Abstract
Background
Lipopolysaccharide (LPS) is a glycolipid that constitutes the Gram-negative bacteria outermost membrane main portion. LPS is frequently of concern in medicine because of nearly all severe sepsis patients have elevated LPS plasma levels, which cause life-threatening organ dysfunction. Consequently, the potential protective benefit of acriflavine (Ac) in limiting LPS-induced acute inflammatory response and the possible underlying mechanisms were investigated.
Methods
Male albino mice were treated with i.p. Ac 4 or 8 mg/kg/day for 2 weeks, then received a single i.p. LPS (10 mg/kg) at day 14.
Results
Ac administration ameliorated hepatic, pulmonary, and testicular dysfunction, as confirmed by attenuation of pathological changes and amendment of oxidative stress parameters. This was associated with inhibition of protein kinase R-like endoplasmic reticulum kinase/phosphatidylinositol 3-kinase (PERK/PI3K) endoplasmic reticulum (ER) stress pathway; toll-like receptor 4/nuclear factor kappa B (TLR4/NF-κB) and active caspase-1/gasdermin D (GSDMD)-N-terminal as well as interleukin-1 beta (IL-1β) inflammatory and pyroptotic signals.
Conclusion
Our results highlighted the protective potential of Ac in a mouse model of LPS-mediated systemic inflammatory response, which paves the way for its clinical application in sepsis.
Introduction
Lipopolysaccharide (bacterial endotoxin) is the main outer membrane constituent of Gram-negative bacteria. 1 LPS can stimulate non-sterile inflammatory response, which accounts for its pathogenicity in many organs such as liver, 2 lung, 3 and testis. 4 In several clinical studies, elevated levels of plasma endotoxin have been detected in approximately 70 to 80% of all individuals with severe sepsis, as well as septic shock.5,6 Epidemiologically, according to WHO, it is documented that the sepsis prevalence worldwide represents about 48.9 million, amongst which 11.0 million patients died representing 19.7% of all deaths globally. 7 Yet, management of sepsis is limited to anti-infection as well as life-supporting measures. 8 Hence, the identification of targeted therapies for treatment and/or prevention from sepsis is crucial. Targeting LPS-mediated immune responses could be a hopeful approach for sepsis treatment.
The inflammatory response of immune cells is originated by germline-encoded pattern recognition receptors (PRRs) that identify the molecules of damaging cells and compounds of microorganisms. The pathogen-associated molecular patterns (PAMPs) are deemed as microbe-specific molecular compounds involving bacterial LPS. 9 The PRRs include toll-like receptors (TLRs). 10 Amongst numerous TLRs, TLR4 is an important receptor to perceive bacterial LPS and generates inflammatory response. TLR4 can use downstream adaptor molecules such as myeloid differentiation primary-response protein 88 (MyD88) to conduct a chain of reaction that leads to nuclear factor kappa B (NF-κB) translocation into the nucleus. NF-κB, in turn, accounts for the pro-inflammatory cytokine expression such as interleukin-1 (IL-1), tumor necrosis factor-α (TNF-α), and interferon (IFN)-γ 11 and triggers the production of NOD-like receptor protein 3 (NLRP3).12,13
Acriflavine, an over-the-counter medicine, is utilized for the microbial infections treatment in fishes and humans, mainly as an antiseptic agent having a quaternary ammonium moiety recognized to have antiseptic mechanisms of action comprising inhibition of the synthesis of deoxyribonucleic acid (DNA), protein and cell-wall.14–16 Of note, the widespread available clinical studies on Ac have not shown any major side effects after the long-term systemic administration of large doses. 17 Furthermore, Ac is US Food and Drug Administration (FDA)-approved hypoxia inducible factor-1 (HIF-1) inhibitor for non-oncological human utilization. 18 An earlier study, which tested the impact of Ac in a mouse model of colorectal cancer, has reported the inhibitory effect of Ac in multiple immune cell types in the case of inflammation-driven cancers. 19 By inhibiting the phosphatidylinositol 3-kinase (PI3K)/phosphoinositide-dependent kinase-1 (PDPK1) pathway, Ac inhibits crucial adaptive mechanisms essential for cell survival during metabolic stress. 20 Moreover, a previous experiment revealed that Ac could inhibit hepatocellular carcinoma (HCC) growth and enhance antitumor efficacy in vivo. 21 Furthermore, it inhibited severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and other beta coronaviruses. 22
In addition to conventional use of Ac as antiseptic, we hypothesized that Ac could ameliorate LPS-induced multi-organ injury via modulation of pyroptosis, inflammation, and endoplasmic reticulum (ER) stress pathways. This study is the first to investigate the possible underlying mechanisms by which Ac exhibited its effect against LPS-induced acute inflammatory response. Thus, the current study was implemented to assess the potential therapeutic benefits of Ac in limiting the acute inflammatory response following exposure to the non-sterile microbial component LPS in mice.
Materials and methods
Experimental animals, drugs, chemicals and study design
Experimental animals
Seventy-six adult male albino mice weighing 25 ± 5 g were kept in cages randomly (six mice/cage) for 2 weeks to acclimate to laboratory conditions. They were kept in an animal house at 25°C with 50% humidity and maintained under 12-h light/dark cycle. Mice had free access to plenty of standard food and water supply. Animals were purchased from “Vacsera: The Egyptian Organization for Biological Products and Vaccines”, Giza, Egypt.
Ethical considerations
The research procedure was authorized by Mansoura University Animal Care and Use Committee (MU-ACUC (RHARM.PhD.23.03.20)) and reported in accordance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines.
Drugs and chemicals
LPS from Escherichia coli O111:B4 (Cat. No. L2630) and Ac (Cat. No. A8126) were obtained from Sigma Aldrich chemical Co. (St Louis, MO, USA). LPS was dissolved in normal saline (NaCl 0.9%) in situ. Ac was dissolved in phosphate buffered saline (PBS). The used reagents as well as chemicals in biochemical tests were of the highest analytical grades.
Study design
Albino mice were weighed and assigned into six groups in this manner:
Group (1): Control (C) group: Mice (n = 12) received intraperitoneal (i.p.) injection of normal saline for 2 weeks.
Group (2): Ac 4 group: Mice (n = 12) received once daily i.p. injection of Ac at a dose of 4 mg/kg 23 for 2 weeks.
Group (3): Ac 8 group: Mice (n = 12) received Ac for 2 weeks (8 mg/kg, daily, i.p.). 24
Group (4): Lp group: Mice (n = 16) received single i.p. injection of LPS at a dose of 10 mg/kg 25 at day 14.
Group (5): Ac 4 + Lp group: Mice (n = 12) received Ac for 2 weeks (4 mg/kg, daily, i.p.), then received single i.p. injection of LPS (10 mg/kg) at day 14 after an hour from Ac administration.
Group (6): Ac 8 + Lp group: Mice (n = 12) received Ac for 2 weeks (8 mg/kg, daily, i.p.), then received single i.p. injection of LPS (10 mg/kg) at day 14 after an hour from Ac administration.
Twenty-four hours post LPS injection, the number of dead animals were recorded and the survived mice were weighed, then anesthetized by injection of secobarbital (40 mg/kg, i.p.). Half the number of remaining animals in each group (n = 4-6) underwent bronchoalveolar lavage for collecting bronchoalveolar lavage fluid (BALF). The BALF then was centrifuged for 20 min at 4000 rpm at 4°C using cooling centrifuge (Sigma, Germany). The supernatant was gathered and utilized for total protein (TP) assessment and the cell pellets were used for total leucocyte count (TLC) and differential cell count determination. 26
Blood samples were collected from all alive mice (n = 8-12) through cardiac puncture and serum samples were obtained by leaving the blood to clot on ice for 20 min then centrifugation for 15 min at 3000 rpm for alanine aminotransferase (ALT), aspartate aminotransferase (AST) and testosterone estimation.
