Abstract
Macrolide antibiotics have been shown to protect airway epithelial cells and macrophages from sulfur mustard (SM)–induced cytotoxicity. In the current study, the efficacy of roxithromycin in ameliorating SM-induced respiratory injury was further evaluated in a rat model. Anesthetized rats (N = 8/group) were intratracheally exposed to SM by vapor inhalation. For the drug treatment groups, rats were orally given 10, 20, or 40 mg/kg roxithromycin one hr prior to exposure and every twenty-four hr thereafter. After one, three, or seven days of treatment, sections of the lung were examined and scored for histopathological parameters. Treatment with roxithromycin ameliorated many of the symptoms caused by SM in some animals. In particular, treatment at 40 mg/kg for three days showed significant improvements (p < .05) over the untreated group. When the evaluation was focused on trachea, treatment with roxithromycin for three days showed a trend of dose-dependent protection; moreover, the groups treated with 20 or 40 mg/kg of roxithromycin were statistically different (p < .001 and p < .05, respectively) from the untreated group. These results suggest that roxithromycin protects against some damages associated with SM injury in the lung, particularly in the upper respiratory tract.
Introduction
Sulfur mustard (2,2’-dichlorodiethyl sulfide, SM) is a chemical warfare agent that has been used numerous times in history since World War I, causing injuries and deaths to a large number of victims (Szinicz 2005; Kehe and Szinicz 2005). Being the major chemical warfare agent during World War II, SM was produced and stockpiled by many countries and is probably still the most distributed chemical warfare agent in the world (Szinicz 2005). Due to the ready availability, ease of production and storage, as well as persistence and stability, SM also represents a potential terrorist agent that poses a significant threat to civilians as well.
Exposure to SM causes blistering of the skin as well as damage to the eyes and the respiratory tract. Respiratory damage due to SM inhalation has been found to be dose dependent (Eisenmenger et al. 1991; Papirmeister et al. 1991). At low to moderate doses, the upper respiratory tract is mostly affected, whereas at higher doses, damage is seen in the lower lung, including the alveoli. SM-induced epithelial cell injury and cell death in the tracheobronchial tree leads to inflammation and sloughing of the mucosa during acute stages. The clinical signs of SM inhalation include asthma, chronic bronchitis, bronchiectasis, and pulmonary fibrosis (Emad and Rezaian 1997). Pulmonary damage and associated secondary infections have been responsible for most fatalities (Papirmeister et al. 1991).
Despite decades of research, no completely satisfactory explanation of the toxic mechanism and the associated pathophysiological processes exists. As a result, there is currently no effective therapy for respiratory lesions caused by SM exposure. Nonetheless, it is generally believed that SM acts as an electrophile that alkylates cellular and extracellular components of living tissue such as DNA, RNA, proteins, and other biomolecules. As a result, complex cellular events develop, including cell cycle arrest, the synthesis and release of inflammatory mediators, and cytotoxicity. Following these cellular effects are tissue responses such as inflammation and tissue damage (Amir et al. 2000). The acute injury is associated with a rapid and massive release of destructive enzymes such as proteases and mediators of inflammation such as proinflammatory cytokines, nitric oxide, and so on (Cowan and Broomfield 1993; Smith et al. 1995; Cowan et al. 2003; Kehe et al. 2009).
Macrolides are a group of antibiotics that were initially discovered for their antibacterial properties. The name macrolide is derived from their structure, which is characterized by a macrocyclic lactone ring with various amino sugars attached (Bryskier et al. 1993). In recent years, an increasing number of studies have suggested that macrolide antibiotics exhibit anti-inflammatory functionalities by modulating the production of proinflammatory cytokines (Rubin 2004). Although the mechanisms underlying this effect are still unclear, macrolides have been shown to affect several pathways of the inflammatory process, including the migration of neutrophils, the oxidative burst in phagocytes, and the production of proinflammatory cytokines (Konno et al. 1994). Our previous in vitro studies have demonstrated that macrolide antibiotics protect airway epithelial cells and macrophages from SM-induced cytotoxicity through the suppression of inflammatory responses (Gao et al. 2007, 2008, 2010). The effectiveness of the macrolide antibiotics in our in vitro studies prompted us to further test them in an in vivo model. In the current study, we tested the efficacy of a common clinically used macrolide antibiotic, roxithromycin (Young, Gonzalez, and Sorkin 1989; Markham and Faulds 1994), in ameliorating SM-induced respiratory injury in a rat model.
Materials and Method
Reagents
Sulfur mustard (2,2’-dichlorodiethyl sulfide; 4 mM) was acquired from the U.S. Army Edgewood Research, Development and Engineering Center (Aberdeen Proving Ground, MD). Roxithromycin was obtained from Sigma (St. Louis, MO).
Animals
Male rats (Crl:CD SD BR), weighing 230 to 260 g, were purchased from Charles River Laboratories (Wilmington, MA) and maintained in an animal care facility that is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The “Guide for the Care and Use of Laboratory Animals” (1996) was followed during the conduction of the research described in this report.
SM Exposure
The exposure model has been described previously (Anderson et al. 1996, 2000). Briefly, rats were anesthetized with an intramuscularly (i.m.) administered combination of ketamine (80 mg/kg) and xylazine (10 mg/kg) and intubated with a modified glass Pasteur pipette (ca. 5 cm long) using a laryngoscope to visualize the larynx and a piece of PE90 tubing as a guide tube. The length of the glass endotracheal tube was sufficient for distal placement in the trachea at a point between the larynx and the bifurcation of the trachea. Once in place, the endotracheal tube was secured by wrapping a piece of porous tape around the tube and the rostrum of the rat. A glass endotracheal tube was necessary to minimize absorption of SM. SM (0.25 mg) in absolute ethanol (100 µl) or ethanol alone were placed in a water-jacketed (37°C) glass vapor generator custom fabricated by Atmar Glass (Kennett Square, PA). Spontaneously breathing rats were connected to these devices and exposed accordingly for fifty min. By the end of the exposure period, the SM in ethanol was completely vaporized and inhaled. This passive exposure system includes an inlet one-way respiratory check valve from Hans Rudolph (Kansas City, MO) to ensure that the only source of air for the animal during the exposure was through the vapor generator. Exhaled air passes through a two-way non-rebreathing Rudolph valve and then through a charcoal-filtered bleach trap to decontaminate any exhaled SM. At the conclusion of the fifty-min exposure, the rats were disconnected from the vapor generator, the endotracheal tube was removed, and the rats were returned to their cage.
