Abstract
Background
Dexmedetomidine (DEX) can offer protection to the nervous, urinary and circulatory systems. It can alleviate local oxidative stress, reduce inflammatory responses, inhibite cellular autophagy and decrease apoptosis.
Aim
To explore the potential protection and possible mechanisms of DEX against propofol (PPF)-induced memory impairment in developing rats.
Material and methods
The effects of DEX on spatial learning and passive avoidance abilities of rats were evaluated using eight-arm mirror maze and passive avoidance experiments. mRNA levels were detected using RT-qPCR analysis while protein levels were determined using western blot. A network pharmacology approach was used to predict potential targets of DEX against PPF-induced memory impairment. The cell autophagy and apoptosis were detected using commercial kits.
Results
DEX improved the impairment of developing rats on spatial learning and passive avoidance caused by PPF exposure. DEX regulates autophagic activity to inhibit neuronal apoptosis. RARα and Src were potential targets for DEX against memory impairment caused by PPF exposure. DEX upregulated the expression levels of Bdnf, p-CREB/CREB, p-Akt/Akt, and p-TrkB/TrkB proteins.
Conclusion
DEX may regulate Bdnf/TrkB and activate the activity of the PI3K/Akt signaling pathway by targeting RARα and Src, thereby inhibiting excessive autophagy and alleviating memory impairment.
Introduction
General anesthetics (GA) are critical medications for surgical procedures, which facilitate successful surgery by inducing analgesia and reducing pain and anxiety. 1 Over 300 million complex and painful surgical procedures are being performed unconscious and insensitivities to pain under various anesthetic interventions, alone or in combination. 2 Millions of infants and children are exposed to anesthetic drugs every year for diagnostic and surgical purposes. 3 However, general anesthesia drugs, such as propofol (PPF), have been reported to cause apoptosis and inflammatory responses in nerve cells. 4 In addition, general anesthetics can induce neuroapoptotic degeneration of developing neurons. 2 Propofol is a widely used general anesthetic for infants in clinical anesthesia. Previous studies have found that propofol can cause damage to the immature nervous system, which may lead to neurodevelopmental disorders and cognitive dysfunction in infants and children. 5 It can induce neuronal death in the developing brain, leading to cognitive impairment, such as memory, learning, reasoning, and planning abilities. 6 In pediatric anesthesia, the solutions to the neurotoxicity of propofol have received a great deal of attention.
Dexmedetomidine (DEX), an α-2adrenoceptor agonist, is widely used in clinical treatment for its sympatholytic, sedative, amnestic, and analgesic properties. 7 Notably, DEX has potential properties in neuroprotection, cardioprotection, and renoprotection. 7 Studies have shown that DEX can inhibit the apoptotic factors (Bax and Cleaved Caspase-3) and promote the anti-apoptotic factors (Bcl-2), thus suppressing neuronal apoptosis. 8 DEX can recover the desflurane-induced impairment in motor function and memory. 9 DEX can reduce PPF-induced hippocampal neuron injury in vitro. 10 DEX has been reported to alleviate PPF-induced neural injury in developing rats by activating the Akt/CREB/BDNF signaling pathway. However, the specific targets of DEX in neuroprotection remain to be further elucidated.
This study aimed to explore the potential mechanism of action of DEX in alleviating PPF-induced memory impairment in rats by constructing animal and cellular models. The common DEX targets and genes related to PPF-induced neurological injury were predicted using network pharmacology. We speculated that DEX targets RARα and Src to regulate Bdnf/TrkB/P13K/Akt signaling pathway, and inhibits excessive cellular autophagy, thereby reducing neuronal apoptosis.
Materials and methods
Materials
DEX and PPF were obtained from MedChemExpress (MCE, USA). Bovine serum albumin was obtained from ThermoFisher (USA). Other materials were of the highest grade available.