Then, the remaining animals were sacrificed by cervical dislocation and liver (n = 8-12), lungs (n = 4-6), and testes (n = 8-12) were isolated, weighed and divided. Sperm samples were collected from cauda epididymis of both testes, which were suspended in 1 ml normal saline (NaCl 0.9 %) at room temperature and were used immediately for sperm analysis. 27 Pieces of liver and left lung were fixed in 10% buffered formalin and left testis was fixed in Davidson’s solution 28 for upcoming histopathological assessment and immunohistochemical analysis. The rest of the liver, right lung, and right testis were conserved for nuclear extract using Abcam Nuclear Extraction Kit (Abcam Inc., Waltham, USA, Cat. No. ab113474) and tissue homogenate preparation at −80°C. Protein content of the homogenates and the nuclear extract were determined based on the method of Bradford 29 by means of Genei protein assessment kit (Bangalore, India, Cat. No. 2624800021730) (Supplemental 1).
Assessment of relative organ/body weight ratio
At the end of experiment, terminal body weight and absolute weight of organs were recorded in all groups. Relative organ/body weight ratio was determined to normalize the variation of actual organ weights because of animals’ body weights fluctuations, by using of the subsequent equation:
Biochemical assessments
Determination of hepatic enzymes
Kinetic determination of serum ALT and AST activity was performed using a commercial kit (ELITech Clinical Systems SAS, Sees, France, Cat. No. ASSL-0250 and Cat. No. ALSL-0250, respectively).
Determination of TLC and differential cell count
Cell pellets of BALF were resuspended in 0.5 ml normal saline for TLC and differential cell count 30 determination by using hemocytometer and autohematology analyzer (Diagon D-cell 60, Hungary).
Assessment of TP
This assay is based on colorimetric determination of protein in the BALF sample using a commercial kit (Vitro Scient, Alsharkia, Egypt, Cat. No. 13302).
Sperm analysis
To calculate sperm concentration, a well-mixed sample was diluted with normal saline (NaCl 0.9 %) as 1 + 19 (1:20), then the hemocytometer was loaded with diluted sample allowing spermatozoa to settle in a humid chamber. Samples were assessed within 10-15 min at ×200 lens (Leica Microsystems, Watzlar, Germany). Sperm count was expressed as concentration of million/ml. Sperm viability was performed using one step eosin-nigrosine procedure and presented as percent, as described previously. 31
Determination of serum testosterone
This assay is based on a competitive test principle using a high affinity monoclonal antibody specifically directed against testosterone using commercial kit (Roche Diagnostics, Rotkreuz, Switzerland, Cat. No. 08946353190).
Estimation of oxidative stress markers
The lipid peroxidation end product, malondialdehyde (MDA), was estimated in tissue samples in the form of thiobarbituric acid reactive substances (TBARS) following the method of Ohkawa et al. (1979). 32 Total nitrate/nitrite (NOx) in tissue homogenates was assessed using colorimetric assay kit provided by BioVision (CA 95035, USA, Cat No. K262-200). Total antioxidant capacity (TAC) was assessed by colorimetric assay kit (Biodiagnostic, Giza, Egypt, Cat. No. TA2513) in homogenate samples, in accordance to the manufacturer’s guidelines. 33
Determination of tissue levels of protein kinase R-like endoplasmic reticulum kinase (PERK), phosphatidylinositol 3-kinase (PI3K), toll-like receptor 4 (TLR4), and interleukin-1 beta (IL-1β)
PERK, PI3K, TLR4, and IL-1β were estimated by sandwich enzyme-linked immune-sorbent assay (ELISA) in hepatic, pulmonary, and testicular tissue homogenates by commercial kits from Antibodies-online (PA 19464, USA, Cat. No. ABIN5594541), Cusabio Technology (TX 77054, USA, Cat. No. CSB E08419 m), Novus Biologicals (Briarwood Avenue, USA, Cat. No. NBP2-76570), and Wuhan Fine Biotech Co. (Hubei, China, Cat. No. EM0109), respectively. These biomarkers were evaluated in duplicate in accordance with the provided instructions by the manufacturers.
Determination of nuclear factor kappa B (NF-κB) in nuclear extract
NF- κB was measured in duplicate in hepatic, pulmonary, and testicular tissue nuclear extracts using commercially available ELISA kit (Wuhan Fine Biotech Co., Hubei, China, Cat. No. EM1230), as per the manufacturer’s instructions.
Determination of gasdermin D (GSDMD)-N-terminal by western blot
Concisely, hepatic, pulmonary, and testicular tissue were homogenized in cocktails of inhibitors and RIPA buffer (Sigma Aldrich chemical Co., MO, USA, Cat. No. PPC1010 and 20188, respectively) then centrifuged (15000 rpm, 5 min). Supernatants were aspirated and proteins separation was performed using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and the gel blotted into polyvinylidene fluoride membrane. Then, membrane was incubated overnight at 4°C with the primary antibody against GSDMD-N-terminal (Abcam Inc., MA 02453, USA, Cat. No. ab215203) followed by the horseradish peroxidase (HRP)-conjugated secondary antibody (Novus Biologicals LLC, Briarwood Avenue, USA, Cat. No. NB7160). Thereafter, the chemiluminescent substrate was added to the blot. The assessment was carried out in triplicate. The GSDMD-N-terminal band intensity was read against beta actin (β-actin) (Sigma Aldrich chemical Co., MO, USA, Cat. No. SAB5600204) using the ChemiDoc MP imaging system (BioRad, Hercules, California, USA).
Histopathological examination
Criteria for histopathologic scoring of liver.

Criteria for histopathologic scoring of lung.

Immunohistochemical determination of active caspase-1
Pyroptosis and inflammation were detected through immunohistochemical (IHC) staining against active caspase-1. 37 Briefly, in formalin-fixed paraffin-embedded tissues, antigen retrieval was performed, after deparaffinization and rehydration, by using water bath as heating method. Rabbit polyclonal anti-active caspase-1 antibody (1:200); (Bioss Inc., Massachusetts 01801, USA, Cat. No. bs-10442R) was performed on sections then incubated at 4°C overnight. The specimens were incubated with biotinylated anti-rabbit/mouse secondary antibody (GBI Labs, WA 98021, USA, Cat. No. D01-6) at room temperature for 10 min. Horseradish peroxidase (HRP) labeled streptavidin was then applied to interact with biotinylated secondary antibody for 10 min to produce a complex. Then 3,3’ diaminobenzidine (DAB) chromogen/substrate was added to completely cover tissue and incubated for 5 min then counterstained with Mayer’s Haematoxylin. The HRP enzyme of the streptavidin complex catalyzed the substrate/chromogen DAB reaction to form a brown color deposit at the antigen site. Slides were examined under light microscope (Olympus CX31, Tokyo, Japan) using ×100 magnification, scale bar = 100 μm. The positively stained areas percentage of the total area of tissue within the field of view was semi-quantitatively measured in 12 fields blindly by means of ImageJ analysis software version 1.53c (National Institutes of Health, USA). The average percentage of three sections were calculated. To eliminate non-specific binding and background interferences, various controls were used.
Statistical analysis
Data were expressed as mean ± standard error of the mean (SEM), where n equals the number of mice in each group. Statistical significance was set at p < 0.05. One-way analysis of variance (ANOVA) followed by post-hoc test, Tukey’s multiple comparison test, was used for statistical comparison of parametric values between groups. Kruskal–Wallis analysis followed by Uncorrected Dunn’s test was performed for evaluation of non-parametric values. GraphPad Prism software (GraphPad Software Inc. version 8, San Diego, California, USA) was utilized to perform statistical analysis and graphing.
Results
Neither Ac 4 nor Ac 8 treatment had significant consequence on organ/body weight ratio, mortality percentage, hepatic, pulmonary, and testicular parameters, in addition to oxidative stress markers when compared with control group; and accordingly, Ac control groups were excluded from the results section for simplicity (Supplemental 2).
