Abstract
Mercury (Hg) represents a ubiquitous environmental heavy metal that could lead to severe toxic effects in a variety of organs usually at a low level. The present study focused on the liver oxidative stress, one of the most important roles playing in Hg hepatotoxicity, by evaluation of different concentrations of mercuric chloride (HgCl2) administration. Moreover, the protective potential of curcumin against Hg hepatotoxic effects was also investigated. Eighty-four rats were randomly divided into six groups for a three-days experiment: control, dimethyl sulfoxide control, HgCl2 treatment (0.6, 1.2, and 2.4 mg kg−1 day−1), and curcumin pretreatment (100 mg kg−1 day−1) groups. Exposure of HgCl2 resulted in acute dose-dependent hepatotoxic effects. Administration of 2.4 mg kg−1 HgCl2 significantly elevated total Hg, nonprotein sulfhydryl, reactive oxygen species formation, malondialdehyde, apoptosis levels, serum lactate dehydrogenase, and alanine transaminase activities, with an impairment of superoxide dismutase and glutathione peroxidase in the liver. Moreover, HgCl2 treatment activated nuclear factor-E2-related factor 2-antioxidant response element (Nrf2-ARE) signaling pathway in further investigation, with a significant upregulation of Nrf2, heme oxygenase-1, and
Introduction
Mercury (Hg) is a naturally occurring metal that is considered as one of the most toxic metals with regard to human health. Hg usually occurs in three different chemical states in the environment, elemental or vapor, inorganic, and organic, all of which are toxic to animals and humans. 1 Large population is currently exposed to low levels of Hg owing to occupational exposure. Individuals involved in the disposal or recycling of Hg-contaminated wastes are also at risk of exposure. 2 Hg compounds always accumulate gradually in central nervous system or kidney in bodies of mammals, leading to Hg-induced neurotoxicity or nephrotoxicity, either. 3 –7 Both elemental and organic Hg are metabolized into inorganic Hg, mainly mercuric chloride (HgCl2), accumulates slowly in fundamental organs. 8 Although organic or inorganic Hg toxicity has been extensively studied, the molecular mechanisms remain not completely understood.
Liver is an important organ actively involved in many metabolic functions and is a frequent target for a number of toxicants. Owing to its role in bile production, liver has been implicated in the deposition and excretion of Hg. However, only a few studies focused on Hg-induced hepatotoxicity. 9 Oxidative stress, always accelerated by a combination of reactive oxygen species (ROS) overproduction and endogenous antioxidant capacity impairment, is widely accepted as one of the major mechanisms implicated in hepatotoxins-induced liver injuries. 10 –12 ROS overgeneration has been reported to play important roles mediating HgCl2-induced liver oxidative stress. 13 On the other hand, the interaction between the nuclear factor-E2-related factor 2 (Nrf2) and Kelchlike ECH-associating protein 1 (Keap 1) in cytoplasm would be disrupted upon oxidative stress. Nrf2 that translocate to nucleus interact with an antioxidant response element (ARE) to initiate the transcription of target genes, and their encoded proteins serve to detoxification and antioxidation. 14 Although Nrf2 signaling pathway has proved the existence of biological defense mechanisms contributing to antagonize exogenous or endogenous toxins-induced oxidative damage, 15 –18 the protective mechanisms have not yet been clarified, and only few studies focused on Nrf2 pathway activation under heavy metal-induced liver oxidative stress. 19,20
Since oxidative stress might play important roles in liver injury, the endogenous or exogenous antioxidants and their antioxidant mechanisms should be paid more attention to prevent Hg hepatotoxicity. Curcumin is a natural phenolic compound isolated as a yellow pigment from turmeric (
Taken together, oxidative stress may play important roles in Hg-induced hepatotoxicity. A rat model of different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2 exposure was established for investigation of Hg hepatotoxicity focusing on liver oxidative stress. Levels of apoptosis, lactate dehydrogenase (LDH), alanine transaminase (ALT), nonprotein sulfhydryl (NPSH), superoxide dismutase (SOD), glutathione peroxidase (GPx), ROS formation, malondialdehyde (MDA), as well as Nrf2-ARE signaling pathway activation were selected for investigating the toxic effects after different concentrations of HgCl2 exposure in the liver. In addition, as an exogenous antioxidant, curcumin was selected to verify whether it could antagonize Hg-induced hepatotoxicity via antioxidation abilities, which may be helpful for full evaluation of the Hg hepatotoxicity mechanisms.
Materials and methods
Chemicals
HgCl2 (≥97%) was purchased from Beijing Chemical Industry (Beijing, China). Curcumin (≥98%) was provided by Sigma (Saint Louis, Missouri, USA). Analysis kits of LDH, ALT, NPSH, SOD, GPx, and MDA were provided by Jiancheng Bio-engineering Institute (Nanjing, China). Dimethyl sulfoxide (DMSO), 2′,7′-dichloride fluorescent yellow double acetate (DCFH-DA), nicotinamide adenine dinucleotide (NAD), and Folin phenol reagent were obtained from Sigma. Annexin V-FITC/PI apoptosis detection kit was obtained from Beyotime Institute of Biotechnology (Nantong, China). RNAiso Plus, PrimeScript® RT reagent kit with gDNA Eraser, and SYBR® Premix Ex TaqTM II analysis kits were provided by TaKaRa Biotechnology Company (Dalian, China). Goat polyclonal primary antibodies developed against Nrf2, heme oxygenase-1 (HO-1),
Animals
Adult wistar rats with an initial body weight ranged from 160 g to 180 g were provided by Laboratory Animal Center of China Medical University (SPF grade,
Experimental design
Rats were randomly divided into six groups (control group, DMSO control group, HgCl2-treatment groups, and curcumin pretreatment group), with 14 animals (seven males and seven females) in each group. Rats in control and HgCl2-treatment groups were subcutaneously (s.c.) injected with saline; rats in DMSO control group were s.c. injected with DMSO; rats in curcumin pretreatment group were s.c. injected with 100 mg kg−1 curcumin. Curcumin was dissolved in DMSO before injection. Two hours later, rats in control and DMSO control groups were intraperitoneally (i.p.) injected with saline; rats in HgCl2-treatment groups were i.p. injected with 0.6, 1.2, or 2.4 mg kg−1 HgCl2 that dissolved in saline, respectively; while rats in curcumin pretreatment group were i.p. administered with 2.4 mg kg−1 HgCl2, either. These concentrations of HgCl2 were selected according to the previous studies. 28 All the pretreatment or HgCl2 administration were given every day at a volume of 5 mL kg−1, for up to 3 days.
Sample collections
At 24 h after the last injection, all rats in each group were killed via decapitation after anesthetized. First, for biochemical analysis, six rats (three males and three females) in each group, the blood was collected through the abdominal aorta to measure activities of LDH and ALT in serum. Hundred milligrams of the livers were digested by nitric acid for 12 h in order to determine total Hg levels. The remaining livers were prepared for 5% or 10% homogenate for determination of endogenous nonenzymatic or enzymatic antioxidants (NPSH, SOD, and GPx) and MDA levels. All the substance contents and enzyme activities were normalized to the protein amount, using bovine serum albumin as standard. Second, four rats (two males and two females) in each group, the livers were prepared for single-cell suspension in order to investigate the ROS levels and cell apoptosis. Third, four rats (two males and two females) that remained in each group, the total RNA and protein in the liver were extracted for the determination of Nrf2, HO-1,
Body weight and coefficients of liver
Body weight of all rats was recorded after killing by an excess dose of anesthetic ether and cervical dislocation. Livers were removed and weight was measured immediately after removal. The coefficients of liver weight to body weight were calculated as the ratio of tissue wet weight (g) to body weight (g). (Liver coefficient = liver wet weight/body weight ×100%.)
Determination of total Hg levels in liver
The Hg concentrations were detected with cold vapor atomic fluorescence spectrometry as described previously. 29 The liver tissue (100 mg) was digested with 2.0 mL of nitric acid for 12 h. Then 2.0 mL of 50% sulfuric acid and 3.0 mL of saturated potassium permanganate were added in, at 90°C for an hour. After cooling, the oxidant excess was reduced by the addition of 50% hydroxylamine. This solution was raised up to 10 mL, homogenized, then 1.0 mL was taken, to which 1.0 mL of dehydrated alcohol and 2.0 mL of 20% stannous chloride were added. Hg levels were analyzed using F732 Mercury Analyzer, Huaguang Instrument Factory of Shanghai, China (excitation/emission wavelength: 254 nm; zero: auto; measured by: peak height; blank: sulfuric acid/nitric acid, hydrochloric acid, potassium bromide/potassium bromate, hydroxylamine hydrochloride; detection limits: ≤0.05 μg L−1) immediately, via a standard calibration curve prepared by HgCl2. The instrument was calibrated with Hg standards prior to sample analysis. The levels of total Hg in the liver were expressed as microgram per gram wet liver tissue.