Subjects
All animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of The First People’s Hospital of Lanzhou City (Approval No: 2023016) and conducted following the ARRIVE guidelines and the NIH Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize animal suffering, and euthanasia was performed following AVMA guidelines. 7-day-old Sprague Dawley (SD) SPF-grade male rats (14–17 g), which were purchased from SiPeiFu (Beijing) Biotechnology Co. (Animal License No.: SCXK (Beijing) 2019-0010). The rats were housed in an SPF-grade animal laboratory center. The rats were divided into sham group, PPF group, DEX + PPF group, and DEX + PPF + AgoRARα/antiSrc group. The PPF group was given an intraperitoneal injection of 100 mg/kg propofol. The DEX + PPF group was given an intraperitoneal injection of 25 μg/kg dexmedetomidine for five consecutive days followed by an intraperitoneal injection of 100 mg/kg propofol. After the intervention, animals were euthanized using intraperitoneal administration of 100 mg/kg propofol following humane euthanasia protocols recommended by the AVMA. Efforts were made to minimize suffering, including prior anesthesia where necessary. Hippocampal tissue was removed immediately from half of the rats in each group, and the other half of the rats were routinely fed for 7 days after awakening before removal of the hippocampal tissue.
In vitro experiment: Human neurons-hippocampal (HN-h) primary cells (ScienCell, USA) were divided into a control group, PPF group, DEX + PPF group, and DEX + PPF + AgoRARα/antiSrc group. The DEX concentration was 10 μM. Rat hippocampal neurons were isolated from the obtained hippocampal tissues.
Behavior tests
Eight-Arm Radial Maze (ARM)
The experimental animals were tested in a standard 8-ARM (Med Associates Inc., St. Albans, VT) to assess memory function. Before performing the 8-ARM test, the experimental animals received seven sessions of habituation training. During habituation training, reinforcers were randomly distributed on the central platform and arms, or only at the end of each arm. The rats were first allowed to familiarise themselves with the maze on the central platform for 2 min, and then all the doors were automatically opened to allow the rats to explore the arms freely for 10 min. After habituation training, the experimental rats were allowed to perform a standard radial arm task five times. After completing each test task, (a) the total errors, (b) the total time, and (c) the number of correct choices before the first error were recorded.
Passive avoidance test
Using a passive avoidance task to assess aversive memory. The experimental animals were placed in an apparatus with light and dark areas. During training, rats were placed in a lighted area and allowed to explore freely. When rats moved into a dark area, their feet suffered from a slight shock. Latency to darkness was recorded during the training phase. Latency to darkness and shock duration were recorded during the retention phase.
RT-qPCR
Total RNA from hippocampal tissues of experimental rats was extracted by using Trizol reagent (ThermoFisher, Beijing, China), and cDNA was synthesized by reverse transcription. Primer sequences were synthesized by Beijing Qingke Biological Co. PCR amplification was performed using GAPDH as an endogenous reference gene. The relative expression levels of target mRNA was calculated using 2−ΔΔCt.
Western blot
After the behavioral test, rats were sacrificed. The whole brain was carefully removed from the skull and stored at −80°C until used. Hippocampal tissues were lysed with cell lysis solution on ice for 20 min and centrifuged at 8000g for 8 min at 4°C. Proteins from cell lysates were separated by SDS-PAGE followed by western blot analysis. The gels were stained with a vinylidene difluoride membrane and then exposed to the corresponding antibodies (LC3-II, LC3-I, Beclin-1, p62, RARα, Src, Bdnf, p-CREB, CREB, pAkt, Akt, p-TrkB, Trk). After coupling of the corresponding antibodies with horseradish peroxidase, the protein bands were visualized by ECL (MCE, Shanghai, China). Pictures were taken with a chemical imager and then analyzed on a grey scale using Image J software.
Apoptosis analysis
Cell apoptosis were assayed by TUNEL kit (BIORN, Nanjing, China), Bax kit (MCE, Shanghai, China), and Bcl-2 kit (MCE, Shanghai, China) according to the manufacturer’s protocol. The cells were then analyzed by flow cytometry.
Autophagy analysis
A CYTO-ID® Autophagy Detection Kit (Megazyme, Shanghai, China) was used to determine the autophagy level of the cells. Approximately 1 × 105 cells were stained with CYTO-ID green detection dye at 37°C for 30 min. After staining, the cells were assayed by flow cytometry analysis on BD FACSymphony A1 Cell Analyzer (Becton Dickinson, United States).
Statistical analysis
All experiments were conducted with at least three biological replicates (n = 3), with results reported as mean ± SD. Statistical significance was determined using one-way ANOVA followed by an LSD test, with exact p-values reported where applicable. All raw data, including full-length Western blot images, are available upon request.