Effect of Ac administration on LPS-induced changes on mortality percentage and relative organ/body weight ratio
Percentage of mortality in Lp group was significantly elevated compared to control mice. Pretreatment with Ac (4 mg/kg) or Ac (8 mg/kg) significantly decreased mortality percentage by 1.26 and 1.45-folds, respectively relative to Lp animals. Nevertheless, there was still significant difference between Ac 4 + Lp and Ac 8 + Lp groups in comparison with control group. Pretreatment with the Ac (8 mg/kg) significantly showed reduction in mortality percentage by 24.99% when compared to the low dose-pretreated group (Figure 1(A)). Effect of Ac administration on LPS-induced changes on mortality percentage and relative organ/body weight ratio.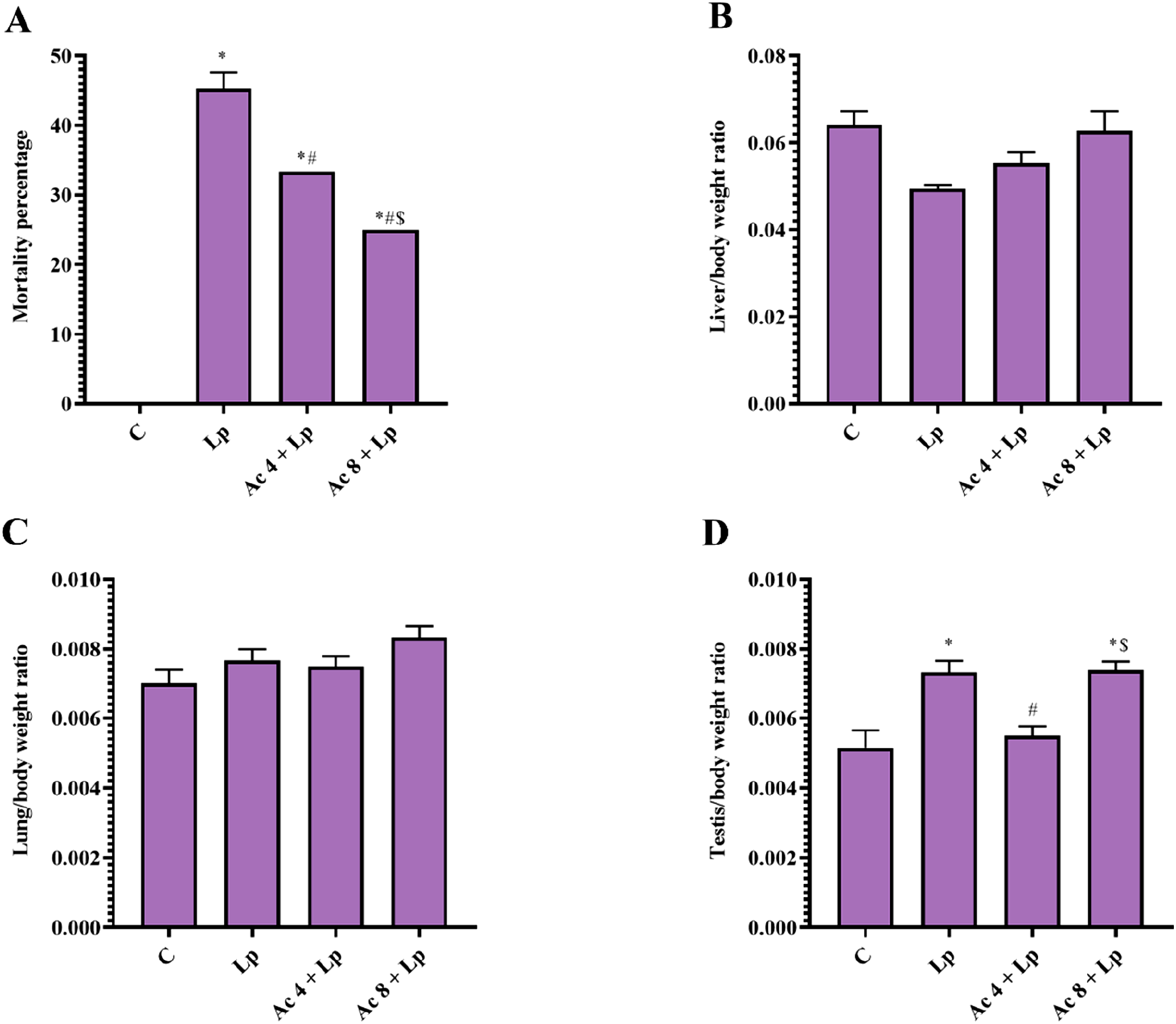
There were no statistical differences between all groups in terms of relative liver/body weight ratio and relative lung/body weight ratio (Figure 1(B), (C)).
However, relative testis/body weight ratio in Lp group was significantly raised by 1.43-folds in comparison to control mice. Ac (4 mg/kg) administration once daily for 2 weeks significantly normalized relative testis/body weight ratio as the results were decreased by 25% as compared to Lp group. In contrast, pretreatment with Ac (8 mg/kg) in Lp mice could not restore normal relative testis/body weight ratio (Figure 1(D)).
Effect of Ac administration on LPS-induced changes on serum liver enzymes
ALT
Lp significantly augmented serum ALT relative to control mice by 3.16-folds. Pretreatment with Ac 4 + Lp or Ac 8 + Lp mice significantly diminished the serum ALT level relative to Lp group by 41.71% and 50.89%, respectively. However, there was still significant difference between Ac 4 + Lp and Ac 8 + Lp groups compared to control group. Injection of the higher dose of Ac significantly showed reduction in serum ALT level when compared to the low dose-pretreated group by 15.75% (Figure 2(A)). Effect of Ac administration on LPS-induced changes on serum liver enzymes.
AST
Lp significantly elevated serum AST level relative to control mice by 2.32-folds. Pretreatment with Ac (4 mg/kg) or Ac (8 mg/kg) significantly decreased the serum AST level relative to Lp group by 33.13% and 43.92%, respectively. There was still significant difference between Ac 4 + Lp and Ac 8 + Lp groups compared to control group. Ac (8 mg/kg) pretreatment significantly declined serum AST level by 16.13% compared to Ac (4 mg/kg)-pretreated group (Figure 2(B)).
Effect of Ac administration on LPS-induced changes on BALF samples
TLC
TLC in BALF was significantly boosted in Lp mice in comparison with control mice by 3.22-folds. Pretreatment with Ac (4 mg/kg) or Ac (8 mg/kg) reverted TLC to control values as indicated by significant decreased TLC relative to Lp group by 54.55% and 56.44%, respectively (Figure 3(A)). Effect of Ac administration on LPS-induced changes on BALF samples.
Neutrophils percentage (N%)
Lp group showed significant increase in N% in BALF relative to control group by 1.37-folds. Injection of Ac (4 mg/kg) or Ac (8 mg/kg) restored normal values as indicated by significant decrease in N% relative to Lp group by 20.51% and 22.12%, respectively (Figure 3(B)).
Lymphocytes percentage (L%)
Lp group showed significant decline in L% in BALF compared to control group by 72.99%. Mice received Ac (4 mg/kg) or Ac (8 mg/kg) pretreatment showed a significant increase in L% by 2.65 and 2.95-folds, respectively relative to Lp mice. Though, there was still a significant difference between Ac 4 + Lp group relative to control group (Figure 3(C)).
TP
TP level in Lp group was significantly raised compared to control mice by 4.73-folds. Mice pretreated with Ac (4 mg/kg) or Ac (8 mg/kg) revealed a significant suppression in TP level relative to Lp group by 56.15% and 66.54%, respectively. There was still a significant difference between Ac 4 + Lp and Ac 8 + Lp groups relative to control group. Pretreatment with Ac (8 mg/kg) significantly declined TP concentration by 23.68% compared to Ac (4 mg/kg)-pretreated group (Figure 3(D)).
Effect of Ac administration on LPS-induced changes on sperm analysis and serum testosterone levels
Sperm count
Sperm count in Lp group was significantly declined compared to control group by 71.14%. Administration of Ac (4 mg/kg) or Ac (8 mg/kg) significantly increased sperm count relative to Lp group by 2.61 and 2.51-folds, respectively. There was still a significant difference between Ac 4 + Lp and Ac 8 + Lp groups relative to control group (Figure 4(A)). Effect of Ac administration on LPS-induced changes on sperm analysis and serum testosterone levels.
Sperm viability
Changes in sperm viability were measured by determining the area under the curves (AUCs) of each group. Sperm viability AUC in Lp mice was significantly decreased relative to control mice by 63.00%. Pretreatment with Ac (4 mg/kg) or Ac (8 mg/kg) improved sperm viability AUC. Statistical analysis showed significant increase relative to Lp group by 2.38 and 2.36-folds, respectively. Still, there was a significant difference between Ac 4 + Lp and Ac 8 + Lp groups relative to control group (Figure 4(B), (C)).
