Abstract
Objectives:
Baicalein, a flavonoid derived from the roots of Scutellaria baicalensis Georgi, demonstrates multifarious pharmacological effects due to its high antioxidant activity. However, the latent mechanisms remain insufficiently resolved. In the present research, we evaluated the therapeutic effects of baicalein on isoprenaline (ISO)-induced heart failure and investigated the possible underlying mechanisms.
Methods:
Toxicity was analyzed in zebrafish embryos and mouse atrial myocytes HL-1. The MTT assay was used to evaluate the effectiveness of baicalein. DCFH-DA was used as a fluorescence probe to detect intracellular reactive oxygen species (ROS). Superoxide dismutase (SOD), malondialdehyde (MDA), and glutathione peroxidase (GSH-Px) levels were measured using SOD, MDA and GSH-Px commercial kits. Adult BALB/c mice were randomized into six groups of ten animals each. Cardiac function was analyzed by echocardiographic images. Structural changes were analyzed by hematoxylin & eosin (HE) staining, Masson staining and TUNEL staining. The mechanism of baicalein was investigated by analyzing relative signaling pathways through western blotting.
Results:
Our studies show that baicalein both significantly reduces ISO-induced oxidative stress, apoptosis and cardiac fibrosis in vitro and vivo, this phenomenon was related to mitochondrial fusion/fission balance and inhibiting GRP78/CHOP pathway.
Conclusions:
Our results suggested that baicalein controls mitochondrial fusion/fission balance and inhibits GRP78/CHOP pathway, thus exerting therapeutic effects in ISO-induced heart failure in HL-1 cells and BALB/c mice. These results suggested that baicalein may be a potential therapeutic agent for heart failure.
Graphic abstract
Treatment mechanism of baicalein in ISO-induced heart failure.

Introduction
Cardiovascular disease (CVD) is the leading cause of global fatalities. By 2035, more than 130 million adults (45.1%) are expected to have some form of CVD in the United States, with a heavy financial burden. 1 Heart failure (HF) is a complex clinical syndrome characterized by abnormal cardiac structure or function. It has a 5-year survival rate similar to that of malignant tumors and is the final stage of various severe heart diseases. 2 This unacceptably high mortality rate has become a global consensus and calls for a reassessment of cardiac biology to identify new strategies for heart failure prevention and treatment.
It is well known that mitochondria are power houses in the body. The most metabolically diligent organ is the heart, which possesses the highest mitochondrial content of any tissue. 3 Mitochondrial dysfunction plays a prominent role in many cardiac disorders, such as heart failure. 4 Dynamic organelles and mitochondria constantly shape themselves through fusion and fission in response to changes in energy requirements. 5 Pathological mitochondrial remodeling, characterized by increased fission and reduced fusion, is associated with increased mitochondrial oxidative stress and increased cardiomyocyte apoptosis. Considering the impact of the mitochondrial morphology on mitochondrial behavior and cardiomyocyte performance, altered mitochondrial dynamics could be expected to induce or exacerbate the pathogenesis and progression of heart failure. 6 Mitochondrial fusion is facilitated by mitofusion (Mfn)-2 and optic atrophy (OPA)-1, 7 and mitochondrial fission is driven by dynamin-related protein-1 (Drp1) mitochondrial fission factor (fis)-1. 8 Simultaneously, Drp1 is widely expressed in the heart in humans, 9 it has been reported that Drp1 is enhanced under pathological conditions in the heart. 10 Moreover, the maintenance of cellular homeostasis involves the involvement of multiple organelles. Mitochondria-associated ER membrane (MAM) is located between the two organelles, ER and mitochondria, and is the most direct connection medium. 11 As a MAM-related protein, Drp1 can be closely involved in signal transduction from ER to mitochondria.
The ER is the site for protein synthesis, folding, and secretion. Prolonged pathological stimuli such as ischemia and oxidative stress impair ER function, triggering ER stress. When ER stress is prolonged or severe, the overexpression of CHOP and GRP78 induces apoptosis to eliminate unhealthy cells, contributing to the process of cardiac hypertrophy. 12 Prolonged cardiac hypertrophy causes dilated cardiomyopathy, heart failure, arrhythmias, and sudden death. 13 Although the specific roles of mitochondrial dysfunction and ER stress in the pathophysiology of HF are not clear, ISO-induced models of HF provide clues regarding the interplay between mitochondrial dysfunction, ER stress and HF pathogenesis.