Results
Effect of dexmedetomidine on spatial learning and passive avoidance response
To demonstrate that DEX can alleviate the damage of memory function caused by PPF in experimental rats, we assessed the spatial learning in rats using the Eight-Arm Radial Maze. The results show that the total error in the PPF group was dramatically higher than in the sham group, while the total error in the DEX + PPF group was significantly lower than in the PPF group (Figure 1(A)). The results of the experiment in groups of rats visiting all 8 arms showed that the time was higher in the PPF group compared to the sham group, while the time was lower in the DEX + PPF group compared to the PPF group (Figure 1(B)). In addition, the correct choice before first error was significantly less in the PPF group compared to the sham group, while the correct choice before first error was more in the DEX + PPF group compared to the PPF group (Figure 1(C)). The above experimental results suggested that DEX can affect the spatial learning ability of rats. To further demonstrate the protective effect of DEX on memory function, we evaluated the aversive memory of experimental rats using a passive avoidance task. The results showed that there was no difference in latency to the dark between the groups of experimental rats in the training task (Figure 1(D)). However, in the retention task, the latency to the dark was shorter in the PPF group compared to the sham group, and the latency to the dark was prolonged in the DEX + PPF group compared to the PPF group (Figure 1(E)). In addition, we assayed the shock duration in the retention task. The results showed that the shock duration was longer in the PPF group compared to the sham group, while the shock duration was shorter in the DEX + PPF group compared to the PPF group (Figure 1(F)). These results indicated that DEX can alleviate PPF-induced memory impairment in rats. Effect of dexmedetomidine on spatial learning and passive avoidance response (A) Total errors to visit all 8 arms, (B) Time taken to visit all 8 arms, (C) The number of correct responses made before the first error, (D)The latency to the dark in training task of the passive avoidance test, (E) The latency to the dark in retention task of the passive avoidance test, (F) The shock duration in retention task of the passive avoidance test. n = 4.
Effect of dexmedetomidine on autophagy
Previous studies have shown that almost all general anesthetics in children cause apoptosis of neuronal cells.11,12 In addition, some researchers have found that PPF can lead to massive apoptosis in neurons and astrocytes, which contributes to the development of postoperative cognitive dysfunction.
13