Measurement of LDH and ALT activities in serum
Activities of LDH and ALT in serum were analyzed with detection kits. LDH determination is based on the principle that LDH catalyzes the oxidation of NAD to Nicotinamide Adenine Dinucleotide Hydrogen (NADH). Briefly, NADH and 0.1% of pyruvate were added and the serum was incubated at 37°C for 15 min. The samples were then incubated with the coloring reagent at 37°C for 15 min. The reaction was stopped by the addition of 0.4 mol L−1 sodium hydroxide (NaOH). LDH activity was proportional to the increase in absorbance due to the reduction of NAD into NADH. The absorbance at 440 nm for LDH was read using a spectrophotometric assay kit type 722 (detection limits: 330–800 nm; measurement range of absorbance: 0–1.999A). LDH level (in percentage) was calculated as: (ASample − ABlank) (AControl − ABlank) × 100%. For ALT activity determination, 0.1 mL of serum was mixed with 0.5 mL of reaction matrix liquid, and the incubation was carried out at 37°C for 30 min. Then 0.5 mL of 2,4-dinitrobenzene hydrazine was added, followed by a 20 min-incubation at 37°C. The reaction was stopped by the addition of 0.4 mol L−1 NaOH. The absorbance at 505 nm for ALT was read using a spectrophotometric assay kit type 722 (detection limits: 330–800 nm; measurement range of absorbance: 0–1.999A). The values were expressed in units per liter of serum.
Evaluation of NPSH contents in liver
NPSH contents were measured with analysis kits in accordance with the 5, 5′-dithiobis-(2-nitrobenzoic acid) (DTNB) colorimetric method.
30
The liver tissue was homogenized to the concentration of 10% in phosphate-buffered saline (PBS). A mixture of 0.1 mL of the supernatant and 0.9 mL of 5% trichloroacetic acid was mixed and centrifuged (2300×
Measurement of enzymatic antioxidants in liver
SOD and GPx activities in the liver were measured with analysis kits, according to the manufacturer’s instructions. Briefly, SOD activity determination was based on the generation of superoxide radicals produced by xanthine and xanthine oxidase, which reacts with 2-(4-iodophenyl)-3-(4-nitrophenol)-5-phenyltetrazolium chloride, to form a red formazan dye. SOD activity was estimated by the degree of inhibition of the reaction. GPx activity was determined, based on the principle that GPx catalyzes the oxidation of glutathione by cumene hydroperoxide. In the presence of glutathione reductase and reduced nicotinamide adenine dinucleotide phosphate (NADPH), the oxidized glutathione could immediately convert to the reduced form with a contaminant oxidation of NADPH to NADP+. Absorbance was recorded at 560 nm for SOD and 340 nm for GPx, respectively, using a spectrophotometric assay kit type 722 (detection limits: 330–800 nm; measurement range of absorbance: 0–1.999A). The values were expressed in units per milligram of protein. The protein determination was performed according to the procedure of Lowry et al. using bovine serum albumin as the standard.
Quantification of ROS formation in liver
The liver cell suspension was prepared according to the method described by Villalba et al. 31 ROS formation was evaluated with a modified method reported previously, 32 which was monitored using the oxidation-sensitive fluoroprobe DCFH-DA, a nonfluorescent compound that is freely taken up into cells. DCFH-DA reveals the intracellular production of redox-active substances and has been used widely to investigate oxidative damage in intact cells. It is a nonfluorescent lipophilic ester that easily crosses the plasma membrane into the cytosol. The acetate group is rapidly removed by unspecific esterases. When DCFH-DA was deacetylated and oxidized to form the fluorescent compound, fluorescence intensity was increased in a concentration-dependent manner. In the present study, cells (1 × 106 mL−1) were incubated with 10 μM DCFH-DA at 37°C for 30 min. The fluorescence increase, which is due to the hydrolysis of DCFH-DA to DCF by nonspecific cellular esterase and its subsequent oxidation by peroxides, was measured by flow cytometry using a FACScan flow cytometer (Becton-Dickinson, Germany) with an excitation wavelength of 488 nm and emission wavelength of 525 nm. The amount of intracellular ROS was proportional to the intensity of DCF fluorescence, and the fluorescence intensity was recorded directly to indicate the relative amount of ROS. The ROS levels were normalized against the control group.
Determination of MDA contents in liver
MDA is a by-product of the oxidative degradation of cell membrane lipids and serves as an index of the intensity of oxidative stress. It was measured as the production of lipid peroxide according to a modified method previously described,
33
based on the principle that MDA could combine with thiobarbituric acid (TBA) to form a pink chromogen compound. In brief, 10% liver homogenate was mixed with the reaction mixture consisting of 0.1 M ice-cold PBS and 0.1 M ferrous chloride in a total volume of 1.0 mL (pH 7.4). The reaction was then stopped by 1.0 mL of 10% trichloroacetic acid, followed by 1.0 mL of 0.67% TBA. After centrifugation at 2500×
Apoptosis analysis
The apoptosis levels were measured by flow cytometry according to Annexin V-FITC/PI double-stained method. 34 Annexin V is a calcium-dependent phospholipids-binding protein that has a high affinity for phosphatidylserine; Annexin V-FITC is a sensitive probe for identifying cells that are undergoing apoptosis, while propidium iodide (PI) is a nonspecific DNA dye that is excluded from living cells with intact plasma membranes but incorporated into the nonviable cell. The liver cells (1 × 106 cells mL−1) of treated rats were collected, resuspended in 200 μL ice-cold binding buffer, and 5 μL Annexin V-FITC and 5 μL PI were added. Untreated cells were washed twice with PBS, collected, resuspended in 200 μL binding buffer, and 5 μL Annexin V-FITC and 5 μL PI were added, which served as the control. After 15-min incubation in darkness, the apoptosis rate was determined immediately. The data was quantified and analyzed using a FACSCalibur flow cytometer (BD Biosciences, Franklin Lakes, New Jersey, USA). Discrimination of cells was performed as the early apoptotic cells (Annexin V+/PI−).
Quantitative real-time RT-PCR for Nrf2, HO-1, and γ -GCSh mRNA expression
For total RNA isolation, the liver tissues were homogenized in RNAiso Plus, and the total RNA was isolated according to the method as previously described.
35
The absorbance of the RNA solution was determined using NanoPhotometer (IMPIEN, Germany) at 260 and 280 nm, respectively. OD260/OD280 ratio was between 1.6 and 1.8, respectively. The first-strand of cDNA was synthesized from 1 μg of total RNA by reverse transcriptase performed on Applied Biosystems 2720 Thermal Cycler (Applied Biosystems, Singapore) with PrimeScript RT reagent Kit. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed in SYBR
Primer sequences used for the amplification of each gene in the present study.a
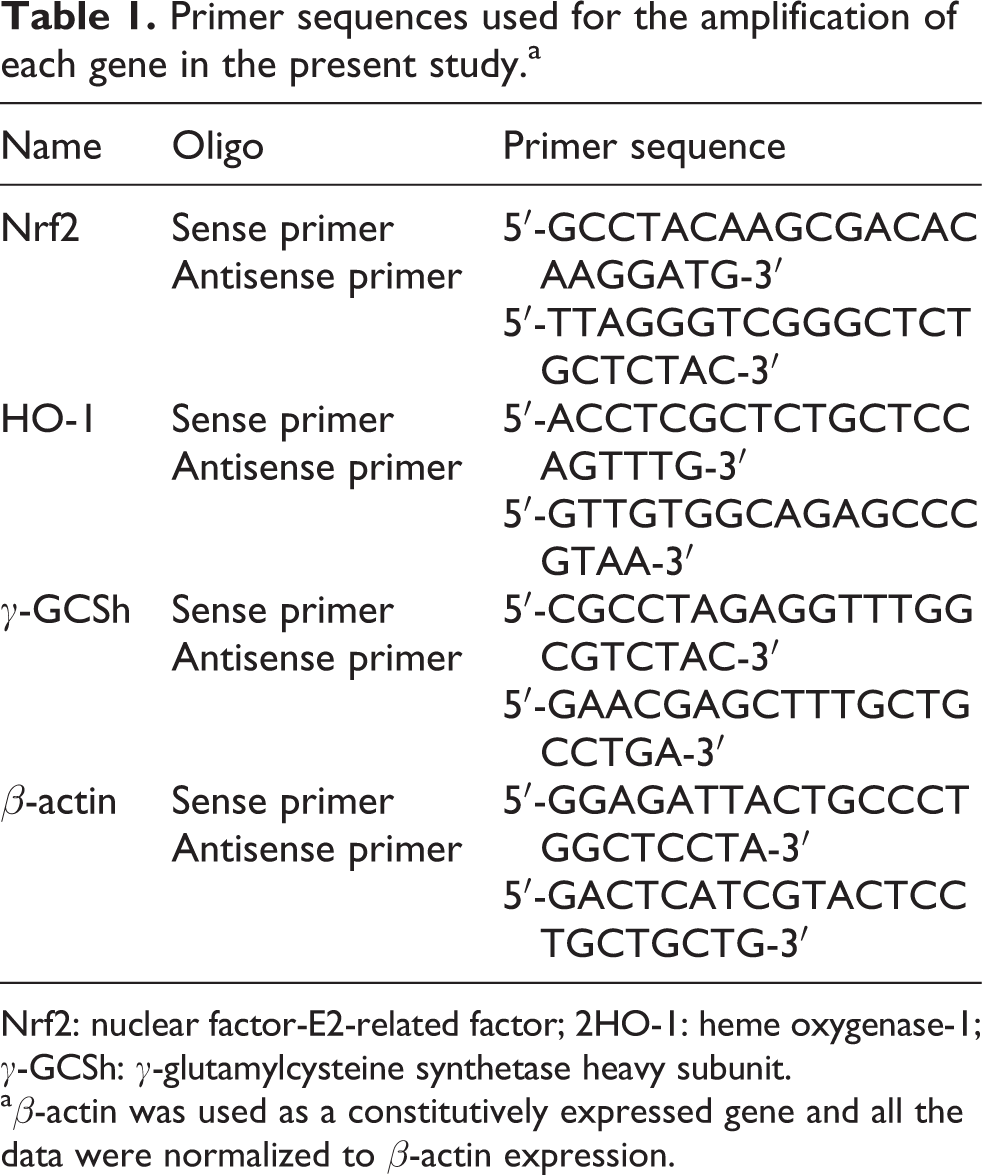
Nrf2: nuclear factor-E2-related factor; 2HO-1: heme oxygenase-1;
a
Western blotting for Nrf2, HO-1, and γ -GCSh protein expression
Protein extraction and immunoblot analysis were conducted as described by Guerguerian et al.
