects of eplerenone, empagliflozin or colchicine (Figure 2(e)). These findings imply that the activation of mitochondrial fusion mediated by eplerenone, empagliflozin or colchicine plays a pivotal protective role against the myocardial fibrosis induced by doxorubicin in H9c2 cells. Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) significantly ameliorate doxorubicin-induced mitochondrial fusion in H9c2 cells. (a) Following exposure to the target drug, H9c2 cells incubated with 100 nM MitoTracker Deep Red (MTR) for 30 min. Image shown is representative of n = 3 independent experiments (duplicate wells each). (b) The expression of MFN2 was analyzed by western blot to assess its protein levels. Blot representative of n = 3 biological replicates, bar graph: fold-change mean ± SD. Doxorubicin versus control, *p = 0.0234; eplerenone versus doxorubicin, #p = 0.0467; empagliflozin versus doxorubicin, #p = 0.0303; colchicine versus doxorubicin, #p = 0.0375. (c) H9c2 cells were immunostained with anti-MFN2 antibodies and stained with MTR. Nuclei were counterstained with DAPI. The fold change in MFN2 fluorescence density is expressed. (n = 4, mean ± SD). Doxorubicin versus control, ***p = 0.0002; eplerenone versus doxorubicin, #p = 0.0125; empagliflozin versus doxorubicin, #p = 0.0314; colchicine versus doxorubicin, #p = 0.0433. (d) The transfection efficiency was determined by western blot analysis, which allowed for the quantification of protein expression levels. si-MFN2-1 versus si-GAPDH, *p = 0.0223; si-MFN2-2 versus si-GAPDH, p = 0.1074, si-MFN2-3 versus si-GAPDH, *p = 0.0319. (e) The protein levels of MMP9 and α-SMA were evaluated by western blot analysis. The data presented represent the mean ± SD from three independent experimental replicates. NS, not significant compared with si-MFN2-1 group. si-MFN2-1 versus si-GAPDH, *p = 0.0422 (α-SMA), *p = 0.0246 (MMP-9); si-MFN2-1 + doxorubicin versus si-MFN2-1,p = 0.8830 (α-SMA), *p = 0.9963 (MMP-9); si-MFN2-1 + doxorubicin + eplerenone versus si-MFN2-1+ doxorubicin, p = 0.8830 (α-SMA), p = 0.9963 (MMP-9); si-MFN2-1 + doxorubicin + empagliflozin versus si-MFN2-1+ doxorubicin, p = 0.7733 (α-SMA), p = 0.6644 (MMP-9); si-MFN2-1 + doxorubicin + colchicine versus si-MFN2-1+ doxorubicin,p = 0.8778 (α-SMA), p = 0.8949 (MMP-9).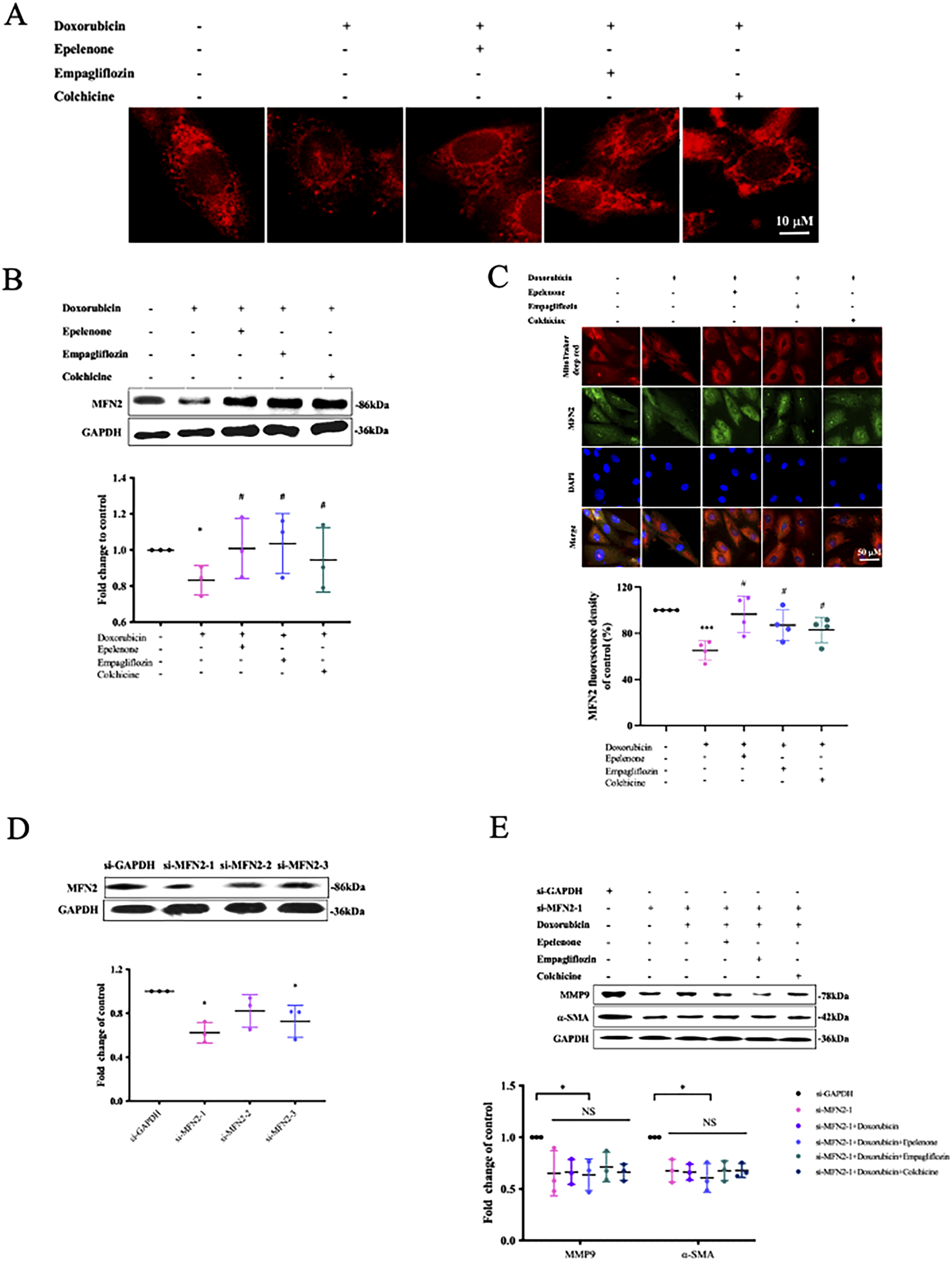
Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) effectively downregulate doxorubicin-induced elevation of mitochondrial fission in H9c2 cells
Mitochondrial dynamics play a pivotal role in maintaining the morphology and function of mitochondria during cellular stress.
19
During periods of cellular stress, mitochondrial dynamics could be significantly changed, resulting in changes to the mitochondrial morphology and function.20,21 Notably, DRP1 (also referred to as DNM1L) is considered a key regulator of mitochondrial fission.22,23 To explore whether the expression of molecules involved in mitochondrial fission could be modulated by eplerenone, empagliflozin, or colchicine treatment, western blot analyses are used to assess the expression levels of DRP1 protein. As shown in Figure 3(a), the elevation of DRP1 protein expression induces by doxorubicin is significantly