Abstract
Introduction
Sodium dodecyl sulfate (SDS), a widely used surfactant in detergents, has raised concerns due to its potential health risks, particularly in children. This study evaluates the impact of SDS exposure on GH secretion in GH3 cells, focusing on oxidative stress as a key mechanism.
Methods
GH3 cells were treated with varying concentrations of SDS (0.001–10 mM) for 24 or 48 h. Cell viability was assessed using the MTT assay, while GH secretion was quantified via ELISA. Oxidative stress levels were evaluated through ROS fluorescence assays, and gene expression of Nrf2, IL-6, TNF-α, and caspase-3 was analyzed using qPCR. Additionally, the antioxidant N-acetylcysteine (NAC) was used to determine its protective effects against SDS-induced oxidative stress.
Results
SDS exposure led to a dose-dependent decrease in GH secretion and cell viability, with oxidative stress identified as a primary driver. Nrf2 exhibited a biphasic response, showing transient upregulation at low doses but suppression at higher concentrations, exacerbating oxidative damage. NAC treatment reduced ROS levels and partially restored GH secretion, confirming the role of oxidative stress in SDS-induced toxicity.
Discussion
These findings suggest that SDS exposure may disrupt endocrine function, warranting further risk assessment of its safety in consumer products. Given SDS’s prevalence in household products, future research should focus on the long-term effects of SDS exposure to children and potential therapeutic interventions to mitigate oxidative damage.
Keywords
Introduction
Detergents, widely used in households and industries, have become a significant concern regarding their impact on children’s health. Over the past decades, there has been growing global attention to the adverse effects of detergents. These chemicals are commonly recognized for causing severe allergic reactions in the respiratory and gastrointestinal tracts and damaging epithelial tissues.1,2 Such effects have raised alarms about the safety of detergent exposure, particularly in vulnerable populations like children. 3
Recent reports have highlighted that the harm from detergent exposure extends beyond allergies and tissue damage. Emerging evidence suggests that children exposed to detergents may experience more severe consequences, including vomiting, respiratory distress, and even fatal outcomes.4–6 These findings indicate that the ingestion or exposure to detergents might lead to unforeseen and systemic health implications, necessitating a deeper investigation into the mechanisms underlying these effects. Additionally, cases of accidental detergent ingestion are not uncommon in children, increasing the risk of both acute poisoning and chronic exposure, which could have long-term developmental implications.7–9 It is noteworthy that detergents have been reported to be harmful for endocrine system, emphasising the significance to analyse its effects on childhood growth and development. 10 A key component of many detergents is sodium dodecyl sulfate (SDS), a surfactant widely used for its effective cleaning and emulsifying properties. SDS has been identified as a potential contributor to the adverse effects observed in detergent exposure. 11 Studies have shown that SDS can lead to endothelial cell damage, primary hepatocyte injury in mice, and severe allergic reactions through mechanisms involving increased inflammation and oxidative stress.11,12 Given its prevalence in household products and its potential to interact with biological systems, understanding SDS’s impact is particularly crucial, especially in contexts related to childhood development.
In this study, we focused on the effects of SDS on growth hormone secretion using the GH3 cell line, a well-established in vitro model for anterior pituitary function and growth hormone release.13–15 Previous studies suggest that SDS exposure can lead to cellular toxicity, but its specific impact on endocrine function remains unclear. Given the role of GH3 cells in anterior pituitary hormone secretion, this study aims to elucidate the molecular mechanisms underlying SDS-induced disruption of GH secretion. By evaluating oxidative stress and the Nrf2 pathway, we aim to provide insights into the endocrine risks associated with detergent exposure, particularly in paediatric populations. The findings of this work provide valuable insights into the potential risks posed by detergents and contribute to developing strategies for mitigating these risks.
Materials and methods
Cell culture
GH3 rat pituitary tumor cells were cultured in complete medium (Ham’s F-12K supplemented with 15% horse serum, 2.5% fetal bovine serum, and 1% penicillin-streptomycin) under standard conditions (37°C, 5% CO2). Cells were subcultured when reaching 90% confluence.