36
Total protein was extracted from the liver tissue with radioimmunoprecipitation assay buffer containing protease inhibitors at 4°C. Protein concentrations were quantified with the bicinchoninic acid assay reagent. Equal amounts of protein (30 μg per lane) was separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, Massachusetts, USA). PVDF membranes were subsequently blocked overnight at 4°C in Tween 20 Tris-buffered saline (TBST) containing 5% nonfat powdered milk, followed by briefly rinsed in TBST and incubated with Nrf2 (1:300), HO-1 (1:300),
Statistical analysis
SPSS for Windows, version 16.0, was used for statistical analysis. Results were expressed as mean ± standard deviation. Differences among the means were determined with one-way analysis of variance followed by a Student–Newman–Keuls test for multiple comparisons. The difference at either
Results
Behavioral observation and liver weight coefficient
Throughout the experiment, there were no behavioral changes observed in any experimental group and no animal showed any unusual response. In addition, no significant alteration was found in the liver weight coefficients among all the treatment groups (Figure 1).
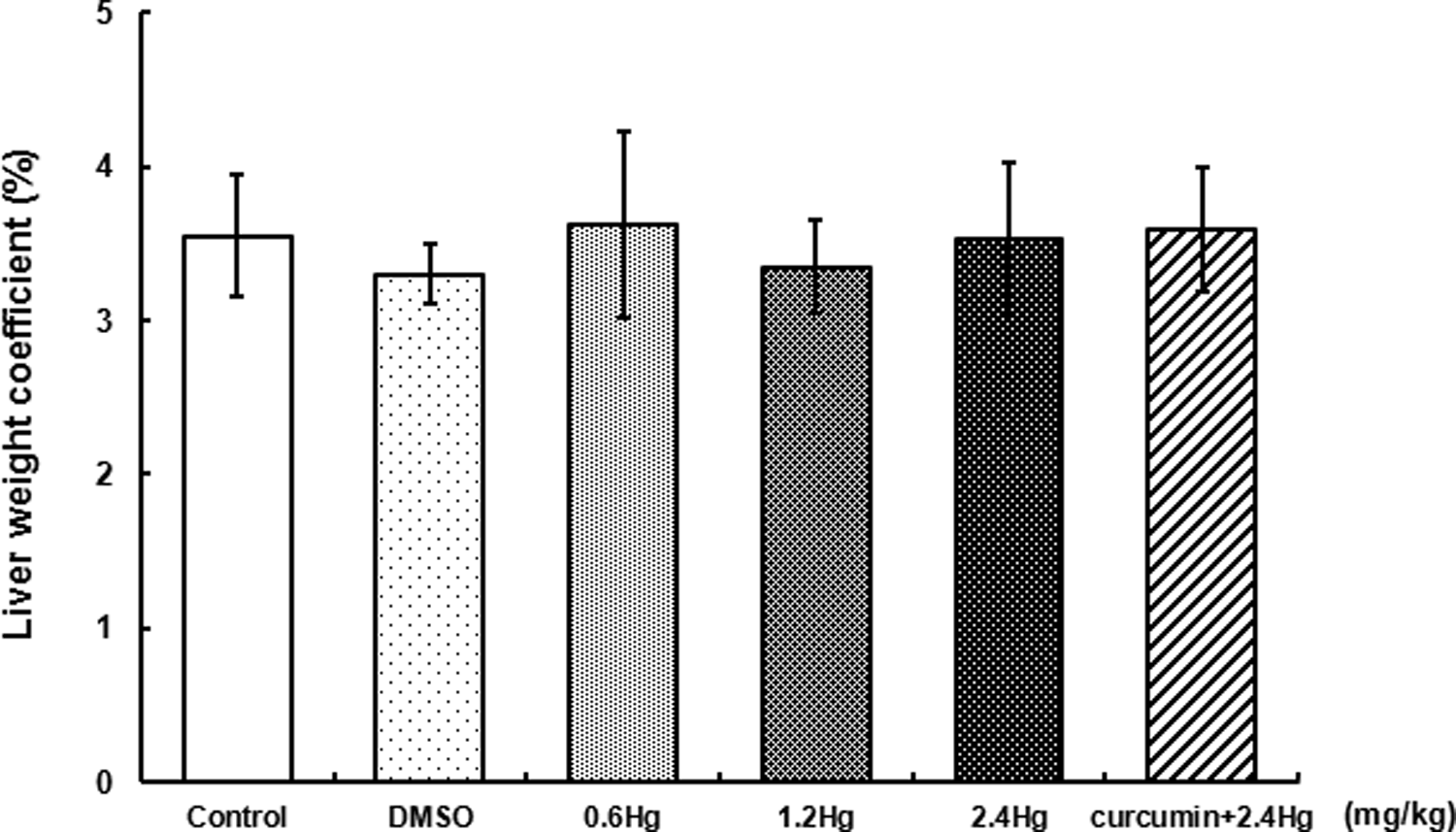
Effects of HgCl2 exposure and curcumin pretreatment on liver weight coefficient. Notes: Rats were administrated with different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2, or pretreated with curcumin (100 mg kg−1) followed by 2.4 mg kg−1 HgCl2 exposure. Data were presented as mean ± SD. of 14 animals in each group and analyzed by one-way ANOVA. HgCl2: mercuric chloride; ANOVA: analysis of variance.