reduced by eplerenone, empagliflozin or colchicine. Moreover, the analysis of immunofluorescence microscopic images analyses have revealed that these treatments significantly attenuated the expression of DRP1 protein (Figure 3(b)). To further validate these findings, DRP1-siRNA is employed. The transfection efficiency of the siRNA is confirmed through western blot analyses (Figure 3(c)). In alignment with the aforementioned observations, the expression levels of MMP9 and α-SMA remain unaltered by doxorubicin, eplerenone, empagliflozin or colchicine after the silencing DRP1 (Figure 3(d)). Collectively, these findings implicate that the inhibition of mitochondrial fission can exert a suppression effect of myocardial fibrosis, thereby indicating a promoting role of mitochondrial fission in myocardial fibrosis in H9c2 cells. Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) effectively downregulate doxorubicin-induced elevation of mitochondrial fission in H9c2 cells. (a) Band densities were quantified with Image Pro-Plus. Bar graph shows fold-change mean ± SD from three independent experiments (triplicate wells each). Doxorubicin versus control, *p = 0.0247; eplerenone versus doxorubicin, #p = 0.0331; empagliflozin versus doxorubicin, #p = 0.0379; colchicine versus doxorubicin, #p = 0.0406. (B) Fluorescence microscopy was used to visualize H9c2 cells immunostained with anti-DRP1 antibodies. The fold change in DRP1 fluorescence density is expressed. (n = 4, mean ± SD). Doxorubicin versus control, **p = 0.0013; eplerenone versus doxorubicin, #p = 0.0439; empagliflozin versus doxorubicin, #p = 0.0285; colchicine versus doxorubicin, #p = 0.0210. (c) The transfection efficiency was analyzed by western blot, which provided a quantitative measure of protein expression. si-DRP1-1 versus si-GAPDH, *p = 0.0241; DRP1-2 versus si-GAPDH, p = 0.0654; si- DRP1-3 versus si-GAPDH, *p = 0.0454. (d) Expression levels of MMP9 and α-SMA were evaluated by western blot analysis. Band densities of the specific proteins were normalized and expressed relative to the control condition. Data are means ± SD of three independent experiments; NS, not significant. si-DRP1-1 versus si-GAPDH, *p = 0.0293 (α-SMA), *p = 0.0349 (MMP-9); si-DRP1-1 + doxorubicin versus si-DRP1-1, p = 0.1892 (α-SMA), p = 0.2196 (MMP-9); si-DRP1-1 + doxorubicin + eplerenone versus si-DRP1-1 + doxorubicin, p = 0.2457 (α-SMA), p = 0.1957 (MMP-9); si-DRP1-1 + doxorubicin + empagliflozin versus si-DRP1-1+ doxorubicin, p = 0.3431 (α-SMA), p = 0.2323 (MMP-9); si-DRP1-1 + doxorubicin + colchicine versus si-DRP1-1+ doxorubicin, p = 0.1637 (α-SMA), p = 0.3759 (MMP-9).
Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) can potentially restored mitochondrial biogenesis and mitochondrial membrane potential in a doxorubicin-induced myocardial fibrosis in vitro model of H9c2 cells
