Abstract
Introduction
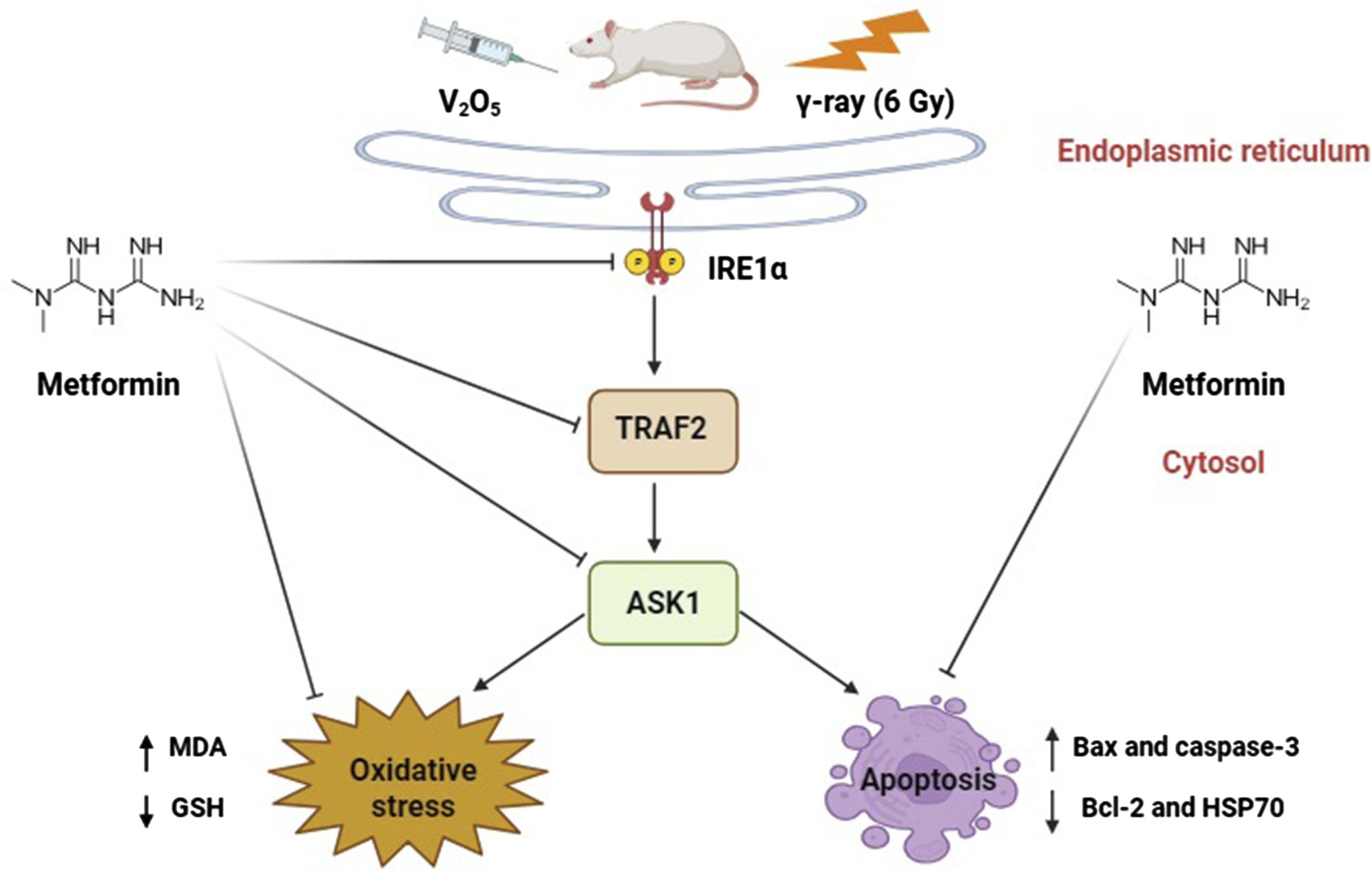
This work aims to validate the ameliorative influence of metformin against endoplasmic reticulum stress (ERS)-prompted apoptosis caused by vanadium pentoxide (V2O5) or gamma-irradiation (γ-irradiation) in hepatic tissues of male rats.
Methods
There were six groups of rats: the control, metformin (100 mg/kg body weight, i.p.), V2O5 (12.5 mg/kg body weight, i.p), V2O5 plus metformin, γ-irradiation group (acute dose 6 Gy), and γ-irradiation plus metformin; for 2 weeks. Hepatic malondialdehyde (MDA) and reduced glutathione (GSH) levels were evaluated. Additionally, the protein expression of certain endoplasmic reticulum stress-related (ERS) biomarkers; Inositol requirement enzyme 1α (IRE1α), TNF receptor-associated factor 2 (TRAF2), and Apoptosis signal-regulating kinase 1 (ASK1); were estimated in hepatic tissues. Moreover, apoptosis-associated biomarkers; Bax, Bcl-2, caspase-3 and HSP70 levels have been assessed. Furthermore, histopathological changes in hepatic tissues were observed.
