Abstract
Methamphetamine (METH) is an illicit dopaminergic neurotoxin and is an extremely addictive psychostimulant drug that influences monoamine neurotransmitter system of the brain and is responsible for enhancing energy and satisfaction and feelings of alertness. Long-lasting exposure to METH causes psychosis and increases the risk of Parkinson’s disease. Studies have revealed that obestatin (OB) is a novel endogenous ligand, which may have neuroprotective effects. Hence, we hypothesized that OB might appropriately limit METH-induced neurotoxicity via the control of apoptotis and autophagy. In the current study, PC12 cells were exposed to both METH (0.5, 1, 2, 3, 4, and 6 mmol/L) and pretreatment OB (1, 10, 100, and 200 nmol/L) in vitro for 24 h to determine appropriate dose, and then downstream pathways were measured to investigate apoptosis and autophagy. The results have shown that OB reduced the apoptotic response post-METH exposure in PC12 cells by developing cell viability and diminishing apoptotic rates. Furthermore, the study has exhibited OB decreased gene expression of Beclin-1 by real-time polymerase chain reaction and LC3-II by Western blotting in METH-induced PC12 cells, which demonstrated that autophagy is reduced. The study is proposed that OB is useful in reducing oxidative stress, which may also play an essential role in the regulation of METH-triggered apoptotic response. So these data indicate that OB could potentially alleviate METH-induced neurotoxicity via the reduction of apoptotic and autophagy responses.
Introduction
Methamphetamine (METH) is an illicit cationic lipophilic dopaminergic neurotoxin and is an extremely addictive psychostimulant drug that influences monoamine neurotransmitter system of the brain and is responsible for enhancing energy and satisfaction and feelings of alertness. 1 –3 Recent studies have shown that METH has a chemical structure similar to amphetamine, and long-term METH abuse directs to neurodegenerative changes in the human brain through neuroimaging analyses, including damage to dopamine and serotonin axons, gray matter atrophy followed by hypertrophy of the white matter and microgliosis in various brain regions, such as prefrontal cortex, striatum, hippocampus, and midbrain. In addition, this drug enhances motor activity, satisfaction, happiness, consciousness, and respiration and decreases appetite. 4 –6 Meanwhile, narcotics such as marijuana or heroin are derived from plants, even though METH can simply be provided by the chemical available in illegal laboratories at low cost, which causes widespread use throughout the world. 7 In the World Drug Report (2015), METH users increased to 158% over the past 5 years, with popular areas of use evolving, including North America and Europe. 8,9 Furthermore, Iran is the fifth country in the world after Mexico, the United States, China, and Thailand for METH abuse. 10,11 Epidemiological studies have suggested that METH use increased the risk of neurodegenerative diseases such as Parkinson’s disease. Nevertheless, the molecular mechanisms of METH’s role in these diseases remain to be interpreted. 12 –14
Research evidence has shown that METH neurotoxicity generates reactive oxygen species (ROS), which emerges in the cortex or other regions of the brain. 15,16 On the other hand, METH can increase inflammatory factors such as tumor necrosis factor α and apoptosis caused by the expression of apoptotic genes and activation of apoptotic pathways. 15,17 Moreover, some studies on animal models have been reported that METH use can prompt apoptosis and autophagy in dopaminergic neurons. 18 –20 Recent research studies have revealed that autophagy is provoked by METH through Akt/mTOR-dependent pathways. 21,22 Understanding the mechanisms involved in the neurotoxicity of METH can lead to the discovery of new strategies to prevent the neurotoxic and neurodegenerative processes.