Total Hg levels in liver with HgCl2 exposure or curcumin pretreatment
Total Hg levels in the liver tissue were measured after three days of HgCl2 treatment or curcumin pretreatment. As shown in Figure 2, there was a concentration-dependent elevation of the liver Hg levels after different dosages of HgCl2 exposure, which were significantly higher than those in control (
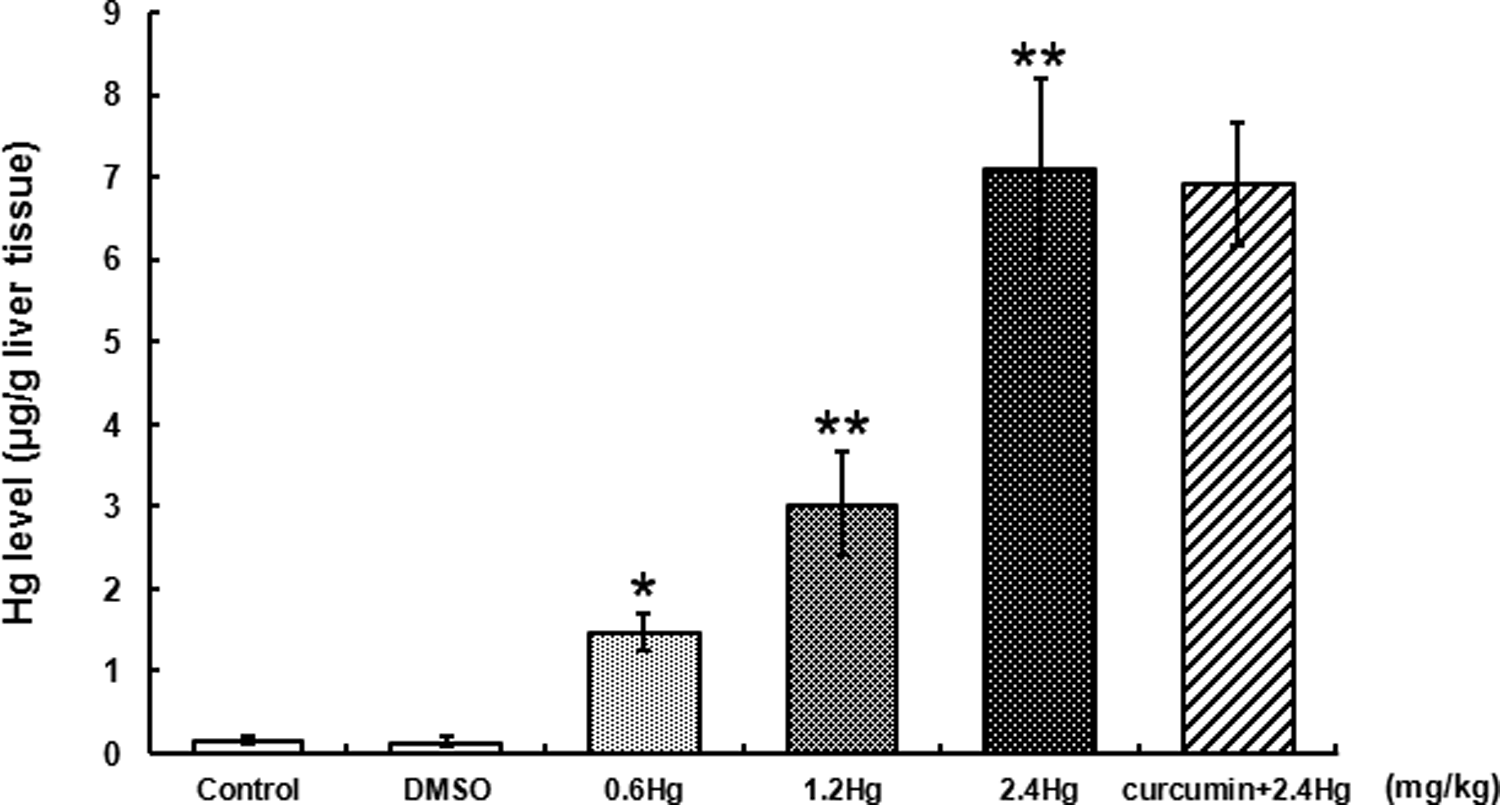
Effects of HgCl2 exposure and curcumin pretreatment on total Hg levels in liver. Notes: Rats were administrated with different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2, or pretreated with curcumin (100 mg kg−1) followed by 2.4 mg kg−1 HgCl2 exposure. Data were expressed as mean ± SD of six independent experiments for each treatment group and analyzed by one-way ANOVA. Significant difference was defined as *
LDH and ALT activities in serum with HgCl2 exposure or curcumin pretreatment
LDH and ALT activities were selected as two enzymes that could reflect the function of liver in the present study. Based on the findings of Figure 3(a) and (b), compared to those in control, HgCl2 administration resulted in LDH and ALT activities elevated concentration, dependently and significantly, especially in the 1.2 mg kg−1 (1.8-fold, 3.4-fold,
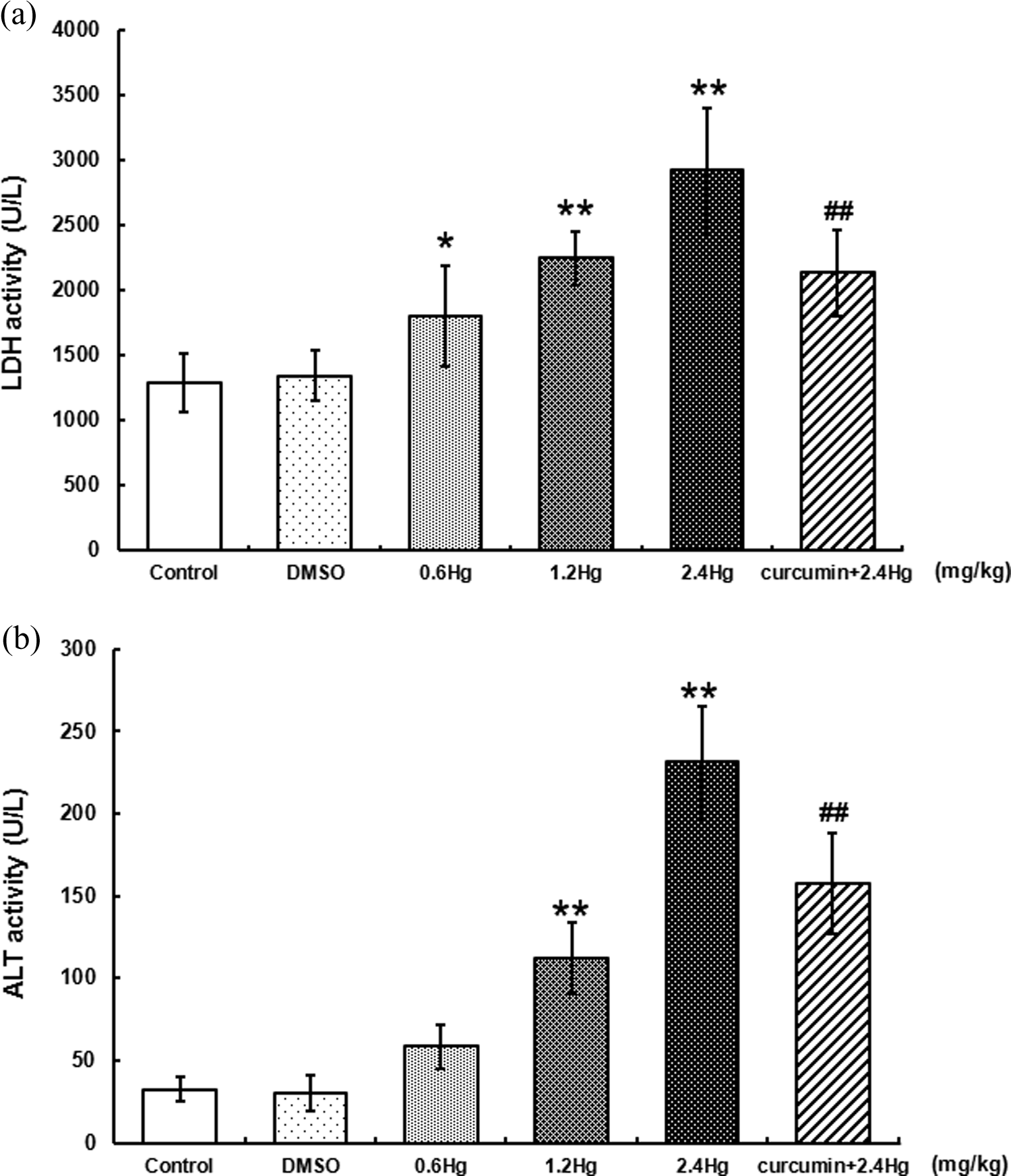
Effects of HgCl2 exposure and curcumin pretreatment on LDH (Figure 3(a)) and ALT (Figure 3(b)) activities in serum. Notes: Rats were administrated with different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2, or pretreated with curcumin (100 mg kg−1) followed by 2.4 mg kg−1 HgCl2 exposure. Data were expressed as mean ± SD of six independent experiments for each treatment group and analyzed by one-way ANOVA. Significant difference was defined as *
NPSH contents in liver with HgCl2 exposure or curcumin pretreatment