Results
Metformin with V2O5 or γ-irradiation significantly decreased MDA, IRE1α, TRAF2, ASK1, Bax, and caspase-3 compared with V2O5 or γ-irradiated groups. Meanwhile, it significantly elevated GSH, Bcl-2, and HSP70 levels compared to exposure to V2O5 or γ-irradiation groups. Interestingly, the obtained results concur well with histological alterations.
Discussion
Our findings demonstrate the protective influence of metformin against ER stress-induced apoptosis through enhancing GSH and reduction of ERS and apoptosis suggesting that metformin may have positive impacts as a potential radiation protector beyond its glucose-lowering effect.
Introduction
The endoplasmic reticulum (ER) is an imperative intracellular organelle that helps preserve cellular homeostasis. 1 Endoplasmic reticulum stress (ERS) and the unfolded protein response (UPR) are triggered due to body exposure to external stressors that cause damage. 2 ERS can initiate various cell death processes like ferroptosis, pyroptosis, autophagy, and apoptosis. 3 These cell death pathways have a crucial impact on the onset and progression of neurodegenerative diseases, inflammation, diabetes, osteoporosis, and malignancies. They can also lead to liver conditions including fatty liver disease, hepatitis, alcoholic liver disease, and liver neoplasms. 4 Therefore, there is great potential for treating liver damage by modulating ERS.
Drinking water, soil, and air all have high levels of heavy metal pollution. Heavy metals can also be found in building materials, processed foods, pharmaceuticals, and cosmetics. As a result, it is extremely difficult for individuals to stay safe from exposure to any of the numerous dangerous heavy metals. 5 The fifth most prevalent element in the crust of the earth is vanadium (V) which is known as an important industrial and environmental pollutant that harms living organisms. 6 Burning fuel oils, mining, and the decomposition of rock and soil containing vanadium all release vanadium compounds into the atmosphere, soil, and water. 7 Vanadium can build up in the lungs, liver, and bone, inhibiting important enzymes involved in cell metabolism like adenylcyclases, kinases, ribonucleases, phosphatases, and ATPases. Vanadium toxicity is mainly caused by high reactive oxygen species (ROS) levels, oxidative stress, metabolic disruption, and mitochondrial dysfunction. 8 The most widely used and available form of vanadium is vanadium pentoxide (V2O5). 9 Dust produced by burning solid and liquid fuels as well as municipal waste heavily pollutes the environment with V2O5, increasing the risk of exposure for anyone living nearby. 10
Metformin is the first-line therapy for controlling type 2 diabetes via lowering hepatic gluconeogenesis. 11 It exhibited antioxidant, 12 anti-inflammatory, and immunomodulatory properties. 13 Researchers are currently examining metformin for desired defense in a variety of disease models including aging, cardiovascular, and neurological diseases besides malignancies. 14 Additionally, metformin has an effective role in protecting liver cells in nonalcoholic fatty liver disease (NAFLD), nonalcoholic steatohepatitis (NASH), and hepatic carcinoma. 15 Previous studies indicated its potential impact in preventing heavy metal toxicity. Metformin was shown to reduce mitochondrial damage and ROS formation in mouse hepatocytes exposed to arsenic. 16 Additionally, metformin suppressed the expression of hexokinase 2, which in turn prompts lipocalin 2 (LCN2) to prevent nickel-induced autophagy and cell death through the AMPK pathway in human airway epithelial cells. 17 Furthermore, metformin decreased neuron cell death induced by cadmium in lab experiments. 18 Metformin also improved the development of hippocampus neurons, neuron structure, and spatial memory in mice treated with aluminum chloride. 19 Metformin exhibited protection against liver and kidney toxicity caused by aluminum phosphide by reducing oxidative stress and inflammation. 20 Moreover, metformin reduced kidney damage in rats caused by lead through lowering the accumulation of D-lactate and methylglyoxal in kidney tissue. 21
High-dose ionizing radiation (IR) exposure causes biologically harmful side effects. Both direct and indirect effects of IR cause radiation damage. Enzymes frequently repair damage to biomolecules caused by both processes.22,23 As a result of the indirect effects of reactive free radical generation, cell biomolecules suffer oxidative damage which impairs biological functions. IR, being an oxidative stressor and DNA-damaging agent, induces tissue damage. 24 Furthermore, IR arrests the cell cycle by utilizing p53 which is activated by the DNA damage response, resulting in the induction of cellular senescence. 25 The proliferative capability and structural function of the affected tissues appear to be assessing factors controlling responses against IR in vivo, given that the IR response is dose-dependent and specific to certain tissues. 26 The use of antioxidants like metformin has been suggested since oxidative stress is critical to the pathophysiology of heavy metal toxicity and ionizing radiation.27,28
While there is some information on oxidative stress and toxicity caused by V2O5, there appears to be limited published researches on the biological effects of V2O5 on liver tissue. Furthermore, to our knowledge, no experimental studies have examined the protective role of metformin against apoptosis related to ERS in the liver, prompted by either V2O5 or γ-irradiation. Therefore, this investigation explored for the first time, the potential protective effect of metformin against apoptosis triggered by ERS in male rats’ livers, caused by exposure to V2O5 or γ-rays.