Baicalein (5,6,7-trihydroxy-2-phenyl-4H-1-benzopyran-4-one) is a natural flavonoid component derived from the roots of the traditional Chinese herbal medicine Scutellaria baicalensis Georgi. Clinically, Scutellaria baicalensis Georgi is widely used in the treatment of heart failure, myocardial hypertrophy, atherosclerosis, hypertension, and other cardiovascular diseases. Previous studies have shown that baicalein undertakes multiple pharmacological activities, such as antioxidative damage, 14 anti-inflammatory. 15 It exerts a cardioprotective effect by hindering inflammation, apoptosis and fibrosis, thereby reducing damage from heart failure. 16 However, the mechanisms underlying these effects are poorly understood. In the present research, we analyzed the therapeutic effects of baicalein on ISO-induced HF in vivo and in vitro and explored whether the cardio-protective potential exerted by baicalein was mediated by controlling mitochondrial fusion/fission balance and inhibiting ER stress related pathway-GRP78/CHOP pathway.
Materials and methods
Drugs and reagents
Baicalein (CAS: 491-67-8, purity ≥98%) was obtained from Chengdu Desite Biotechnology Co., Ltd. (Chengdu, China). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) and isoprenaline (ISO) were procured from Sigma–Aldrich. Antibodies against GAPDH, caspase3, caspase9, cleaved PARP, Drp1, MFF, OPA1, mtTFA, ATF4, CHOP, GRP78, p-PERK, and PERK were procured from Cell Signaling Technology (Danvers, MA, USA). Fetal bovine serum (FBS) and Dulbecco’s modified Eagle’s medium (DMEM) were obtained from Gibco (Grand Island, NY, USA).
Animals
BALB/c mice can better exhibit clinically relevant features of HF, So we chose BALB/c as the experimental animal. 17 BALB/c mice were obtained from Yis Experimental Animal Technology Co Ltd. in Changchun (No. 202000035165). Animal custody and study protocols were endorsed by the Laboratory Animal Welfare & Ethics Committee of Zunyi Medical University (No: ZMU21-2404-006). BALB/c mice were arbitrarily divided into six groups (n = 10): control group, ISO group, ISO + Met (10 mg/kg metoprolol) group, treatment groups: ISO + HSL (12.5 mg/kg baicalein), ISO + HSM (25 mg/kg baicalein), and ISO + HSH (50 mg/kg baicalein). They were domiciled under a 12-h light/dark period at 25 ± 2°C. The ISO group, ISO + Met group and the treatment groups were injected subcutaneously with 2.5 mg/kg/d ISO for 7 days, 18 and each treatment group underwent gastric administration for 28 days. 19
Assessment of cardiac function
After being anesthetized by intraperitoneal injection with 0.3 mL/100 g 2% pentobarbital sodium in mice, Vevo3100LT high resolution small animal ultrasound imaging system (Visual Sonics, Canada) was used to test fractional shortening (FS) and left ventricular ejection fraction (EF%).
Histological analysis
Heart tissues were fixed in 10% formalin and processed into paraffin sections. Then, the sections were stained following a hematoxylin & eosin (H&E) or Masson staining protocol. A TUNEL kit was used to recognize DNA fragments from apoptotic cells, following the manufacturer’s instructions. Images were observed and captured using a Nikon COOLPIX A900 digital camera. The levels of fibrosis in cardiomyocytes were analyzed by Masson staining, and the levels of apoptosis in cardiomyocytes were identified by TUNEL assay using ImageJ software.
Cell culture
HL-1 cells are mouse atrial myocytes that are commonly used for the establishment of in vitro models of heart-related diseases. HL-1 cells were cultured in a cell incubator with 5% CO2 at 37°C. HL-1 cells were divided into control group, ISO group, ISO + Met (1.5 µmol/L metoprolol) group, treatment groups: ISO + HSL (12.5 µmol/L baicalein), ISO + HSM (25 µmol/L baicalein), and ISO+HSH (50 µmol/L baicalein). The ISO group, ISO + Met group and the treatment groups supernatant was discarded after 24 h of exposure to ISO (50 µmol/L), followed by 3% FBS medium prepared with baicalein (12.5, 25, 50 µmol/L) and metoprolol (1.5 µmol/L) for 24 h. Equal amounts of 3% FBS medium were added to the control group.