SDS and antioxidant NAC treatment
Sodium dodecyl sulfate (SDS) stock solution (1 mol/L) was prepared by dissolving 288 mg of SDS powder in 10 mL of sterile medium, followed by filtration for sterilization. For experiments, cells were divided into control (0 mM SDS) and experimental groups treated with different concentrations of SDS (0.001 mM, 0.01 mM, 0.1 mM, 1 mM, 10 mM) for 24 or 48 h. Low concentrations (0.001–0.1 mM) reflect potential residual SDS exposure from personal care products, while higher concentrations (1–10 mM) model acute exposure from ingestion. 12 The 24-h exposure duration is a “worst-case” scenario of prolonged SDS exposure during a working day. 2 Considering that accidental ingestion in children may occur frequently, persist over time, and often go unnoticed, the 48-h exposure duration was included to evaluate the potential cumulative endocrine toxicity effects of sustained SDS exposure.
For antioxidant treatment, N-acetylcysteine (NAC, Merck, Germany, A0150000) stock solution (100 mmol/L) was prepared by dissolving 16.3 mg of NAC powder in 1 mL of DMSO, followed by filtration for sterilization. In experimental setups, NAC was diluted to a working concentration of 2 mM in the medium and added 1 h prior to SDS treatment to evaluate its protective effects against SDS-induced oxidative stress. Experimental groups were categorized as control (0 mM SDS), SDS-only (0.1 mM SDS), and SDS + NAC (0.1 mM SDS +2 mM NAC) treated for 24 h.
Cell viability assay (MTT)
Cell viability was evaluated using the MTT assay. Cells were seeded in 96-well plates at 100 μL/well and incubated for 24 h. After SDS treatment, 10 μL of MTT solution was added to each well, followed by 4 h of incubation at 37°C. Formazan crystals were dissolved by adding 100 μL of DMSO, and absorbance was measured at 570 nm using a microplate reader.
Growth hormone detection (ELISA)
Cell supernatants were collected after SDS treatment, and growth hormone levels were measured using a rat growth hormone ELISA kit (RX302355R, Ruixin Biotech). The assay followed the manufacturer’s instructions. Standards and samples (50 μL) were added to a 96-well plate pre-coated with anti-GH antibodies, followed by the addition of HRP-conjugated detection antibodies. Substrate reactions were performed with TMB, and absorbance was measured at 450 nm using a microplate reader.
Reactive oxygen species (ROS) detection
Intracellular ROS levels were assessed using DCFH-DA fluorescent probes. After SDS treatment, cells were collected and suspended in 10 μmol/L DCFH-DA diluted in PBS. Cells were incubated at 37°C for 20 min, with periodic mixing. Excess probe was removed by washing cells three times with PBS. Fluorescence was observed and photographed using a confocal microscope.
Quantitative PCR (qPCR)
Total RNA was extracted using Trizol reagent, and RNA concentration and purity were assessed using a NanoDrop spectrophotometer. Reverse transcription was performed using Hifair® III 1st Strand cDNA Synthesis SuperMix (Shanghai Yisheng), and qPCR was conducted using Hieff® qPCR SYBR Green Master Mix (Shanghai Yisheng). Primers targeting TNF-α (Forward: CCGATTTGCCATTTCCATACCAG; Reverse: TCACAGAGCAATGACTCCAAAAG), IL-6 (Forward: GGATTTCCGTTTCTACCTGG; Reverse: GCCGAGTAGACCTCATAGTG), Caspase-3 (Forward: GGACAACAACGAAACCTCCG; Reverse: CATTGCGAGCTGACATTCCA), and Nrf2 (Forward: TGACTCTGACTCCGGCATTT; Reverse: CCCCAGAAGAATGTGTTGGC) were used to analyze gene expression. Relative expression levels were calculated using the ΔΔCT method.
Analytical method
All experiments were performed in triplicate, and results are presented as mean ± standard deviation (SD). Statistical significance was determined using one-way ANOVA followed by Tukey’s post-hoc test for multiple comparisons (GraphPad Prism 9.0). Outliers were assessed using Grubbs’ test and removed if p < .05. A p-value of < .05 was considered statistically significant.