Material and methods
Chemicals and drugs
Both V2O5 and metformin were gotten from Sigma-Aldrich Co., USA, St Louis, Mo. High analytical-grade chemicals were also used in this experiment.
Experimental model species
For this investigation, 48 male Wister rats, weighing between 120 and 150 g at 3 months of age, were taken from the Egyptian Atomic Energy Authority’s animal house. Throughout the trial, the animals were provided with tap water on a free-choice basis and fed commercial pellets.
Ethical considerations
The research procedure has been authorized by the Institutional Animal Care and Use Committee and is being conducted in compliance with the guidelines established by the Research Ethical Committee of Tanta University, Egypt’s Faculty of Science (IACUC-SCI-TU-0302). This protocol complies with the US National Institutes of Health’s Guide for Care and Use of Laboratory Animals (NIH Publication No. 85–23, revised 2011). This study is reported in accordance with ARRIVE guidelines.
The process of radiation
Rats were exposed to a high dosage of γ-radiation over their entire bodies. The radiation dose was 6 Gy, carried at a rate of 0.36 Gy per minute, using a Cesium-137 irradiator offered by the National Center for Radiation Research and Technology. 29 This device, supplied by Candy Minimal Atomic Energy (gamma cell, 40), allows for uniform irradiation of small biological samples without posing external radiation risks to operators.
Treatments and doses
Rats were administered with V2O5 at a saline-diluted dosage of 12.5 mg/kg body weight.30,31 A solution containing 100 mg/kg of metformin was prepared using saline as a solvent and was given to specific groups of rats. 32
Experimental design
After allowing the rats to acclimatize in their cages for 2 weeks, they were erratically assigned into six groups of eight rats each: Group 1 (control): given certain volume of saline injections equivalent to metformin dose. Group 2 (Met): received 100 mg/kg metformin injections intraperitoneally (i.p.) daily for 14 consecutive days. Group 3 (V2O5): received 12.5 mg/kg V2O5 injections i.p. daily for 14 consecutive days. Group 4 (V2O5 + Met): given 12.5 mg/kg V2O5 and 100 mg/kg metformin IP together daily for 14 days. Group 5 (IR): exposed to 6 Gy of whole-body γ-radiation as a single dose. Group 6 (IR + Met): exposed to 6 Gy whole-body γ-irradiation as Group 5, then given 100 mg/kg metformin i.p. daily for the next 14 consecutive days.
Liver tissue sampling
After anesthetizing rats by injecting ketamine (50 mg/kg body weight) intraperitoneally, 33 the liver tissue was removed and divided into three parts after rinsing with ice-cold isotonic saline. The first part was centrifuged at 3000 rpm for 5 minutes at 4°C after weighing and homogenizing it (10% w/v) in ice-cold phosphate buffer (0.05 M, pH 7.4). The resulting supernatant was stored at −80°C for the later examination of the parameters associated with oxidative stress. To test ERS and other apoptotic biomarkers later, the second part was stored at −80°C. To enable histological examination, the third part was fixed in 10% neutral formalin.
Biochemical analysis
Assessment of oxidative stress markers
Lipid peroxidation as malondialdehyde (MDA) was assessed according to the procedure described. 34 Lipid peroxidation was measured by thiobarbituric acid assay, which is based on malonaldehyde (MDA) reaction with thiobarbituric acid forming thiobarbituric acid reactive substances (TBARS), a pink color complex exhibiting a maximum absorption at 532 nm. The results were expressed as nmol of MDA/g wet tissue. Furthermore, the content of reduced glutathione (GSH) was determined by the outlined method. 35 Where, GSH content was measured photometrically at 412 nm using 5, 5-dithiobis-2-nitrobenzoic against a reagent blank within 5 minutes using spectrophotometer, UNICAM 5625, UV/ VIS, England. The results were stated as mmol of GSH/g wet tissue.