MTT detection of cell viability
HL-1 cells were cultured in 96-well plates at 8 × 103 cells/well. Cells were cultured using the cell culture method described above. To detect the cell survival rate, 5 mg/mL thiazole blue tetrazolium bromide (MTT) was added to the cells for 4 h. Following the elimination of MTT, DMSO was added to each and every well and detected at optical densities of 570 and 630 nm by an enzyme labelling instrument.
Reactive oxygen species in cells
After different treatments of HL-1 cells, the cell culture medium was removed and replaced with diluted DCFH-DA at a dilution ratio of 1:1000 with an ultimate concentration of 10 µmol/L. The cells were incubated at 37°C for 20 min. Cells were cultured three times with cell culture medium to thoroughly remove intracellular DCFH-DA. The wells were assayed via fluorescence microscopy and quantified by ImageJ software.
Biochemical indicators
Enzyme-linked immunosorbent (ELISA) assays were used for quantification of oxidative stress factor levels in line with the manufacturers’ protocols. After the end of drug intervention, mice in all groups were sacrificed, whole blood was taken and allowed to rest for approximately 1 h at 3000 r/min, then centrifuged for 10 min, and serum was collected. HL-1 cells were treated with ISO with/without baicalein and metoprolol, and then the cell culture supernatant was collected. The levels of SOD, MDA, and GSH-Px in serum and cell culture were absorbed by using a Tecan microplate reader at 450 nm.
Western blotting
Cell lysate samples were assayed using RIPA lysis buffer with phosphatase and protease inhibitors, followed by centrifugation at 12,000 rpm for 15 min and separation of the lysate samples at 4°C. Protein concentration in the lysate samples was quantified by the BCA protein assay. The amount of protein sample added per pore was 40 µg. Accurate concentrations of protein samples were determined so that each specimen was uniform at 15 µL per well. Running electrophoresis was set at 80 V for 30 min and 120 V for 40 min. After completion, the membrane was transferred to a PVDF membrane and closed for an hour and a half. Primary antibodies (GAPDH, caspase3, caspase9, cleaved PARP, Drp1, MFF, OPA1, mtTFA, ATF4, CHOP, GRP78, p-PERK, and PERK) were diluted at a proportion of 1:1000. Secondary antibodies were diluted at 1:2000. The blots were subsequently detected by chemiluminescence.
Embryo treatments
Zebrafish (Series Daniorerio, Series AB) were maintained in a circulating aquaculture system (Beijing Aisheng Technology Co Ltd., Beijing, China). The fish were maintained in the zebrafish system for light-dark cycling (14 h: 10 h) at 28.5°C ± 1°C. Zebrafish embryos (4 h) were evenly assigned to 6-well plates, 10 tails with fixed capacity to 5 mL. Ten tails were used in each group: the control group, DMSO group and treatment groups (12.5, 25, and 50 µmol/L baicalein). Zebrafish embryos of each group were cultured in a 28°C incubator, and the survival at 24, 48, 72, 96, and 120 h was recorded. Zebrafish were selected for this study, and all animal experiments were conducted in accordance with the guidelines published by the Institutional Animal Care and Use Committee of Zunyi Medical University.
Statistical analysis
Experimental results were displayed as the mean ± SD. Means were compared by one-way ANOVA followed by Duncan’s post-hoc tests at p ≤ 0.05. GraphPad Prism 7 software was used for data analysis.
Results
Effects of baicalein on zebrafish growth and cell viability
The chemical structure of baicalein is shown in Figure 1(a). The MTT assay showed that there was no inhibitory effect of 12.5–50 µmol/L baicalein on the viability of HL-1 cells after treatment (Figure 1(b)). Zebrafish have tissues, organs and systems similar to humans, and its genes and signalling pathways are more than 85% similar to humans. We found that the survival rates of zebrafish were start to reduced after treatment with 50 µmol/L baicalein (Figure 1(c)). We also found a certain probability of the minor deformity phenomenon on zebrafish embryos in 50 µmol/L baicalein (Figure 1(d)). However, the dose of 50 µmol/L in vitro was not toxic, but through the zebrafish model also predicted that 50 µmol/L may have potential toxicity, and less than 50 µmol/L should be selected in future studies.

Baicalein has no acute-toxicity and cardio-toxicity to zebrafish and HL-1 cells. (a) The chemical structure of baicalein. (b) The toxicity of baicalein on HL-1 cells was measured by MTT. Zebrafish embryos were treated with different concentrations baicalein for different times. The mortalities (c) of embryo-larva were measured. (d) Teratogenic effects on zebrafish embryos and pericardial edema in 50 µmol/L baicalein. Results were presented as mean ± SD (n ≥ 3). ###p < 0.001, versus CON, *p < 0.05, **p < 0.01, ***p < 0.001 versus ISO.