Roxithromycin Treatment
Based on treatment time (duration from the start of the treatment to the time of sacrifice), the experiments were divided into three major time groups: one day, three days, and seven days. In each time group, rats were assigned to one of five treatment groups (N = 8/group): ethanol-exposed/no drug treatment (control); SM-exposed/no drug treatment (SM); SM-exposed/treatment with 10 mg/kg roxithromycin (RXM 10); SM-exposed/treatment with 20 mg/kg roxithromycin (RXM 20); SM-exposed/treatment with 40 mg/kg roxithromycin (RXM 40). In all, 120 rats were used in the study. Roxithromycin powder was suspended in saline by vigorous mixing and administered p.o. (10 ml/kg) at doses of 10, 20, or 40 mg/kg in respective groups of rats one hr prior to SM exposure and every twenty-four hr thereafter. The control group and the SM group received an equal volume (10 ml/kg) of vehicle.
Histopathology of the Lungs
Animals were sacrificed at one day, three days, or seven days of roxithromycin treatment. After euthanasia, the lungs were inflated with neutral buffered formalin, fixed, processed, and stained with hematoxylin and eosin. Six sections of lung were taken from each rat, including right cranial lobe, right medial lobe, right caudal lobe, left lung, right accessory lobe, and trachea. Each section was examined and scored on the following parameters: bronchial/bronchiolar exudates, bronchial/bronchiolar epithelial necrosis, bronchial/bronchiolar neutrophil infiltrates, bronchial associated lymphoid tissue (BALT) necrosis, alveolar exudate/edema, alveolar hemorrhage, alveolar neutrophil infiltrates, alveolar epithelial necrosis, perivascular fibrin/edema, and increase in alveolar macrophages. Each parameter was given a score from 0 to 3: 0 = normal; 1 = minimal, present in 1–10% of the section; 2 = moderate, present in 10–50% of the section; 3 = severe, present in >50% of the section.
Histopathology of Trachea
The trachea section from each animal was examined and scored on the following parameters: tracheal exudates, tracheal neutrophil infiltrates, tracheal epithelial attenuation, and tracheal epithelial necrosis or loss. Each parameter was given a score from 0 to 3: 0 = normal; 1 = minimal, present in 1–10% of the section; 2 = moderate, present in 10–50% of the section; 3 = severe, present in >50% of the section. In some cases, as tracheal epithelium was injured and individual cells were lost, the remaining cells took on a flattened or attenuated appearance, thus covering the same surface area with fewer cells. In order to account for these changes, both necrosis/loss and epithelial attenuation were scored and the average was used. In cases where there was no epithelium left, the score for the necrosis/loss was used as the average of the two.
Data Analysis
Data are shown as mean ± standard deviation. Group comparisons were conducted using one-way ANOVA followed by Tukey’s post hoc multiple comparison test. Pathology data were analyzed across groups with a chi-square test. For significance analysis, all groups treated with roxithromycin were compared to the group exposed to SM only using a Fisher’s exact test. Significant results were identified when p < .05 or smaller.
Results
Overall Histopathological Changes
The most profound histopathological changes in the SM-exposed animals were found in the trachea, bronchi, and bronchioles. These changes consisted of exudates, epithelial necrosis and loss, and neutrophilic infiltrates. In trachea, exudates composed of sloughed cells, fibrin, necrotic debris, and inflammatory cells often filled the lumen. Tracheal epithelium was often necrotic or lost. Remaining epithelial cells were markedly attenuated, and epithelial attenuation increased over time. Often neutrophils were present in the mucosa and lamina propria. Figure 1 highlights some histopathological sections with typical changes seen in SM-exposed animals in the trachea area. Similar to the trachea, the lumen in bronchi and bronchioles was often filled with sloughed cells, fibrin, necrotic debris and inflammatory cells. Epithelium was frequently necrotic, attenuated, or lost, and attenuation increased over time. Neutrophilic infiltrates were often present in the mucosa and lamina propria. Some of these changes are shown in Figure 2 .