Determination of IRE1α, TRAF2, and ASK1 protein expression in hepatic tissue by western blotting analysis
The defined protocol was followed for performing the western blot analysis. 36 The Bradford assay was used to assess the protein concentrations after the extraction of the total proteins from hepatic tissues. 37 The protein extracts were electrophoretically transferred in equal volumes onto polyvinylidene difluoride (PVDF) membranes (Bio-Rad) after sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Dried skim milk (5% w/v) in Tris Buffered Saline Tween (TBST) (10 mM Tris-Hcl, pH 7.5, 100 mM NaCl, and 0.1% Tween 20) was utilized to block the membranes for 2 hours at room temperature. Subsequently, the membranes were incubated overnight at room temperature with the primary antibodies: IRE1α (#3294), TRAF2 (#4724), ASK1 (#8662), and β-actin (#4967) acquired from Cell Signaling Technology, USA, each with a 1:1000 dilution. The membranes were first washed with TBST, then they were incubated for 2 hours at room temperature with appropriate horseradish peroxidase (HRP)-linked secondary antibody (#7074, Cell Signaling Technology, USA) followed by washing again with TBST. Chemiluminescence detection was performed using the Bio-Rad detection kit in accordance with the manufacturer’s protocol. The resulting bands were examined employing Image J software (1.49v) to determine alterations in protein expression levels, and each protein’s intensity was adjusted relative to β-actin.
ELISA assessment of Bax, Bcl-2, caspase-3, and HSP70 expression in hepatic tissues
The levels of Bax (Biovision, #E4513), Bcl-2 (CUSABIO, Wuhan, Hubei, China, CSB-E08854r), caspase-3 (Biovision, #E4592), and Hsp70 (ENZO Life Sciences, ADI-EKS-715) were measured in liver homogenates using rat ELISA kits following the manufacturer’s protocols. The intensity of color is measured spectrophotometrically at 450 nm in a microplate reader.
Histopathological examination of hepatic tissues
The hepatic sections that were dehydrated with ethanol; were cut into 4-6 μm thick slices, after clearing in xylene and embedding in paraffin blocks. The sections have been extracted, deparaffinized with xylene, and dyed with hematoxylin and eosin (H&E) for light microscopy histopathological analysis consistent with Bancroft. 38 For each sample, cross-sections from three slides were chosen. An Olympus XC30 microscope was utilized to analyze tissue sections from the slides. Tokyo, Japan. As previously documented, semi-quantitative assessments were utilized to determine the incidence and severity of liver lesions as follows: normal architecture (no damage) is grade 0, hepatocyte swelling is grade I, hepatocyte ballooning is grade II, lipid droplets in hepatocytes is grade III, and hepatocyte apoptosis and necrosis is grade IV. 39
Statistical analysis
The Kolmogorov-Smirnov test was used to determine data normality, while Bartlett’s test was used to determine homogeneity. Parametric data were analyzed using one-way ANOVA and Tukey’s multiple-comparisons test. Results were provided as mean ± standard error of the mean (SEM). The Kruskal-Wallis test followed by the post-hoc test (Dunn’s test) was used to examine hepatic damage scores, which were reported as medians (interquartile ranges). The statistical analyses were carried out using GraphPad Prism software (version 6.01). All statistical tests had a predefined significance level of p ≤ .05. The analytical methodology followed the approach described. 40
Results
Metformin ameliorates oxidative stress induced by V2O5 or IR in hepatic tissues
Levels of MDA and GSH were assessed in liver tissues (Figure 1). V2O5 and IR groups exhibited a significant rise in MDA levels and a drop in GSH levels (p ≤ .05) in comparison with the control group. Conversely, treatment with metformin along with V2O5 and IR revealed a substantial decline in MDA and a rise in GSH levels as compared to both V2O5 and IR groups. The expected values exposed that metformin has a strong antioxidant effect against gamma radiation or V2O5-induced hepatotoxicity. The effect of metformin administration on hepatic malondialdehyde (MDA) and reduced glutathione (GSH) levels in rats. Data presented as mean ± SE (n = 6), (a) significantly different from the control group (p ≤ .05). (b) significantly different from the V2O5 group (p ≤ .05). (c) significantly different from the IR group (p ≤ .05).
