Abstract
Background
Glufosinate ammonium (GLA) is a widely used herbicide in rural Vietnam, often associated with severe toxicity. Given the limited diagnostic resources at primary care levels, identifying simple and effective predictors of severity is critical for early triage and intervention.
Objective
To identify clinical and biochemical predictors of severe outcomes in patients with glufosinate ammonium poisoning using multivariable logistic regression.
Methods
We conducted a retrospective study of 83 patients with confirmed glufosinate ammonium poisoning admitted to our poison control center at Bach Mai hospital between March 2023 and October 2024. Demographic, clinical, and laboratory data were collected. Severe outcomes were defined as the need for mechanical ventilation, inotropic support, Glasgow Coma Score ≤8, or in-hospital mortality. Logistic regression was used to identify significant predictors and evaluate model performance.
Results
The mean age was 48.6 ± 17.3 years; 62.7% were male; 88% of cases resulted from intentional ingestion; six patients died (7.2% mortality). Significant predictors of severe outcomes included glufosinate ammonium ingestion >100 ml, (Odds Ratio (OR) 4.5, p < 0.001), time to hospital >6 h (OR 3.2, p = 0.004), pH < 7.35 (OR 5.7, p < 0.001), lactate >4.0 mmol/L (OR 6.2, p < 0.001), creatinine >110 µmol/L (OR 4.3, p = 0.005), and NH3(ammonia) > 100 µmol/L (OR 5.4, p = 0.002). The final logistic regression model including pH < 7.35, time to hospital >6 h, NH3 > 100 µmol/L, and ingested volume >100 mL demonstrated good discrimination (AUC = 0.84, 95% CI 0.74–0.93; p < 0.001) and acceptable calibration by the Hosmer–Lemeshow test (p = 0.47).
Conclusion
Readily available clinical and laboratory indicators, such as pH < 7.35, delayed hospital arrival, high ammonia levels, and large ingested volume, were strong predictors of severe outcomes in glufosinate ammonium poisoning. The simple logistic model (AUC = 0.84) may support early triage in low-resource settings, but prospective validation is needed.
Introduction
Glufosinate ammonium is a non-selective herbicide widely used worldwide, with increasing application since 2012 after paraquat was banned. 1 Chemically, glufosinate ammonium is the ammonium salt of phosphinothricin, a glutamate analog. The tripeptide often confused here is bialaphos, which releases glufosinate after metabolism. The preferred International Union of Pure and Applied Chemistry (IUPAC) name is the ammonium salt of 2-amino-4-[hydroxy(methylphosphinoyl)]butanoic acid, a structural analog of glutamic acid. Its toxic effects are mediated through inhibition of glutamine synthetase, leading to elevated extracellular glutamate, neurotoxicity, and disruption of the Krebs cycle, which results in metabolic acidosis and hyperlactatemia.2,3 Several prognostic factors for severe glufosinate ammonium poisoning have been studied globally, including the ingested dose,3–5 time elapsed from ingestion to hospital admission, 6 blood ammonia levels, 7 altered artery blood gas, 5 seizure episodes, 2 brain damage, 3 and altered consciousness.1,8
In Vietnam, especially in rural areas where herbicide usage is widespread, glufosinate ammonium poisoning presents a significant challenge due to the limited diagnostic and therapeutic capacity at local health facilities.4,9 GLA is a neurotoxic agent that may lead to metabolic acidosis, respiratory failure, and multi-organ dysfunction if not managed promptly. 9 Patients often initially present to district or community health centers with limited diagnostic and treatment resources.9,10 Therefore, identifying simple, reliable predictors of severity can support frontline physicians in making decisions regarding treatment or referral. Our study provides simple, easy-to-perform clinical and paraclinical factors with quick results to predict severity and provide treatment directions for patients with glufosinate ammonium poisoning in emergency units.
Methods
This retrospective observational study was conducted at the Poison Control Center of Bach Mai hospital. We reviewed medical records of 83 consecutive patients admitted between March 2023 and October 2024 with confirmed glufosinate ammonium poisoning. This retrospective study was approved by the Bach Mai Hospital Institutional Review Board (approval number: BMH/IRB/2023-1675/QD-DHYD). Patient data were de-identified prior to analysis, only authorized personnel accessed the dataset, stored on secured institutional servers.
Inclusion criteria: History of glufosinate ammonium ingestion Urinary detection of glufosinate ammonium was performed within 24 h of admission using an in-house validated HPLC/LC-MS/MS method. The lower limit of detection was approximately 0.05 mg/L, consistent with previously published methods (Hori YH et al., 2002
11
; Ohara T et al., 2021
12
). Among 83 patients, 80 (96.4%) had laboratory confirmation by urinary HPLC. In three patients, urine samples were unavailable due to delayed referral or prior catheterization, diagnosis in these cases was based on a reliable ingestion history confirmed by family members and verification of the original product container. Only patients with complete clinical, biochemical, and outcome data were included.