Results
SDS reduced cell viability and decreased growth hormone secretion
MTT assays revealed that SDS exposure significantly reduced GH3 cell viability. At 24 h, SDS concentrations ≥1 mM caused a notable decrease in cell viability (p < .05). After 48 h, SDS concentrations as low as 0.1 mM significantly reduced cell viability (p < .05). Higher concentrations (10 mM) led to nearly complete cell death (Figure 1(a) and (b)). Dose-Dependent Effects of SDS on GH3 Cell Viability and Growth Hormone Secretion. SDS exposure significantly reduced GH3 cell viability and growth hormone (GH) secretion in a dose-dependent manner. (a, b) MTT assays showed that 24-h exposure to 1 mM SDS led to a ∼50% reduction in cell viability, while 10 mM SDS nearly abolished viability (p < .001). At 48 h, SDS concentrations as low as 0.1 mM significantly reduced cell viability (p < .001). (C, D) ELISA results demonstrated that GH secretion decreased after SDS treatment. At 24 h, 0.1 mM SDS caused a significant reduction in GH secretion (∼20%, p < .001), and 1 mM SDS further suppressed GH secretion by over 50% (p < .001). At 48 h, GH suppression was even more pronounced. Data are expressed as mean ± SD (n = 6). Statistical significance was determined using one-way ANOVA followed by Tukey’s test. ***p < .001, **p < .01 indicate significance compared to the control group.
ELISA results demonstrated that SDS exposure led to a significant reduction in growth hormone secretion. At 24 h, SDS at concentrations ≥0.1 mM reduced GH secretion by approximately 20% compared to the control group (p < .05). After 48 h, concentrations ≥1 mM suppressed GH secretion by more than 50% (p < .01) (Figure 1(c) and (d)). Based on these findings, a 24-h exposure to SDS was selected as the experimental condition for subsequent assays, as it caused significant GH suppression at 0.1 mM and could also affect cell viability at higher concentrations, allowing for further investigation into the underlying mechanisms of SDS-induced toxicity.
SDS induced oxidative stress and modulated gene expression
Figure 2(a) confirmed the results of the MTT assay in Figure 1, demonstrating that SDS significantly reduced GH3 cell viability and proliferation after 24 h. However, the reduction in growth factor secretion at 0.1 mM SDS was not associated with decreased cell viability or proliferation, suggesting that other mechanisms such as oxidative stress or inflammation may be involved. At 0.1 mM SDS, ROS levels were significantly elevated, as indicated by increased fluorescence intensity in DCFH-DA assays (Figure 2(b)). Quantitative analysis confirmed that ROS mean fluorescence intensity increased intensively compared to the control group at 0.1 mM(p < .001), indicating that the decrease in growth factor level may relate to ROS increase. SDS-Induced Oxidative Stress and Inflammatory Gene Expression in GH3 Cells. SDS exposure induced oxidative stress and modulated gene expression in GH3 cells. (a) SDS treatment significantly reduced GH3 cell proliferation in a dose-dependent manner. At 1 mM, cell viability was reduced by approximately 50% (p < 0.01; n = 6). (b) Representative fluorescence images of ROS generation in GH3 cells under different SDS concentrations. (c) Quantification of ROS mean fluorescence intensity showed a significant increase at 0.1 mM SDS (p < .001), suggesting oxidative stress as a key mechanism of GH levels decreasing. (d–f) SDS significantly upregulated the expression of pro-inflammatory cytokines IL-6 and TNF-α and apoptosis marker caspase-3 at 0.1 mM (p < .05), indicating inflammation and apoptosis may also contribute to SDS-induced toxicity. Data are shown as mean ± SD (n = 3). *p < .05, **p < .01, ***p < .001, compared to control.
qPCR analysis further revealed that at 0.1 mM SDS, the expression of pro-inflammatory cytokines IL-6 and TNF-α increased significantly. IL-6 and TNF-α expression levels were elevated approximately 6- and 8-fold, respectively, at 10 mM SDS (p < .001, Figure 2(d) and (e)). Caspase-3 expression, a marker for apoptosis, was also significantly upregulated, increasing nearly 5-fold at 10 mM SDS (p < .001, Figure 2(f)). These findings suggest that oxidative stress and inflammatory pathways may also be critical mediators of SDS-induced suppression of growth hormone secretion.
SDS activated Nrf2 expression
Nrf2 (nuclear factor erythroid 2–related factor 2) is a master regulator of cellular antioxidant responses. It plays a pivotal role in maintaining redox homeostasis by controlling the expression of numerous antioxidants and cytoprotective genes.
