Abstract
Glufosinate ammonium poisoning can cause neurological complications even after a symptom-free period. We prospectively investigated the predictors of neurologic complications in acute glufosinate ammonium poisoning and the change of serum ammonia level as a predictor of patient’s presence and recovery of neurologic complication. This prospective observational study collected data from consecutive patients diagnosed with acute glufosinate ammonium poisoning between September 2014 and June 2016. Serum ammonia was serially measured. The patients were divided into two groups: the neurologic complication group and the nonneurologic complication group. We also defined 25 other insecticide- or herbicide-poisoned patients as controls. The neurologic complication group included 18 patients (72.0%). The latency period for neurologic complications was within 48-h postingestion. The peak ammonia level was statistically higher in the neurologic complication group than in the control group (p < 0.001) and the nonneurologic complication groups (p = 0.001). There was a statistical difference between the nonneurologic complication group and the neurologic complication group (p = 0.0085) in terms of ingested amount. The peak ammonia was the only predictor for the development of neurologic complications (the optimal cutoff: 90 μg/dL). In patients with mental changes, the mean serum ammonia levels before and after recovery of the mental changes were statistically different (p = 0.0019). In acute glufosinate ammonium poisoning, serial serum ammonia level measurements are needed and a serum peak ammonia level greater than 90 μg/dL is a predictor of neurologic complications. Also, it is important to treat the hyperammonemia in acute glufosinate ammonium poisoning.
Introduction
Glufosinate, a phosphorus-containing amino acid, is an active ingredient in nonselective herbicides and is a glutamate analog that inhibits glutamine synthetase (GS) in plants and blocks glutamine synthesis from glutamate and ammonia. The associated intracellular accumulation of ammonia causes tissue necrosis and death. 1 –3 Glufosinate ammonium–containing herbicides are formulated with the ammonia salt of glufosinate, an anionic surfactant, such as sodium polyoxyethylene alkylether sulfate and propylene glycol as a solvent. 4,5
Glufosinate ammonium herbicide is now available in many countries, and its use has gradually increased. 6 Since 2013, glufosinate ammonium herbicide has seen greater use as a nonselective herbicide in Korea because sales of Gramoxone® (paraquat) were banned in Korea in November 2012; glufosinate ammonium is advertised to have a low toxicity to both humans and the environment. However, previous reports indicate that glufosinate ammonium poisoning can cause severe complications, such as serious disturbances of consciousness, seizures, and amnesia, after latent periods of 4–60 h following ingestion. 1,2,7 –11 Therefore, glufosinate-poisoned patients should be carefully observed for several days after ingestion, even if the clinical signs are not remarkable on admission.
In particular, it would be helpful for clinicians if a laboratory test could predict neurologic complications. Recently, serum hyperammonemia has been suggested to indicate neurologic complications in glufosinate ammonium poisoning. However, existing reports regarding the serum ammonia level as a probable predictor of neurologic complications are limited to case reports or retrospective studies. 1,3,12 –14 We also reported that the initial serum ammonia level, which is performed at the emergency department (ED), may predict neurologic complications. 12
We prospectively investigated serial serum ammonia level and occurrence of neurologic complications in glufosinate ammonium poisoning and attempted to determine a trend of serum ammonia levels according to the presence and recovery of neurologic complications.
Methods
Patients
This prospective observational study collected data from consecutive patients diagnosed with acute glufosinate ammonium poisoning between September 2014 and June 2016. The ED was located in a single, urban, tertiary-care hospital with more than 43,000 annual visits that is staffed 24 h a day by board-certified emergency physicians. This study was approved by the Institutional Review Board of Yonsei University Wonju College of Medicine (approval number: YWMR-14-7-059). Each participant received information regarding the study aims, and they all provided written informed consent.
Acute glufosinate ammonium poisoning was confirmed by patient or guardian statements, and a physician verified the agent and transcribed the bottle label into the patient records. All patients with hyperammonemia upon ED arrival received a lactulose enema to lower the ammonia level, as high levels have been suggested to cause neurologic complications. 12 All glufosinate ammonium–poisoned patients were admitted to the intensive care unit (ICU) for close observation for more than 1 day.