Serum testosterone level
Serum testosterone concentration in Lp mice was significantly declined by 93.50% compared to control group. Ac (4 mg/kg) or Ac (8 mg/kg) pretreatment significantly elevated the serum testosterone concentration by 5.67 and 6.98-folds, respectively compared to Lp mice. However, the effect of Ac on serum testosterone by both doses still significantly differed from control group (Figure 4(D)).
Effect of Ac administration on LPS-induced changes on tissues oxidative stress biochemical parameters
MDA
Lp group showed a significant raise in hepatic, pulmonary, and testicular MDA content by 2.06, 2.31, and 2.34-folds, respectively relative to control mice. Pretreatment with Ac (4 mg/kg) significantly reduced hepatic, pulmonary, and testicular MDA level relative to Lp group by 28.09%, 30.96%, and 15.97%, respectively. In addition, pretreatment with Ac (8 mg/kg) significantly suppressed hepatic, pulmonary, and testicular MDA level relative to Lp group by 45.41%, 48.78%, and 42.22%, respectively. Nevertheless, the effect of Ac on tissue MDA still significantly differed from control group in liver, lung, and testis tissues in low dose, while in high dose in testicular tissue. Pretreatment with high dose significantly reduced tissue MDA level by 24.10%, 25.81%, and 31.23% compared to lower dose-pretreated group in liver, lung, and testis samples, respectively (Figure 5(A)). Effect of Ac administration on LPS-induced changes on tissues oxidative stress biochemical parameters.
NOx
Hepatic, pulmonary, and testicular NOx content was significantly boosted in Lp mice by 3.69, 3.31, and 3.08-folds, respectively relative to control group. Mice received Ac (4 mg/kg) or Ac (8 mg/kg) pretreatment significantly reduced the hepatic, pulmonary, and testicular NOx level relative to Lp group by 27.34%, 30.83%, and 13.14%, respectively. Moreover, pretreatment with Ac (8 mg/kg) significantly lowered hepatic, pulmonary, and testicular NOx level relative to Lp group by 64.57%, 69.01%, and 60.15%, respectively. However, the effect of low dose on tissue NOx still significantly differed from control group in liver, lung, and testis tissue. Pretreatment with high dose significantly decreased tissue NOx level by 51.24%, 55.20%, and 54.13% in liver, lung, and testis samples, respectively compared to lower dose-pretreated group (Figure 5(B)).
TAC
Hepatic, pulmonary, and testicular TAC content in Lp group was significantly suppressed by 77.86%, 83.09%, and 76.60%, respectively relative to control group. Lp mice under Ac (4 mg/kg) pretreatment showed a significant elevation in hepatic, pulmonary, and testicular TAC level relative to Lp group by 2.36, 2.44, and 2.16-folds, respectively. Furthermore, pretreatment with Ac (8 mg/kg) significantly increased hepatic, pulmonary, and testicular TAC level relative to Lp group by 3.80, 4.93, and 3.86-folds, respectively. Yet, the effect of Ac on tissue TAC still significantly differed from control group in liver, lung, and testis tissue in low dose, while in high dose in liver and lung tissue. Pretreatment with high dose significantly elevated tissue TAC content by 1.61, 2.02, and 1.79-folds compared to lower dose-pretreated group in liver, lung, and testis samples, respectively (Figure 5(C)).
Effect of Ac administration on LPS-induced changes on PERK levels
In Lp group, hepatic, pulmonary, and testicular PERK content was significantly elevated in comparison with control mice by 4.72, 3.70, and 4.43-folds, respectively. Pretreatment with Ac (4 mg/kg) restored PERK level, evidenced by significant decline in hepatic, pulmonary, and testicular PERK level by 31.81%, 34.48%, and 31.62%, respectively relative to Lp mice. Additionally, pretreatment with Ac (8 mg/kg) elucidated a significant decrease in hepatic, pulmonary, and testicular PERK level by 74.05%, 69.18%, and 71.23%, respectively relative to Lp group. There was still a significant difference between Ac 4 + Lp group relative to control group in liver, lung, and testis, and between Ac 8 + Lp group relative to control group in lung. Pretreatment with higher dose significantly decreased hepatic, pulmonary, and testicular PERK level by 61.95%, 52.96%, and 57.93%, respectively compared to lower dose-pretreated animals (Figure 6(A)). Effect of Ac administration on LPS-induced changes on protein kinase R-like endoplasmic reticulum kinase (PERK) and phosphatidylinositol 3-kinase (PI3K) levels.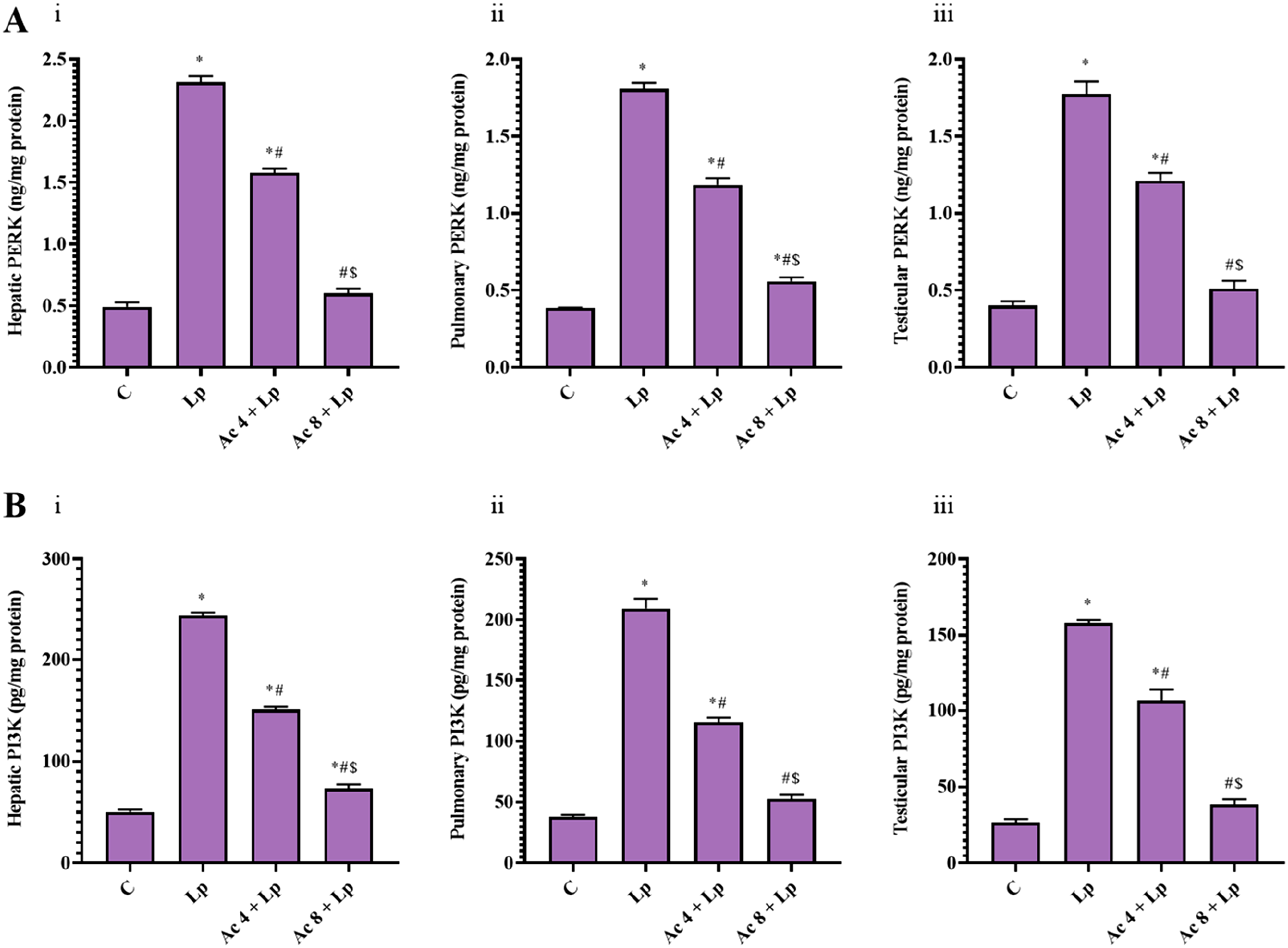
Effect of Ac administration on LPS-induced changes on PI3K levels
Assessment of hepatic, pulmonary, and testicular homogenates demonstrated a significant rise in PI3K level in Lp group by 4.84, 5.53, and 5.96-folds, respectively relative to control mice. Ac (4 mg/kg) pretreatment significantly declined the hepatic, pulmonary, and testicular PI3K level by 38.14%, 44.84%, and 32.62%, respectively relative to Lp group. Additionally, administration of Ac (8 mg/kg) significantly suppressed hepatic, pulmonary, and testicular PI3K level by 70.03%, 74.91%, and 75.74%, respectively relative to Lp group. Of note, there was still a significant difference between Ac 4 + Lp group in liver, lung, and testis, whereas Ac 8 + Lp in liver only relative to control group. Pretreatment with Ac (8 mg/kg) significantly diminished hepatic, pulmonary, and testicular PI3K level compared to Ac (4 mg/kg)-pretreated group by 51.56%, 54.52%, and 64.00%, respectively (Figure 6(B)).