16
Given that SDS exposure leads to the generation of reactive oxygen species (ROS), which are known to cause cellular damage and dysregulate signaling pathways, it is essential to examine the role of Nrf2 in this context. Previous studies have shown that surfactants like SDS can trigger oxidative stress, leading to an imbalance in redox homeostasis and activating cellular defense mechanisms.17,18 Based on these findings, Nrf2 emerges as a key target to understand the cellular response to SDS-induced oxidative stress. SDS treatment had a dual effect on the expression of Nrf2 in GH3 cells, with a slight upregulation observed at lower concentrations and a significant reduction at higher concentrations (Figure 3). At 0.01 mM SDS, Nrf2 expression showed a modest increase compared to the control group, suggesting an initial compensatory response to oxidative stress. However, at concentrations of 0.1 mM or higher, Nrf2 expression was significantly reduced, reaching approximately 80% of the control level at 0.1 mM (p < .05) and nearly 60% at 1 mM and 10 mM (p < .01). This biphasic response indicates that while low concentrations of SDS may activate antioxidant defenses, prolonged or higher exposure likely overwhelms the cellular defense mechanisms, impairing the Nrf2 pathway. This is consistent with previous findings that This is consistent with previous findings that Nrf2 activation follows a biphasic response, where low levels of oxidative stress induce a protective upregulation of Nrf2, while excessive oxidative stress suppresses its expression, leading to impaired antioxidant defense and increased inflammation,
19
and aligns with the concept of hormesis, where mild stressors activate protective pathways, but excessive exposure results in toxicity.
20
These findings highlight the complex relationship between SDS-induced oxidative stress and cellular antioxidant responses. It needs further investigation into the threshold at which oxidative damage becomes irreversible. Modulation of Nrf2 Expression by SDS Exposure in GH3 Cells. Nrf2 expression was slightly upregulated at low SDS concentrations (0.001–0.01 mM) but significantly reduced at higher concentrations. The suppression of Nrf2 at high SDS concentrations suggests an impaired antioxidant defense mechanism, exacerbating oxidative stress-related damage (0.1–10 mM, *p < .05, **p < .01 vs control). Data are presented as mean ± SD (n = 3).
Antioxidant NAC attenuated SDS-induced effects