The intricacies underlying the mechanisms of doxorubicin-induced myocardial fibrosis remain enigmatic, despite several putative explanations proposed. To further investigate the potential influence of doxorubicin on mitochondrial dynamics, biogenesis and function, MitoTracker Green (MTG) is used to label mitochondria independently of their membrane potential and to assess the relative mitochondrial mass. Strikingly, our MTG staining reveal a significant decrease of mitochondrial mass in the eplerenone-, empagliflozin- or colchicine-treated groups compared with the doxorubicin-alone treated group (Figure 4(a)). TFAM, a crucial regulatory factor governing mitochondrial biogenesis, is also thoroughly examined in this study. Our findings have revealed that the doxorubicin-induced upregulation of TFAM could be counteracted by the administration of eplerenone, empagliflozin or colchicine in H9c2 cells, thereby restoring the balance of mitochondrial homeostasis (Figure 4(b)). Mitochondrial health can be evaluated by tracking changes in mitochondrial membrane potential (MMP).
24
MMP was evaluated by using JC-1 staining, followed by fluorescence microscopy. In cells with a high level of MMP, JC-1 exists as a monomer within the mitochondrial matrix and fluoresces red. Conversely, when the MMP level is low, JC-1 exists in an aggregate form outside of mitochondria and appears green.
25
We observed doxorubicin treatment significantly decreased MMP (increased green/red ratio), while eplerenone, empagliflozin, and colchicine effectively restored MMP (Figure 4(c)). These observations suggest that eplerenone, empagliflozin, and colchicine preserve mitochondrial biogenesis and membrane potential in H9c2 cells when co-administered with doxorubicin. Overall, these findings indicate that the cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) protect H9c2 cells from doxorubicin-induced fibrosis (Figure 5). Cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) can potentially restored mitochondrial biogenesis and mitochondrial membrane potential in a doxorubicin-induced myocardial fibrosis in vitro model of H9c2 cells. (a) Histograms show mean fluorescence intensity (MFI) of gated live cells, bar graph gives MitoTracker Green (MTG)-MFI fold-change (mean ± SD) from n = 3 independent experiments (duplicate wells each). Doxorubicin versus control, **p = 0.0026; eplerenone versus doxorubicin, #p = 0.0467; empagliflozin versus doxorubicin, p = 0.7479; colchicine versus doxorubicin, ##p = 0.0061. (b) Western blot analysis was employed to assess the expression level of the TFAM protein. Band densities of specific protein were expressed as ratios to control. Data are means ± SD of three independent experiments. Doxorubicin versus control, *p = 0.0192; eplerenone versus doxorubicin, ##p = 0.0045; empagliflozin versus doxorubicin, ##p = 0.0049; colchicine versus doxorubicin, #p = 0.0465. (c) JC-1 fluorescence microscopy provided a visual and qualitative assessment of mitochondrial membrane potential. Quantitative analysis of the ratio of green/red fluorescent intensity. Data are presented as means ± SD of three independent experiments. NS, not significant. Doxorubicin versus control, *p = 0.0171; eplerenone versus doxorubicin, #p = 0.0182; empagliflozin versus doxorubicin, #p = 0.0483 colchicine versus doxorubicin, #p = 0.0456. Schematic representation of the mechanism of myocardial fibrosis alleviation through mitochondrial dynamics modulation by cardiovascular agents (i.e. eplerenone, empagliflozin and colchicine) in H9c2 cells.