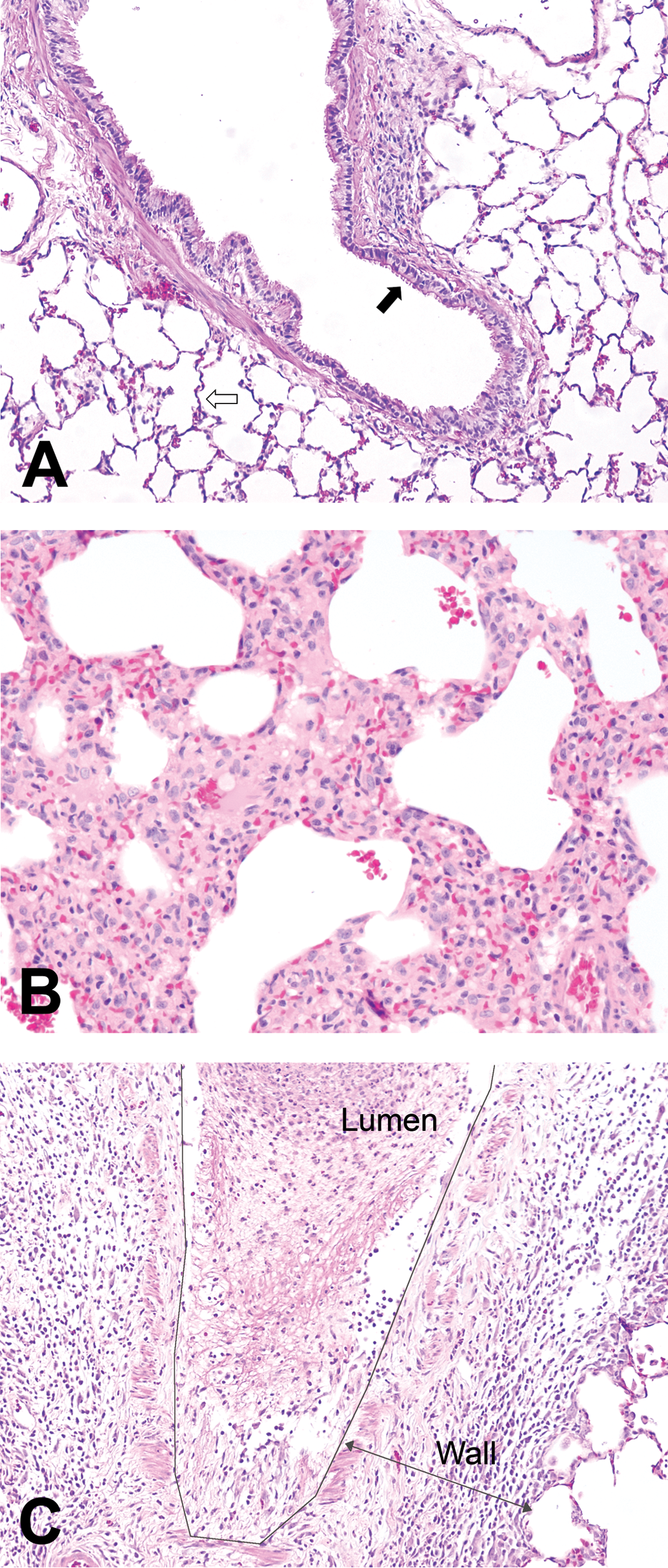
Histopathological sections showing typical changes seen in sulfur mustard (SM)–exposed animals in bronchiole and alveolar areas. (A) Normal (control) animals (20×). The open arrow points to the alveoli. Note the thickness of the septa and the absence of debris or cells within alveolar spaces. The closed arrow indicates bronchiole. Note the uniform pseudostratified columnar epithelial cells lining the
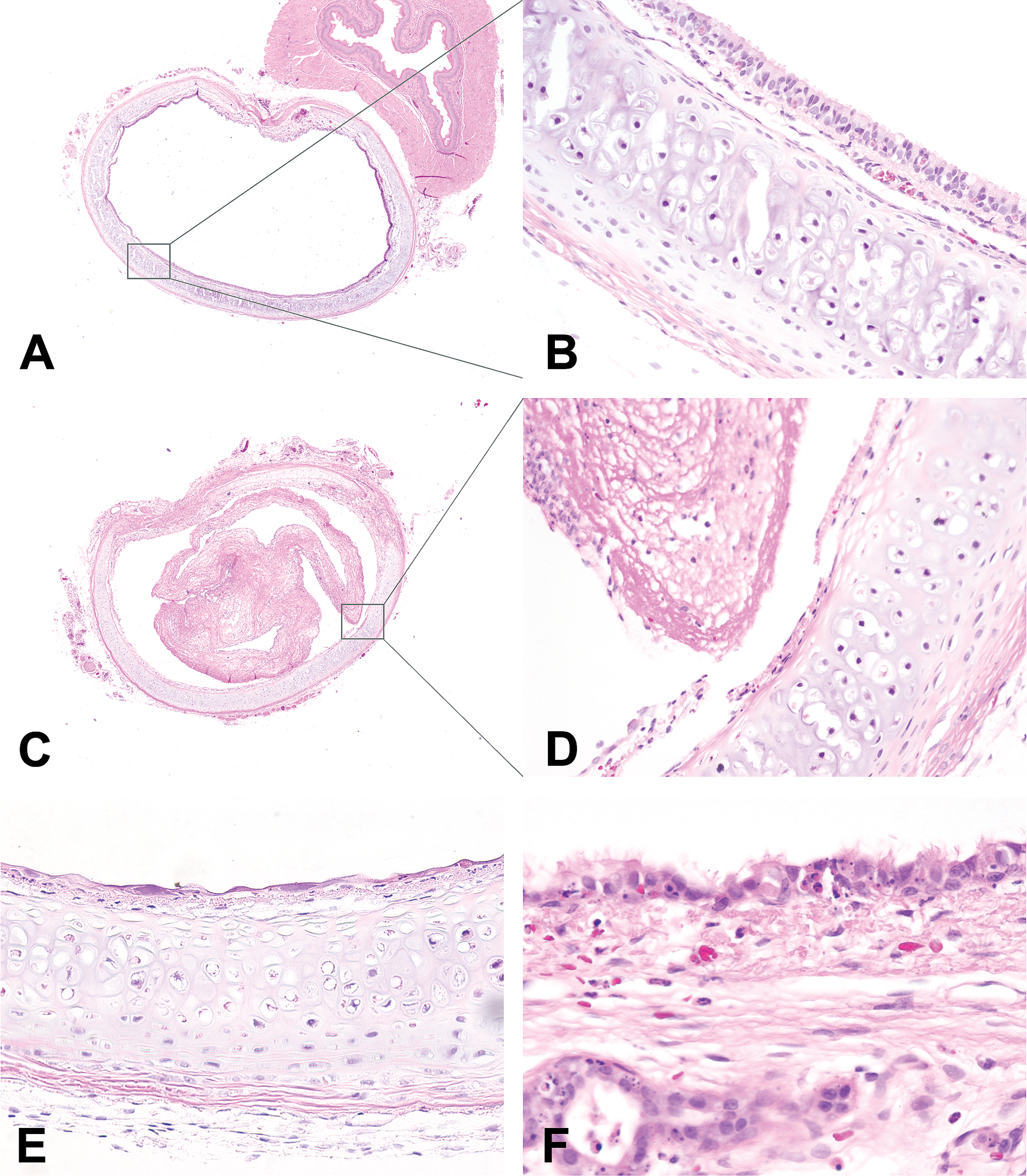
Histopathological sections showing typical changes seen in sulfur mustard (SM)–exposed animals in the trachea area. In normal animals, the trachea lumen was free from exudate and debris (A, 2×), and the respiratory epithelium was intact with a normal pseudostratified columnar morphology, and no inflammatory cells were present (B, 20×). In SM-exposed animals, the tracheal lumen was filled with exudates (C, 2×), which were composed of fibrin, necrotic debris, and inflammatory cells (D, 20×). Most of the respiratory epithelium was lost, and the remaining epithelial cells were markedly attenuated (E, 20×), with the necrotic epithelial cells scattered along the surface (F, 40×).