Patients were excluded for the following reasons: (1) poisoning with any additional material except for alcohol; (2) liver diseases, chronic renal disease, or cardiac arrest before ED arrival, chemotherapy, current urinary tract infection, gastrointestinal hemorrhage, or multiple myeloma, which can cause hyperammonemia; (3) a history of seizure; (4) presentation more than 12 h following ingestion; and (5) refusal to participate.
Study variables and definitions
The following clinical parameters were assessed: age, sex, ingested amount, intentionality of poisoning, co-ingestion of alcohol, time elapsed from ingestion to ED arrival, use of gastric irrigation or charcoal, medical history, initial vital signs, initial symptoms, and neurologic and nonneurologic complications upon admission, such as mental changes (Glasgow Coma Scale, GCS < 13), seizures, amnesia, intubation and mechanical ventilation, respiratory failure, shock (systolic blood pressure <90 mmHg), pneumonia, cardiac arrest during hospitalization, and inhospital mortality. We defined the amount of ingested glufosinate ammonium as follows: “a little” or “a spoonful” as 5 mL, “a mouthful” as 25 mL, “a small cup” as 100 mL, and “a bottle” as 300 mL. 15 Respiratory failure was defined as hypoxia (partial pressure of arterial oxygen (PaO2) < 70 mmHg) or hypercapnia (partial pressure of arterial carbon dioxide (PaCO2) > 60 mmHg). Intubation and mechanical ventilator care were initiated if the patient required airway protection or respiratory failure treatment.
Laboratory parameters included serum ammonia level, arterial blood gas analysis, lactate, alanine transaminase (ALT), and creatinine (Cr). The serum ammonia level was obtained using a Dimension Vista® System (Siemens, Munich, Germany). Venous or arterial blood was collected in a tube containing lithium heparin to measure the ammonia concentrations. The tube was completely filled, tightly capped, stored on ice, and centrifuged without delay. The samples were analyzed within 30 min of centrifugation. The Dimension Vista Ammonia method is an enzymatic method that uses glutamate dehydrogenase and a stabilized nicotinamide adenine dinucleotide phosphate oxidase analog. Ammonia reacts with α-ketoglutarate and reduced cofactor to form
The patients were divided into two groups: the neurologic complication group (patients with mental changes, seizures, and amnesia during their hospital course) and the nonneurologic complication group (patients without these complications). We also evaluated patients poisoned with 25 other insecticides or herbicides that do not produce hyperammonemia to compare their serum ammonia level with glufosinate ammonium–poisoned patients; these patients were the control group.
Study end point
The primary outcome was to investigate the predictors of neurologic complications in acute glufosinate ammonium poisoning. The secondary outcome was evaluating the change of serum ammonia level as a predictor of patient’s presence and recovery of neurologic complication.
Statistical analysis
Data are expressed according to the properties of the variable. Continuous variables are presented as the mean and standard deviation or the median and range. Categorical variables are shown as a frequency and percentage. To compare the three groups, we performed a χ 2 test (Fisher’s exact test), analysis of variance, or Kruskal–Wallis test as appropriate. We analyzed multiple comparisons using Bonferroni correction, Sheffé’s method, and/or Dunn’s procedure. If patients had neurologic complications, we conducted a paired t-test to compare their serum ammonia levels before and after recovery. Logistic regression analysis identified factors that predict neurologic complications, and the results were expressed as odds ratio (OR) with 95% confidence interval (CI). The area under the receiver operating characteristic (ROC) curve was used to evaluate the ability of serum ammonia for classifying neurologic complications. We suggested an optimal cutoff of serum ammonia using Youden’s Index. A p value less than 0.05 was statistically significant, and all statistical analyses were conducted using SAS version 9.4 (SAS Inc., Cary, North Carolina, USA).