Metformin ameliorates ERS induced by V2O5 or IR in hepatic tissues
The relative expression of ERS-related proteins; IRE1α, TRAF2, and ASK1 in liver tissues was detected using western blotting (Figure 2). V2O5 and IR groups revealed a significant elevation in IRE1α, TRAF2, and ASK1 protein expression (p ≤ .05) as compared to normal rats. In contrast, the administration of metformin besides V2O5 and IR exhibited a significant decline in IRE1α, TRAF2, and ASK1 protein expression (p ≤ .05) as compared to groups exposed to V2O5 or IR alone. Consistent with the estimated values, metformin may have beneficial impacts against gamma radiation or V2O5-induced hepatotoxicity, besides its glucose-lowering effect. The effect of metformin administration on ERS biomarkers in liver tissues of rats exposed to V2O5 or IR. Data presented as mean ± SE (n = 6), (a) significantly different from the control group (p ≤ .05). (b) significantly different from the V2O5 group (p ≤ .05). (c) significantly different from the IR group (p ≤ .05).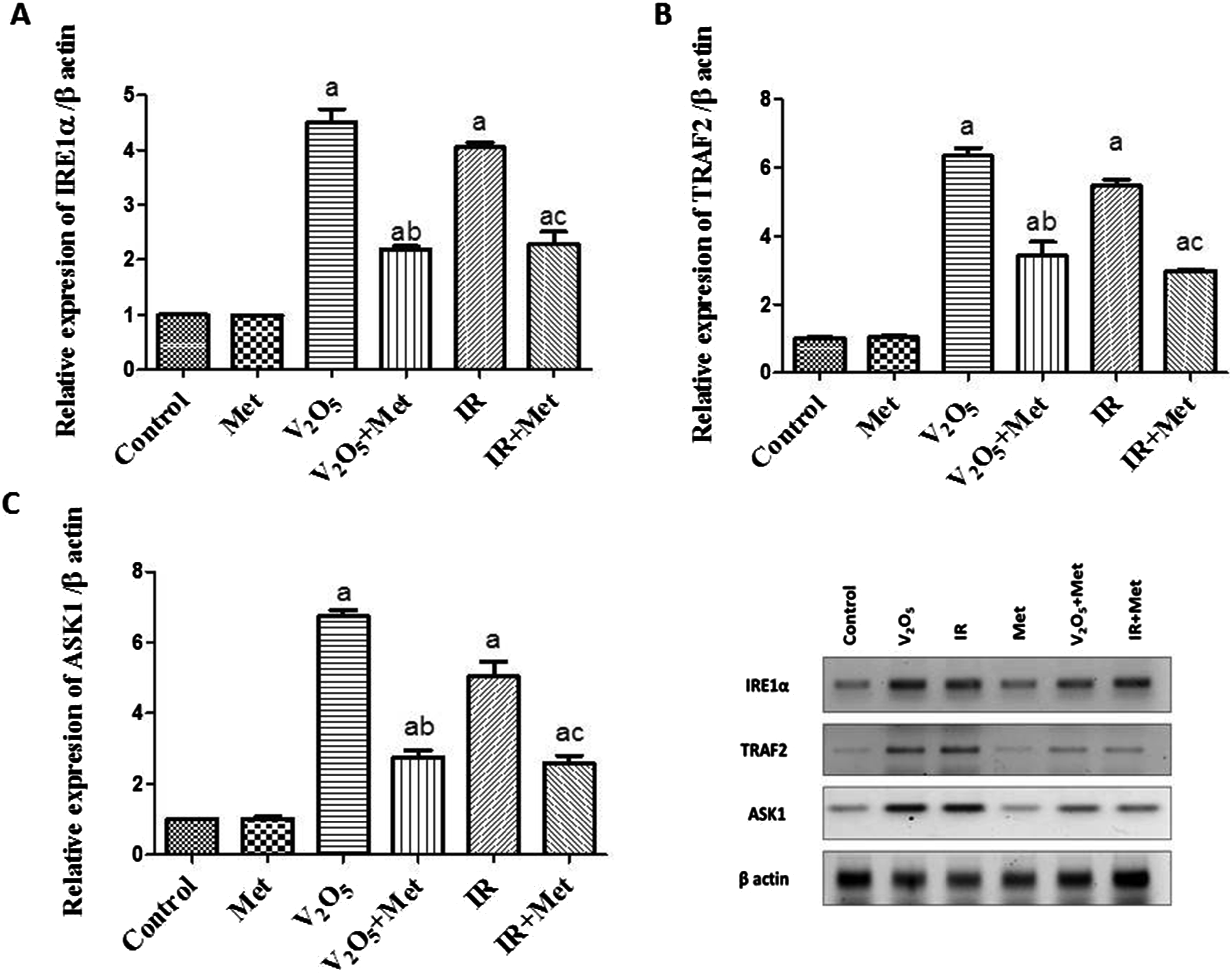
Metformin decreases ERS-induced apoptosis caused by V2O5 or IR in hepatic tissues
The exposure of rats to V2O5 or IR exhibited a significant elevation in Bax and caspase-3 and a significant decline in Bcl-2 expression in hepatic tissues (p ≤ .05) in comparison with normal rats. Interestingly, the treatment with metformin accompanied by V2O5 or IR significantly decreased Bax as well as caspase-3 and significantly raised Bcl-2 expression in liver tissues (p ≤ .05) when compared with groups administered with V2O5 or IR alone (Figure 3). The effect of metformin administration on apoptotic biomarkers in liver tissues of rats exposed to V2O5 or IR. Data presented as mean ± SE (n = 6), (a) significantly different from the control group (p ≤ .05). (b) significantly different from the V2O5 group (p ≤ .05). (c) significantly different from the IR group (p ≤ .05).