Consistent with previous in vivo studies using this model (Anderson et al. 1996), changes in the alveoli were not as prominent as they were in the trachea, bronchi, and bronchioles. The most common and profound changes were the filling of alveolar spaces with edema and fibrin or an increased number of alveolar macrophages (Figure 2). Less commonly, there was necrosis of alveolar epithelium or neutrophilic infiltrates into the alveolar spaces. Frequently in SM-exposed animals, the perivascular spaces were expanded by edema and fibrin.
Table 1 shows group averages for each parameter used in the pathological evaluation. The trachea was included in the columns for bronchial epithelial necrosis and bronchial neutrophil infiltrates. SM exposure resulted in higher scores in all areas, but with only minimal differences in BALT necrosis, alveolar hemorrhage, and alveolar epithelial necrosis. It was noticed that lesions were more consistent and more severe in larger airways (trachea, bronchi, and bronchioles) than they were in alveoli. Lesions in these larger conducting airways increased in severity, on average between days one and three. By day seven, the lesions in the larger airways were less severe than day three. Lesions were not severe in all sections of the lungs in many of the animals, but all lobes were affected in some animals.
Average score per animal in each experimental group for all pathological parameters.a
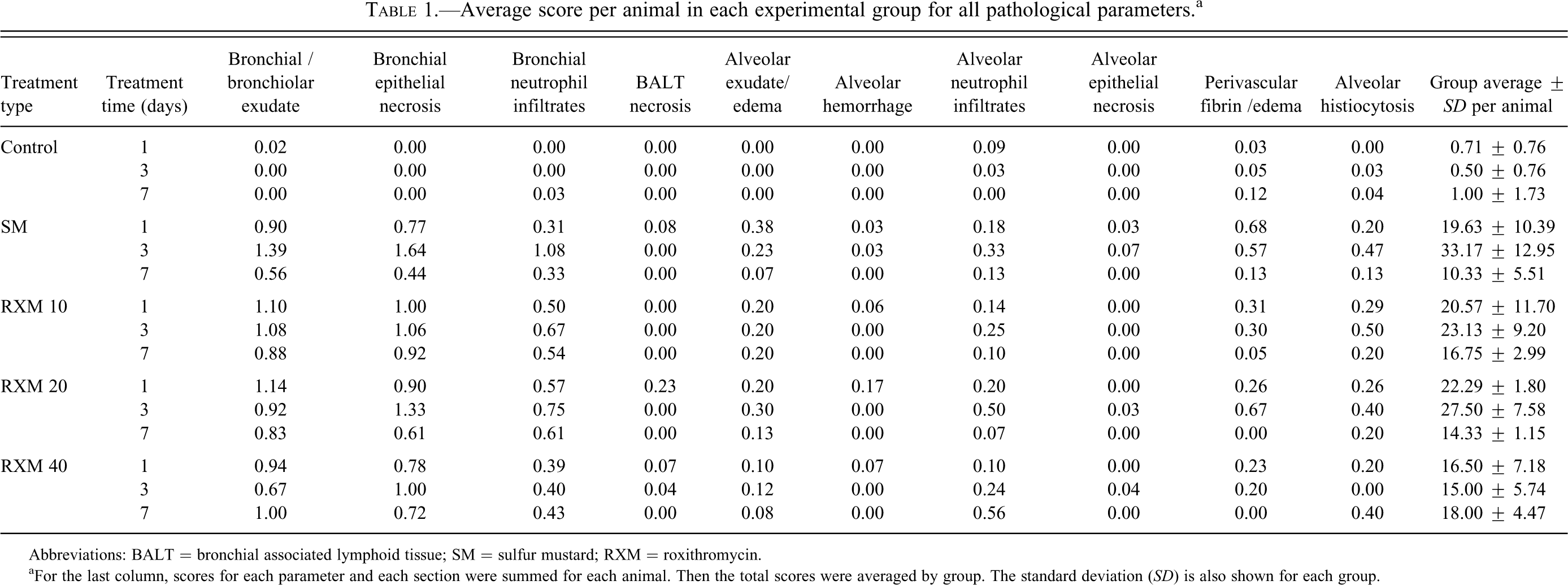
Abbreviations: BALT = bronchial associated lymphoid tissue; SM = sulfur mustard; RXM = roxithromycin.
aFor the last column, scores for each parameter and each section were summed for each animal. Then the total scores were averaged by group. The standard deviation (SD) is also shown for each group.
Similar to the SM group, the groups treated with roxithromycin also had higher scores in the larger conducting airways when compared to the alveoli. The two groups treated with 10 or 20 mg/kg roxithromycin also had a pattern of increased severity on day three when compared to days one and seven. The magnitude of difference appeared to decrease as the dose increased until at the highest dose (40 mg/kg) there was no difference between days one, three, and seven.