Baicalein alleviated ISO-induced apoptosis in HL-1 cells
As shown in Figure 2(a), the MTT assay revealed that the viability of HL-1 cells was decreased after exposure to ISO (50 µmol/L) compared with the control. However, the viability of HL-1 cells incubated with baicalein was markedly increased in a concentration-dependent manner. We also measured the apoptosis-regulating factors caspase3, caspase9, and cleaved PARP. ISO stimulation can increase intracellular expression of caspase3, caspase9, and cleaved PARP, compared with the ISO group, the level of caspase3, caspase9, and cleaved PARP in the treatment group was significantly decreased (Figure 2(b–e)).

Apoptosis of HL-1 cells upon ISO stimulation, a protective effect exerted by baicalein. (a) The effect of baicalein on the viability of HL-1 cardiomyocytes was measured via MTT assay. (b–e) The protein levels of Caspase3, Caspase9, and Cleaved-PARP in HL-1 cells were determined by western-blot. Outcomes are displayed as average ±SD (n ≥ 3). ###p < 0.001, versus CON, *p < 0.05, **p < 0.01, ***p < 0.001 versus ISO.
Baicalein attenuated ISO-induced oxidative stress in HL-1 cells
Excessive ROS can cause the cell REDOX balance to be broken, which leads to oxidative stress reaction. 20 We examined the level of ROS production in cells exposed to ISO with or without baicalein. HL-1 cells were exposed to ISO for 24 h and then treated with baicalein for 24 h. Following treatments, cells were stained with DCRH-DA. Quantification of fluorescence intensity confirmed significantly reduced levels of ROS in HL-1 cells treated with baicalein compared to the ISO group (Figure 3(a)). Intracellular MDA content increases when oxidative stress occurs in cells. The oxidative stress response caused by ROS will lead to the content of a series of redox-related substances in cells changes, such as lipid oxidation products MDA, SOD, GSH-Px. 21 ISO stimulation can increase intracellular MDA content, compared with the ISO group, the level of MDA in the treatment group was significantly decreased, it is worth noting that ISO + HSH group was reduced by 88%. (Figure 3(c)). On the contrary, ISO stimulation can decrease intracellular GSH-Px and SOD content, compared with the ISO group, the level of GSH-Px and SOD in the treatment group was markedly increased (Figure 3(b and d)).

Baicalein alleviated oxidative stress in HL-1 cell. (a) ROS expression in HL-1 cells was analyzed by fluorescent staining. (scale bars, 100 μm; n ≥ 3 in each group). 12.5, 25, and 50 µmol/L baicalein have differences compared with ISO group. (b–d) Expression of biomarkers MDA, GSH, SOD of oxidative stress was detected by ELISA. ##p < 0.01, versus CON, *p < 0.05, **p < 0.01, versus ISO.
Baicalein inhibited ISO-induced mitochondrial fusion/fission unbalance and GRP78/CHOP pathway in HL-1 cells
We examined the effects of baicalein on mitochondrial biogenesis-related proteins (Drp1, MFF, OPA1, and mtTFA) and ER stress-related proteins (ATF4, P-PERK, PERK, CHOP, and GRP78). Figure 4 shows that the expression of Drp1 and MFF significantly increased and OPA1 and mtTFA decreased in ISO group compared with those in the control group. However, treatment groups resulted in a decrease in the protein expression of Drp1 and MFF and an increase in the protein expression of OPA1 and mtTFA (Figure 4(a–f)). Relative to control group, GRP78, p-PERK, ATF4, and CHOP levels in ISO-induced HL-1 cells were increased but were remarkably decreased by treatment with baicalein (Figure 5(a–e)). Our results indicated that baicalein ameliorated mitochondrial fusion/fission unbalance and GRP78/CHOP pathway.

Western-blot expression of mtTFA (a, c), OPA1 (a, d), Drp1 (b, e), and MFF (b, f) was appraised in HL-1 cells treated by indicated concentration of baicalein or met within/without ISO. Results were presented as mean ±SD (n ≥ 3). ##p < 0.01, ###p < 0.001, versus CON, *p < 0.05, **p < 0.01, ***p < 0.001 versus ISO.