Treatment with the antioxidant N-acetylcysteine (NAC) significantly mitigated SDS-induced oxidative stress and reduced its inhibitory effect on growth hormone secretion (Figure 4). ROS levels, as shown in Figure 4(a) and quantified in Figure 4(b), were elevated in the SDS-treated group compared to the control group (p < .001). NAC treatment effectively reduced ROS levels, bringing them closer to control levels (p < .01). Similarly, growth hormone secretion (Figure 4(c)), which was markedly suppressed in the SDS group (p < .001), was partially restored with NAC treatment (p < .05). These findings are based on experiments using 0.1 mM SDS, a concentration previously shown to disrupt growth hormone secretion without significantly affecting cell viability or proliferation. This highlights the relevance of moderate SDS concentrations in studying oxidative stress and the potential of NAC to counteract these effects. These findings suggest that oxidative stress plays a central role in SDS-induced suppression of growth hormone secretion, and NAC alleviates these effects by reducing oxidative stress. Protective Effects of NAC Against SDS-Induced Oxidative Stress and GH Suppression in GH3 Cells. NAC treatment reduced oxidative stress and partially restored GH secretion in 0.1 mM SDS-exposed GH3 cells. (a) Representative fluorescence images of ROS generation in control, SDS-treated, and SDS + NAC-treated cells. (b) Quantification of ROS intensity showed a significant reduction in ROS levels in NAC-treated cells compared to the SDS-only group (p < .01; n = 3). (c) Growth hormone secretion, which was significantly suppressed by SDS exposure (p < .001; n = 6), was partially restored by NAC treatment (p < .05). These results suggest that oxidative stress plays a key role in SDS-induced toxicity and that NAC exerts protective effects. Data are presented as means ± SDs. Statistical significance: ###p < .001 versus control; *p < .05 and **p < .01 versus SDS-treated group.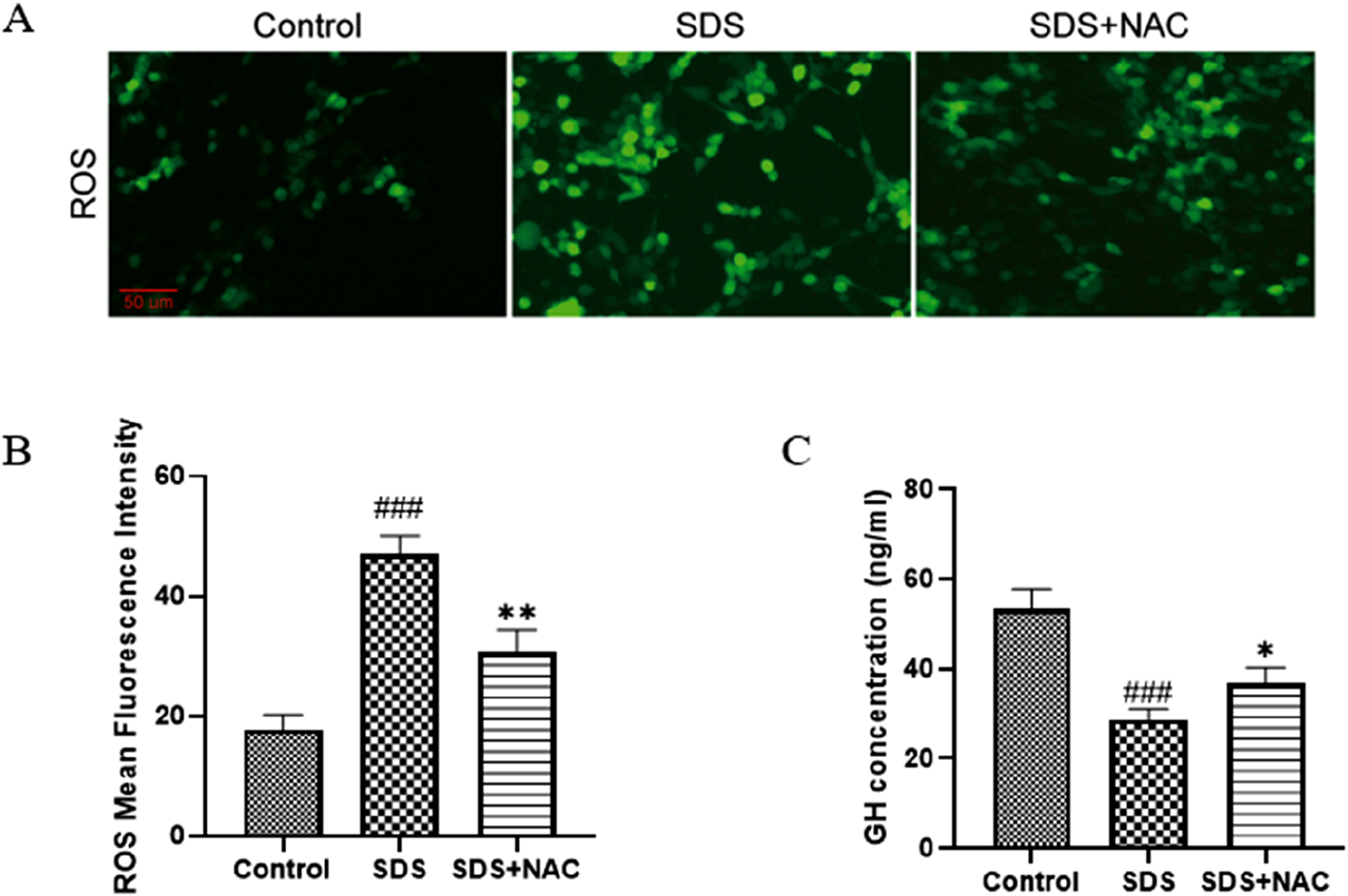
Discussion
This study investigated the effects of SDS, a key component in many detergents and personal care products, on GH3 rat pituitary tumor cells. In this study, we show for the first time that SDS exposure can significantly reduce GH secretion and increase oxidative stress in a dose-dependent manner. These findings are particularly relevant to children, who are more likely than adults to experience prolonged and high-dose exposure to SDS-containing products due to higher rates of accidental ingestion and acute poisoning incidents. This emphasizes the need for further research on SDS’s potential impacts on growth and development in children, as well as its implications for recovery and treatment strategies in cases of poisoning.