Therefore, we detected apoptosis in the hippocampal cells of 7-day-old rats exposed to PPF using TUNEL assay. The results showed that PPF caused a dramatic increase in the rate of apoptosis compared to the sham group, while treatment with DEX reversed to some extent the damage caused by PPF to the neuronal cells (Figure 2(A)). To further support our conclusions, we assessed the mRNA levels of Bax and Bcl-2. The results showed that PPF exposure upregulated Bax mRNA expression compared to the sham group; interestingly, co-administration of DEX reversed the effects of PPF (Figure 2(B)). In contrast, PPF exposure downregulated the expression of the Bcl-2 mRNA; however, co-administration of DEX reversed the effects of PPF (Figure 2(B)). Previous studies have shown that autophagic activity plays a crucial role in maintaining the homeostasis of neuronal cells. Therefore, we assessed the level of autophagy in hippocampal tissue cells from each group of rats. The results showed that the relative expression of LC3II/LC3I was dramatically elevated in the PPF group compared to the sham group, which was reversed by co-administration of DEX (Figure 2(C), Supplemental Figure 1). To further demonstrate our findings, we assayed the expression levels of the autophagy markers, including Beclin-1 and p62, by western blot. The results showed that PPF elevated the Beclin-1 protein level but reduced the p62 protein level (Figure 2(D)). Interestingly, co-administration of DEX reversed the effects of PPF (Figure 2(D), Supplemental Figure 1). These results indicated that DEX could inhibit PPF-induced excessive autophagy of neuronal cells. Effect of dexmedetomidine on apoptosis and autophagy (A) Apoptotic cells were detected by the TUNEL kit, (B) mRNA expression levels of Bax and Bcl-2 were detected by RT-qPCR, (C) Relative expression of LC3-II/LC3-I was detected by western blot, (D) Relative expression of Beclin-1 and p62 were detected by western blot. n = 4.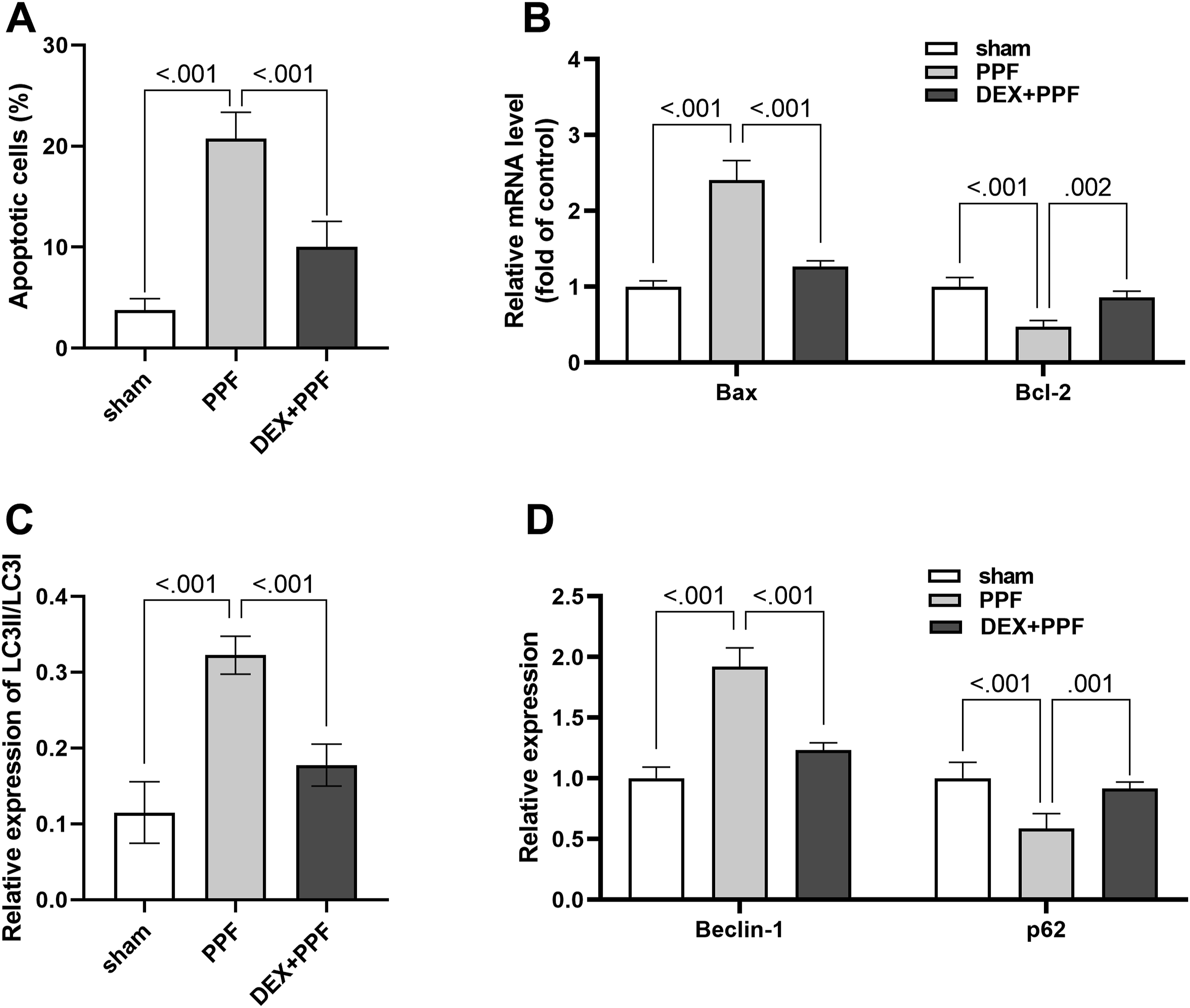
The potential targets of dexmedetomidine on memory impairment
Using the GSE101724 database, 3288 relevant targets of memory impairment were collected; while 158 potential targets of DEX were collected from the PharmMapper database. The intersection yielded 14 common action targets (Figure 3(A)). In addition, we performed a PPI network analysis of these 14 targets using the STRING database (Figure 3(B)). Among them, SRC and RARA are the most critical genes (Figure 3(B)). Therefore, we detected the expression levels of RARα and SRC proteins in the hippocampal tissues from each group of experimental rats by western blot. We found that the RARα expression was elevated in the PPF group compared to the sham group; however, co-administration of DEX reversed the effects of PPF (Figure 3(C)). The SRC expression level showed an opposite trend to that of RARα (Figure 3(D)). Excitingly, the results of the cell experiments were consistent with the results of the animal experiments (Figure 3(E) and (F)). Therefore, we hypothesized that RARα and SRC are critical targets for DEX on memory impairment. The potential targets of dexmedetomidine on memory impairment (A) Venn diagram of predicted DEX targets and memory impairment genes from GSE101724 databases. (B) Interaction network of proteins encoded by the 11 target genes for DEX against memory impairment, with each node representing a protein and the edges representing protein-protein interactions. RARα (C) and Src (D) protein expression levels of rat hippocampus were detected by western blot. RARα (E) and Src (F) protein expression levels of HN-h were detected by western blot. n = 4.