Metformin ameliorates HSP70 levels in hepatic tissues
The groups that received V2O5 or IR exhibited a significant downregulation in HSP70 levels in liver tissues (p ≤ .05) as compared to the control group. On the contrary, the administration of metformin combined with V2O5 and IR significantly upregulated hepatic HSP70 levels (p ≤ .05) in comparison to groups administered with V2O5 or IR alone (Figure 4). The effect of metformin administration on HSP70 levels in liver tissues of rats exposed to V2O5 and IR. Data presented as mean ± SE (n = 6), (a) significantly different from the control group (p ≤ .05). (b) significantly different from the V2O5 group (p ≤ .05). (c) significantly different from the IR group (p ≤ .05).
Metformin mitigates the histopathological alterations in hepatic tissues
The histopathology of liver sections from all experimental animals is illustrated in Figure 5. The liver tissue of the control group displayed the typical histological pattern of liver cords and lobules, as well as the presence of the central hepatic vein. Anastomosing plates connected polygonal hepatocytes; the boundaries of these hepatocytes faced either the sinusoids or adjacent hepatocytes (grade 0). Additionally, liver tissue sections from the metformin-treated group also exhibited normal hepatocytes organized in cords surrounding the central vein (grade 0). Moreover, liver sections from the V2O5 group displayed hepatic cord disorganization and necrobiotic alterations in hepatocytes, which were typified by nuclear pyknosis and vacuolation of the cytoplasm with central nuclei. Hepatocytes displayed clear cytoplasm indicating glycogen infiltration. Apoptosis of hepatocytes appeared as eosinophilic bodies scattered throughout the hepatic lobules. Hepatic sinusoidal narrowing with Kupffer cells hyperplasia was also detected (grade IV). Interestingly, the liver tissue sections from the group treated with V2O5 and metformin showed ballooning degeneration of hepatocytes in the peripheral zone. Narrowing of hepatic sinusoids with hyperplasia of Kupffer cells was also noticed (grade II). Moreover, hepatic sections of the animal group exposed to radiation revealed hepatic cord disorganization with dilatation of the central vein. Numerous numbers of apoptotic eosinophilic bodies were scattered all over the hepatic lobules, and hepatic sinusoidal dilatation Kupffer cells hyperplasia was seen (grade IV). The liver sections of the group that received metformin treatment and radiation, on the other hand, displayed modest hepatocyte swelling and cytoplasmic granularity, with centrally located vesiculated nuclei and peripheral nuclear chromatin condensation in the periphery zone. Hepatic sinusoidal dilation with Kupffer cells hyperplasia was also noted in (grade I). Histopathology of hepatic tissue sections stained by H&E, X200. (A) The control group shows strands of polygonal cells with prominent round nuclei and eosinophilic cytoplasm (Red arrow). (B) The Met group shows the normal histological structure of hepatic lobules and organization of hepatic cords (Red arrow). (C) The V2O5 group shows glycogen infiltration of hepatocytes with clear cytoplasm (Red arrow). (D) V2O5 + Met group showing ballooning degeneration of hepatocytes in the peripheral zone (Red arrow). (E) IR group showing disorganization of hepatic cords with apoptosis of hepatocytes and dilatation of sinusoids (Red arrow). (F) IR + Met group showing mild swelling of hepatocytes and dilation of sinusoids (Red arrow), Scale bar = 20 
Discussion
One vital intracellular organelle that carries out various biological functions is the ER. It is involved in cholesterol and phospholipid synthesis to produce cell membranes, calcium homeostasis, processes, folds, transports proteins within cells and considered the main metabolic organ in the body is the liver. 41 It is the primary site of protein synthesis, contains many enzymes, and carries out additional tasks including metabolism and secretion. 42 Therefore, its extremely developed ER system assists the hepatocytes in performing their physiological activities. Harmful factors that affect the liver disrupt its metabolism and synthesis, triggering ERS and a cascade of events that damage the liver to varying degrees, posing a major threat to human health. 43 As a result, there is a lot of promise for treating liver damage by targeting ERS. This study examines the possible hepatoprotective role of metformin against ERS-prompted apoptosis caused by V2O5 administration or γ-irradiation exposure in rats.
