Abstract
This study investigated whether ammonia concentrations can predict delayed neurotoxicity development and neurotoxicity latency in glufosinate ammonium (GLA) herbicide-poisoned patients presenting with an alert mental state and stable hemodynamics. This retrospective observational case study included 26 patients divided into 2 groups: neurotoxicity during hospitalization (complicated group) and without neurotoxicity (noncomplicated group). Thirteen patients (50.0%) experienced neurotoxicity at 16 h post-ingestion. Although ammonia concentrations at presentation did not differ significantly between the two groups, the ammonia level in the complicated group increased significantly at the next measurement and remained significantly higher than that in the noncomplicated group until 48 h after ingestion. The peak ammonia concentration before neurotoxicity development was an independent predictor of neurotoxicity (odds ratio: 1.047, 95% confidence interval: 1.010–1.087, p value = 0.014), and the optimal cutoff value of peak ammonia concentration for predicting neurotoxicity was 101.5 μg/dL. The rate of ammonia increase was not associated with the time latency from ingestion to neurotoxicity development. This study showed that serial ammonia measurements in GLA-poisoned patients may identify those who are at high risk of developing neurotoxicity. However, as this study enrolled few patients, further qualified trials are required to confirm our results and to reveal the etiology of hyperammonemia and its causality in neurotoxicity.
Introduction
Glufosinate (GLF) irreversibly inhibits glutamine synthetase (GS), which catalyzes the synthesis of glutamine from glutamate and ammonia. GS inhibition leads to the accumulation of intracellular ammonia and to a deficiency of glutamine, which is phytotoxic. 1 Glufosinate ammonium (GLA)-containing herbicides are formulated with the ammonia salt of GLF, the anionic surfactant sodium polyoxyethylene alkyl ether sulfate and propylene glycol ether.
GLA herbicide is now available in many countries, including the United States, Canada, Germany, the United Kingdom, and Korea, and its use has gradually increased. 2 The increased use of GLA herbicide has been accompanied by an increased incidence of human poisoning cases in Asian countries, such as Korea, Japan, and Taiwan. 1,3
The characteristic features of acute GLA herbicide poisoning in humans are primarily classified into two categories: neurotoxicity and hemodynamic toxicity. 4 Hemodynamic toxicity can likely be attributed to the formulated surfactant, 5 whereas neurotoxicity, including seizure, an altered mental state, amnesia, and central apnea requiring mechanical ventilation support, is thought to be caused by GLF. 2
Neurotoxicity frequently appears after an asymptomatic interval of several hours and can be as delayed as 52 h post-ingestion. 6 –8 This incidence of delayed neurotoxicity is as high as 50%. 6,9 Therefore, a major challenge for physicians is to identify those patients who are likely to develop delayed neurotoxicity during the early stage after ingestion. Extremely few clinical markers have been shown to be useful for the early identification of these patients. 9 Recently, hyperammonemia was suggested as an indicator of severe GLA herbicide poisoning in a case report; however, the blood ammonia levels were measured after the development of neurotoxicity. 7 No subsequent studies investigating whether hyperammonemia is the cause or the effect of neurotoxicity and whether hyperammonemia can predict neurotoxicity have been performed.
This study investigated whether ammonia concentrations in the blood can predict patients who present a score of 15 on the Glasgow Coma Scale (GCS) and stable hemodynamics after GLA herbicide ingestion and who are likely to develop neurotoxicity following hospital admission.
Materials and methods
Study design
This investigation was a single-institution, retrospective observational case study performed by chart review. The study design was approved by the institutional review board at Chonnam National University Hospital, Gwangju, South Korea.
Subjects
The inclusion criteria allowed the participation of patients who were 18 years or older who presented to our emergency department (ED) with a score of 15 on the GCS and a systolic blood pressure (BP) of more than 90 mmHg without vasopressor infusion after GLA herbicide ingestion between 2010 and 2015 Feburary. The patients who were discharged with T60 codes, which were classified according to the International Classification of Disease (ICD)-10, were selected using the hospital electronic medical record system, and their medical records were reviewed. A diagnosis of GLA herbicide poisoning was made by a history of GLA herbicide ingestion provided by the patient and by a verified record of the ingested agent by a physician.