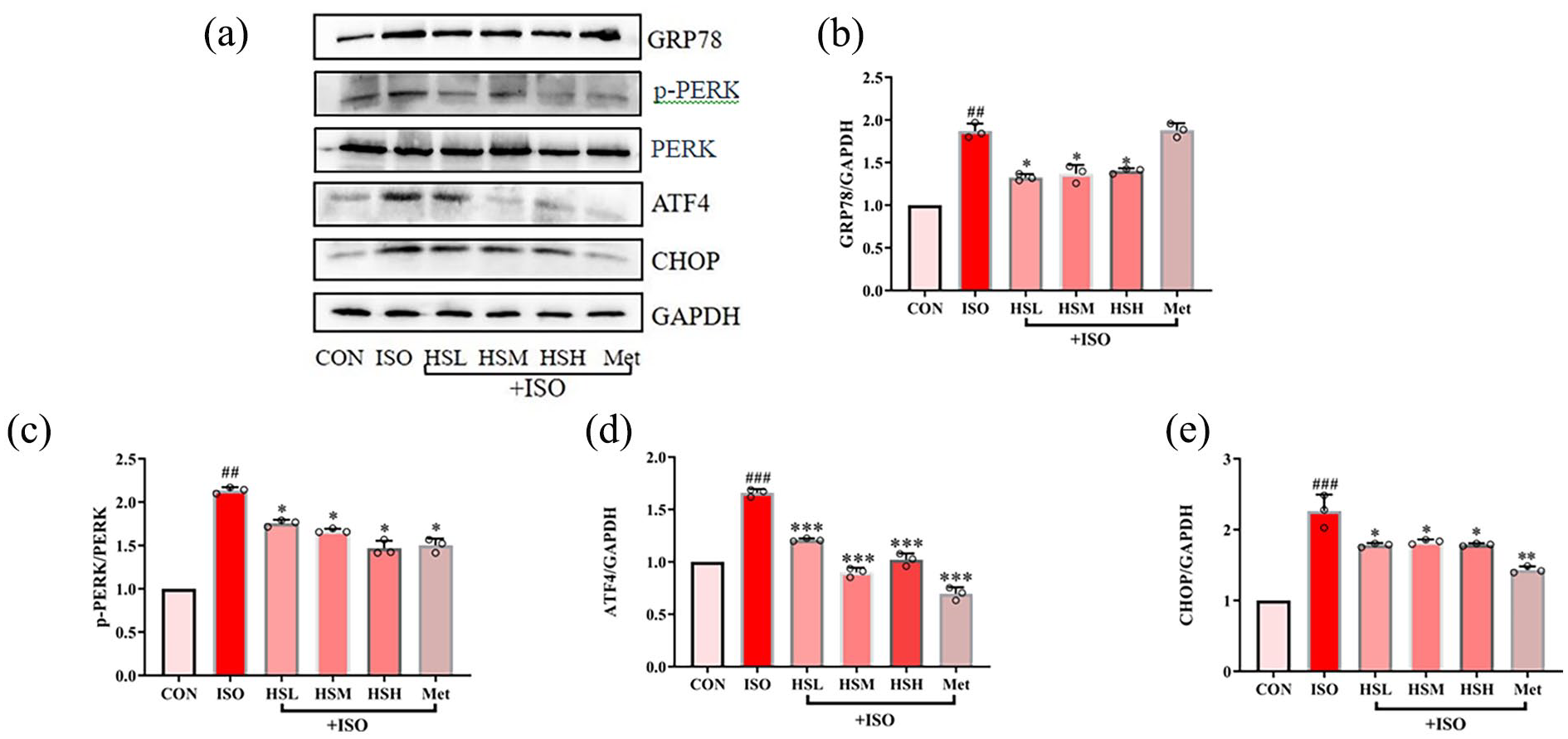
The expression of GRP78 (a, b), p-PERK (a, c) ATF4 (a, d) and CHOP (a, e) was analyzed in HL-1 cells by western blotting. Results were presented as mean ±SD (n ≥ 3). ##p < 0.01, ###p < 0.001, versus CON, *p < 0.05, **p < 0.01, ***p < 0.001 versus ISO.
Baicalein improved cardiac functional and structural deficits in heart failure mice
To analyze the effect of baicalein on heart failure, we gastrically administered baicalein to ISO-induced heart failure mice. Cardiac functional parameters were evaluated by echocardiography. Compared with the control group, the ejection fraction (LVEF) and fractional shortening (LVFS) of the mice were reduced by 24% and 18% under iso stimulation, but the LVEF and LVFS of the ISO+HSH group were only reduced by 8% and 6% compared with the control group (Figure 6(a–c)).