The peptide hormone obestatin (OB), first discovered in 2005, which encoded a 23-amino acid amidated peptide by the ghrelin gene together through posttranslational processing of preproghrelin peptide, is produced in the oxyntic mucosa of the stomach. 23,24 It has several scales of anti-ghrelin physiological actions such as suppressed food intake, slowed absorption of food, diminished intestinal peristalsis, and thereby prompting a reduction in body weight. 23,25 Alloatti et al. have discovered that OB could initiate some antiapoptotic signaling pathways and preserve the cardiac cell against myocardial damage and apoptosis provoked by ischemia–reperfusion (I/R). 26 It also plays a significant role in cardiac physiology and function. OB develops myocardial function and decreases cell death and apoptosis of cardiomyocytes after I/R in separated rat heart. 27,28 Some studies have revealed that OB relieves renal, gastrointestinal, and hemorrhage-induced brain damage in rats. 29 Moreover, it has been shown that OB promotes the proliferation of human retinal epithelial cells and rat dopaminergic cells. 30,31 Considering the neuroprotective effects of OB against METH-induced toxicity has been unknown and the lack of sufficient therapeutic strategies to share out with METH-induced neurotoxicity, the present study aimed to investigate the neuroprotective activity and potential mechanisms of OB in METH-induced PC12 cells. To this end, we examined the expression of Beclin-1 and LC3-II in catecholaminergic PC12 cells to analyze the effect of OB on autophagy in METH-induced PC12 cells. In addition, to evaluate the effect of OB on apoptosis, we used apoptosis assay using flow cytometry.
Methods and materials
Reagents
The PC12 cell line was obtained from Buali (Avicenna) Research Institute (Mashhad, Iran). RMPI-1640 medium, fetal bovine serum (FBS), trypsin–ethylenediaminetetraacetic acid (EDTA) were purchased from Gibco (Carlsbad, California, USA). OB, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and phosphate-buffered saline (PBS) were purchased from Sigma Chemical Corp. (St Louis, Mussouri, USA). PARIS™ Kit was purchased from Life Technologies, Thermo Fisher Scientific Inc. (Waltham, Massachusetts, USA). PrimeScript real-time polymerase chain reaction (RT-PCR) kit and bicinchoninic acid (BCA) assay kit were purchased from Takara Biotechnology (Shiga, Japan). MAP LC3β and β-actin antibody were purchased from Santa Cruz Biotechnology, Inc. (Dallas, Texas, USA). The Annexin V–fluorescein isothiocyanate (FITC) apoptosis detection kit was obtained from Invitrogen (Carlsbad, California, USA).
Cell culture and drug treatments
To evaluate the neuroprotective effects of OB on METH-induced cells, rat differentiated PC12 cell line was prepared in the current study. PC12 cells were grown in RPMI-1640 medium supplemented with 10% FBS, and 100 U/mL penicillin/streptomycin (PAN-Biotech GmbH, Aidenbach, Germany) at 37°C. In addition, 0.25% trypsin-EDTA was applied for detaching cells, and they were passaged every 2–3 days. PC12 cells were exposed to some concentrations of METH for 24 h. OB was added to the culture medium at determined concentrations 1 h before METH treatment.
MTT assay and microscopic morphological changes
Cell viability was evaluated by MTT assay. In summary, 104 cells/well PC12 cells were seeded in a 96-well plate. After one overnight incubation, METH was added in different concentration (0–6.0 mM) to discover the proper METH concentrations for inducing cells in the following studies. Then, 1, 10, 100, and 200 nM OB were added to cells 1 h prior to METH exposure to show the neuroprotective role of OB in METH-induced neurotoxicity. The morphology of PC12 cells was taken using an inverted microscope (Olympus IX71, Central Valley, Pennsylvania, USA). MTT added in a volume of 10 μl to each well (0.5 mg/mL final concentration) and incubated for 4 h. The MTT-containing medium was replaced by 100 μL of dimethylsulfoxide (Santa Cruz, Dallas, Texas, USA), and the plates were incubated for 10 min at 37°C. Absorbance was obtained at 570 nm with background subtraction at 690 nm using Biotek Cytation 5 imaging reader (Biotek Instruments, Winooski, Vermont, USA). All of the experiments were done in triplicate.
RNA extraction and complementary DNA synthesis
PC12 treatment groups were harvested, and total RNA was isolated using the PARIS Kit (Life Technologies, Thermo Fisher Scientific Inc.) according to the manufacturer’s directions. The quantity and quality of isolated RNAs were evaluated by Picodrop microliter spectrophotometer (OEM, UK) and electrophoresis on a 0.8% agarose gel. Eventually, PrimeScript™ RT reagent kit (Takara Bio, Shiga, Japan) was utilized to synthesize complementary DNA from 1 μg of total RNA according to the manufacturer’s instruction.