Discussion
Using an in vitro H9c2 model, we showed that eplerenone, empagliflozin and colchicine protect against doxorubicin-induced cardiotoxicity by preserving mitochondrial dynamics, enhancing biogenesis and maintaining membrane potential.
Indeed, these cardiovascular agents, i.e. eplerenone, empagliflozin and colchicine, have been previously studied for their potential to mitigate the chemotherapy related cardiotoxicity induced by doxorubicin.26,27 Empagliflozin, a sodium-glucose cotransporter 2 inhibitor, is primarily utilized for managing type 2 diabetes. However, recent studies hint at its potential for offering cardioprotective benefits beyond mere glucose control. Comprehensive research has demonstrated that empagliflozin can enhance cardiac performance, mitigate oxidative stress, and mitigate myocardial fibrosis in animal models of doxorubicin-induced cardiotoxicity. These benefits may arise from the modulation by empagliflozin of multiple signaling pathways that underlie cardiac injury and remodeling. 28 Eplerenone, a selective aldosterone receptor antagonist, is widely used in the treatment of hypertension and heart failure. Aldosterone, a hormone that can exacerbate inflammation, fibrosis, and oxidative stress in the heart, is effectively countered by eplerenone. By blocking the harmful effects of aldosterone, eplerenone offers protection to the heart against the deleterious consequences of doxorubicin-induced cardiotoxicity. Numerous studies have shown that eplerenone significantly mitigates myocardial damage, inflammation, and fibrosis in animal models of doxorubicin-induced cardiotoxicity.29–31 Colchicine, an anti-inflammatory medication, is employed to treat various conditions, including gout and pericarditis. In the context of doxorubicin-induced cardiotoxicity, colchicine has garnered interest for its potential in mitigating inflammation and fibrosis in the heart. Robust studies have demonstrated that colchicine effectively diminishes myocardial injury, inflammation, and fibrosis in animal models of doxorubicin-induced cardiotoxicity. These beneficial effects are likely mediated by the anti-inflammatory and anti-fibrotic properties of these agents. 32 Collectively, eplerenone, empagliflozin, and colchicine have emerged as the cardiovascular agents that demonstrate promise in mitigating doxorubicin-induced cardiotoxicity. This is achieved through various mechanisms, including the attenuation of inflammation, oxidative stress, and fibrosis in the heart. These agents offer potential therapeutic avenues for the prevention and treatment of doxorubicin-related cardiac damage. However, the primary objective of this experiment was to compare the relative efficacy of these three novel agents under identical experimental conditions. Future studies will incorporate the positive controls to further contextualize the efficacy of the promising candidates identified here.
The dysregulation of mitochondrial dynamics, a crucial aspect of the mitochondrial quality control, bioenergetics, and cellular function, can significantly contribute to the cardiac toxicity and various cardiovascular diseases. Mitochondria, being highly dynamic organelles, undergo the continuous processes of fusion, fission, and mitophagy to maintain their integrity and functionality. Imbalances in these dynamic processes, however, can lead to the mitochondrial dysfunction, oxidative stress, and impaired energy metabolism, thereby promoting the cardiac toxicity and cardiovascular pathology.33,34 The disruptions in the mitochondrial quality control and bioenergetics due to the deregulated dynamics further exacerbate the oxidative stress, further compromising the cardiac function and overall cardiovascular health. Therefore, elucidating the underlying mechanisms of the mitochondrial dynamics and the exploring therapeutic strategies to restore mitochondrial homeostasis offer promising avenues for the prevention and treatment of cardiac dysfunction and cardiovascular disorders. Targeting the mitochondrial dynamics to maintain the mitochondrial health and function holds the significant potential in mitigating the cardiac toxicity and improving the cardiovascular outcomes.