NPSH contents were determined as one of the primary endogenous nonenzymatic antioxidants in the present study. As shown in Figure 4(a), HgCl2 exposure resulted in an elevation of NPSH contents, which was represented as a dose-dependent manner. Compared to that in control, a significant increase in NPSH contents was observed (36.4%,
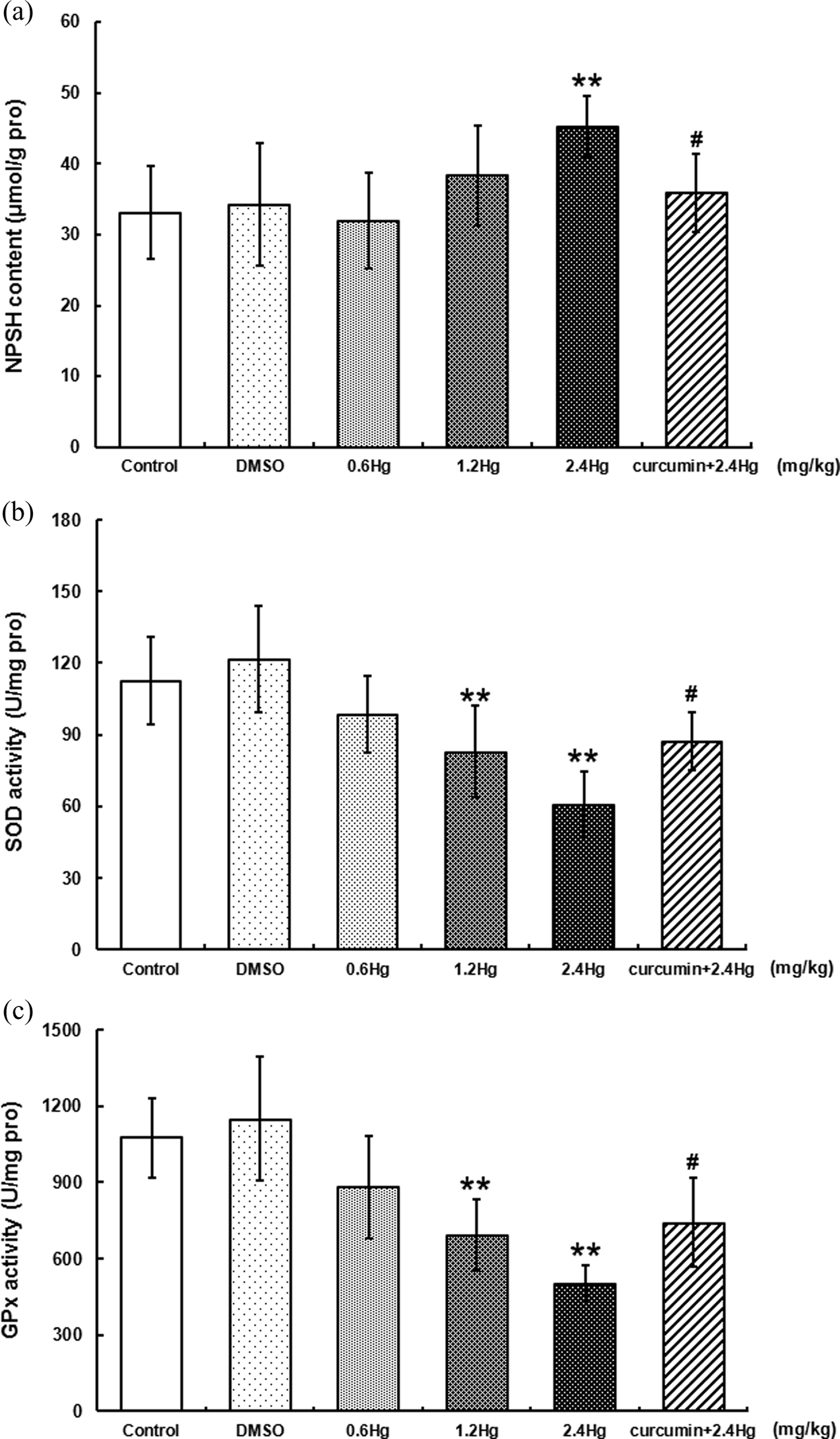
Effects of HgCl2 exposure and curcumin pretreatment on endogenous nonenzymatic (NPSH, Figure 4(a)) or enzymatic antioxidants (SOD and GPx, Figure 4(b) and (c)) levels in liver. Notes: Rats were administrated with different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2, or pretreated with curcumin (100 mg kg−1) followed by 2.4 mg kg−1 HgCl2 exposure. Data were expressed as mean ± SD of six independent experiments for each treatment group, and analyzed by One-way ANOVA. Significant difference was defined as **
Activities of enzymatic antioxidants in liver with HgCl2 exposure or curcumin pretreatment
SOD and GPx were determined as two important endogenous antioxidant enzymes in the liver. As represented in Figure 4(b) and (c), HgCl2 exposure resulted in a dose-dependent decrease in either SOD or GPx activity. Compared to those in control, the significant decrease in SOD and GPx was observed both in the 1.2 mg kg−1 (26.4%, 35.4%,
ROS formation in liver cells with HgCl2 exposure or curcumin pretreatment
ROS formation was detected to investigate the liver oxidative stress induced by HgCl2 administration. As shown in Figure 5, compared with the control, HgCl2 exposure resulted in a dose-dependent elevation of ROS formation in the liver cells, the significant increase was observed in all dosages of HgCl2-treated groups, 1.5-fold (
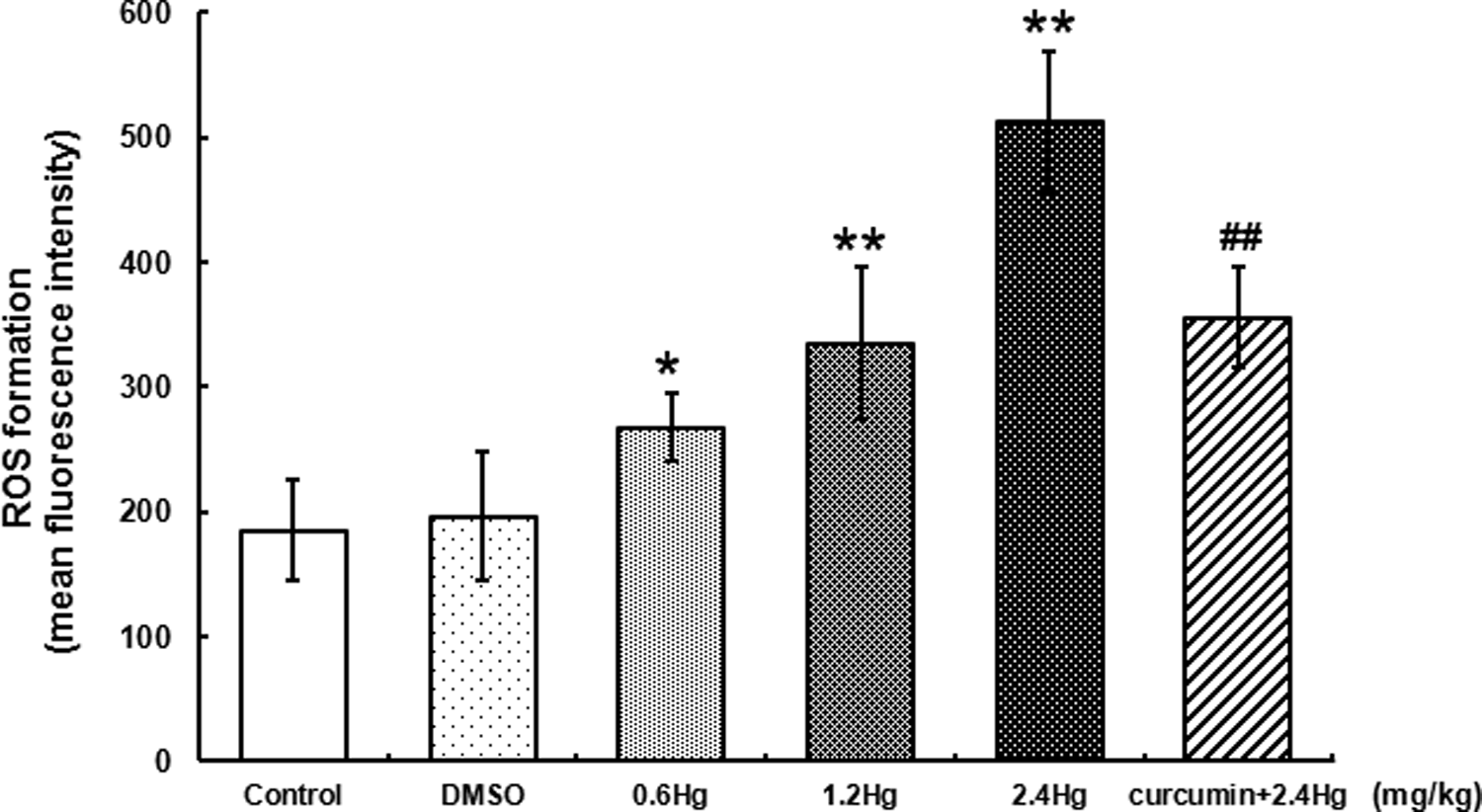
Effects of HgCl2 exposure and curcumin pretreatment on ROS formation in liver cells. Notes: Rats were administrated with different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2, or pretreated with curcumin (100 mg kg−1) followed by 2.4 mg kg−1 HgCl2 exposure. Data were expressed as mean ± SD of four independent experiments for each treatment group, and analyzed by One-way ANOVA. Significant difference was defined as *