For inclusion in the study, the patient’s ammonia levels should have been measured at presentation and should have been measured more frequently than every 12 h during the first 2 days after ingestion.
The exclusion criteria included the following: a history of hepatic dysfunction; hematological malignancy; recent surgery, epileptic seizure, or renal failure; current urinary tract infection; antiepileptic drug use; presentation after 24 h following ingestion or co-ingestion with another drug; and transfer before establishing a final outcome or discharge against medical advice.
Finally, the enrolled patients were classified into the following two groups according to the development of neurotoxicity during hospitalization: the complicated group, which included patients who experienced neurotoxicity, and the noncomplicated group, which included patients who did not present neurotoxicity. Neurotoxicity was defined as seizures, central respiratory failure requiring mechanical ventilation support, or altered mentality (GCS < 13).
Data collection
The following data were collected during the medical chart review: age; gender; amount of ingested GLA herbicide; time interval from ingestion to arrival at our ED; electrocardiogram (ECG) data at presentation; laboratory results, including venous ammonia at presentation and during the first 48 h after ingestion; development of neurotoxicity; onset time of neurotoxicity after ingestion; duration of hospitalization; and survival outcome.
When a patient developed neurotoxicity later than 48 h after ingestion, the ammonia concentrations from 48 h after ingestion to the development of neurotoxicity were also collected. Serum alanine aminotransferase (ALT), arterial base excess (BE), and creatinine (Cr) levels were collected during the first 48 h after ingestion to investigate the cause of elevated ammonia. The amount of GLA herbicide ingested was estimated in quantities as a spoonful (5 mL), mouthful (25 mL), cup (100 mL), or bottle (300 mL). ECG data were interpreted by an emergency medicine physician.
Venous blood was collected in a tube-containing lithium heparin to measure ammonia concentrations. The samples were immediately transported to the emergency clinical laboratory and centrifuged at 3400g for 15 min at 4°C. The ammonia concentrations in the separated plasma were determined by an enzymatic method in which glutamate dehydrogenase catalyzes the reaction of ammonium with β-nicotinamide adenine dinucleotide and α-ketoglutarate to form glutamate (β-NADP+) and water using a UniCel Dex 800 (Beckman Coulter, Brea, California, USA). The normal range of ammonia concentrations at our hospital is 19–60 μg/dL.
The ammonia concentration at presentation and the peak ammonia concentration were evaluated to predict the development of delayed neurotoxicity in patients who were poisoned with GLA herbicide and who appeared healthy on presentation. Because the preceding ammonia level before the development of neurotoxicity is useful as a clinical predictor, the peak ammonia concentration was designated as the highest level before the development of neurotoxicity in the complicated group. Because an observation period of 48 h is recommended in asymptomatic GLA herbicide-poisoned patients, 2 the peak ammonia concentration in the noncomplicated group was defined as the highest level during the first 48h after ingestion.
Additionally, we assessed whether the rate of ammonia increase is associated with the latency of neurotoxicity after ingestion. This rate was calculated using the following formula: the increased ratio of the ammonia concentration at presentation to the peak concentration/log 10 (time interval from presentation to the peak ammonia concentration (h).
The serial ammonia level during the first 48 h after ingestion was assessed according to the following time categories to compare the change in the ammonia level after GLA herbicide ingestion over time between the two groups: at presentation, at presentation < ammonia level ≤ 12 h after ingestion, 12 h after ingestion < ammonia ≤ 24 h after ingestion, and 24 h after ingestion < ammonia ≤ 48 h after ingestion. If multiple measurements were performed during this period, the highest level was used.
Statistical analyses
The baseline patient characteristics are presented as frequencies for categorical variables and as medians and interquartile ranges for continuous variables.