Exclusion: Co-ingestion of other toxic substances or missing critical data.
Collected data: Demographics: age, sex, occupation Exposure: volume ingested, time to admission Clinical: vomiting, abdominal pain, seizure, Glasgow coma scale (GCS), vital signs Labs: pH, HCO3, lactate, NH3, creatinine, Aspartate Aminotransferase (AST), Alanine Aminotransferase (ALT), sodium, potassium, hemoglobin, glucose Severe outcomes: Mechanical ventilation, inotropic support, Glasgow coma scale ≤8, or death. The ingested volume was primarily estimated based on patient or family self-report. When available, this estimate was cross-checked against the residual volume in the original container brought to the hospital. If discrepancies occurred, the lower of the two estimates was recorded. No biochemical back-calculation was performed.
Statistical analysis
Data were analyzed using IBM SPSS Statistics version 27. Continuous variables were expressed as mean ± standard deviation (SD) or median (interquartile range, IQR) as appropriate, and categorical variables as frequencies and percentages. Between-group comparisons were performed using the independent-samples t-test or Mann–Whitney U test for continuous variables and the Chi-square or Fisher’s exact test for categorical variables.
Univariate logistic regression was first used to identify potential predictors of severe outcome. Variables with p < 0.10 in univariate analysis were entered into a multivariable logistic regression model after checking for multicollinearity (Variance Inflation Factor <2.0). Four predictors were retained in the final model based on clinical relevance and statistical significance: pH < 7.35, time from ingestion to hospital >6 h, NH3 > 100 µmol/L, and ingested volume >100 mL. The number of severe outcomes was 31, with four predictors in the final model, resulting in an events-per-variable (EPV) ratio of 7.8. Although the EPV was slightly below the conventional threshold of 10, the retained predictors were clinically grounded and model overfitting risk was mitigated by prior univariate screening and assessment of multicollinearity.
Model performance was assessed by the area under the receiver operating characteristic (ROC) curve (AUC) with 95% confidence intervals calculated using asymptotic methods. Calibration was evaluated by the Hosmer–Lemeshow goodness-of-fit test. Due to the limited sample size, cross-validation and bootstrap resampling were not applied. All analyses were two-tailed, and results were considered statistically significant at p < 0.05.
Results
Demographics: Mean age: 48.6 ± 17.3 years Male: 62.7% Intentional ingestion: 88% The median time from ingestion to hospital arrival was 6.1 h (IQR 4.0–9.5 h). Mean volume ingested: 95.4 ± 72.1 mL
Clinical/Lab Findings: Nausea/vomiting: 94%, abdominal pain: 61%, seizures: 33%, GCS <13: 45% Heart rate >100 beats per minute (bpm): 21%, <60 bpm: 15%, respiratory rate >24 breaths per minute: 18% pH <7.35: 56%, lactate >4.0: 63%, creatinine >110: 41%, NH3 > 100: 23.1%
Grouped analysis by ingested volume:
Association between ingested volume and severe outcomes.

Severe outcomes include mechanical ventilation, inotropic support, GCS ≤8, or death.
Severe outcomes (composite) occurred in 31 of 83 patients (37.3%). Individual components of the composite outcome were: mechanical ventilation in 18 patients (21.7%) inotropic support in 12 patients (14.5%) Glasgow Coma Scale ≤8 in 20 patients (24.1%) and in-hospital mortality in six patients (7.2%).
Because components overlapped, the sum of individual counts exceeds the number of patients with the composite outcome. Specifically, 15 patients experienced exactly one severe component, 10 patients experienced two components, four patients experienced three components, and two patients experienced all four components.
Univariate logistic regression analysis of candidate predictors.

Abbreviations: OR: odds ratio; CI: confidence interval; GLA: glufosinate ammonium; bpm: beats per minute; GCS: Glasgow Coma Scale.
Variables with p < 0.10 in univariate analysis were entered into the multivariable logistic regression model.