Results
Patient characteristics
Between September 2014 and June 2016, 74,000 patients visited our hospital’s ED; 29 consecutive patients with acute glufosinate ammonium poisoning were identified, and 25 were finally included. The exclusion criteria were poisoning with zolpidem (one patient), liver diseases or renal disease that can cause hyperammonemia (two patients), and refusal to participate in this study (one patient).
All patients had ingested an herbicide containing 18.0% glufosinate; 56.0% were men, and the mean age was 55.3 years (range: 27–82 years). The poisoning was intentional in 96.0% of cases, and patients arrived at the ED a median of 2.0 h after ingestion. All patients presented to our hospital within 6 h of ingestion. Hypertension (28.0%) was the most common past medical condition. At their initial presentation, patients complained of nausea (63.0%), vomiting (31.8%), and dyspnea (31.8%). All patients received gastric irrigation or gastric lavage and activated charcoal.
We also analyzed the control group which contained 25 patients with poisoning from other insecticides or herbicides, including pyrethroid (nine patients, 36%), organophosphate (six patients, 24%), carbamate (five patients, 20%), glyphosate (three patients, 12%), organochlorine (one patient, 4%), and sethoxydim (one patient, 4%). Of these 25 patients, 48.0% were men. The mean age was 56.4 years (range: 32–80 years). The poisoning was intentional in 88.0% of cases, and the median duration between ingestion and ED arrival was 3.0 h (Table 1).
Baseline and clinical characteristics of 25 patients poisoned with glufosinate ammonium herbicide and 25 patients defined as the control group.

Cx: complications; ED: emergency department; DM: diabetes mellitus; HTN: hypertension; SBP: systolic blood pressure; PR: pulse rate; RR: respiratory rate; BT: body temperature; PaO2: partial pressure of arterial oxygen; PaCO2: partial pressure of arterial carbon dioxide; ALT: alanine transaminase.
aMean ± standard deviation.
bMedian (interquartile range).
cNonneurologic Cx group versus neurologic Cx group (ingested amounts: p value = 0.009, serum peak ammonia: p value = 0.001).
dControl group versus neurologic Cx group (serum peak ammonia: p value < 0.001).
Incidence and patterns of complications in acute glufosinate ammonium poisoning
Neurologic complications during hospitalization after glufosinate ammonium poisoning were mental change (18 patients, 72.0%), seizure (7 patients, 28.0%), and amnesia (8 patients, 34.8%). All neurologic complication group patients experienced mental changes. All patients reported neurologic complications within admission day 1 (within 48 h postingestion). Of the 18 patients with mental changes, 10 patients (58.8%) reported the changes (GCS < 13) on admission day 0 (within 24 h postingestion). Of these 10 patients, seven patients arrived at the ED with an alert mental status and reported the mental change while in the ED; the other three patients arrived at the ED with a decreased mental status. The remaining eight patients (44.4%) reported a mental change on admission day 1 and experienced no neurologic complications on admission day 0. The median recovery time was 2.5 days in 17 of 18 patients with mental changes; and one patient did not recover a normal mental status due to hypoxic brain injury following cardiac arrest during admission before recovery from a decreased mental status. Seven patients experienced seizures: two (28.6%) on admission day 0 (within 24 h postingestion) and five (71.4%) on admission day 1 (within 48 h postingestion).
Nonneurologic complications included intubation and mechanical ventilation (12 patients, 48.0%), shock (four patients, 16.0%), pneumonia (seven patients, 28.0%), and cardiac arrest (two patients, 8.0%). One patient died from pneumonia during hospitalization (Table 2).
A comparison of nonneurologic complications according to the presence of neurologic complications.

Cx: complications; ICU: intensive care unit.
aMedian (interquartile range)
Comparisons between the control group, nonneurologic complication group, and neurologic complication group
A comparison of patient characteristics is shown in Table 1. The neurologic complication group comprised 18 patients (72.0%) with glufosinate ammonium poisoning. Patients in each group differed in terms of ingested amount (p = 0.018) and peak ammonia level (p < 0.001). Statistical differences were observed for the ingested amounts between the nonneurologic complication group and the neurologic complication group (p = 0.009) in multiple comparison. The peak ammonia level was statistically higher in the neurologic complication group than in the control group (p < 0.001) and nonneurologic complication groups (p = 0.001) in multiple comparison (118.33 μg/dL vs. 26.36 μg/dL vs. 56.29 μg/dL; Table 1 and Figure 1).