The continuous variables that were obtained at presentation were compared using the Mann–Whitney test. Multivariate logistic regression analysis was performed to investigate whether ammonia at presentation and peak ammonia are independent predictors of neurotoxicity. Ammonia variables with significant differences between the two groups and those variables in the univariate analysis with a significance level <0.05 that could easily be assessed at presentation were considered sufficient for inclusion in the logistic regression analysis. A receiver operating characteristic (ROC) curve was generated to obtain the area under the curve (AUC) with the 95% confidence interval (CI) as well as the sensitivity and specificity of the ammonia variables that were selected in the logistic regression analysis for predicting patients who developed neurotoxicity. The Youden index (sensitivity + specificity − 1) was obtained, and the ammonia concentration with the maximum Youden index value was selected as the optimal cutoff value. The serial ammonia, ALT, Cr, and BE levels during the first 48 h after ingestion were compared using the Friedman test, and a post hoc comparison was performed using the Wilcoxon rank test with Bonferroni’s adjustment. Correlations between the rate of ammonia increase and the latency of neurotoxicity were determined using Spearman’s rank method. The values of p < 0.05 were considered statistically significant. All statistical analyses were performed using the Statistical Package for the Social Sciences version 21.0 software.
Results
Of the 42 patients who met the inclusion criteria, 26 were included in the subsequent analyses after applying the exclusion criteria. All patients ingested an herbicide containing 18.0% GLA to attempt suicide and presented to our hospital within 3 h after ingestion with a GCS score of 15 and a median systolic BP of 120.0 mmHg. Thirteen (50.0%) patients experienced neurotoxicity at a median of 16 (12.0–30.5) h after ingestion, and two patients died.
Table 1 presents the baseline and clinical characteristics of the enrolled patients. The patients in the complicated group were older and ingested a larger amount of GLA herbicide than the patients in the noncomplicated group (Table 1). The patients in the complicated group also had a higher white blood cell (WBC) count at presentation.