Effect of Ac administration on LPS-induced changes on TLR4 levels
Hepatic, pulmonary, and testicular TLR4 concentration in Lp group was significantly increased by 5.18, 3.84, and 5.53–folds, respectively compared to control mice. Injection of Ac (4 mg/kg) significantly reduced hepatic, pulmonary, and testicular TLR4 level relative to Lp group by 30.69%, 27.69%, and 25.70%, respectively. Furthermore, pretreatment with Ac (8 mg/kg) significantly lowered the hepatic, pulmonary, and testicular TLR4 level relative to Lp group by 68.50%, 60.85%, and 67.68%, respectively. There was still a significant difference between Ac 4 + Lp group in liver, lung, and testis, while Ac 8 + Lp in lung and testis in relative to control group. Pretreatment with Ac (8 mg/kg) significantly reduced hepatic, pulmonary, and testicular TLR4 level by 54.55%, 45.86%, and 56.50%, respectively compared to Ac (4 mg/kg)-pretreated group (Figure 7(A)). Effect of Ac administration on LPS-induced changes on toll-like receptor 4 (TLR4), nuclear content of nuclear factor kappa B (NF-κB), and interleukin-1 beta (IL-1β) levels.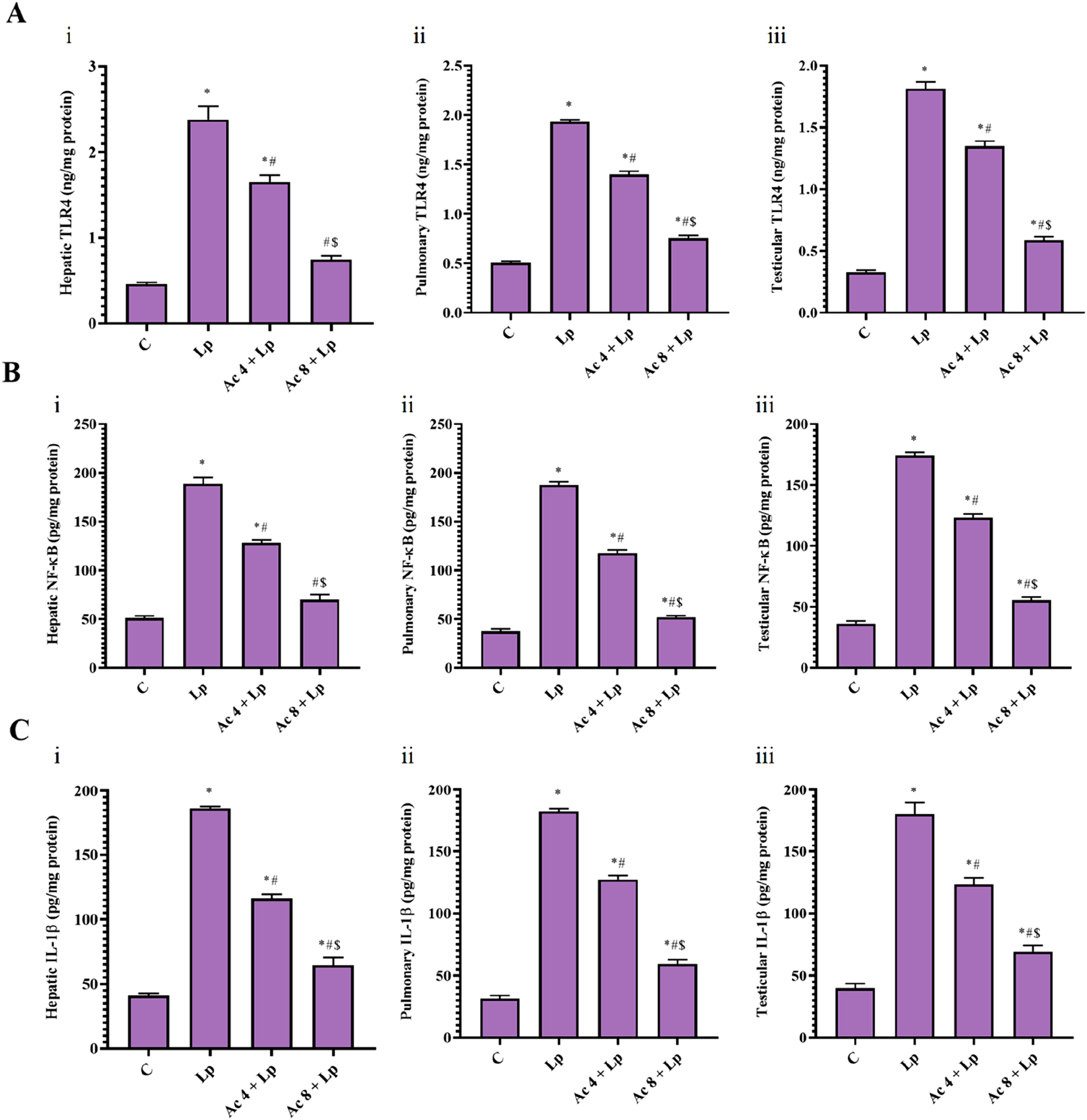
Effect of Ac administration on LPS-induced changes on nuclear NF-κB levels
Lp significantly boosted hepatic, pulmonary, and testicular NF-κB concentration by 3.70, 5.01, and 4.80-folds, respectively relative to control mice. Administration of Ac (4 mg/kg) significantly mitigated the hepatic, pulmonary, and testicular NF-κB by 32.11%, 37.16% and 29.28%, respectively relative to Lp group. As well as, the higher dose administration significantly lessened the hepatic, pulmonary, and testicular NF-κB by 63.05%, 72.42%, and 68.12%, respectively relative to Lp group. Of note, there was still a significant difference between Ac 4 + Lp group relative to control group in liver, lung, and testis, but between Ac 8 + Lp and control groups in lung and testis only. Pretreatment with Ac (8 mg/kg) significantly alleviated hepatic, pulmonary, and testicular NF-κB level by 45.57%, 56.12%, and 54.93%, respectively compared to lower dose-pretreated group (Figure 7(B)).
Effect of Ac administration on LPS-induced changes on IL-1β levels
Lp mice demonstrated a significant rise in hepatic, pulmonary, and testicular IL-1β level by 4.54, 5.81, and 4.52-folds, respectively related to control group. Pretreatment with Ac (4 mg/kg) ameliorated hepatic, pulmonary, and testicular IL-1β levels through a significant reduction in tissue IL-1β levels by 37.68%, 30.26%, and 31.61%, respectively relative to Lp group. Besides, injection of Ac (8 mg/kg) significantly lowered the hepatic, pulmonary, and testicular IL-1β levels by 65.32%, 67.47%, and 61.68%, respectively relative to Lp mice. Yet, there was still a significant difference between Ac 4 + Lp and Ac 8 + Lp groups relative to control group in liver, lung, and testis. Pretreatment with Ac (8 mg/kg) significantly decreased tissue IL-1β content by 44.36%, 53.36%, and 43.97%, respectively compared to lower dose-pretreated group in liver, lung, and testis (Figure 7(C)).