The body excessive production of free radicals and antioxidant capacity are impacted by various environmental pollutants. 44 MDA is a critical biomarker that appears in the final lipid peroxidation stages, which is elicited by an overabundance of ROS. However, GSH is crucial for maintaining redox balance and protecting cells from oxidative stress and the toxicity of foreign substances. 45 In this study, there was a substantial elevation in the oxidant (MDA) and a significant decline in the antioxidant (GSH) levels in the V2O5 and IR groups. These findings align with previous studies reporting the induction of lipid peroxidation and reduced GSH levels by ammonium metavanadate46–48 and sodium metavanadate 49 in animal models. Vanadium toxicity is primarily manifested through the damaging effects of high ROS levels, 50 which alter oxidative stress biomarkers like lipid peroxidation and GSH.51,52 Furthermore, MDA elevation could be explained by the presence of reduced free transition metal ions like ferrous (Fe2+ ) or vanadyl (V4+ ) ions, which then induce the formation of vanadate (V5+ ) and highly reactive hydroxyl radicals in the existence of hydrogen peroxide (H2O2) via the Fenton reaction.8,53 Also, vanadate formation can interact with superoxide ions to form peroxovanadyl radical, which is reduced by NADPH to vanadyl hydroperoxide. The decomposition of vanadyl hydroperoxide by bioreduction enhances H2O2 formation, exacerbating oxidative stress damage. Additionally, MDA elevation by γ-ray agrees with a prior study reporting that γ-ray inhibited SOD activity, leading to increased superoxide accumulation in cellular compartments, and subsequent MDA level increases. Nonetheless, the decrease in GSH levels might be attributable to its overuse because of the increased generation of ROS enhanced by exposure to γ-rays. 54
Several investigations have demonstrated that administering antioxidants is an effective strategy for lowering oxidative stress levels. 55 Earlier research identified the liver as a major site where metformin acts. 13 In this study, treating with metformin ameliorated the changes in MDA and GSH levels compared to groups exposed to V2O5 and γ-ray. These findings can be explained by metformin antioxidant properties against oxidative stress-induced liver damage, consistent with previous works.56,57 The results also align with a prior study showing that metformin enhanced mitochondrial viability and decreased mitochondrial membrane potential in mouse hepatocytes exposed to arsenic. It also suppressed the generation of ROS in conjunction with MDA reduction and GSH elevation on isolated mitochondria from mice hepatocytes. 16 Furthermore, metformin suppressed hepatic lipid peroxidation in rats exposed to aluminum phosphide. 20 Additionally, another study demonstrated that metformin, independent of AMPK, lowered ROS formation and prevented DNA damage and mutation in paraquat-exposed mice. 58 Metformin antioxidant effect may stem from several mechanisms, including direct scavenging of oxidants like hydroxyl radicals, activating NRF2-mediated antioxidant enzymes like catalase, the primary H2O2 decomposer, and constraining the transcription from NADPH oxidase four in long-term metformin administration.59,60
Numerous modes of cell death, such as apoptosis, are recognized to be stimulated by ERS through three main transmembrane receptors located on the inner membrane of ER: protein kinase-like endoplasmic reticulum kinase (PERK), activating transcription factor 6 (ATF6), and inositol requirement enzyme 1α (IRE1α). 61 These receptors can directly detect unfolded proteins within the ER lumen. After the unfolded proteins attach to these receptors, oligomerization, and autophosphorylation are triggered, and appropriate downstream signals are initiated. All eukaryotes have IRE1α, a type I transmembrane protein, in their ER which is the most evolutionarily conserved ERS sensor. 61 Here, the V2O5 and IR groups exhibited a significant rise in IRE1α, TRAF2, and ASK1 protein expression in liver tissues, which explained the induction of ERS. These findings are consistent with previous work reporting that excessive vanadium encouraged ERS in renal tubular epithelial cells of duck through the PERK/ATF4/CHOP pathway. 62
However, this current work found that protein levels of IRE1α, TRAF2, and ASK1 significantly decreased after treating rats exposed to V2O5 and IR with metformin. This indicates that metformin attenuated ERS-triggered by V2O5 and IR exposure. These findings are consistent with previous research showing that metformin reduced ERS and apoptosis in hepatocytes 63 and pancreatic cells 64 downregulating ERS proteins and regulating the insulin receptor substrate 1 (IRS-1) phosphorylation. Other studies have also demonstrated that metformin overwhelms ERS through activation of the AMPK-PI3K-c-Jun pathway. 65 Additionally, metformin has been found to reduce status epilepticus-induced ERS via the PERK-eIF2α-CHOP pathway. 66 Metformin has also been reported to alleviate ERS in hepatocytes of diabetic rats.32,67
There is strong evidence that apoptosis is a key process for sustaining life functions and is triggered mainly by two pathways: the internal pathway and the external death ligand-receptor pathway. 68 When ERS occurs, apoptosis results from the activation of IRE1α, which interacts with CHOP via XBP1 and activates the production of the downstream c-JNK or p38 mitogen-activated protein kinases (p38-MAPK) gene via the TRAF2-ASK pathway. 3 This study found that V2O5 or IR considerably elevated Bax and caspase-3 and dropped Bcl-2 expression in liver tissue. The researchers attributed these results to impaired electron transfer in mitochondria; which lead to ROS generation and induction of apoptosis and align with previous studies.69,70 However, treatment with metformin markedly lowered Bax and caspase-3 and increased Bcl-2 levels compared to V2O5 or IR groups. This suggests metformin inhibits V2O5 and IR induced apoptosis in rat liver which may be linked to its aptitude to mitigate oxidative stress and consistent with other works.71,72
The endoplasmic reticulum oxidoreductase 1α (ERO1α) protein is involved in the apoptosis caused by CHOP. ERO1α can upregulate the endoplasmic reticulum reductase gene which acts as an oxidative enzyme for protein disulfide isomerase (PDI) and generates high levels of H2O2 in the ER. 73 The hyperoxidized condition of the ER causes H2O2 to leak into the cytosol, resulting in oxidative stress and triggering inflammatory reactions and apoptosis. 74 ERO1α activates the ER membrane’s calcium channel IP3R1, causing calcium release into the cytoplasm. This in turn activates plasma membrane proteins CaMKII and NADPH oxidase, stimulating further ROS formation and initiating cell death. 75 These findings align with our results demonstrating that metformin reduced oxidative stress resulting from ERS.