The group average total score (last column in Table 1, which is the sum of all parameters and all sections averaged by group) is graphically displayed in Figure 3 , divided into three groups based on treatment time. In most cases, there were no significant differences between animals treated with roxithromycin and their respective untreated counterparts (exposed to SM only). However, for the three-day groups, there was a significant difference between the SM-exposed animals and those treated with 40 mg/kg of roxithromycin (p < .05), suggesting the effectiveness of the drug at the current dose and duration of treatment.
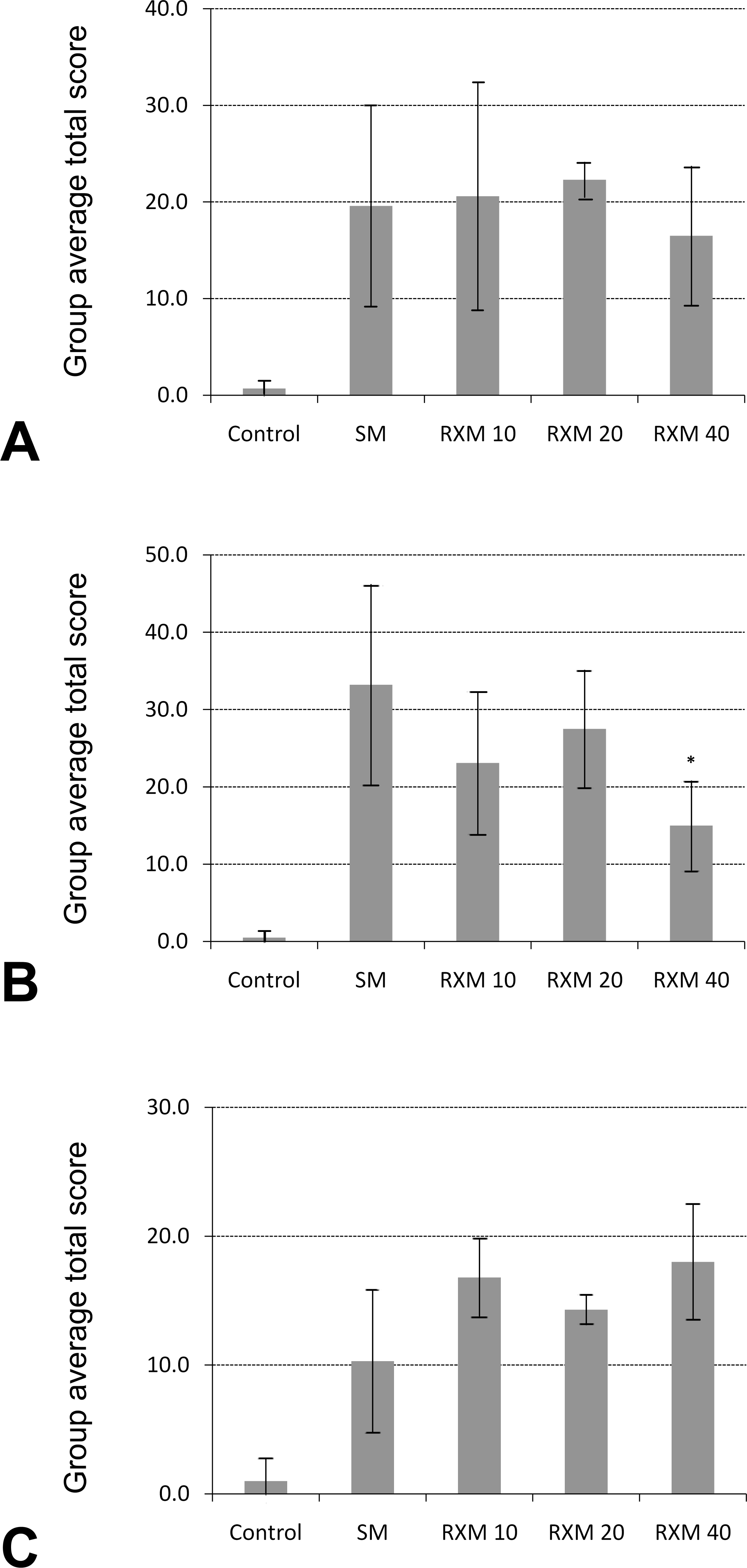
Graphical representation of group average total score of histopathology arranged by treatment time. (A) One-day treatment group. (B) Three-day treatment group. (C) Seven-day treatment group. The group average total scores were calculated by summing the scores of all parameters and all sections for each animal (animal total score) and then averaging the animal total scores by group. Comparisons were made between animals treated with different concentrations of roxithromycin versus their respective untreated counterparts exposed to sulfur mustard only (SM). *p < .05.