Quantitative RT-PCR
GAPDH 32 and Beclin-1 33 primers were obtained from the referenced manuscript, and the sequences were as follows: Beclin-1, forward 5′-ATCCTGGACCGAGTGACCATTC-3′, and reverse 5′-TCTCCTGAGTTAGCCTCTTCCTCC-3′; and GAPDH, forward 5′-GGCTGCCTTCTCTTGTGACAA-3′ and reverse 5′-TGCCGTGGGTAGAGTCATACTG-3′. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed using the SYBER Master Mix without ROX (Ampliqon, 5230 Odense M, Denmark) in a CFX96TM real-time system (Bio-Rad, Foster City, California, USA). All samples were done in duplicate. Relative expression of target genes was normalized to GAPDH as an internal control. Relative gene expression and fold changes were computed using the 2−ΔΔCt method.
Western blotting
Total proteins were isolated from different experimental groups using the PARIS Kit (Life Technologies, Thermo Fisher Scientific Inc.). The total protein concentration was determined based on the reference manual using a BCA assay kit (Takara, Shiga, Japan). Experimental group proteins were boiled for 10 min with loading buffer and separated by 15% sodium dodecyl sulfate–polyacrylamide gel electrophoresis. Proteins were transferred onto polyvinylidene fluoride membranes. Membranes were consequently blocked for 1 h at room temperature in blocking buffer (3% bovine serum albumin in Tris-buffered saline with Tween 20 (TBST)). Then, the membranes were incubated for 4°C overnight with anti-LC3B and anti-β-actin antibody (1:1000 dilution, Santa Cruz, Dallas, Texas, USA). Then, the membranes were washed three times with TBST, and the membranes were incubated with secondary antibody (1:1000 dilution, peroxidase-labeled mouse IgG-κ) at room temperature for 2 h, washed with TBST for three times, visualize with enhanced chemiluminescence immunoblotting detection system. The intensity of each band was quantitatively calculated by the Image J software (NIH, Bethesda, Maryland, USA).
Flow cytometry analysis
Annexin V-FITC/propidium iodide (PI) double staining was performed with the Annexin V-FITC apoptosis detection kit to identify the apoptosis of PC12 cells in experimental groups. Therefore, PC12 cells were seeded in six-well plates at a density of 5 × 105 cells/well and incubated at 37°C overnight. After 24 h of drug incubation, cells were centrifuged to remove the medium. Then, the samples were washed twice with PBS and stained with Annexin V-FITC and PI according to the instructions. The percentage of apoptotic cells was quantified by flow cytometry (Attune NxT, Thermo Fisher Scientific, Eugene, Oregon, USA).
Statistical analysis
Statistical analyses were implemented using GraphPad Prism 6 software (GraphPad Software Inc., San Diego, California, USA). Data were shown as the means ± standard error of the mean (mean ± SEM) and compared by one-way analysis of variance, followed by Turkey’s post hoc analysis to explore the significance of the data. The value of p < 0.05 was considered statistically significant. All the experiments were replicated at least three times.
Results
The effect of OB on the viability in METH-induced PC12 cells
First, we examined the effects of METH at different concentrations (0.5, 1, 2, 3, 4, and 6 mM) on the PC12 cell viability through MTT assay to find the proper concentration for evaluating the neuroprotective effects of OB. Data in Figure 1(a) indicated that METH dose dependently inhibited the proliferation of PC12 cells when incubated for 24 h. The chosen concentration value of METH was about 4 mM such that the concentration was utilized in some experiments. 34,35 Then, we investigated whether OB protects against METH-induced PC12 cells viability loss. As shown in Figure 1(b), when OB (1-200 nM) was added in the cell culture medium 1 h prior to 4 mM METH treatment, the survival of PC12 cells significantly increased in a dose-dependent manner. The maximal rescue occurred at a concentration of 200 nM OB, which we have chosen that for downstream experimental. Moreover, treatment of OB (200 nM) alone showed no effect on PC12 cell viability.