Baseline and clinical characteristics of 26 patients poisoned with glufosinate ammonium herbicide.
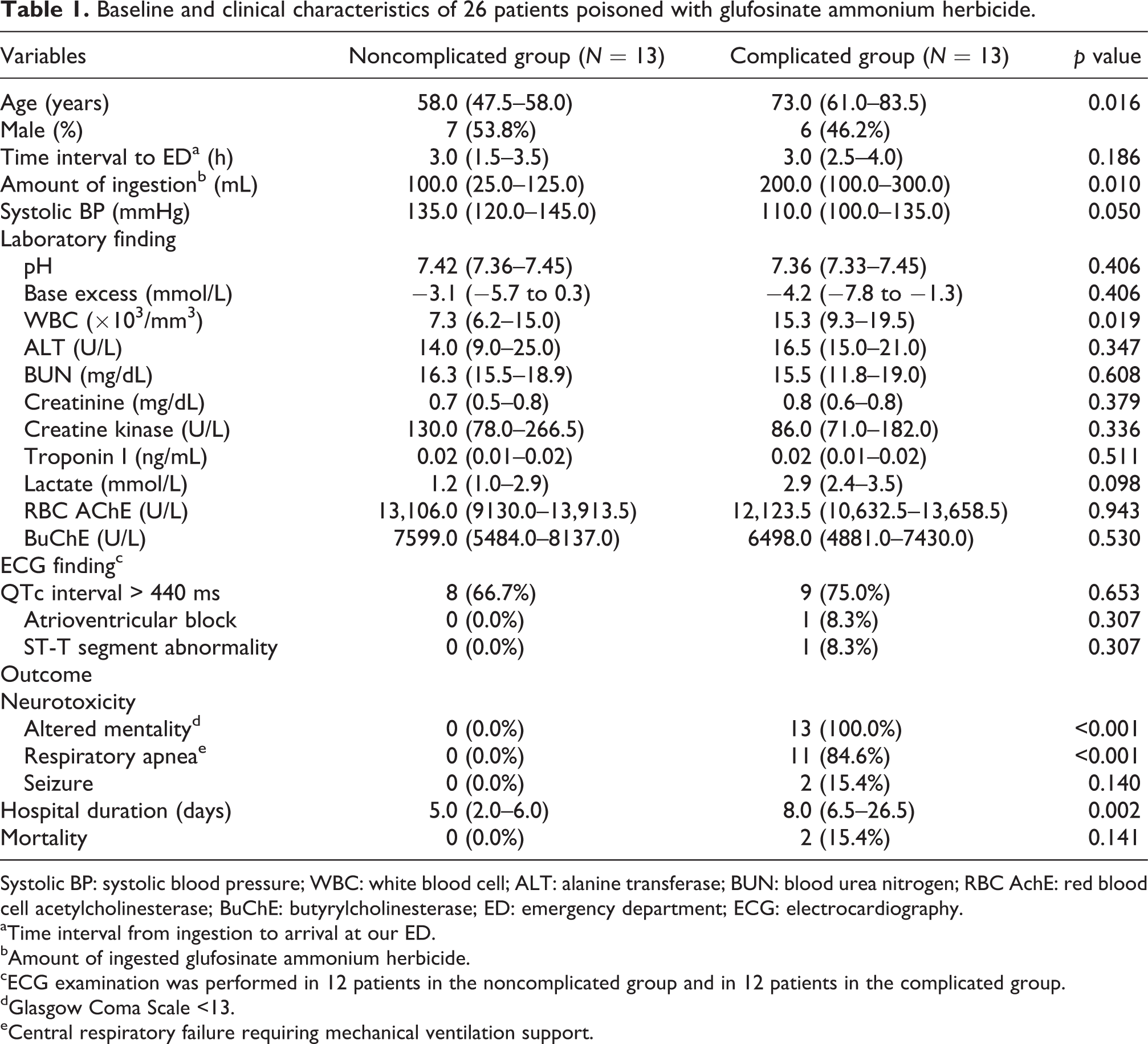
Systolic BP: systolic blood pressure; WBC: white blood cell; ALT: alanine transferase; BUN: blood urea nitrogen; RBC AchE: red blood cell acetylcholinesterase; BuChE: butyrylcholinesterase; ED: emergency department; ECG: electrocardiography.
aTime interval from ingestion to arrival at our ED.
bAmount of ingested glufosinate ammonium herbicide.
cECG examination was performed in 12 patients in the noncomplicated group and in 12 patients in the complicated group.
dGlasgow Coma Scale <13.
eCentral respiratory failure requiring mechanical ventilation support.
The initial ammonia level at presentation did not significantly differ between the two groups, but the ammonia level in the complicated group was significantly higher than that in the noncomplicated group at the next measurement within 12 h after presentation. This was also observed at 12–24 h after ingestion and at 24–48 h after ingestion (Table 2). As shown in Figure 1, the complicated group showed a tendency of increasing ammonia over time, which was in contrast to the decreasing and normalized tendency of the ammonia level in the noncomplicated group. However, the ALT, BE, and Cr levels did not differ during the first 48 h after ingestion between the two groups. The complicated group also had a higher peak ammonia than the noncomplicated group (Figure 2). Logistic regression analysis that included the peak ammonia concentration, age, the amount of ingested GLA herbicide, and WBC demonstrated that peak ammonia was an independent predictor of neurotoxicity (odds ratio = 1.047, 95% CI = 1.010–1.087, p value = 0.014). Based on the ROC curve analysis, the peak ammonia concentration had an AUC of 0.905 (95% CI: 0.793–1.00) for the development of neurotoxicity and an optimal cutoff value of 101.5 μg/dL, with 100% sensitivity and 69.2% specificity, respectively.
Venous ammonia concentration over time in 26 patients poisoned with glufosinate ammonium herbicide.