Histopathology on Trachea Only
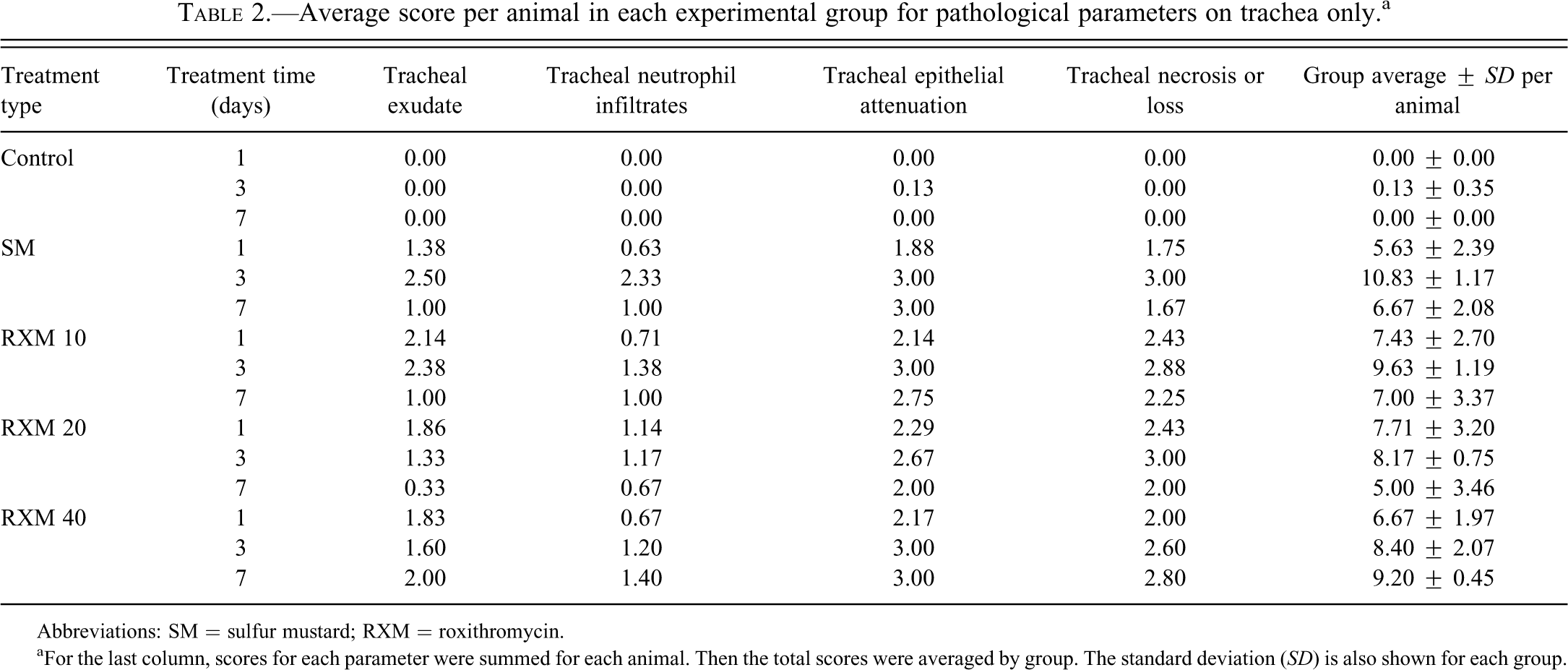
Because the most consistent and most severe lesions were found in larger airways, the trachea was evaluated separately from the rest of the lung. The trachea section from each animal was examined and scored on the following parameters: tracheal exudates, tracheal neutrophil infiltrates, tracheal epithelial attenuation, and tracheal epithelial necrosis or loss. Table 2 shows the averages of histopathological scores from all treatment groups. The trends are similar to those seen in the complete set of data (Table 1). In the SM-exposed groups as well as the groups treated with 10 or 20 mg/kg of roxithromycin, the severity was highest on day three. However, for the groups treated with 40 mg/kg of roxithromycin, lesions in the trachea increased in severity with time.
Average score per animal in each experimental group for pathological parameters on trachea only.a
Abbreviations: SM = sulfur mustard; RXM = roxithromycin.
aFor the last column, scores for each parameter were summed for each animal. Then the total scores were averaged by group. The standard deviation (SD) is also shown for each group.
The group average score (sum of all parameters for each animal averaged by group) shown in the last column of Table 2 is graphically displayed in Figure 4 , divided into three groups based on treatment time. Treatment with roxithromycin for three days showed a trend of dose-dependent improvement, and the groups treated with 20 or 40 mg/kg of roxithromycin were statistically different (p < .001 and p < .05, respectively) from the untreated group (Figure 4), suggesting that treatment with roxithromycin reduces some of the damage caused by SM inhalation in the trachea.