Effect of dexmedetomidine on the expression of BDNF and p-CREB in the hippocampus
The expression levels of Bdnf, p-CREB/CREB, p-Akt/Akt, and p-TrkB/TrkB were detected using western blot. The results of animal experiments showed that PPF inhibited the levels of Bdnf, p-CREB/CREB, p-Akt/Akt, and p-TrkB/TrkB proteins, which was offset by co-administration of DEX (Figure 4(A)–(D), supplemental Figure 2). The results of the cell experiments showed concordance with the results of the animal experiments (Figure 4(E)–(H), supplemental Figure 2). Effect of dexmedetomidine on expression of BDNF and p-CREB in the hippocampus Bdnf (A), p-CREB/CREB (B), p-Akt/Akt (C), and p-TrkB/TrkB (D) protein expression levels of rat hippocampus were detected by western blot, Bdnf (E), p-CREB/CREB (F), p-Akt/Akt (G), and p-TrkB/TrkB (H) protein expression levels of HN-h were detected by western blot. n = 4.
Rescue experiment
To ensure the reliability of the experiments, we performed validation experiments. The results showed that PPF significantly upregulated the expression of RARα compared with the control group (Figure 5(A)). Notably, exposure to AgoRARα/antiSrc reversed the effects of DEX compared to the DEX + PPF group (Figure 5(A)). PPF significantly downregulated the expression of Src compared to the control group (Figure 5(B)). Whereas, exposure to AgoRARα/antiSrc reversed the effects of DEX compared to the DEX+PPF group (Figure 5(B)). To further demonstrate that DEX can inhibit neuronal apoptosis by modulating the level of autophagy. We validated our experiments on apoptosis and autophagy. The results showed that PPF exposure significantly increased the apoptosis rate compared to the control group (Figure 5(C)). However, exposure to AgoRARα/antiSrc