Effect of Ac administration on LPS-induced changes on GSDMD-N-terminal levels
Lp group presented with a significant augmentation in relative hepatic, pulmonary, and testicular expression of GSDMD-N-terminal by 4.68, 2.17, and 2.03-folds, respectively related to control mice. Pretreatment with Ac (4 mg/kg) ameliorated relative expression of hepatic and testicular GSDMD-N-terminal level through a significant decline in the tissue relative expression of GSDMD-N-terminal by 33.82% and 26.47%, respectively relative to Lp animals. Furthermore, administration of Ac (8 mg/kg) significantly reduced the relative hepatic, pulmonary, and testicular expression of GSDMD-N-terminal by 58.11%, 53.70%, and 39.83%, respectively relative to Lp mice. Nevertheless, there was still a significant difference between Ac 4 + Lp mice relative to control animals in liver, lung and, testis, whereas between Ac 8 + Lp group relative to control group in liver. Pretreatment with Ac (8 mg/kg) significantly lessened hepatic and pulmonary relative expression of GSDMD-N-terminal level by 36.71% and 45.62%, respectively compared to lower dose-pretreated group (Figure 8(A)–(D)). Effect of Ac administration on LPS-induced changes on gasdermin D (GSDMD)-N-terminal levels.
Effect of Ac administration on LPS-induced histopathological changes
H&E stained liver sections from control mice exhibited normal histological appearance of hepatocytes (Figure 9(A)–(C)). Lp group showed mild to moderate hepatic necrosis with high hepatic vacuolation and multifocal to coalescing perivascular inflammatory aggregations (Figure 9(D)–(F)). Ac (4 mg/kg)-pretreated group showed mild scattered necrotic hepatocytes adjacent to mild microvesicular steatosis, inset, minimal to mild intacytoplasmic microvesicular vacuoles admixed with few necrotic cells with pyknotic nucleus (Figure 9(G)). Ac (8 mg/kg)-pretreated group showed moderate microvesicular steatosis with few single cell death, inset, many intracytoplasmic microvesicular fat vacuoles (Figure 9(H), (I)). Effect of Ac administration on LPS-induced hepatic histopathological changes.
Lung representative photomicrograph from control group showed normal histological appearance of lung alveoli and bronchiolar architecture (Figure 10(A)–(C)). Lp group showed thickening of interalveolar septa with many inflammatory infiltrates beside focal interstitial hemorrhage (Figure 10(D)). Lp group showed diffuse peribronchiolar mononuclear cellular infiltrates extending into interstitial alveolar septa and admixed with interstitial fibrosis and minimal interstitial congestion leading to severe interalveolar thickening, inset, interstitial aggregation of lymphocytes, macrophages and fibroblast (Figure 10(E), (F)). Ac (4 mg/kg)-pretreated group showed mild, focal peribronchiolar inflammatory cells admixed with congested and hemorrhagic blood vessels (Figure 10(G), (H)). Ac (8 mg/kg)-pretreated group showing moderate to severe interalveolar thickening with many mononuclear cell infiltrates, inset, interstitial aggregation of macrophages, lymphocytes, plasma cells, fibroblasts and RBCs (Figure 10(I)). Effect of Ac administration on LPS-induced pulmonary histopathological changes.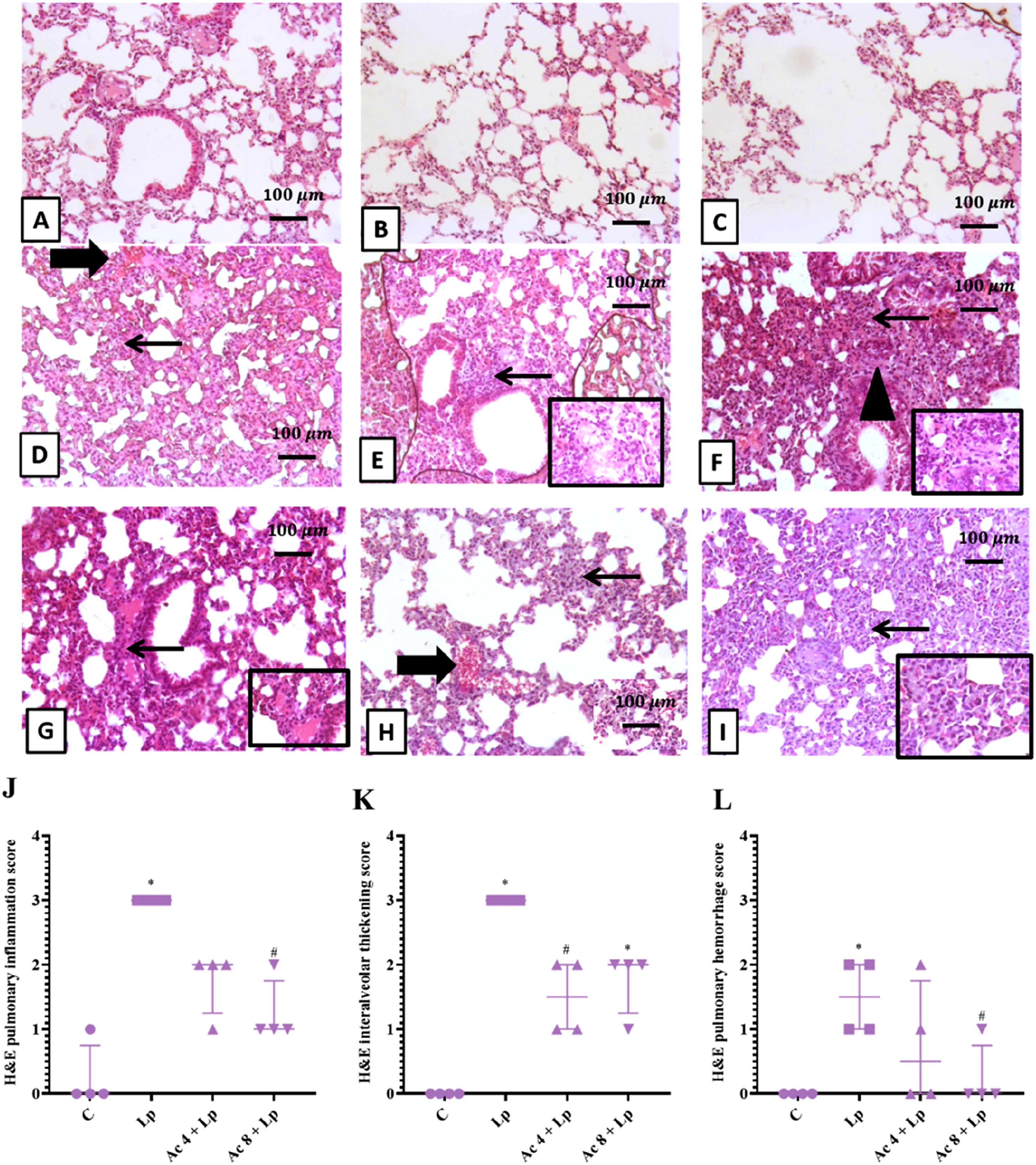
Representative photomicrograph of testes from control group demonstrated a compact and regular arrangement of spermatogenic cells in the seminiferous tubules (Figure 11(A)–(C)). Lp mice showed distorted testicular architecture with interstitial edema admixed with many inflammatory cells widely separated many spermatogenic germ cell layers’ necrosis, some atrophied tubules and other lined with only one cell layer (Figure 11(D), (E)). Lp group showed edema widely separated vacuolated seminiferous tubules that lined with few spermatogenic cells (Figure 11(F)). Ac (4 mg/kg)-pretreated group showed regular appearance of most tubular architecture with few tubules showing necrosis of all spermatogenic cells (Figure 11(G), (H)). Ac (8 mg/kg)-pretreated showed most cells showing complete spermatogenesis except few ones that showed necrosis of spermatogonial stem cells and spermatocytes with loss of many spermatids (Figure 11(I)). Effect of Ac administration on LPS-induced testicular histopathological changes.
A semi-quantitative scoring of the difference in histopathological alterations amongst all groups was represented for hepatic tissues in Figures 9(J)–(L), lung tissues in Figures 10(J)–(L), and for testicular tissues in Figure (11(J).