The mean peak ammonia level in each groups. Cx: complications.
A subgroup analysis of serum ammonia level was based on the presence of neurologic complications and the day they developed and revealed that patients with neurologic complications on admission day 0 (within 24 h postingestion) had a statistically higher serum ammonia level than the nonneurologic complication group. In patients with neurologic complications on admission day 1 (within 48 h postingestion), an increase in the serum ammonia level was seen the next day, although there was no statistical difference among the three groups (Table 3 and Figure 2).
Serial patterns of serum ammonia in each group.

Cx: complications; ammonia 0: serum ammonia level checked on admission day 0; ammonia 1: serum ammonia level checked on admission day 1.
aMean ± standard deviation.
bNonneurologic Cx group versus neurologic Cx group on admission day 0 (ammonia 0: p = 0.037).
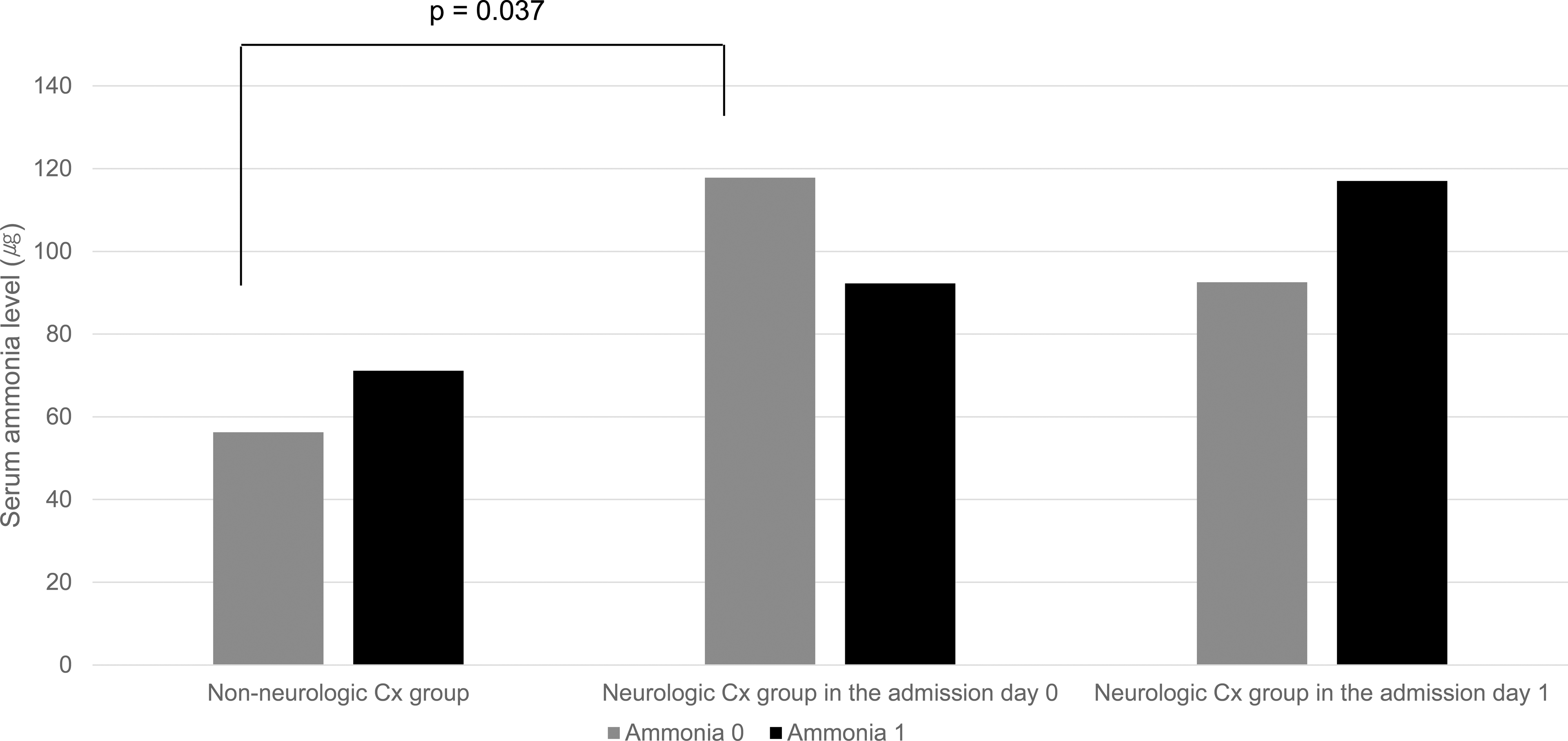
Serial pattern of serum ammonia in each groups. Cx: complications; ammonia 0: serum ammonia level, which was performed on admission day 0 (within 24 h postingestion); ammonia 1: serum ammonia level, which was performed on admission day 1 (within 48 h postingestion).