OB protect against METH-induced cells viability loss in PC12 cells. (a) Effects of METH on cell viability. PC12 cells were treated with the indicated concentration of METH (0.5, 1, 2, 3, 4, and 6 mM) and the cell viability was measured after treatment of 24 h by MTT assay. (b) Effects of OB on METH-induced cell viability loss. Cells were treated with 4 mM METH for 24 h in the absence or presence of OB (1, 10, 100, and 200 nM) treatment at the indicated concentrations 1 h prior to METH treatment, and a dose-dependent effect of OB was assessed by MTT assay. (c) The cells were treated with 4 mM METH, 4 mM METH plus 200 nM OB or 200 nM OB alone for 24 h. Morphological changes have shown in response to OB treatment. Results are expressed as a percentage of value in untreated control cultures and are mean ± SEM of three replicate values obtained from three independent experiments. **p <0.01, ***p <0.001, and ****p <0.0001 versus control; # p < 0.05, ## p < 0.01 versus METH treatment alone. METH: methamphetamine; OB: obestatin; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; SEM: standard error of the mean.
Morphological changes of METH-induced PC12 cells in response to OB treatment
Microscopic study of morphological changes of PC12 cells after treatment of cells with METH and OB after 24 h under an inverted microscope have shown that the METH-induced PC12 cells have changed compared to the control group. The shape of the cells under the influence of METH has been rounded, which presents a significant change in the normal state of the cell membrane and reduced cell volume. In addition, a number of cells were singled from the flask bottom and floated. While 1 h of pretreatment of cells with OB led to stability of the cells. Moreover, there is no change in the morphology of the cells in the control and OB groups (Figure 1(c)).
The effect of OB on autophagy in METH-induced PC12 cells
In this part of the study, to evaluate the effect of OB on autophagy in METH-induced PC12 cells, and the neuroprotective effects of OB, the mRNA expression of Beclin-1 level and LC3-II protein level were examined. To investigate the expression of Beclin-1, RT-qPCR was used, and the result has determined that Beclin-1 mRNA expression in METH-induced PC12 cells was significantly increased compared with the control group (1.16 ± 0.13, p < 0.001) as presented in Figure 2(a). While pretreatment with OB decreased Beclin-1 mRNA expression in METH-induced cells (0.58 ± 0.0.05, p < 0.01). On the other hand, 200 nM OB alone decreased the mRNA expression level of Beclin-1 (0.37± 0.0.09, p < 0.01). Furthermore, the LC3-II protein expression was analyzed through Western blotting. The result has shown that the protein expression of LC3-II in METH-induced PC12 cells was significantly increased compared with the control group (1.45 ± 0.06, p < 0.05). In contrast, pretreatment with 200 nM OB decreased protein expression of LC3-II in METH-induced PC12 cells (0.19 ± 0.06, p < 0.01). The 200 nM OB decreased the protein expression level of LC3-II the most to 0.43 ± 0.12 (p < 0.01) in comparison with the METH-only treated group (Figure 2(b)).

Effect of OB on autophagy in METH-induced PC12 cells. The cells were treated with 4 mM METH, 4 mM METH plus 200 nM OB, or 200 nM OB alone for 24 h. RNA and proteins were extracted and analyzed by real-time and Western blot analyses: (a) real-time PCR analysis for Beclin-1 gene expression (n = 8) and (b) Western blot analysis for LC3-II gene expression (n = 4). Values are means ± SEM. Our results were analyzed by one-way ANOVA, followed using a Tukey’s post hoc test. Statistical significance: *p < 0.05 compared to the control group, ## p < 0.01, ### p < 0.001: compared to the METH group. METH: methamphetamine; OB: obestatin; PCR: polymerase chain reaction; ANOVA: analysis of variance.
The effect of OB on apoptosis in METH-induced PC12 cells
Apoptosis assay was performed to examine the effect of OB on apoptosis in METH-induced PC12 cells through Annexin V-FITC/PI kit. As shown in Figure 3(a) and (b), the control group has 4.53 ± 0.80% apoptosis and 2.5. ± 2.08% necrosis. While the METH group has 21.84 ± 0.22% apoptosis and 9.63 ± 3.27% necrosis. On the other hand, PC12 treated with OB has 5.55 ± 0.24% apoptosis and 1.21 ± 0.31% necrosis. However, when the cells were pretreated with OB prior to exposure to 4 mM METH, the observed apoptosis rate was significantly attenuated, and the apoptosis rate was 10.63 ± 1.09% apoptosis and 3.38 ± 0.73% necrosis. Together, results have explained that apoptosis and necrosis regularly increased in the METH group compared with the control group (p < 0.001, p < 0.05, respectively) and as indicated in Figure 3(b), this experiment has proved that OB significantly reduced cell apoptosis on METH-induced PC12 cells compared to the METH group (p < 0.05).