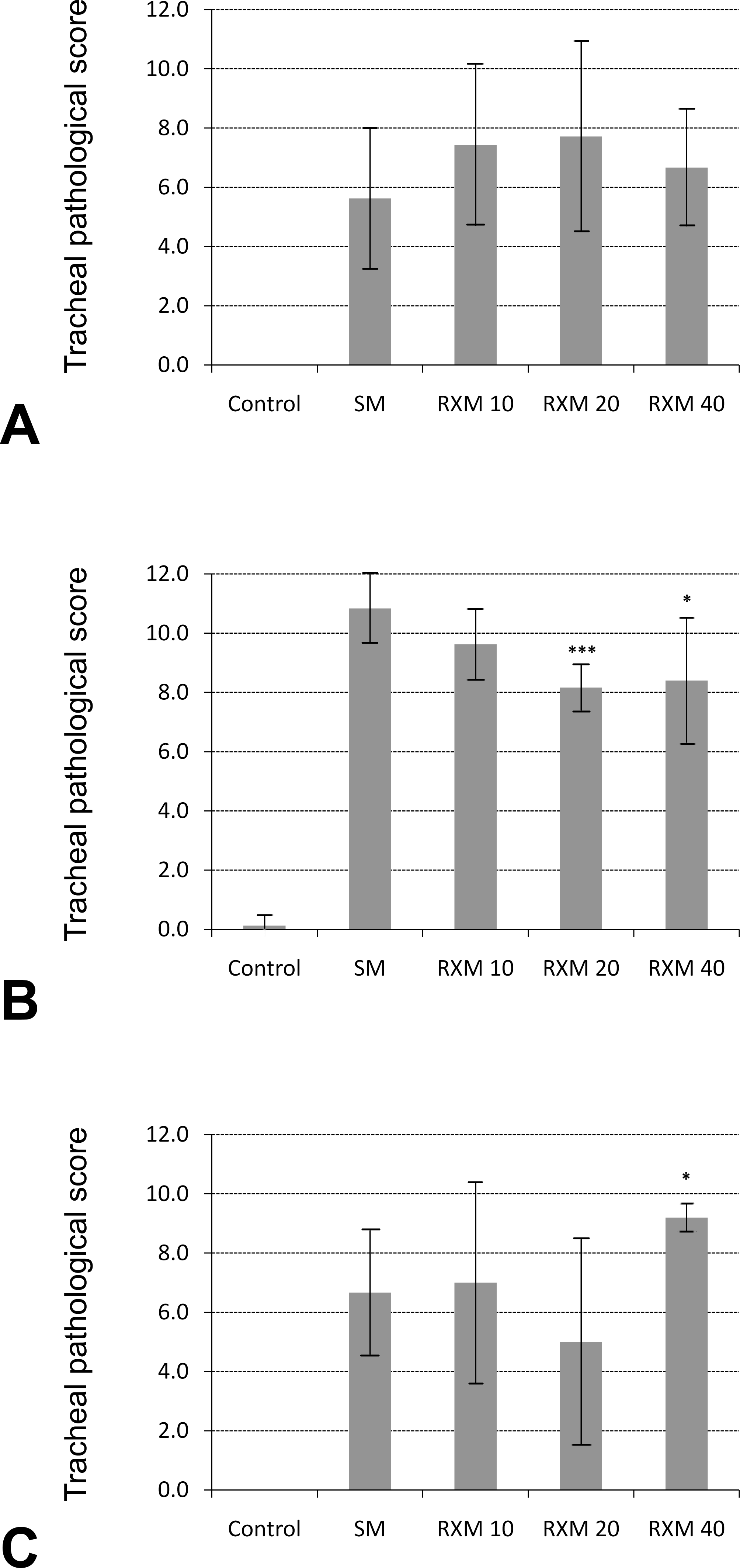
Graphical representation of group average score of pathological parameters focusing on trachea, arranged by treatment time. (A) One-day treatment group. (B) Three-day treatment group. (C) Seven-day treatment group. The scores were calculated by summing the scores of all parameters for each animal (animal total score) and then averaging the animal total scores by group. Comparisons were made between animals treated with different concentrations of roxithromycin versus their untreated counterparts exposed to sulfur mustard only (SM). *p < .05. ***p < .001.
Surviving Animal Counts
The number of surviving animals at the end of the experiment in each group is listed in Table 3 . In general, there were some gains in the groups treated with roxithromycin except for those treated with a dose of 10 mg/kg, where the number remained the same for all groups exposed to SM. In all cases, the animals died within three days after SM exposure; however, compared with animals exposed to SM only, treatment with roxithromycin not only increased the number of survivors, but also delayed the dying of the animals to some extent (Figure 5 ).
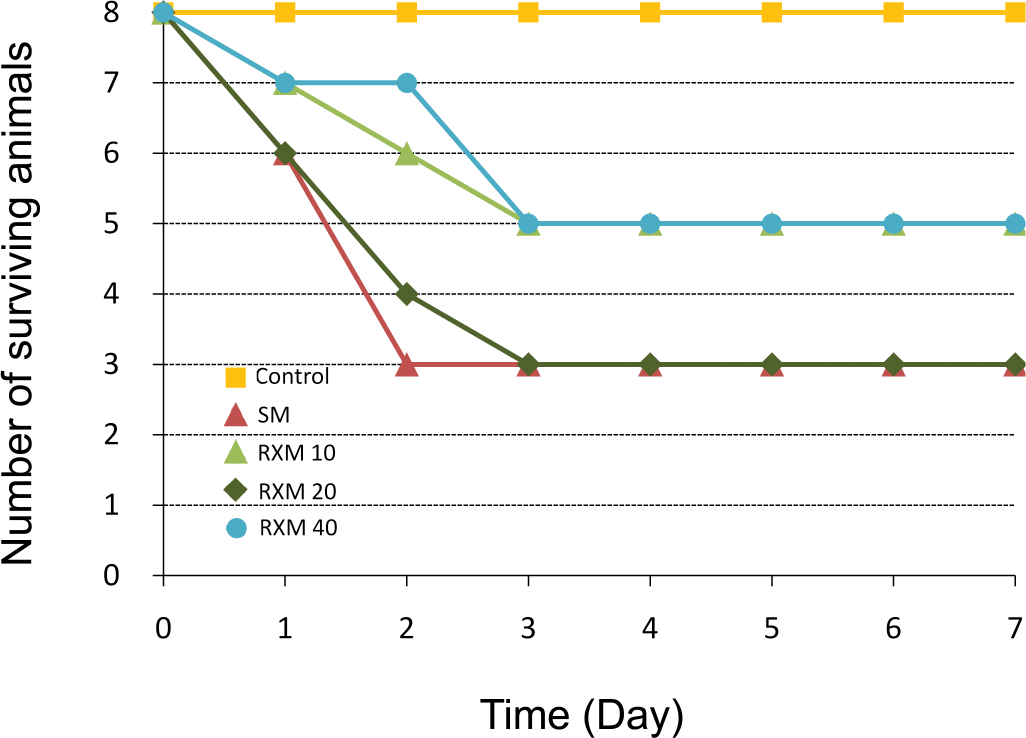
Number of surviving animals at each day in each group in the seven-day drug treatment groups.
Number of surviving animals in each experimental group.a

Abbreviations: SM = sulfur mustard; RXM = roxithromycin.
aEach group had eight animals (N = 8) at the beginning of experiment.