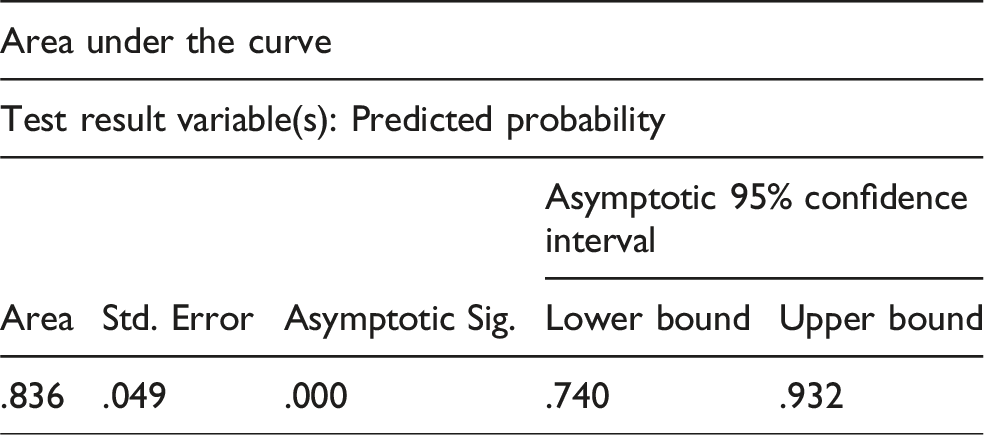

ROC curve of the final logistic model (pH < 7.35, NH3 > 100 µmol/L, volume >100 mL, time to hospital) predicting severe outcomes (AUC = 0.84, 95% CI 0.74–0.93; p < 0.001).
Of the 83 patients, 31 (37.3%) experienced at least one severe outcome component (mechanical ventilation n = 18, inotropic support n = 12, GCS ≤8 n = 20, in-hospital mortality n = 6). These 31 events formed the basis of prediction modeling. In logistic regression, the model showed fair discrimination, with an AUC of 0.84 (95% CI 0.74–0.93, p < 0.001). The ROC curve is presented in Figure 1. This analysis used asymptotic 95% confidence intervals without cross-validation or bootstrap resampling due to the limited sample size. The final logistic regression model including pH < 7.35, time to hospital >6 h, NH3 > 100 µmol/L, and ingested volume >100 mL demonstrated good discrimination (AUC = 0.84, 95% CI 0.74–0.93; p < 0.001) and acceptable calibration by the Hosmer–Lemeshow test (p = 0.47). Logistic Model Equation (TRIPOD requirement):
The final multivariable logistic regression model included four predictors. The estimated coefficients were: Intercept = −3.42 pH < 7.35 = β = 1.74 NH3 > 100 µmol/L = β = 1.71 Ingested volume > 100 mL = β = 1.52 Time to hospital > 6 h = β = 1.28 Thus, the predicted probability of a severe outcome is: P = 1/(1 + e^–(–3.42 + 1.74·(pH<7.35) + 1.71·(NH3>100) + 1.52·(Volume>100) + 1.28·(Delay>6h)). Example:
For a patient with pH < 7.35, NH3 > 100 µmol/L, ingested volume >100 mL, and delayed arrival: Linear predictor = −3.42 + 1.74 + 1.71 + 1.52 + 1.28 = 2.83
Predicted probability = 0.944 (94.4% risk).
Discussion
This study identified several independent predictors of severe outcomes among patients with glufosinate ammonium poisoning, including large ingestion volume, delayed hospital arrival, metabolic acidosis (pH <7.35), and elevated ammonia levels. These factors are biologically plausible and consistent with previous studies.3–5,7,13,14
Glufosinate ammonium, a glutamate analog, inhibits glutamine synthetase, resulting in hyperammonemia, excitotoxicity, and mitochondrial dysfunction.2,3,7 Consequently, metabolic acidosis and hyperlactatemia are markers of systemic hypoxia and correlate strongly with poor outcomes.5,8,14 Time to hospital arrival and ingested dose reflect exposure severity and access to timely medical care.3,4,6,13
Compared with general severity scores such as APACHE II or SOFA, 15 our simplified model uses readily available clinical and biochemical variables and is therefore practical for frontline triage in resource-limited settings. Although internal validation (bootstrap or cross-validation) was not performed due to sample size constraints, model discrimination remained statistically significant (AUC = 0.84). External validation in larger prospective cohorts is warranted. 16
Conclusion
We developed a simple and practical severity prediction model based on basic clinical and laboratory parameters. This model may assist frontline clinicians in early risk assessment and triage, particularly in low-resource settings, but requires further prospective validation.
Footnotes
Acknowledgements
Poison Control Center, Bach Mai Hospital.
Ethical considerations
This retrospective study was approved by the Bach Mai Hospital Institutional Review Board (approval number: BMH/IRB/2023-1675/QD-DHYD). Due to the retrospective nature using de-identified medical records, individual informed consent was waived in accordance with institutional guidelines.
Consent for publication
Individual informed consent was waived by the Bach Mai Hospital IRB due to the retrospective use of de-identified medical records.
Author contributions
D.D. Nguyen: conception, writing; L.H.A. Nguyen: data collection, methodology; B. Nguyen: formatting.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The de-identified dataset supporting the conclusions of this article is available from the corresponding author upon reasonable request.