Baicalein inhibits the effect of ISO on cardiac dysfunction in vivo. (a) Representative echocardiographic images of the left ventricle showing ventricular dilation and cardiac dysfunction in heart failure mice, while baicalein inhibited ventricular dilation and cardiac dysfunction. In the ISO group, ejection fraction (b) and shortening fraction (c) decreased significantly, after baicalein intervention, cardiac function increased. Results were presented as mean ±SD (n ≥ 3). ###p < 0.001, versus CON, *p < 0.05, **p < 0.01, versus ISO. (d) HE staining in mouse hearts at day 28 in the different group (scale bars, 50 μm; n ≥ 3 in each group). (e) Masson staining of fibrosis and area quantitation in mouse hearts at day 28 in the different group (scale bars, 50 μm; n ≥ 3 in each group).
HE staining and Masson staining showed normal heart architecture in control group. This revealed obvious structural changes, such as massive infiltration of inflammatory cells, necrosis and fibrosis, in heart tissue from ISO group. Treatment groups showed less tissue damage and fibrosis than ISO group significantly (Figure 6(d and e)).
Generally, cardiac injury and cardiac fibrosis in heart failure are seen alongside increased apoptosis in the heart. We analyzed apoptosis in mouse hearts by using a TUNEL assay. As shown in Figure 7(a and b), the number of TUNEL-positive cells of treatment group was significantly lower than ISO group. Meanwhile, the beneficial effect of baicalein was reflected by the normalization of the increase in caspase-3, caspase-9, and cleaved PARP induced by ISO in mouse hearts (Figure 7(c–f)).

Baicalein inhibits the effect of ISO on apoptosis and oxidative stress in vivo. (a–b) The quantification of TUNEL-positive cell number per high-power field (scale bars, 50 μm; n ≥ 3 in each group). #p < 0.05, versus CON, *p < 0.05, **p < 0.01 versus ISO. n ≥ 3. The protein levels of Caspase3 (c, d), Caspase9 (c, e), Cleaved-PARP (c, f) in cardiac of mice were determined by Western-blot. Outcomes are displayed as average ±SD (n ≥ 3). ##p < 0.01, versus CON, *p < 0.05, **p < 0.01, ***p < 0.001 versus ISO. n ≥ 3. The expression of MDA (g), GSH (h) and SOD (i) during oxidative stress in the mouse serum was determined by ELISA (n ≥ 3). #p < 0.05, ##p < 0.01, ###p < 0.001, versus CON, *p < 0.05, **p < 0.01, versus ISO.
To assess oxidative stress in heart tissues, we analyzed the levels of MDA, GSH-Px, and SOD in mouse serum. GSH-Px and SOD were decreased and MDA was increased in ISO-induced heart failure, while baicalein administration reversed these effects in mouse serum (Figure 7(g–i)).
Baicalein suppresses ISO-induced mitochondrial fusion/fission unbalance and GRP78/CHOP pathway in the heart
We also confirmed the inhibitory effect of baicalein on mitochondrial biogenesis-related proteins (Drp1, MFF, OPA1, and mtTFA) (Figure 8(a–f)) and GRP78/CHOP pathway related proteins (GRP78, p-PERK, ATF4, and CHOP) in heart failure mouse hearts (Figure 8(g–k)).

Baicalein inhibits the expression of mitochondrial fusion/fission related protein and ER stress related protein. Western-blot expression of Drp1 (a, c), MFF (a, d), mtTFA (b, e), and OPA1 (b, f) was appraised in rat cardiacs (n ≥ 3). ##p < 0.01, ###p < 0.001, versus CON, *p < 0.05, **p < 0.01 versus ISO. Western blot expression of GRP78 (g, h), p-PERK (g, i), ATF4 (g, j), and CHOP (g, k) was analyzed in cardiac of rats (n ≥ 3). ##p < 0.01, ###p < 0.001, versus CON, *p < 0.05, **p < 0.01, ***p < 0.001 versus ISO.