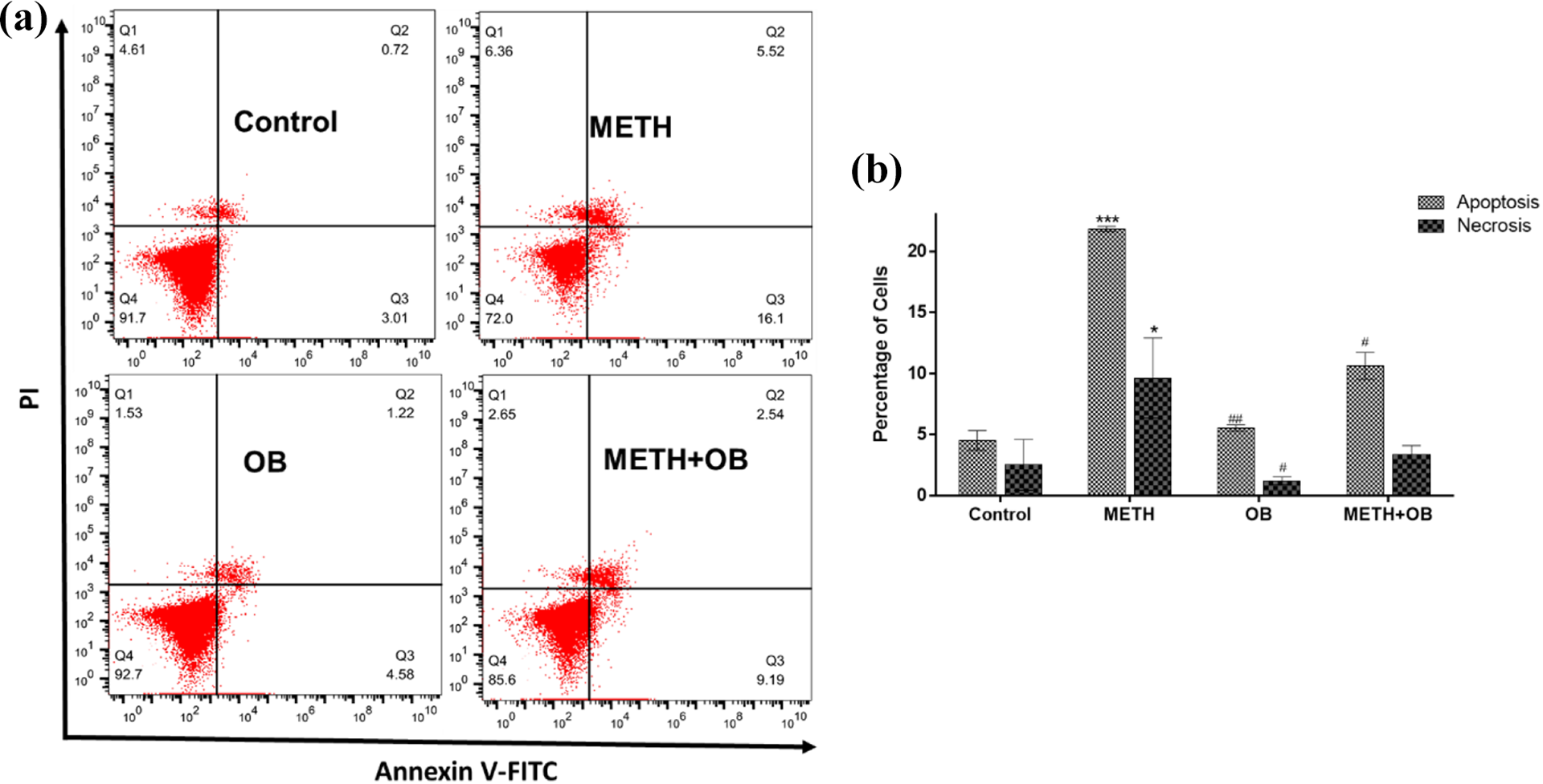
OB protect against METH-induced PC12 cell apoptosis. Cells were treated with 4 mM METH for 24 h in the absence or in the presence of 200 nM OB treatment 1 h prior to METH treatment. (a) Apoptosis results are shown in logarithmic fluorescence intensity with the x-axis (annexin V-FITC) versus y-axis (PI). Four quadrants represent necrosis cells (Q1), late apoptotic cells (Q2), viable cells (Q3) and early apoptotic cells (Q4). (b) The percentage of apoptotic PC12 cells in each group. Data are presented as mean ± SEM (n = 3). *p < 0.05 and ***p < 0.001 compared to the control group, #p < 0.05 and ##p < 0.01 compared to the METH group. METH: methamphetamine; OB: obestatin; FITC: fluorescein isothiocyanate; SEM: standard error of the mean.