reversed the inhibitory effect of DEX on apoptosis compared to the DEX+PPF group (Figure 5(C)). The assay of autophagic activity showed that autophagic flux (MFI) was enhanced in the PPF group compared to the control group (Figure 5(D)). Notably, AgoRARα/antiSrc exposure reversed the inhibitory effect of DEX on autophagic activity compared to the DEX+PPF group (Figure 5(D)). Rescue experiment RARα (A) and Src (B) protein expression levels of HN-h were detected by western blot with/without AgoRARα/antiSrc, (C) Apoptotic cells were detected by the TUNEL kit with/without AgoRARα/antiSrc, (D) Autophagic fux (MFI) was detected by the CYTO-ID® Autophagy detection kit with/without AgoRARα/antiSrc, (E) Possible mechanism of DEX against memory impairment induced by PPF. n = 4.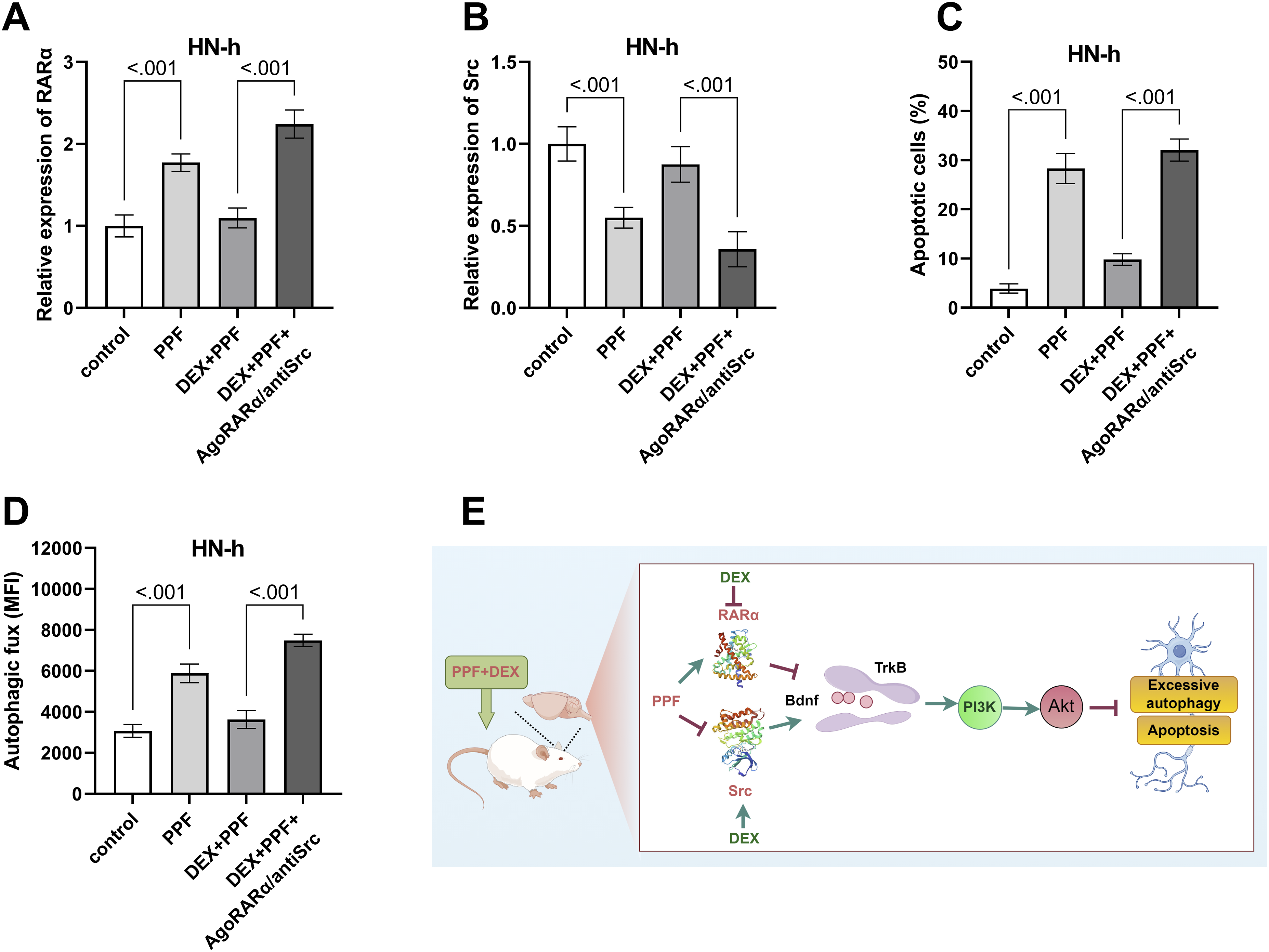
Discussion
Mammals are born with an underdeveloped nervous system, and the immature brain must undergo a period of rapid development called “synaptogenesis”. 14 Previous studies have shown that the use of the intravenous anesthetic PPF during the “synaptogenesis” period causes memory impairment, learning and behavioral deficits in the developing brain.15,16 This study explored the effects and mechanisms of DEX on memory impairment in the developing brain. Our results suggest that DEX can improve spatial learning and passive avoidance abilities of rats. From the perspective of cellular mechanisms, DEX can inhibit apoptosis and modulate autophagic activity. Using the network pharmacological method, the potential targets of DEX on memory protection were analyzed. The potential mechanism predicted for DEX to alleviate memory impairment was to target RARα and Src, thereby affecting activity of PI3K/Akt signaling pathway (Figure 5(E)).