In conclusion, beyond the notable enhancement in the clinical prognosis of patients with cardiovascular diseases, agents such as eplerenone, empagliflozin, and colchicine hold significant promise in another critical area. They may play a pivotal role in restoring mitochondrial dynamics and alleviating doxorubicin-induced myocardial fibrosis, thereby potentially offering a cardioprotective effect against the cytotoxicity associated with anti-cancer agents. However, it is crucial to recognize that these preclinical findings do not automatically translate into clinical cardioprotection. Before any therapeutic applications can be considered, a series of sequential challenges must be overcome: First, validation in disease-relevant in vivo models is essential to confirm the agents’ efficacy in counteracting both structural and functional cardiotoxicity. Next, pharmacokinetic and pharmacodynamic modeling is necessary to ensure that the free plasma concentrations achievable in humans do not undermine the cytotoxic potency of doxorubicin. Safety profiling is also imperative when these agents are used in combination with standard chemotherapeutic regimens, to rule out adverse effects such as QT-prolongation, electrolyte imbalances, or immunosuppression, which could potentially worsen oncological outcomes. Furthermore, prospective biomarker-driven trials should be conducted, measuring troponin and NT-proBNP, as early endpoints, before initiating large-scale outcome studies. Only after successfully navigating these steps can we accurately determine the true clinical utility of these agents in mitigating doxorubicin-induced cardiotoxicity.
Limitations
It is important to note that H9c2 cells are undifferentiated myoblasts derived from embryonic heart tissue. Thus, the results obtained from this model should be interpreted cautiously, as they may not fully represent the responses of mature cardiomyocytes in vivo.
A recognized cardioprotective positive control (e.g., dexrazoxane) was not included in this experiment, which could limit the direct validation of the results of this study. Therefore we plan to include a positive control in forthcoming experiments to rigorously validate the therapeutic value of our intervention against the established cardioprotective standards.
While our study demonstrated the favorable modulation of the fibrosis-related markers and mitochondrial-protein expression, the direct functional indices such as the ATP content and the oxygen consumption rate (OCR) were not assessed. In pilot experiments conducted 48 h post-doxorubicin exposure, we observed that the extensive mitochondrial damage rendered ATP and OCR signals highly variable and near the lower limit of detection, compromising the measurement reliability. Consequently, these assays were not included in the final experimental design; future work will optimize the earlier time points or the lower doxorubicin doses to the enable robust quantification of the mitochondrial function.
Supplemental material
Suppplemental Material - Alleviation of the mitochondrial dynamics impairment in doxorubicin-induced cardiomyocyte toxicity by cardioprotective agents
Suppplemental Material for Alleviation of the mitochondrial dynamics impairment in doxorubicin-induced cardiomyocyte toxicity by cardioprotective agents by Jing Yang, Shuyun Qing, Yirong Wang, Xiang Ye, Qin He, Junbo Xu, Gang Huang in Human & Experimental Toxicology
Footnotes
Ethical considerations
This study used only the commercially available H9c2 cell line (Catalogue No GNR5; Cell Bank, Type Culture Collection, Chinese Academy of Sciences, Shanghai, China). The cell bank certifies that the line was originally obtained and distributed in full compliance with the institutional animal-care and biosafety guidelines of the Chinese Academy of Sciences and with the Convention on Biological Diversity. No human or live-animal subjects were involved; therefore, formal ethical approval by our institutional review board was not required. All cell-culture work was conducted under the Biosafety Level 2 (BSL-2) standards and institutional biosafety protocols.
Author contributions
Jing Yang, Qin He, Junbo Xu and Gang Huang designed the research; Jing Yang, Shuyun Qing and Yirong Wang performed the research; Junbo Xu and Gang Huang contributed some reagents; Jing Yang and Xiang Ye analyzed the data; Jing Yang wrote the paper; Junbo Xu and Gang Huang revised the paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sichuan Science and Technology Program (2024YFFK0282), Sichuan Health Commission (23LCYJ044, 24LCYJZD01), Department of Human Resources and Social Security of Sichuan Province (2021-11), The Third People’s Hospital of Chengdu Clinical Research Program (CSY-YN-01-2023-011, 076, 077), The Third People’s Hospital of Chengdu Scientific Research Project (2023PI17), Chengdu Municipal Health Commission (2021200, 2022392).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Research integrity and publishing ethics
All authors confirm that this manuscript presents original research. We have rigorously avoided plagiarism and have accurately cited and referenced all sources. Raw western-blot images and the complete numerical dataset are available upon reasonable request to ensure full transparency.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