Discussion
METH is a stimulant and sympathomimetic drug that easily passes through the blood–brain barrier and induces the central nervous system. Some studies have shown that METH exposure can cause cell death, neurotoxicity, and substantial injury to the dopaminergic axon terminals which have been associated with oxidative stress and the imbalance between the production of free radicals and antioxidants. 36,37 On the other hand, METH is known as a neurotoxic model, which new research studies have published that it is involved in inflammatory–neurological processes and brain dysfunction due to abuse of the drug. Indeed, glial cells develop to be activated in response to METH, although its effects on microglial cells are not fully elucidated. 38 However, there are some studies which have shown that oxidative stress, 36 apoptosis, necrosis, 39,40 excitotoxicity, and calcium deregulation are associated with METH neurotoxicity. 41 These pieces of evidence have motivated researchers to study the neurotoxicity effects of METH and the study of composites that decrease its toxic consequences. The antioxidant function of OB has been suggested as a plausible mechanism for its neuroprotective potential. 42 Hence, the current study examined the novel neuroprotective effects of OB on METH-induced neurotoxicity in the catecholaminergic PC12 cell line, which is a suitable model for neurological and neurobiological studies. First, the results suggest that OB treatment increase cell viability and proliferation of PC12 cells. Furthermore, it was shown that OB decreases autophagy and apoptosis which is raised by METH toxicity.
Recent studies have shown that OB is a new peptide hormone that is encoded by the identical gene as ghrelin. It is an anorexigenic peptide, which reverses the outcomes of ghrelin by reducing appetite and weight gain. In addition, it is involved in different physiological functions such as myocardial functional improvement in response to risk factors, participation in cardiovascular regulations by ghrelin, improved cardiac function, suppression of apoptosis of myocardial cell and protection of cardiac cell from cardiac damage. 25,43 According to the studies, our study confirmed the neuroprotective effects of OB, which has shown that it could reduce the neurotoxic effects of METH.
Recent studies have presented that METH exposure can cause a reduction in cell viability. Pitaksalee et al. have published that proliferation and cell viability of SH-SY5Y cells diminished significantly after 24 h of METH treatment in a dose-dependent manner. 44 On the other hand, Shen et al. have investigated the protective role of OB in MES23.5 dopaminergic cells against 1-methyl-4-phenylpyridinium (MPP+)-induced neurotoxicity. They reported that OB pretreatment prevented or even changed the MPP+-induced reduction of proliferation in cells. 31 In the current research, MTT assay showed that METH reduced proliferation and cell viability significantly versus the control group, while cell death could be obstructed when the cells were pretreated with different concentrations of OB between 1 nM and 200 nM. Therefore, the results have found that decreased cell viability induced by METH can be rescued by OB.