The choice of SDS concentrations and exposure durations in this study aimed to simulate realistic exposure scenarios and their potential biological effects. SDS doses ranged from 0.001 to 10 mM, covering the spectrum of possible bloodstream exposure level. 21 Low concentrations (0.001–0.1 mM) reflect potential residual SDS exposure from personal care products, while higher concentrations (1–10 mM) model acute exposure from ingestion. 12 The exposure durations were selected to assess both short- and long-term effects. The 24-h exposure duration represents a “worst-case” prolonged exposure scenario during a working day, 2 while the 48-h exposure duration was included to evaluate the potential cumulative endocrine toxicity of sustained SDS exposure, particularly in children, where accidental ingestion may frequently occur, persist over time, and often go unnoticed. These doses were selected to model both acute and prolonged exposure conditions, enabling a detailed evaluation of SDS-induced oxidative stress, growth hormone secretion, and cellular viability in GH3 cells. Previous research on SDS penetration into human skin, such as studies using radiolabeled SDS, highlights that prolonged exposure can lead to dose-dependent penetration of SDS into deeper layers of the stratum corneum and eventually into the bloodstream. This phenomenon arises due to surfactant-induced barrier damage, allowing higher concentrations of SDS to breach the skins protective layers. 12 These findings suggest that prolonged dermal exposure may result in systemic absorption of SDS, raising concerns about long-term health risks. Similarly, gastrointestinal exposure to SDS—such as accidental ingestion of detergent-containing products—may present comparable risks. 22 Although no studies to date have specifically quantified the doses of SDS that could be absorbed into the bloodstream through the digestive tract, accidental ingestion of SDS-containing detergents is not uncommon in children. 3 There have been reports of severe outcomes, including respiratory distress, lethargy, coma, and even death, following such incidents. This underscores the need for further research to understand the potential for SDS absorption through the gastrointestinal tract and its associated systemic effects. 4
The suppression of GH secretion by SDS was observed even at concentrations that did not significantly reduce cell viability or proliferation. This suggests that SDS-induced disruption of GH secretion may involve mechanisms independent of direct cytotoxicity, such as oxidative stress and inflammatory responses. Elevated reactive oxygen species (ROS) levels and the upregulation of pro-inflammatory cytokines (IL-6 and TNF-α) observed in this study support this hypothesis. These results are consistent with prior findings that SDS can promote ROS generation and influence cellular survival by interacting with critical antioxidant enzymes such as catalase (CAT), leading to structural and functional alterations. 12 Additionally, SDS has been reported to impair the balance of cellular redox states, exacerbating oxidative stress and triggering inflammatory cascades. Interestingly, the dual effects of SDS on Nrf2 expression further highlight the complexity of its impact on cellular oxidative stress and inflammation. At low concentrations, a slight upregulation of Nrf2 was observed, suggesting a transient compensatory antioxidant response aimed at mitigating oxidative stress. However, prolonged exposure or higher concentrations of SDS significantly suppressed Nrf2 expression, which may impair its protective role in both antioxidant defense and inflammation regulation. Nrf2 has been shown to play a dual role by not only activating antioxidant pathways but also modulating the expression of genes involved in inflammatory processes, including pro-inflammatory cytokines. The suppression of Nrf2 at higher SDS concentrations could exacerbate inflammatory responses, further contributing to cellular dysfunction. Further research is needed to delineate the specific molecular pathways involved and to explore potential therapeutic strategies for mitigating SDS-induced toxicity, particularly in sensitive populations such as children.
The protective effects of N-acetylcysteine (NAC) further emphasize the central role of oxidative stress in SDS-induced toxicity. 23 NAC, a potent antioxidant, effectively reduced ROS levels and partially restored GH secretion in SDS-treated cells. These findings suggest that therapeutic strategies targeting oxidative stress, such as antioxidant supplementation, could mitigate the adverse effects of SDS on endocrine function.
While the potential risks of SDS exposure are significant, it is important to note that no evidence currently suggests SDS accumulates in any specific organ. Moreover, SDS entering the bloodstream is rapidly cleared from the body. 24 Treatment considerations for SDS-induced toxicity are primarily relevant in cases of substantial ingestion, such as in children who accidentally ingest large amounts. Given the widespread use of SDS in household products, our findings raise concerns about its potential endocrine-disrupting effects, particularly in children who may experience repeated exposure. Stricter measures may be taken to prevent children from coming into contact with detergents. While this study provides critical mechanistic insights, further in vivo research is needed to assess systemic SDS toxicity. Future studies should incorporate physiologically based pharmacokinetic (PBPK) modeling to estimate real-life risk and inform regulatory guidelines on SDS exposure limits in consumer products.
Overall, this study demonstrates that SDS exposure reduces GH secretion via oxidative stress and Nrf2 pathway suppression, potentially impacting endocrine function. Given SDS’s widespread presence in household products, further toxicological assessments and regulatory reviews are warranted to determine its safety for pediatric exposure.
Footnotes
Statements and declarations
Author Contributions
Hua Tang designed the study, conducted experiments, and wrote the manuscript. Lanlan Li supervised the study and contributed to manuscript revision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Clarify the use of animal-derived reagents
This study used fetal bovine serum (FBS) for cell culture. All reagents derived from animal sources were purchased from commercial suppliers following ethical procurement guidelines.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