ALT, pH, bicarbonate, and Cr levels did not differ during the first 48 h after ingestion between the three groups. There were also no statistically significant differences between the control group and the glufosinate ammonium poisoning group regarding their baseline characteristics (Table 1).
The neurologic complication group was more likely to receive intubation and mechanical ventilator care than the nonneurologic complication group (p = 0.005). The median ICU admission length was statistically longer in the neurologic complication group than in the nonneurologic complication group (6.5 days and 2.0 days, respectively, p = 0.001; Table 2).
Predictors of neurologic complications and change of serum ammonia after recovery of neurologic complications
Multiple logistic regression analyses to determine factors related to the development of neurologic complications identified peak ammonia level (OR: 1.075, 95% CI: 1.004–1.152, p = 0.038) as statistically significant. The optimal point of peak ammonia level was 90 μg/dL (sensitivity: 72%, specificity: 100%, and area under the curve: 0.881) according to ROC curves and the Youden Index (Table 4).
Predictors of neurologic complications as determined by multivariate logistic regression analysis.

OR: odds ratio; CI: confidence interval.
In patients with mental changes, the mean peak ammonia levels were statistically different from the serum ammonia level, which was assessed when the mental changes disappeared (118.33μg vs. 70.65μg/dL, p = 0.002). In the neurologic complication group, the serum peak ammonia level in seizure patients was higher than that in patients without seizure, although there was no statistically significant difference (135.71 μg/dL vs. 107.27 μg/dL, p = 0.279; Table 5).
Change in serum ammonia level between altered mental status and recovered mental status in the neurologic complication group.

aMean ± standard deviation.
Discussion
Predictors of neurologic complications and change of serum ammonia after recovery of neurologic complications
This study prospectively confirmed that the peak ammonia level was an only predictor of neurologic complications in acute glufosinate ammonium poisoning. The mean peak ammonia level of patients with mental changes was statistically different from the serum ammonia level of when the mental change is resolved. Therefore, treating the hyperammonemia in acute glufosinate ammonium poisoning is important to recover the patient’s neurological complications.
Because glufosinate is an inhibitor of synthesis of glutamine from glutamate and ammonia in plants, the intracellular accumulation of ammonia can cause tissue necrosis and death. 1 –3 Although few studies are available regarding the effects of glufosinate on mammals, there may be some similarity between mammalian and plant GS. In those studies, increased hepatic ammonia levels were found at more than a sublethal dose of glufosinate exposure; the increased ammonia level effect can be partially attributed to mammalian GS inhibition. 9,16
The arterial ammonia level is not well correlated with the brain ammonia level because the brain does not significantly take ammonia up from the arterial blood under normal physiological conditions. It has been debated whether ammonia enters the brain through passive diffusion and/or active transport by ion transporters. However, if the arterial ammonia level is elevated, increased uptake of ammonia by the brain can occur. 17,18 In addition, the formulated anionic surfactant contained in glufosinate herbicide may increase the uptake of ammonia into the brain by increasing the permeability of the blood–brain barrier. 19 Therefore, in acute glufosinate ammonium–poisoned patients, increases in the serum ammonia level may raise brain ammonia levels.