Discussion
SM inhalation leads to respiratory damage characterized by quick onset of inflammatory responses and subsequent pulmonary dysfunction. The clinical signs of SM inhalation include asthma, chronic bronchitis, bronchiectasis, and pulmonary fibrosis (Emad and Rezaian 1997). Macrolide antibiotics have shown effectiveness in a variety of chronic respiratory diseases such as diffuse panbronchiolitis (DPB), asthma, cystic fibrosis, chronic bronchitis, and chronic sinusitis (Rubin 2004). Although the underlying mechanisms are still unclear, it is now generally believed that the effectiveness of macrolides is a result of their immunomodulatory function. Macrolides have been shown to affect several pathways of the inflammatory process, including the migration of neutrophils, the oxidative burst in phagocytes, and the production of proinflammatory cytokines (Konno et al. 1994). Our previous in vitro studies have demonstrated that macrolide antibiotics protect airway epithelial cells and macrophages from SM-induced cytotoxicity through the suppression of inflammatory responses (Gao et al. 2007, 2008, 2010). In the current study we have evaluated roxithromycin, a representative macrolide antibiotic, for its efficacy against SM-induced lung injury using a rat SM inhalation model that has been published previously (Anderson et al. 1996, 2000).
The immunomodulatory effects of macrolides have been observed in clinical studies of chronic inflammatory airway diseases (e.g., DPB) after long-term treatment with low dose of macrolides (Rubin 2004). For roxithromycin, a daily dosage of one tablet (150 mg) was usually administered for a period of two to three months (Kimura et al. 1997; Suzuki et al. 1997, 2000). The dosage is equivalent to 1.5 to 3.0 mg/kg, using a body weight for an average patient in the range of 50 to 100 kg. However, this treatment regimen cannot be adopted directly in the current study as, in the case of SM inhalation exposure, airway inflammation is not chronic but rather acute. In severe cases, symptoms develop within hours after exposure (Eisenmenger et al. 1991; Papirmeister et al. 1991). Assuming a balance between drug dosage and treatment duration may exist, in the present study we tested roxithromycin using relatively high doses (10, 20, and 40 mg/kg) for shorter treatment times (one, three, and seven days).
The best results were obtained for the three-day treatment groups with doses of 20 mg/kg and 40 mg/kg, respectively (Figs. 3B and 4B). Clearly, treatment for one day is too short for any therapeutic effect of the drug to be seen (Figs. 3A and 4A). We expected to see more prominent beneficial effect for the seven-day treatment groups. On the contrary, compared to the SM-exposed group, a higher pathological score was obtained for the group treated with 40 mg/kg of roxithromycin (Fig. 4C). This suggests that longer treatment (seven days) at high dose (40 mg/kg) may have adverse effect on pathological outcome of respiratory injuries caused by SM exposure. The doses used in the current study (10–40 mg/kg) are ca. ten to twenty times higher than those used in clinical studies (1.5–3.0 mg/kg). Although macrolides are considered as well-tolerated antibiotics, adverse side effects and toxicity of macrolide antibiotics have been reported in many cases (Jain and Danziger 2004; Periti et al. 1993). Clinical data over a forty-year period in many thousands of adult and pediatric patients on the tolerability of clinically available macrolide antibiotics revealed that gastrointestinal reactions represent the most frequent disturbance due to induction of endogenous release of motilin (Periti et al. 1993). Incidences of adverse reactions in the heart, liver, central nervous system, and immune system, although rare, have also been reported (Periti et al. 1993). The findings of the current study may suggest a possible adverse effect of macrolide antibiotics, when administered at high doses and for long periods of time, on the respiratory system.
The dose of SM used in the exposure (1.0 mg/kg) also appeared to be too high for the experiment. For the seven-day experiment, more than 50% of the animals died within three days after SM exposure in some groups (Figure 5), namely, less than 50% of the animals survived the experiment (Table 3). The number of surviving animals in these groups may be too low to produce meaningful results from pathological evaluations (Fig. 3C).
As previously reported, the SM exposure model used in the current study did not produce even exposure along the entire respiratory system (Anderson et al. 1996). More severe damages were seen in the upper respiratory tract (trachea, bronchi, and bronchioles) than in the deeper lung (alveoli). This may partly explain the result that more prominent effect of roxithromycin was seen when histopathological evaluation was focused on trachea only instead of on the entire respiratory system (Figs. 4B vs. 3B).
To recapitulate, the doses for both SM and roxithromycin used in the current study seemed too high to adequately evaluate the effect of roxithromycin. A sublethal dose of SM should be used so that most, if not all, animals would survive the exposure. In addition, a lower dose but longer term treatment regimen for roxithromycin should be sought so that toxicities associated with high concentrations of the drug could be avoided. However, such a regimen may not be easy to figure out, as there seems to be a balance between the therapeutic effect and the toxic side effects for roxithromycin, which may be true for almost all drugs. In addition, a SM inhalation animal model that can produce even exposure along the entire respiratory tract is desirable so that the protective effect of roxithromycin in the deeper lung (alveoli) area could also be evaluated.
Conclusion
The results presented in the current study indicate that treatment with roxithromycin reduces some of the damages associated with SM-induced inflammation, particularly in the upper respiratory tract. Also, treatment with roxithromycin seemed to improve animal survival time to a certain extent. In view of the fact that there are currently no effective antidotes for SM inhalation injuries, the results of the present study suggest that macrolide antibiotics may serve as potential vesicant respiratory therapeutics.
Footnotes
Acknowledgments
We thank Dr. Radharaman Ray (USAMRICD) for critically reading the manuscript. We also thank Dr. Hiroshi Ishida (WRAIR) and Mss. Michele L. Conti and Danielle C. Paradiso (USAMRICD) for their technical assistance in sulfur mustard exposure, drug administration, and animal processing. The authors are also grateful to MAJs Cary L. Honnold and William L. Wilkins (WRAIR) and LTC Shelley P. Honnold (USAMRIID) for their kind help in preparing new pathological photomicrographs used in the revised manuscript and the many helpful discussions.
This work was supported by the Defense Threat Reduction Agency (DTRA) Project No. 3.F0003_05_WR_C. The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting true views of the U.S. Army or the Department of Defense.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