Effect of Ac administration on LPS-induced changes on active caspase-1 content
Lp mice revealed a significant elevation in positive expression of hepatic, pulmonary, and testicular caspase-1 percentage by 11.65, 4.81, and 5.20-folds, respectively relative to control mice. Injection of Ac (4 mg/kg) ameliorated percentage of caspase-1 positive expression in hepatic, pulmonary, and testicular tissue through a significant reduction in tissue caspase-1 percentage by 77.38%, 64.47%, and 55.38%, respectively relative to Lp group. Additionally, administration of Ac (8 mg/kg) significantly lowered the hepatic, pulmonary, and testicular caspase-1 percentage by 61.95%, 79.22%, and 69.52%, respectively relative to Lp group. Nevertheless, all still demonstrated a significant difference compared to control group except lungs of mice pretreated with high dose. Ac 4 + Lp group showed a significant difference relative to control group in liver, lung, and testis as well as between Ac 8 + Lp group relative to control group in liver and testis. Pretreatment with Ac (8 mg/kg) significantly reduced hepatic, pulmonary, and testicular caspase-1 percentage expression by 68.22%, 41.53%, and 31.70%, respectively compared to Ac (4 mg/kg)-pretreated group (Figures 12–14). Effect of Ac administration on LPS-induced changes on active caspase-1 content. Effect of Ac administration on LPS-induced changes on active caspase-1 content. Effect of Ac administration on LPS-induced changes on active caspase-1 content.


Discussion
Inflammation is a normal physiological response to infection (non-sterile) and injury (sterile). During the first encounter to microbial component, for example LPS, the activated innate immune cells recognize them via PRRs, more precisely, PAMPs. Following this detection, a network of signaling pathways and cellular responses are activated, which if not balanced will result in host tissue damage. 9 LPS causes sepsis, dysregulated immune responses, which is often associated with organ dysfunction or death. 38 It is still accompanied by worldwide high prevalence and mortality. 39 To date, sepsis treatment is restricted to anti-infection and life-supporting techniques. Yet, no specific pharmacotherapeutic option is available. 8 Therefore, in this study, we investigated the therapeutic potential of Ac in an experimental model of acute inflammatory response following exposure to LPS in mice for the aim of discovering new effective agent for management of LPS-induced hepatic, pulmonary, and testicular injuries.
In the current study, male mice were used to reduce the variation and resistance to LPS due to the estrogen effect in females. 40 Injection of LPS (10 mg/kg), intraperitoneally, induced injury to liver, lungs, and testes that were accompanied by elevation in percentage mortality. This observation agreed with prior study by Qiao et al. (2011). 41 Pretreatment with Ac for 2 weeks (4 and 8 mg/kg, daily, i.p.), prior LPS challenge at day 14, significantly alleviated LPS-induced hepatic, pulmonary, and testicular injuries in mice as demonstrated by reduction in the mortality percentage in a dose-dependent manner.
LPS causes damages in multiple organs including liver that leads to hepatic injury as revealed by a promotion in the serum ALT and AST levels as hepatocyte injury circulating markers. The hepatic enzymes are cytoplasmic in nature but are leaked into circulation when hepatocyte damage occurs because of an alteration in membrane permeability. 42 In the present study, induction of liver injury was confirmed by the significant raised ALT and AST activities in serum. Our observation are consistent with previous studies that revealed liver injury in LPS-treated mice indicated by elevation in ALT and AST serum levels in parallel with the deteriorated liver morphological damage and oxidative stress.43,44
In the existing study, Ac administration caused a reduction in the leakage of serum liver enzymes, ALT and AST, in a dose-dependent manner. In the study of Choi et al. (2000), Ac inhibited LPS-mediated activation of NF-kB through suppression of nuclear factor kappa B inhibitor (IκB) degradation and TNF-α production in rat and thus Ac could be accompanied by an anti-inflammatory activity in the liver. 45
LPS is a main trigger of the lung inflammation in the pathogenesis of acute lung injury that mediates a local inflammatory reaction characterized by an acute and massive accumulation of neutrophils.46,47 These stimulated neutrophils migrate into the lung and generate reactive oxygen species (ROS), chemokines, cytokines, and proteolytic enzymes that in turn launch a cascade of events resulting in inflammation escalation that increases protein leakage. 48 The TP level in the BALF is an index of epithelial permeability injury extent and pulmonary edema. 49 In our experiment, the induction of lung injury was revealed by the elevated TLC, the changed differential cell count from normal; increased N% as well as decreased L%, and the increased TP in BALF. These biochemical observations were supported by the histopathological examination. These findings came in concordance with other studies that documented that LPS-treated group exhibited a rise in total cell numbers and TP level in the BALF.50–53
On the other hand, Ac reduced TLC, normalized differential cell count, and decreased TP in BALF in a dose-dependent manner confirming improvement in LPS-induced pulmonary injury. A previous study had a similar observation that documented that administration of Ac prior to lung contusion (LC) could reverse the increased albumin level in the BALF, an indicator of the permeability injury extent, that induced by LC. 54
Systemic microbial infection might damage the epididymal and testicular functions, thus impairing male fertility.55,56 The pathogenesis in the epididymis and testis is likely an indirect impact of microbial infection-induced systemic inflammatory cytokines. This effect could be also attributed to LPS-mediated testicular dysfunction associated with apoptosis in germ cells in seminiferous tubules. 57 In our study, the induction of testicular impairment by LPS was shown by noticeable deficiency of serum testosterone, sperm count, and sperm viability in sperm samples from cauda epididymis of both testes. These results came in line with previous studies that documented a lowering in blood testosterone level and an impairment of spermatogenesis in the group received LPS.57–60 Contrary, Ac elevated serum testosterone, restored sperm count, and sperm viability confirming combatting of LPS-induced injury, which could be due to the anti-inflammatory effect of Ac.
Sepsis progression is linked to the imbalance between the ROS generation and scavenging by the cellular antioxidant system.61–64 Many studies have revealed that excessive ROS and other free radicals are released in sepsis, as H2O2, superoxide anions and NO. These ROS and free radicals are accompanied by inflammation, result in a condition of oxidative stress and contribute to high mortality rates.65,66 Nevertheless, direct detection of ROS and other free radicals is absolutely difficult as they are all highly reactive in a nonspecific manner and short-lived. 67 Therefore, oxidative damage is generally evaluated by secondary products measurement including amino acids, nucleic acids, and lipid peroxidation derivatives. MDA is a product of lipid peroxidation induced by excessive ROS and widely used as a marker of oxidative stress. 68 Also, some antioxidants which are associated with metabolism of free radicals and protection of cells from oxidative stress are usually depleted. In the current study, an elevation in MDA and NOx levels as well as a reduction of TAC in hepatic, pulmonary, and testicular tissue homogenates were found. These results came in concordance with other studies that documented a condition of oxidative stress associated with LPS injection in hepatic,44,69 pulmonary,41,70,71 and testicular tissues. 58
Inversely, Ac amended MDA and NOx levels and increased TAC in hepatic, pulmonary, and testicular tissue homogenates in a dose-dependent manner. These observations are consistent with an earlier study that indicated that the protective impact of Ac on myocardial ischemia/reperfusion injury in rats could be attributed to its antioxidant effects as supported by the decline in MDA and the elevation of glutathione peroxidase levels. 72
Several physiological and pathological circumstances could interfere with protein folding and consequently affect ER homeostasis for instance redox disturbances, xenobiotics, and viruses. 73 Notably, ER stress causes the unfolded protein response (UPR) activation, which lessens the unfolded proteins load and elevates the capacity of protein folding to preserve protein homeostasis. UPR activation includes PERK. 74 Besides impaired protein folding, ER stress might result in a substantial ROS quantity generation. 75 Remarkably, it has been assessed that 25% of ROS generation in a cell caused by the development of disulfide bonds in the ER throughout the normal production of protein. 76 Nevertheless, ROS buildup might induce the resident ER proteins oxidation including the polypeptide folding machinery proteins, subsequently leading to an ER stress as well as producing a vicious cycle. 77 Additionally, PERK stimulates protective autophagy through the PI3K/protein kinase B (Akt)/mammalian or mechanistic target of rapamycin (mTOR) signaling pathway to inhibit apoptosis in addition to inflammation. 78