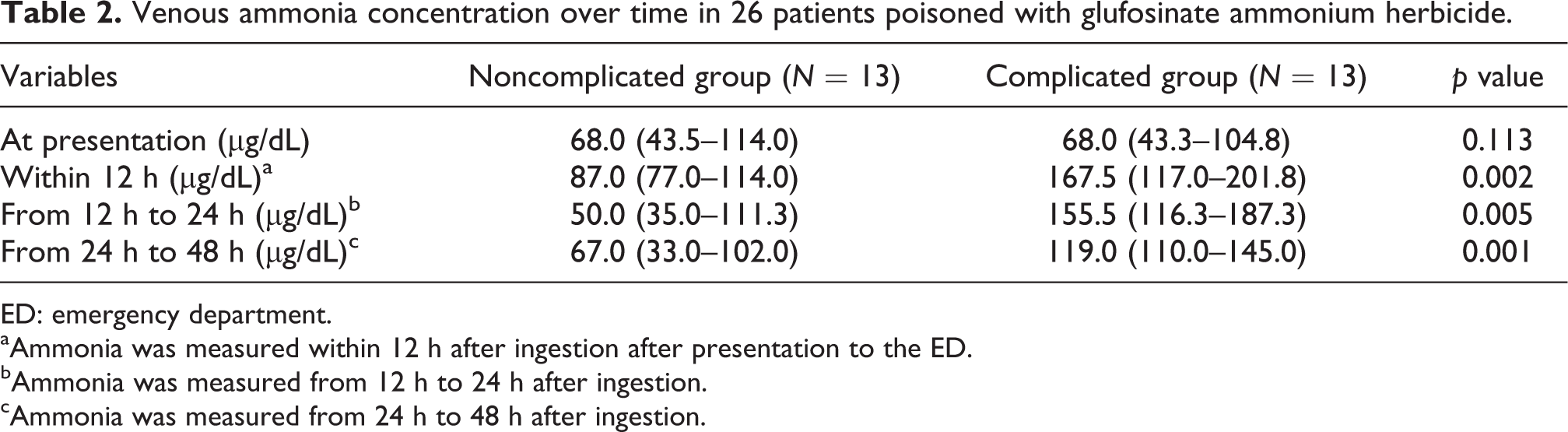
ED: emergency department.
aAmmonia was measured within 12 h after ingestion after presentation to the ED.
bAmmonia was measured from 12 h to 24 h after ingestion.
cAmmonia was measured from 24 h to 48 h after ingestion.

The changes in venous ammonia over the first 48 h after glufosinate ammonium herbicide poisoning. The ammonia level in the complicated group at presentation (68.0 (43.3–104.8) µg/dL) was significantly lower than that at the next measurement within 12 h after presentation (167.5 (117.0–201.8) µg/dL, p = 0.048). The highest ammonia level, which was measured within 12 h after ingestion after presentation to the ED, was plotted as 12 h. Similarly, the highest ammonia levels, measured at the interval from 12 h to 24 h after ingestion and at the interval from 24 h to 48 h after ingestion, are designated as 24 h and 48 h, respectively, on the graph. *p < 0.05: compared with the ammonia concentration at presentation in the same group. ED: emergency department.

The peak ammonia concentration according to the development of neurotoxicity. The complicated group had significantly higher peak ammonia concentrations than the noncomplicated group (158.0 (122.0–203.0) μg/dL in the complicated group vs. 95.0 (43.5–117.0) μg/dL in the noncomplicated group, p < 0.001.). The gray areas indicate the normal range of ammonia at our hospital.
The rate of ammonia increase was not associated with the latency of neurotoxicity from ingestion (r = 0.328, p = 0.274).
Discussion
In our study, neurotoxicity occurred in 50.0% of the patients at a median time of 16 h after ingestion. Consistent with our results, neurotoxicity developed in as many as 50% of patients and was delayed until 4–60 h post-ingestion of GLA herbicide. 6,9,10
Ammonia levels differ depending on the measurement site, and venous ammonia levels are usually lower than the arterial levels due to ammonia uptake by several tissues. 11 –13 Arterial ammonia is considered more reliable than venous ammonia because arterial ammonia most likely better reflects the levels of ammonia reaching the brain circulation. However, recent studies have shown that utilizing arterial blood is not necessary when measuring ammonia in the blood and that venous blood is sufficient. 11,13,14 In addition, venous blood measurements are simpler to perform than arterial blood sampling in clinical practice.