Discussion
HF is a serious disease characterized by myocardial remodelling, hypertrophy and fibrosis. The systolic and or diastolic dysfunction results in the inadequate cardiac output for the body’s metabolic needs in patient with HF. 22 Although people have made considerable progress in the pathogenesis of HF over the past few decades, the mortality of HF is still high. 23 Therefore, the development of new treatment for HF has important implications. As the natural phenolic antioxidant isolated from Scutellaria baicalensis Georgi, baicalein is the most abundant component, and have been found alleviated myocardial remodelling, 24 but specific mechanisms need to be further explored. The focus of this research was to scrutinize the role of baicalein as a cardioprotective emissary in vivo and in vitro and to unveil the latent mechanisms underlying these effects.
It has been reported that BALB/c strain could be better studied as a model of heart failure compared to other mouse strains. 17 In clinic, β receptor blockers are commonly used to treat the symptoms caused by heart failure and other diseases, mainly metoprolol tartrate, propranolole hydrochloride, which have the effect of inhibiting myocardial contractility. In this experiment, metoprolol was used as a positive control drug, and our study found that the therapeutic effect of baicalein on heart failure at a dose of 25 µmol/L was equivalent to that of metoprolol. 25
Cardiac agglomeration of ROS has been observed in many cardiovascular diseases, such as HF in rodents and humans. 26 The unfavorable impacts of ROS in HF include the triggering of apoptosis-related signaling pathways, proliferation of cardiac fibroblasts, mtDNA damage, mitochondrial dysfunction, cardiac hypertrophy and so on. These eventually lead to maladaptive myocardial reshaping and cardiac dysfunction. 27 Downregulation of the ROS scavenging system, such as MDA, GSH, and SOD, has been demonstrated in animal models of heart failure. 28 It has been reported that baicalein attenuates cardiac hypertrophy in mice via suppressing oxidative stress and activating autophagy in cardiomyocytes. 29 In addition, baicalein protects isoproterenol induced myocardial ischemic injury in male Wistar rats by mitigating oxidative stress and inflammation. 30 These studies have confirmed that baicalein fulfills an essential function in protecting cardiac cells by suppressing oxidative stress. Consistent with these studies, we show that baicalein effectively reduced oxidative stress in mice heart tissues and HL-1 cells through restoration of GSH-Px, SOD and MDA activity levels.
Mitochondria have come to be an appealing target for novel therapies against heart failure due to mitochondria critical role on ATP generation, redox balance, Ca2+ homeostasis and cell death. 31 The maintenance of mitochondrial bioenergetics depends on a dynamically integrated quality control axis that allows segregation of damaged mitochondria. Disruption of genes that regulate mitochondrial quality control mechanisms such as mitochondrial fusion or fission lessens both myocardial functionality and viability. Drp1 has been demonstrated to be relevant to the advancement of mitochondrial fission and mitochondrial malfunction in sepsis-induced cardiomyopathy. 32 OPA1 is downregulated in ischemic heart failure in the human heart and rat hearts, and Mfn1/2 and Drp1 are enhanced in human nonischemic dilated cardiac failure. OPA1 influences ROS production and regulates antioxidant gene expression and TFAM, Mfn1/2, Bax, Bak and Nrf2 expression. 33 Lower levels of Mfn2, augmented Fis1 and diminished OPA1 in rat hearts have also been observed in an uncomplicated myocardial infarction pattern. 34 Our study determined that the depletion of the protein levels of Drp1, MFF, OPA1, and mtTFA were significantly attenuated by baicalein. Using in vivo and in vitro model of heart failure, we provided that failing hearts exhibit disrupted mitochondrial quality control, characterized by loss of mitochondrial fusion and fission balance, while baicalein controlled mitochondrial fusion and fission balance.