Likewise, our study showed an enhanced ER stress signal detected in hepatic, pulmonary, and testicular tissue that was confirmed by elevation of PERK and PI3K levels in Lp group. These results are supported by other studies that reported increased PERK and PI3K levels after LPS challenge in the lung tissues. 53 Conversely, Ac is a potent PERK/UPR pathway inhibitor 79 and Akt phosphorylation leading to blockage of the Akt/PI3K pathway. 20 From this standpoint, Ac could reduce LPS-induced inflammation. In the current study, Ac attenuated the ER stress in tissue that was confirmed by reduction of PERK and PI3K levels in hepatic, pulmonary, and testicular tissue homogenates in a dose-dependent manner. Our results regarding Ac are in harmony with previous studies that evidenced that Ac caused inhibition of UPR in different human cancer models.16,79
The inflammatory response of immune cells to LPS is originated by PRRs 9 including TLR4 10 that perceive bacterial LPS. TLR4 binds to myeloid differentiation 2 (MD2) to identify LPS, thereafter LPS-binding protein (LBP) and cluster of differentiation 14 (CD14) reinforce LPS binding to TLR4/MD2 receptor complex. 11 After LPS recognition, phosphorylation of the nuclear factor kappa B inhibitor kinase (IKK) complex is induced, which consists of two catalytic subunits, called IKKα and IKKβ, and a regulatory subunit, called IKKγ as well called nuclear translocation of the transcription factor NF-κB essential modulator (NEMO) that phosphorylates the IκB leading to ubiquitin-directed proteasome-induced degradation of the IκB members therefore, allowing the entrance of the NF-κB p50 and p65 subunits into the nucleus. 80 At the same time, many other similar pathways have an effect on TLR4-induced NF-κB stimulation, involving PI3K. 81 PI3K and its downstream target kinase, Akt, seem to be a critical constituent of LPS-mediated NF-κB activity after its translocation into the nucleus. 82 Free NF-κB enters the nucleus to control transcription and trigger pro-inflammatory cytokines gene expression like IL-1 and triggering the production of NLRP3.12,13,83
Multiple exogenous as well as endogenous stimuli, such as production of ROS and calcium ion imbalance during ER stress, could stimulate the NLRP3 inflammasome.84–86 The active NLRP3 inflammasome hydrolyzes inactive pro-caspase-1 to active caspase-1. Thereafter, activated caspase-1 thereafter triggers inflammasome-specific cytokines production and secretion including IL-1β and IL-18 to enhance proinflammatory responses. 83 Whereas concurrently, activated caspase-1 also mediates cleavage of GSDMD-N-terminal cell death domain. 87 GSDMD-N-terminal forms pores in the plasma membrane leading to pyroptosis, an inflammatory cell death. 37
In parallel, our results revealed a boosted inflammatory and pyroptosis signals in Lp mice, as noticed in hepatic, pulmonary, and testicular tissue that was recognized by elevation of TLR4 levels, nuclear content of NF-κB, GSDMD-N-terminal relative expression, and IL-1β levels. These findings supported by previous studies that reported boosted TLR4 level,53,88 nuclear NF-κB level,53,69,88,89 GSDMD-N-terminal,90,91 and IL-1β level41,53,92 after LPS challenge.
Contrariwise, Ac diminished the inflammation and pyroptosis process in hepatic, pulmonary, and testicular tissue that was emphasized by lowering TLR4, nuclear content of NF-κB, GSDMD-N-terminal relative expression, and IL-1β levels in a dose-dependent manner. Our results are supported by another study that documented the decreased level of IL-1β in Ac-treated animals subjected to LC when compared with corresponding control group that suggests its inhibitory role in the pro-inflammatory cytokines production. 54 Furthermore, another study indicated that Ac effectively inhibited NF-κB stimulation in the rats’ liver that would result in suppressing the transcriptional activation of some inflammatory cytokines as IL-1β, which could be account for its anti-inflammatory impact. 45
IHC staining of hepatic, pulmonary, and testicular tissue sections of Lp group against active caspase-1; an inflammatory and pyroptotic caspase, showed increased percentage of expression of active caspase-1. Our observation came in concordance with previous studies in which LPS produced stimulation of NLRP3, cleaved caspase-1, and downstream IL-1β.90–92
On the contrary, Ac-pretreated groups showed a marked reduction in the expression percentage of hepatic, pulmonary, and testicular active caspase-1 expression. As far our knowledge, no earlier studies have investigated the impact of Ac on caspase-1 in lung, liver, or testis injury induced by LPS.
These effects together are reflected on histopathological changes in Lp mice that revealed hepatic necrosis with high hepatic vacuolation and multifocal to coalescing perivascular inflammatory aggregations. These pathological observations are consistent with other studies that demonstrated hepatocyte swelling and necrosis upon LPS administration.43,44,93
Moreover, Lp group demonstrated pulmonary thickening of interalveolar septa with many inflammatory infiltrates beside focal interstitial hemorrhage. Additionally, diffuse peribronchiolar mononuclear cellular infiltrates extending into interstitial alveolar septa and admixed with interstitial fibrosis and minimal interstitial congestion were observed leading to severe interalveolar thickening, interstitial aggregation of lymphocytes, macrophages and fibroblast. These observations came in concordance with previous data by Mei et al. (2007), 50 Reis Gonçalves et al. (2012), 52 Abdallah et al. (2020), 71 Huang et al. (2020), 53 and Ye and Liu (2020) 94 that showed pulmonary vessels infiltrates and alveolar wall edema in the LPS-treated group.
Additionally, Lp group showed distorted testicular architecture with interstitial edema admixed with many inflammatory cells widely separated many spermatogenic germ cell layers’ necrosis, some atrophied tubules and other lined with only one cell layer. In addition, Lp group sections showed testicular edema and widely separated vacuolated seminiferous tubules that are lined with few spermatogenic cells. These observations are in agreement with other studies that indicated immature male gametes in lumens of seminiferous tubules and epididymis after LPS treatment.58–60
Oppositely, histopathological features of hepatic, pulmonary, and testicular tissue sections were amended following Ac treatment. As far as our knowledge, there are no preceding studies addressed the impact of Ac on those issues upon LPS administration.
Conclusion
The existing study results plainly indicated the dose-dependent protective impact of Ac on hepatic, pulmonary, and testicular function in a model of LPS-mediated multiple organ injury through maintaining antioxidant capacity and reducing ROS, mitigating ER stress, and attenuating inflammatory and pyroptotic signaling that cooperatively protect the histological integrity of the investigated-organs (Figure 15). The overall outcomes of our study support the protective impact of Ac against LPS-induced systemic inflammatory response and boost the possibility of its application clinically on endotoxemia or sepsis. Schematic presentation of the underlying mechanism by which acriflavine exerts its protective effect against LPS-induced multiple organ damage.
Yet, the study has some limitations including the lack of a comparison between the effects of Ac with a standard anti-inflammatory drug like glucocorticoids that is commonly used in the management of sepsis and the absence of NLRP3 expression measurement besides using pathway-specific inhibitors. Thus, this study future directions would be to investigate the impact of NLRP3 or GSDMD inhibitors or gene knockdown on LPS-induced organ injury. Even with the approval of Ac by FDA suggesting its reasonable clinical safety, the use of a mouse model makes it difficult to quantitatively extrapolate to a clinical scenario in humans, despite the absence of bias in the experiment performed.
Supplemental Material
Supplemental Material - Acriflavine protects against LPS-induced sepsis via regulation of pyroptosis, inflammation, and endoplasmic reticulum stress
Supplemental Material for Acriflavine protects against LPS-induced sepsis via regulation of pyroptosis, inflammation, and endoplasmic reticulum stress by Esraa G. El-Waseif, Sara H. Hazem, Dalia H. El-Kashef, and Ghada M. Suddek in Human & Experimental Toxicology
Footnotes
Acknowledgments
The authors are grateful to Dr Iman Ibrahim, Pathology Department, Faculty of Veterinary Medicine, Mansoura University, Egypt, for her plentiful aid with histopathological assessment of tissue sections.
Ethical consideration
The research procedure has been authorized by Mansoura University Animal Care and Use Committee (MU-ACUC (RHARM.PhD.23.03.20)). Animal care and use in the current study complies with ARRIVE guidelines.
Author contributions
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