Autophagy is a cellular mechanism that plays an essential role in the growth, homeostasis, and differentiation of the cell. In addition, it degenerates damaged organelles and misfolded proteins to conserve cellular homeostasis for controlling the suitable level of proteins and intracellular organelles. Furthermore, autophagy can be activated upon physiological stresses such as cell protection response to intracellular stresses, promote cell survival, removal of the cell of damaged organelles, toxic metabolites, and intracellular pathogens. 45 –48 Due to the lack of peculiar control, autophagy may stimulate apoptosis and degrade cells by self-digestion and eventually leading to death. The relations between ROS and autophagy endure in a category of pathological states. Prior studies have shown that improved levels of ROS magnified oxidative stress response, excessive generation of ROS stimulates hypoxia-inducible factor (HIF-1), p53, and nuclear factor erythroid 2–related factor 2 (NRF2). Then, these transcription factors induce the transcription of target genes such as LC3 and BNIP3 and p62. Eventually, They induce autophagy in the cytoplasm, which can be estimated as posttranscriptional regulation. 49 –52 The interaction between antiapoptotic proteins and autophagy proteins performs a crucial role in cellular protection or cell death. Bcl-2 is fundamentally connected to Beclin-1 and providing basal levels of autophagy. Following autophagy-inducing forms, Bcl-2 separates from Beclin-1 and enhances autophagy. 53 Previous investigations have illustrated that various drugs such as cocaine, morphine, heroin, tetrahydrocannabinol, and nicotine can increase autophagy in different experiments. 18,20 On the other hand, Frost and Cadet have suggested that the toxic effects of METH involve a cascade pathway that incorporates the production of hydrogen peroxide, superoxide, and hydroxyl radicals. Besides, they have shown that in vitro experiments have displayed the production of both superoxide radicals and hydrogen peroxide during exposure of cells to METH. 54
Additionally, Ma et al. have proved that METH induces autophagy as a survival response to the death of apoptotic endothelial cells through the kappa opioid receptor. They have reported that autophagy is an early response to the METH-induced stress, and it may have a protective role during exposure to METH. Nevertheless, prolonged exposure to METH is flushing autophagy and dooming the cell to apoptotic death. 55 Besides, Cid-Díaz et al. have reported that OB regulates protein synthesis, autophagy, and ubiquitin–proteasome systems in a interrelated way involving Akt-, PKD/PKCμ-, CAMKII- and AMPK-dependent mechanisms. 56 In the present study, qRT-PCR was used to measure the expression of Beclin-1 gene and Western blotting to estimate the expression of the LC3-II protein. It has revealed that exposing PC12 cells with METH caused a significant increase in the expression of Beclin-1 and LC3-II autophagy genes, following in increased autophagy in this group. However, the expression of these proteins in METH-induced cells can be reduced through OB treatment. This evidence demonstrates the neuroprotective effects of OB against autophagy.
Apoptosis involves multiple pathways driving to a change in the structure of the biomolecules, including lipids, proteins, and nucleic acids. The critical changes during apoptosis include the modification of the membrane structure and consequently, the permeability of the membrane to the different materials, which is based on the assay of apoptosis assay of Annexin V-FITC/PI in determining the percentage of apoptosis in cells. 57 Prior research studies have explained that apoptotic cell death can be associated with the neurotoxicity of drug and psychotropic drugs. Oliveira et al. have demonstrated that heroin was accomplished to induce apoptosis in PC12 cells and rat neurons. 58 Furthermore, METH has been determined to produce neuronal apoptosis in vivo and in vitro. Tian and colleagues described that the amount of apoptosis in PC12-treated cells with METH and heroin alone and their combination significantly increased apoptosis. 59 Formation and accumulation of ROS in the mitochondria open mitochondrial permeability transition pores (mPTP) and develop mitochondrial membrane hyperpolarization. Cytochrome c, as a proapoptotic protein, flows from the damaged mitochondrial membrane into the cytoplasm. Leakage of cytochrome c drives to the activation of caspase 3 and nuclear fragmentation to initiate apoptosis. 53
On the other hand, Tang et al. have investigated the role of OB on porcine preadipocytes, and they indicated that OB was involved in porcine adipocyte metabolism by inhibiting preadipocyte apoptosis through decreasing expression of caspase 3, caspase 7, and caspase 9. In addition, OB decreases the oxidative stress by diminishing the level of malondialdehyde and increasing the level of superoxide dismutase and catalase antioxidants. 60 In our study, the results of the apoptosis assay have shown that exposing PC12 cell to METH significantly increase cell apoptosis, while the apoptosis of METH-induced neurotoxicity can be reduced by OB pretreatment. This evidence confirms the protective effects of OB against apoptosis.
Conclusion
Our investigation found that 24 h treatment of PC12 cells with METH led to apoptosis and autophagy, as presented by decreases in cell viability, increases in apoptosis rate, and increases in Beclin-1 and LC3-II expression. On the other hand, OB is effective in protecting PC12 cells against METH-induced neurotoxicity. OB is a neuroprotective peptide, and it prevents neuronal death by affecting multiple enzymatic and molecular pathways and reduces apoptosis and autophagy. Given the role of METH and OB implicates in psychosis and neurodegenerative disease, these findings in vitro may support to future investigations of other pathways in related models.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shahroud University of Medical Sciences [Grant Number 9859].