ROS overproduction causes ER protein misfolding, leading to ER stress. 35 If the ER is overloaded, ER stress can be induced in heart failure. 36 Perturbation of any of the major functions of ER (protein folding, lipid and sterol synthesis, and Ca2+ homeostasis) results in ER stress via the activation of complex cytoplasmic and nuclear signaling pathways collectively termed the unfolded protein response (UPR) (also known as misfolded protein response). 37 Molecular studies on ER function and proteins indicate that ER-associated functions are critical in cardiac physiology and pathology. 38 The UPR triggers an adaptive response to reestablish ER homeostasis by coordinating reduction in the quantity of protein expressed, with increased production of chaperones to handle accumulation of misfolded protein, as well as promotion of ER-associated degradation to remove misfolded proteins. This initial response of protein synthesis suppression and upregulation of ER resident chaperones is designed to resolve the ER stress, restore homeostasis, and enhance survival. 39 If ER stress is severe or prolonged, the UPR may stimulate apoptosis, a significant feature of Heart failure. 40
At the same time, the maintenance of a dynamic state of mitochondria directed implications in recovering stress in ER. GRP78 is one of the molecular chaperone proteins of the ER, which exists in the ER and the extracellular environment. It plays a key role in maintaining the normal glycosylation and folding of proteins in the endoplasmic reticulum, the correct positioning of secreted and transmembrane proteins, and the interception of proteins with quality problems. 41 Appropriate ER stress can up-regulate the expression of GRP78, which then binds to misfolded proteins and promotes the correct folding of these proteins, thereby protecting cells from apoptosis. 42 Usually, PERK binds to GRP78, but when ER stress occurs, PERK separates from GRP78 to form PERK homodimer, oligomerize, and cause its own phosphorylation and kinase activation of kinase domain, and activate downstream signaling pathways, reduce the synthesis of proteins in the ER cavity, reduce the burden of ER, and maintain the balance of the intracellular environment. 43
CHOP is also an ER stress marker protein. Continuous or severe ER stress can increase the expression of GRP78 and CHOP protein, cause cell apoptosis and tissue damage, and promote ER apoptosis. 44 GRP78 was first found to be increased in the human heart in heart failure. 45 CHOP knockout mice produced less cardiac hypertrophy, fibrosis and heart dysfunction than wild-type mice after TAC, implying that CHOP may also contribute to the changeover from heart hypertrophy to cardiac failure. 46 We found that baicalein markedly suppressed the expression of GRP78, p-PERK, ATF4 and CHOP in vivo and in vitro, suggesting that reduced ER stress related pathway-GRP78/CHOP protects cardiomyocytes.
However, there has certain limitations in our work. First of all, the sample size is small. In order to ensure the uniformity of the initial weight of the experimental animals, at the end of a week of adaptive breeding, we removed the mice with weight difference and only six mice in each group, which can lead to insufficient statistical power and increase the risk of false negative results; therefore, future studies should improve the reliability of statistical conclusions by expanding the sample size. Notably, the main limitation of this study is that the understanding of the mechanism of action of HF is still incomplete. Ulteriorly, the detailed intrinsic mechanisms of baicalein in improving heart failure by regulating mitochondrial dysfunction and ER stress pathways remains to be elucidated.
Conclusion
In the existing research, we assessed the curative effects of baicalein against ISO-induced heart failure in mice and in HL-1 cells and analyzed the cardioprotective latency exerted by baicalein via its intervention in mitochondrial fusion/fission balance and GRP78/CHOP pathway. We verified the importance of mitochondrial fusion-fission in HF pathology. Overall, our findings provide insight into some of the mechanisms by which baicalein controlling mitochondrial fusion/fission balance and inhibiting GRP78/CHOP pathway.
Footnotes
Acknowledgements
We thank Elsevier Language Editing Services for its linguistic assistance during the preparation of this manuscript.
Author contributions
Yu Wang and Guoyou Zhang contributed to the literature search and study design. Zhao Zhang, Xuan Zhang, Yan Yang, HongYang Wang, Yangjun Yang, and Liying Xuan carried out experiments. Zhao Zhang, Xuan Zhang and Yan Yang contributed to data collection and analysis. Zhao Zhang and Xuan Zhang participated in the drafting of the article. Yu Wang and Guoyou Zhang revised the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was endorsed by the National Natural Science Foundation of China (No. 81760780), the Inner Mongolia Natural Science Foundation (No. 2020MS08031, No. 2022MS08080, No. 2022MS08082), the Inner Mongolia Health and Family Planning Foundation (No. 201702114, No. 202202267), Science and Technology Department of Inner Mongolia Scientific Research Fund Project (No. 2021GG0279), The Central Government Guides Local Science and Technology Development Fund projects (No. 2022ZY0159).
Ethics approval
Ethical approval for this study was obtained from *Zunyi Medical University Laboratory Animal Welfare & Ethics Committee (ZMU21-2404-006)*.
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation. BALB/c mice were obtained from Y is Experimental Animal Technology Co Ltd. in Changchun (No. 202000035165). They were housed in a plastic cage with a 12-h light-dark cycle, room temperature (25°C ± 2°C), and humidity (60% ± 10%). They were given a regular pellet feed and unrestricted access to water. All of the mices were cared for by competent staff members, and every attempt was made to minimize the quantity of animals used as well as their suffering.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Code availability
Not applicable.
Availability of data and material
All the data in this study are available upon reasonable request from the corresponding author.