MDA contents in liver with HgCl2 exposure or curcumin pretreatment
MDA contents were measured as a representative production of lipid peroxidation in the liver. Here, as shown in Figure 6, HgCl2 exposure resulted in a dose-dependent increase in MDA contents, relative to control. The significant elevation was observed in the 1.2 and 2.4 mg kg−1 HgCl2-treated groups, 1.5-fold (
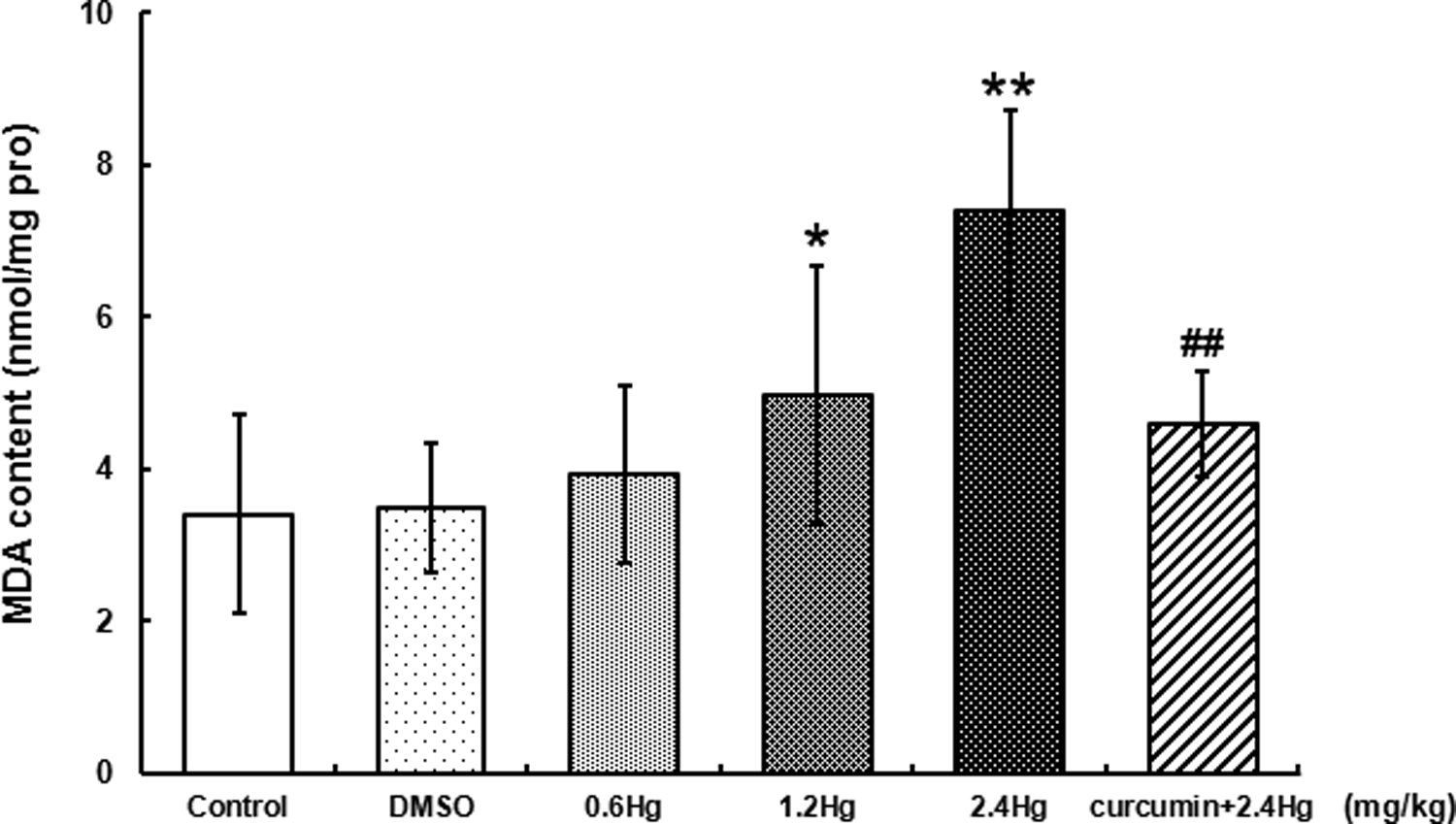
Effects of HgCl2 exposure and curcumin pretreatment on MDA contents in liver. Notes: Rats were administrated with different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2 or pretreated with curcumin (100 mg kg−1) followed by 2.4 mg kg−1 HgCl2 exposure. Data were expressed as mean ± SD of six independent experiments for each treatment group, and analyzed by one-way ANOVA. Significant difference was defined as *
Apoptosis levels in liver cells with HgCl2 exposure or curcumin pretreatment
The apoptosis of the liver cells were analyzed by flow cytometry with double staining of Annexin V-FITC/PI, the results are shown in Figure 7. On account of the present study, there was a dose-dependent increase in apoptosis levels with different concentrations of HgCl2 exposure, the percentage of early apoptotic cells (Q4 quadrant in Figure 7(a)) treated with 2.4 mg kg−1 HgCl2 (48.5 ± 4.9%) was markedly higher than those in the control group (4.1 ± 1.2%,
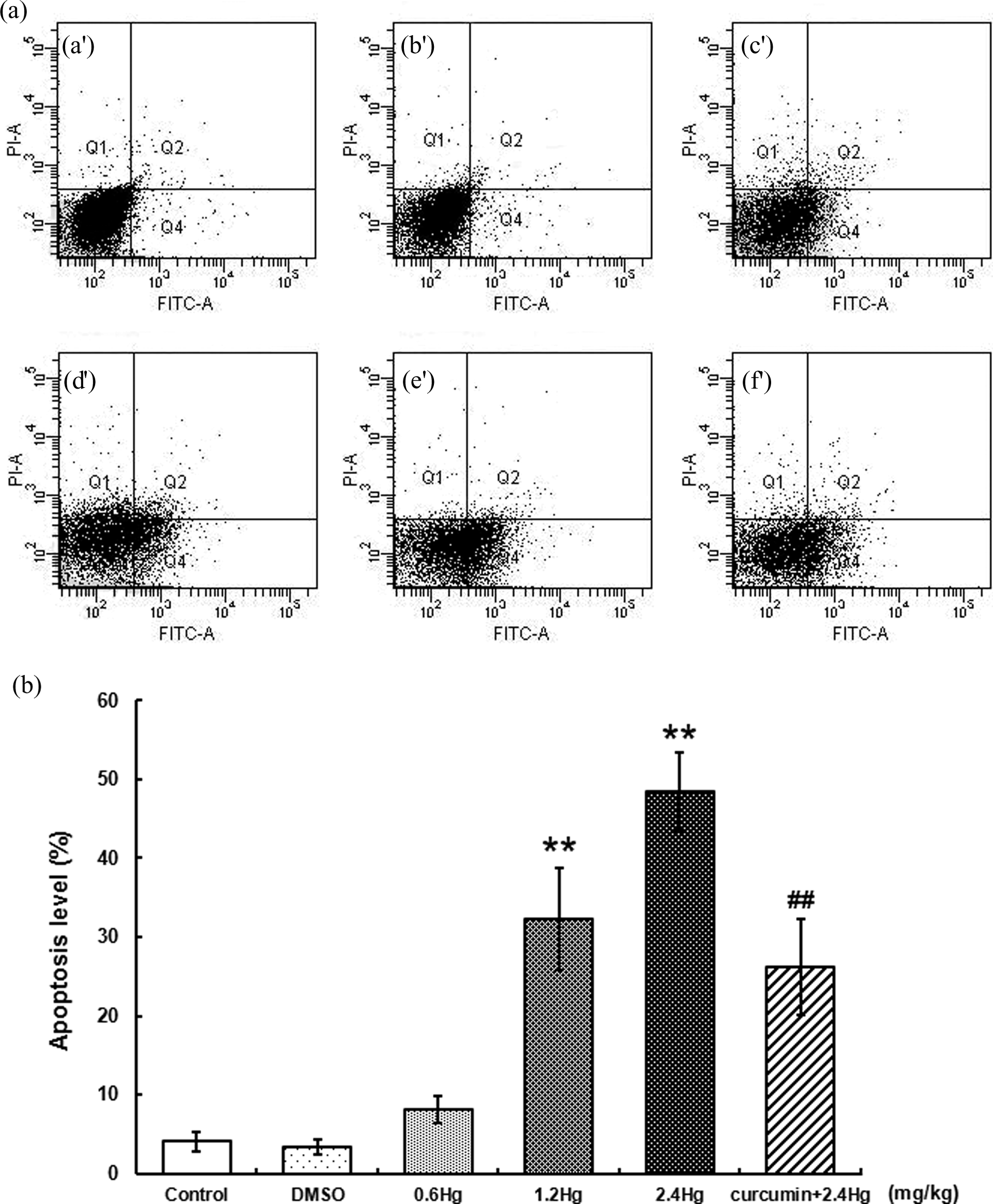
Effects of HgCl2 exposure and curcumin pretreatment on apoptosis levels of liver cells. Notes: Rats were administrated with different concentrations (0, 0.6, 1.2, and 2.4 mg kg−1) of HgCl2, or pretreated with curcumin (100 mg kg−1) followed by 2.4 mg kg−1 HgCl2 exposure. The single positive populations in Q4 