The suggested mechanism of GLA herbicide-related neurotoxicity is GLA itself, GLA metabolites, or N-methyl-
In contrast, neurotoxicity in butterflies that were fed GLA was not reproduced by the intra-arterial injection of ammonia. 23 In rats, an oral lethal dose of GLA, which inhibited cerebral GS and induced seizures, did not increase the cerebral ammonia concentration. 24 Our study demonstrated that the rate of ammonia increase and the latency of neurotoxicity are not associated. These animal studies and no correlation between the latency of neurotoxicity and the rate of increase of ammonia in our study may not be the primary cause of GLA neurotoxicity. Further studies examining the role of ammonia in neurotoxicity are required.
Although the cause of hyperammonemia in GLA herbicide poisoning remains unclear, hyperammonemia can be a secondary phenomenon of acidosis, shock, renal failure, and hepatic dysfunction, which are toxic manifestations of GLA herbicide poisoning. However, the serum ALT and Cr levels and arterial BE levels during the first 48 h after ingestion did not differ between the two groups. GS inhibition by GLA and the direct absorption of the ammonium ion of GLA may account for hyperammonemia. The serum GLF concentration reached its peak level within 3 h after ingestion, and the half-life at α phase of the serum GLF concentration was 1.84 h. 2 In our study, the ammonia concentration at presentation was measured at 3 h after ingestion, and the ammonia level increased continuously after presentation. The rapid absorption of GLF and the delayed elevation of ammonia suggest that ammonia is less likely to originate from the ammonium ion of GLA. In rats, only a lethal dose (1600 mg/kg) of GLA, which significantly inhibited all hepatic, renal, and cerebral GS until 7.5 days after exposure, led to slight increases in the ammonia levels in the liver, but not in the kidneys and brain, at 1 day after exposure. 24 In addition to GS, glutamate dehydrogenase and carbamyl phosphate synthetase are involved in the removal of ammonia in mammals. 25 Considering the alternative mechanisms of ammonia removal and the absence of dose-related increases in ammonia concentrations according to inhibited GS activities in an animal study, 24 the contribution of GS inhibition to hyperammonemia requires further investigation.
This study showed that patients with a risk of neurotoxicity exhibited a tendency of increasing ammonia levels after presentation and that patients with venous ammonia concentrations of more than 101.5 μg/dL, regardless of the measurement time, may be at a high risk of developing neurotoxicity.
In conclusion, serial measurement of the ammonia level in the blood can be helpful in predicting patients who are at a high risk of developing delayed neurotoxicity. However, the etiology of hyperammonemia and the causality between hyperammonemia and neurotoxicity require further investigation.
Limitations
The first limitation of this study is that the number of patients included was too small to draw definite conclusions. This limitation is an inherent pitfall of toxicology reports. This small number of patients may result in the exclusion of significant variables in multivariate analyses; nonetheless, ammonia concentrations at four time points were not overlooked because of the inclusion criteria. Second, the measurement of ammonia can be influenced by the mishandling of the sample or by a delay in the sample reaching the laboratory. 26 Because of the retrospective design of this study, no standardized protocol for ammonia measurement was followed. However, blood samples are transferred as soon as possible to the emergency clinical laboratory in our hospital. In addition, if the results of the ammonia concentration appear abnormal, the laboratory workers recommend that the physician should repeat the analysis. Third, the GLA concentration was not measured. The GLA concentration can confirm the diagnosis of GLA herbicide poisoning and help reveal the etiology of hyperammonemia using GLA concentration-related ammonia levels. Fourth, considering that the fastest onset of neurotoxicity in our study was 5 h after ingestion, the classification of serial ammonia levels using 12-h intervals was too long. A shorter time interval for classification is required to detect significant changes in ammonia levels to help predict patients with a high risk of neurotoxicity at an earlier state. A final limitation was that this study was performed at one center and the results may lack widespread applicability. Further studies to overcome these limitations are necessary.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