Elevated ammonia in the brain can induce the activation of N-methyl-
Incidence of complications and comparisons between the nonneurologic complication group and the neurologic complication group
In this study, 72.0% of the patients were placed in the neurologic complication group. Some differences in the incidence of neurologic complications in previous studies exist 3,5,12,13 that may have occurred due to the definition of complication group and the severity of poisoned patients. However, similarities in the results of recent human studies (including our prospective study) indicate that acute glufosinate ammonium poisoning causes serious complications much more often than previously reported. 3,5,12,13 Accordingly, we should remember that glufosinate ammonium is not safe for human ingestion. This study also reconfirmed that neurologic complications frequently appeared after an asymptomatic interval of several hours. Therefore, we suggest that glufosinate ammonium–poisoned patients with no symptoms upon ED arrival receive close observation to detect serious complications, including mental changes, seizures, and the need for intubation and mechanical ventilation.
In acute glufosinate poisoning, various complications are associated with the ingested amount. In our previous study, as well as those of Mao et al. and Inoue et al., the ingested amounts were not statistically different between the noncomplicated group and the complicated group. 3,12,13 However, in Moon et al.’s study, the ingested amount was significantly higher in the complicated group than in the noncomplicated group. 5 In the present study, the ingested amount was statistically higher in the neurologic complication group than in the nonneurologic complication group. This difference may be because it is difficult to accurately measure ingested amounts, especially in patients with decreased mental status. However, unlike the previous retrospective study, we prospectively investigated the ingested amounts, so the confidence of our results is higher.
Although hyperammonemia can be a secondary phenomenon of acidosis, shock, and renal and hepatic dysfunction, which may be toxic manifestations of glufosinate ammonium poisoning, 5 the serum ALT, Cr, pH, and arterial bicarbonate levels during the first 48 h after ingestion did not differ between the two groups.
In this study, intubation and mechanical ventilation were statistically higher in the neurologic complication group than in the nonneurologic complication group, and there were no nonneurologic complications, including intubation and mechanical ventilator care, in the nonneurologic complication group; only the neurologic complication group demonstrated nonneurologic complications. Therefore, it is important to anticipate neurologic complications, since they may also predict various nonneurologic complications. Intubation and mechanical ventilation may be required in glufosinate ammonium poisoning, such as with neurologic complications like mental changes and seizures, to protect the airway. In addition, respiratory suppression requiring mechanical ventilation may be caused by the glufosinate itself. Reports about respiratory suppression by glufosinate showed that the prognosis of patients significantly depends on the management of respiratory arrest. 6 In this study, 12 patients received intubation and mechanical ventilator care: five intubations for respiratory failure without mental change and seven intubations due to decreased mental changes and uncontrollable seizures. Because mechanical ventilator care and decreased mental status were higher in the neurologic complication group, ICU admission days were statistically longer in the neurologic complication group.
Study limitations
This study had some limitations. First, the number of patients was small due to it being a single-center study. Second, we could not exclude the effects of the contained surfactant. However, the ingested glufosinate concentration was 18% in all patients. Third, patient initial mental status can be affected by the co-ingestion of alcohol and glufosinate herbicide. Although the lack of evaluation of the presence of alcohol co-ingestion using the serum ethanol level in all included patients was a limitation of our study, there were no differences in the incidence of alcohol co-ingestion between the nonneurologic complication group and the neurologic complication group. Further studies are required to clarify these details.
Conclusions
In acute glufosinate poisoning, serial serum ammonia level measurements are needed and a serum peak ammonia level greater than 90 μg/dL is a predictor of neurologic complications. Also, it is important to treat the hyperammonemia in acute glufosinate ammonium poisoning.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