quadrant of Figure 7(a) were considered as the early apoptotic cells (Annexin V+/PI−). (a) control; (b) DMSO; (c) 0.6 mg kg−1 HgCl2; (d) 1.2 mg kg−1 HgCl2; (e) 2.4 mg kg−1 HgCl2; (f) 100 mg kg−1 curcumin + 2.4 mg kg−1 HgCl2. Quantitative analysis of early apoptosis with flow cytometry has been shown in Figure 7(b). Values were expressed as mean ± SD of four independent experiments for each treatment group and analyzed by one-way ANOVA. Significant difference was defined as **
Nrf2, HO-1, and γ -GCSh mRNA expressions with HgCl2 exposure or curcumin pretreatment
qRT-PCR was performed for Nrf2, HO-1, and
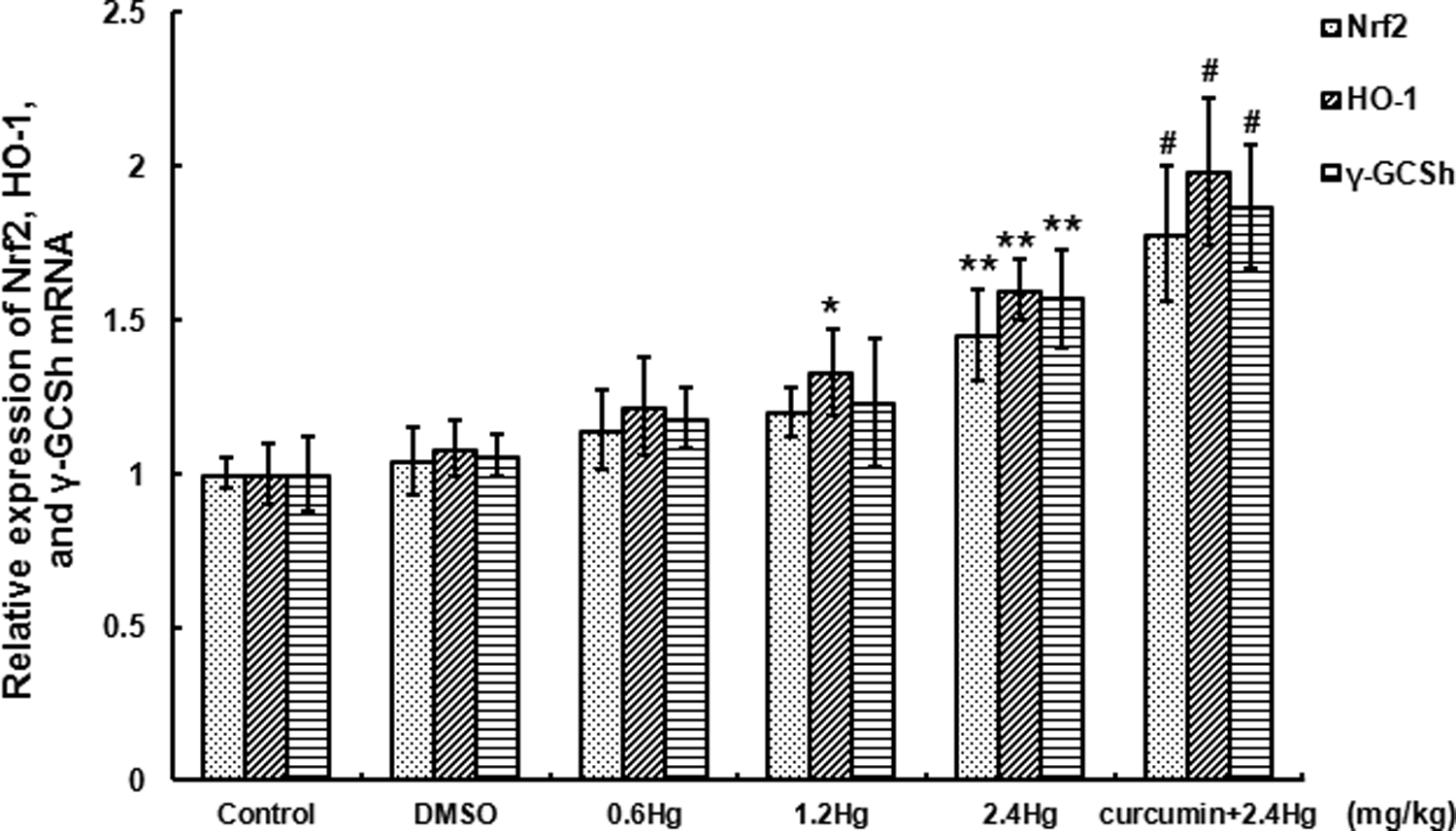
Effects of HgCl2 exposure and curcumin pretreatment on Nrf2, HO-1, and
Expressions of Nrf2, HO-1, and γ -GCSh protein with HgCl2 exposure or curcumin pretreatment
Western blot analysis for Nrf2, HO-1, and
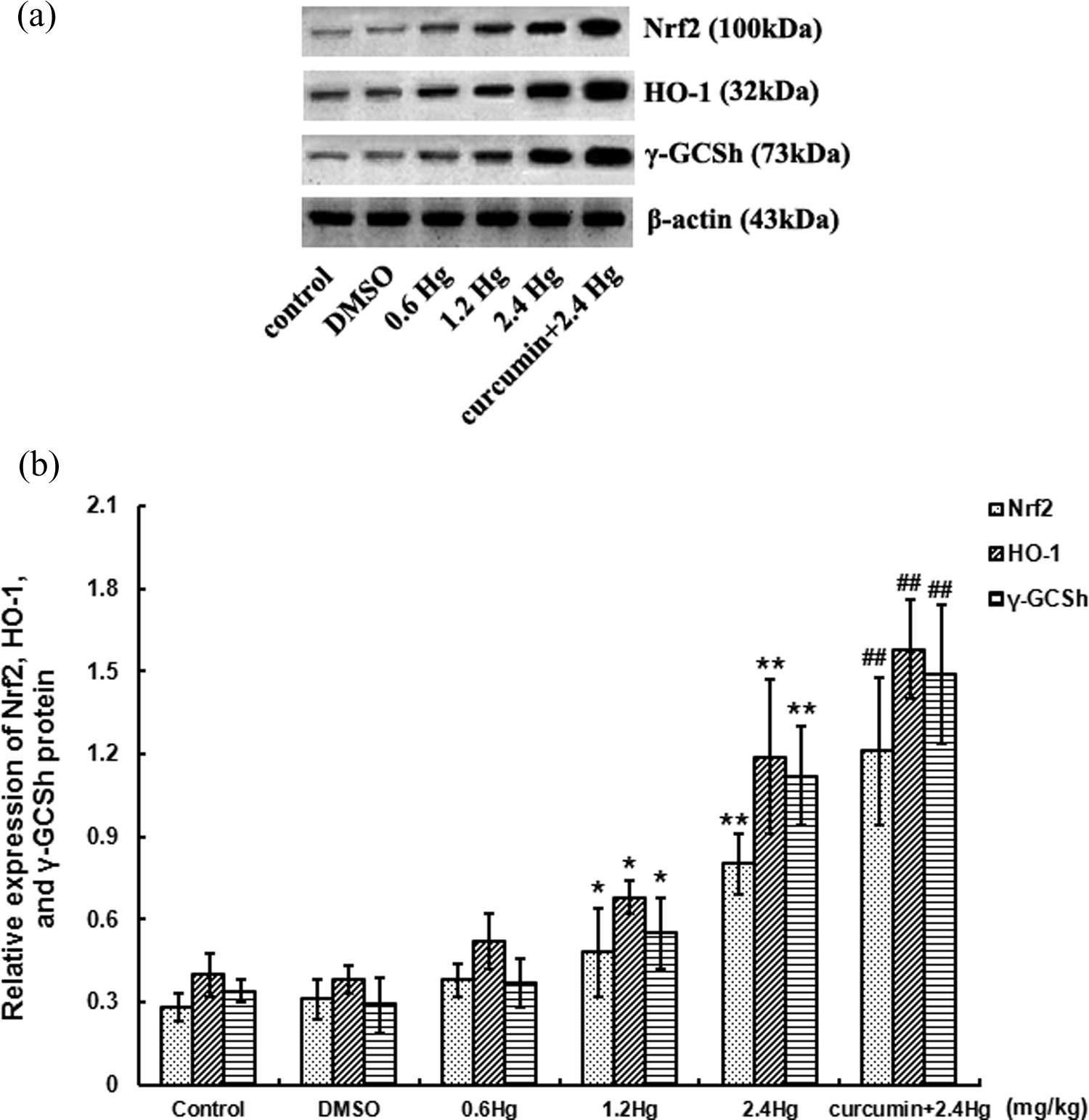
Effects of HgCl2 exposure and curcumin pretreatment on Nrf2, HO-1, and
Discussion
Hg is a ubiquitous highly hazardous environmental pollutant. Overloading Hg pollution resulted from anthropogenic activities or industrialization could cause severe Hg poisoning around the world, such as the “Minamata disease” in Japan, or Songhua River Hg pollution in China. 37 Based on the “National Standard of the People’s Republic of China Sanitary Standard for Drinking Water” (GB 5749-2006), the quality standard for drinking water is 0.0001 mg L−1. In the present study, we selected the concentrations of 0.6, 1.2, and 2.4 mg kg−1 HgCl2 administration at the volume of 5 mL kg−1. These concentrations were chosen, according to previous studies, 28 to keep the minimum number of rats that were used and effectively evaluate Hg hepatotoxicity. In the meantime, there was no gender difference between rats in Hg hepatotoxicity in the concentrations of 0.6–2.4 mg kg−1 for 3 days’ treatment in our preliminary experiments (data not shown).