Apoptosis and autophagy are intracellular processes that can regulate cell survival and death under stress conditions. 17 Recently, there have been identified interactions between autophagy and apoptosis. However, the potential mechanism is unclear. Our team has been working on the relationship between PPF-induced apoptosis of neuronal cells and cellular autophagy. We assayed the expression levels of the pro-apoptotic gene Bax and the anti-apoptotic gene Bcl-2 by RT-qPCR. The results showed that DEX could effectively reverse the changes in Bax and Bcl-2 genes caused by PPF exposure. Meanwhile, we used a western blot to determine the protein expression levels of Beclin-1 and p62. The results showed that DEX could effectively reverse the changes in Beclin-1, and p62 protein expression levels caused by PPF exposure. Previous studies showed that the proportional relationship between Bax/Bcl-2 proteins is a critical factor in deciding apoptosis. 18 In addition, it was found that Beclin-1 is a key regulatory protein in the process of cellular autophagy, which is involved in regulating the formation of autophagic vesicles and is a critical factor in the initiation of autophagy. 19 The upregulation of the p62 protein inhibits autophagic activity. 20 Therefore, we concluded that DEX may regulate excessive cellular autophagy to inhibit apoptosis.
It is well known that astrocytes secrete large amounts of brain neurotrophic factor (Bdnf). Studies have shown that Bdnf combined with TrkB can regulate the complexity of nerve cells and prevents neuropathy.21,22 It has also been shown that upregulation of retinoic acid receptor α (RARα) expression suppresses Bdnf expression. 23 Upregulation of protein kinase Src can increase Bdnf expression. 24 Through network pharmacological analysis we learned that the key targets of DEX against the memory impairment caused by PPF exposure were RARα and Src. Therefore, we hypothesized that DEX targets RARα and Src to regulate Bdnf. It has been found that upregulation of Bdnf expression suppresses autophagy and enhances memory. 25 Additionally, it has been proven that Bdnf inhibits autophagic flux and autophagosomes of hippocampal tissues via TrkB and PI3K/Akt pathways. 26 The molecular regulatory mechanisms of autophagy are complex and involve several different signaling pathways. 27 The PI3K/AKT/mTOR pathway is a classical signaling pathway that promotes cell survival, resists apoptosis, and inhibits cell autophagy.28,29 Therefore, we used a western blot to assess the expression of the major proteins involved in this pathway. Our results showed that PPF exposure down-regulated the expression of Bdnf, p-CREB/CREB, p-Akt/Akt, and p-TrkB/TrkB. Interestingly, co-administration of DEX effectively alleviated the suppressive effects of PPF. Thus, DEX may regulate Bdnf to activate the PI3K/Akt signaling pathway, and thus inhibit cellular autophagic activity.
Some limitations of this study must be considered when interpreting the data presented in our study. Our study of the mechanisms by which DEX protects against PPF-induced neurological damage, such as changes in apoptosis and autophagy, are not exhaustive. The complex perturbation between autophagy and apoptosis may be critical in directing neuronal cell fate, and further studies are needed to elucidate the additional mechanisms.
Conclusion
In summary, DEX can produce memory-protective effects in PPF-exposed rats. DEX can target RARα and Src to upregulate Bdnf/TrkB and activate the PI3K/Akt signaling pathway, thus inhibiting excessive autophagy and reducing apoptosis in neuronal cells. However, the mechanism of action of DEX in treating memory impairment remains to be further elucidated.
Supplemental Material
Supplemental Material - Protective effects of dexmedetomidine against propofol-induced memory impairment in developing rat involved Src and RARα: Dexmedetomidine alleviates memory impairment
Supplemental Material for Protective effects of dexmedetomidine against propofol-induced memory impairment in developing rat involved Src and RARα: Dexmedetomidine alleviates memory impairment by Xiaoyan Xu, Jianmei Yang, Qiang Jia, Qianqian Hu and Huiling Liu in Human & Experimental Toxicology
Footnotes
Statements and declarations
Author contributions
X.Y. Xu contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; J.M. Yang contributed to interpretation and critically revised manuscript; Q. Jia, Q.Q. Hu and H.L.Liu contributed to acquisition, analysis, and interpretation and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Science and Technology Plan Project of Lanzhou Science and Technology Bureau, Gansu Province [Project Number: 2023-4-11]. And Supported by Gansu Provincial Science and Technology Plan, Gansu Provincial Natural Science Foundation [Project number: 23JRRA1455, 24JRRA766].
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article and its supplemental material files. Further enquiries can be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