It has been found that inorganic Hg undergoes extensive biliary-hepatic cycling. Hg secreted into bile and partly reabsorbed into the circulatory system and then returned to the liver. 38 In the present study, it was found that Hg gradually accumulated in the liver with the elevation of HgCl2 concentration. This characteristic of Hg accumulation in the liver might be related to the high affinity between Hg2+ and thiol groups. The Hg–S compounds would go across endothelial cells into the blood and be reabsorbed by the liver. The results of cell apoptosis showed that HgCl2 exposure resulted in apoptosis occurred in hepatocytes in a dose-dependent manner, which verified that HgCl2 caused appreciable cellular injury and apoptosis in the liver. LDH and ALT were determined for investigation of HgCl2-induced hepatic dysfunction. The serum LDH activity is usually selected as a diagnostic indicator under many pathological conditions. 39 Here, a dose-dependent elevation of LDH activity in serum was observed after HgCl2 treatment. This was consistent with the previous findings that an increase in the activity of LDH in serum was always due to the leakage of enzyme from tissues after injury. 40 An elevation of serum ALT activity always seemed as a sensitive indicator of the cytoplasm or mitochondrial membranes injury. 41 In this study, ALT activities were significantly elevated after HgCl2 exposure, especially in the 2.4 mg kg−1 HgCl2-treated group, indicating an obvious hepatic dysfunction under Hg exposure. Results of LDH and ALT activities confirmed liver dysfunction, tissue injury, apoptosis, and necrosis after HgCl2 exposure.
As one of the strongest thiol-binding agents, Hg2+ specifically binds to thiols in cells. 42 The direct chemical interactions among Hg2+ and thiol groups from proteins or nonprotein molecules might play a crucial role in Hg-induced hepatotoxicity, resulting in the impairments of endogenous enzymatic and nonenzymatic antioxidants. NPSH acts as a nucleophilic scavenger of numerous compounds and their metabolites via enzymatic and chemical mechanisms and plays important roles in the protection against oxidative stress caused by ROS. Around 90% of NPSH is glutathione (GSH), which is the most abundant intracellular low molecular weight sulfhydryl compound. 43 GSH may combine with Hg2+ directly and form a complex that prevents Hg2+ from binding to cellular proteins. In addition, GSH could react with free radicals, remove the excessive ROS that generated by oxidants. In the present study, an increased level of NPSH was observed after HgCl2 administration. This might be caused by acute stress reaction and/or the accumulating complexes of Hg-GSH, which was different to previous data that showed NPSH depletion after Hg exposure. 44 Similar results have been reported previously. 28,45 It could be also illustrated that NPSH might act as the first line of defense via binding Hg to serve to antagonize ROS-induced oxidative stress in the liver. SOD and GPx are two important endogenous antioxidant enzymes. SOD always plays in maintaining the balance between oxidation and antioxidation systems through converting the superoxide anion into hydrogen peroxide and hydrogen. GPx is the only antioxidative enzyme that contains selenium in organisms, defending the cell against oxidative stress or damages. 46 The present study indicated that HgCl2 dose-dependently inhibited activities of SOD and GPx. These impairments might aggravate oxidative stress in the liver, leading to the increase in ROS level, which provoke lipid, protein, and DNA oxidation.
Oxidative stress, always accelerated by a combination of endogenous antioxidant-capacity impairment and ROS overproduction, might play vital roles in Hg hepatotoxicity. Mitochondrial was considered as the main intracellular site for ROS generation. Excessive ROS formation may disrupt energy metabolism and impair activities of enzymes that play critical roles in antagonizing oxidative damage. 47 ROS can be scavenged by endogenous nonenzymatic or enzymatic antioxidant systems. When the balance between ROS production and antioxidant defense system is broken, oxidative stress would happen. In the present study, HgCl2 exposure resulted in a significant and dose-dependent elevation of ROS levels in the liver, indicating that the mitochondrial ROS overproduction as well as endogenous antioxidant system deficiency together contributed to the high level of ROS accumulation. Excessive ROS could impair intracellular ionic homeostasis by damaging cellular macromolecules (lipoid, protein, and nucleic acid). MDA is a terminal product of polyunsaturated fatty acid peroxidation, which is often used as a marker of lipid peroxidation. 48 MDA levels after HgCl2 exposure in the present study increased obviously and dose-dependently, indicating a severe lipid peroxidation in the liver owing to ROS-induced oxidative stress. Enhanced lipid peroxidation levels were also reported in Hg toxicity by Agarwal et al. 49 Moreover, excessive ROS can attack mitochondria, calcium, and other apoptosis-related factors would be released from injured mitochondria into the cytoplasm that regulates cell apoptosis. 50
The activation of Nrf2 signaling pathway has been recently thought to be functional as a protective factor against inorganic Hg or methylmercury toxicity.
51,52
However, the protective mechanisms against Hg hepatotoxicity were poorly understood. Once oxidative stress arises, Nrf2 was triggered to dissociate from Keap 1 followed by translocation into the nucleus, where it interacts with ARE to initiate the transcription of target genes and their encoded proteins. On account of the present study, Nrf2 expression upregulated in a concentration-dependent manner both in mRNA and protein levels after HgCl2 exposure, the protein level in the 2.4 mg kg−1 HgCl2 subgroup was almost fourfold of the control. This indicated that HgCl2 could activate the dissociation of Nrf2 from Keap 1 protein, resulting in the induction of cytoprotective proteins. This Nrf2 activation might closely relate to HgCl2-induced liver oxidative stress. In addition, we speculate the combination of Hg2+ and Keap 1 protein thiol, as well as disruption of the thiol groups via oxidative modifications that might contribute to the upregulation of Nrf2 expression. However, it is not yet possible to determine whether the oxidative modifications of Nrf2 and Keap 1 protein thiol were represented as a direct toxic effect of Hg or whether they were represented as a consequence of the pro-oxidative effects of ROS overproduction. Additional findings showed a dose-dependent upregulation of HO-1 and
Curcumin, the bioactive component of spice turmeric and curry, is a lipophilic compound that has strong free radical scavenging potentials. 53,54 Over the past decade, the properties of curcumin concerning antioxidants have been extensively studied. The outstanding antioxidant potential of curcumin is impressive due to its free radical scavenging activity and metal binding property associated with the presence of phenolic and methoxy groups. Since oxidative stress is one of the most important mechanisms mediating Hg-induced liver injury, it is therefore reasonable to presume that curcumin might play some antagonistic roles against HgCl2-induced in vivo hepatotoxicity. In the present study, we found that curcumin pretreatment relieved the HgCl2-induced liver injury in rats. Our results showed that HgCl2 hepatotoxicity, reflected by elevated serum LDH and ALT activities, as well as cell apoptosis, were significantly blocked by curcumin pretreatment. However, the liver Hg level was not obviously reduced by curcumin, indicating that curcumin protects the hepatic function not by counteracting the liver Hg accumulation, but through other protective mechanisms. Further investigation showed that curcumin pretreatment significantly attenuated the liver oxidative stress in rats. Our results observed that the disturbance of GSH balance was consistently reversed by curcumin pretreatment. In addition, the antioxidant capacity deficiency of SOD and GPx caused by HgCl2 was obviously recovered by curcumin, suggesting that curcumin has the protective abilities against Hg-induced endogenous nonenzymatic and enzymatic antioxidant system disruptions, which is helpful to maintain the liver redox homeostasis. Furthermore, ROS level was significantly decreased with curcumin pretreatment when compared to the 2.4 mg kg−1 HgCl2-treated group, which supported that curcumin exerts its antioxidative function partly through the ability of ROS scavenging. The present data was similar to the previous literature that indicated the antioxidant abilities of curcumin to prevent ROS accumulation. 55 As a typical indicator of ROS-mediated lipid peroxidation, the hepatic MDA levels in our study were markedly blocked by curcumin pretreatment versus the 2.4 mg kg−1 HgCl2-treated rats, which supported the ability of curcumin against ROS-induced lipid peroxidation in the liver.
Interestingly, on the other hand, it has been further proved in recent years that an underlying mechanism through which curcumin exerts its antioxidant properties is the Nrf2-ARE pathway activation, promoting the induction of phase II detoxifying and antioxidant enzymes. Previous in vivo studies showed that curcumin supplement could upregulate the expression of various Nrf2 downstream genes encoding detoxifying and antioxidant enzymes, such as glutathione S-transferase, glutathione reductase, quinone oxidoreductase 1, thioredoxin, and HO-1 in the liver and kidney tissues of mice.
56,57
In addition, pretreatment with curcumin has been reported to effectively antagonize hexavalent chromium-induced nephrotoxicity, carbon tetrachloride-induced liver injury, and arsenic-induced neuronal oxidative damage via Nrf2 signaling pathway activation.
53,58,59
In the present study, we investigated the involvement of Nrf2 signaling pathway activation by curcumin in antagonizing HgCl2-induced hepatotoxicity. The results showed that both Nrf2 mRNA and protein, and two typical Nrf2 downstream target genes encoding proteins, HO-1 and
Conclusion
The present study established a rat model of acute HgCl2 poisoning, which provided substantial evidence suggested that oxidative stress plays important roles mediating Hg hepatotoxicity. Additionally, this study also provided a novel mechanistic insight for Nrf2 signaling activation of curcumin to antagonize Hg-induced hepatotoxicity, for deeply exploring the mechanisms of Hg toxicity. In addition, our study may therefore suggest a potential useful chemopreventive dietary component to counteract the occupational Hg poisoning. Moreover, the hepatoprotective roles of curcumin against Hg poisoning will be carried out in an extensive research in our future studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Program for Liaoning Innovative Research Team in University (no. LT2015028).