Heat shock proteins (HSPs) are present in numerous cell compartments like the cytosol, nucleus, mitochondria, and ER. 76 Under normal circumstances, heat shock factor 1 (HSF1) subsists as an inactive monomer in the cytosol or nucleus. However, in response to stress, it translocates into the nucleus where it produces an active trimer that attaches to heat shock elements (HSE) in the promoter region of HSP genes, triggering their gene transcription. 77 This study found that treatment with V2O5 or γ-irradiation caused downregulation of HSP70, likely due to inactivation of certain phosphatases by vanadium ions or γ-rays. This alters the phosphorylation and transcriptional activity of HSF1, keeping it in an inactive monomeric form in the cytosol/nucleus. This prevents HSF1 from binding to HSEs in HSP promoters, inhibiting HSP70 transcription and reducing protein levels.78–80 In contrast, rats treated with metformin and exposed to V2O5 or γ-irradiation showed a substantial rise in hepatic HSP70 levels. This implies metformin may protect against hepatic injury from V2O5 or γ-ray by inducing HSP70, an intracellular cytoprotective protein that assists proper protein folding. Previous studies showed increased HSP70 expression in human esophageal carcinoma cell lines. A clinical trial found a tiny proportion of metformin-administered patients suffered liver fibrosis and steatosis after 2 years.81,82
In summary, histopathological examination of the results indicates that V2O5 and IR have a toxic effect on liver tissue. The histological alterations in hepatocytes induced by V2O5 or IR may be linked to oxidative stress and activation of ERS. These findings concur with prior research showing focal inflammation and significant hepatocyte degeneration after treatment with vanadium-rutin complex. 83 Similar results were seen when animals were exposed to V2O5 inhalation 84 and ammonium and sodium metavanadate. 47 Furthermore, these results align with previous studies describing the histopathological changes linked to structural liver lesions like fibrosis, necrosis, cirrhosis, and hepatocyte death,85,86 as well as hydropic degeneration, inflammatory cell infiltration, and hepatocyte destruction8,31 caused by exposure to vanadium or γ-rays in animal models.87,88 Interestingly, this study found that metformin treatment reduced the histological alterations in liver tissue induced by IR and V2O5, which may be explained by metformin potent antioxidant properties and ability to modulate ERS-associated apoptosis, consistent with previous research.56,89 Earlier work also reported that metformin mitigated liver injury scores caused by aluminum phosphide in rat livers by suppressing oxidative stress and inflammation. 20 Ultimately, we propose that further research should be undertaken to determine whether metformin can protect enhancement of hepatic proteostasis mechanisms such as autophagy, proteasomal degradation pathway, and heat shock protein against protein damage induced by V2O5 or γ-irradiation.
Conclusion
Although this study was limited by using only one dose and assumed timing rather than examining dose and time dependence, the findings support previous evidence V2O5 and γ- irradiation cause toxicity by increasing oxidative stress, ROS generation, and ERS-mediated apoptosis, in addition to histopathological changes. Metformin may protect the liver from the oxidative stress triggered by V2O5 toxicity and γ-irradiation exposure by decreasing lipid peroxidation and improving the antioxidant system. It also protects against ERS-prompted apoptosis resulting from V2O5 or γ-irradiation, thereby preventing liver injury. An important clinical implication is that metformin could be used as a protective agent against liver toxicity induced by heavy metals and ionizing radiation exposure.
Abbreviations
AMP activated protein kinase
Activating transcription factor 6
Endoplasmic reticulum stress
Gamma-irradiation
Glutathione
Gray
Heat Shock Protein 70
Inositol requirement enzyme 1α
Ionizing radiation
Malondialdehyde
Protein kinase-like endoplasmic reticulum kinase
Reactive oxygen species
Unfolded protein response
Vanadium pentoxide
Footnotes
Acknowledgements
This study was conducted at the Egyptian Atomic Energy Authority, National Center for Radiation Research and Technology, Cairo, Egypt. The authors acknowledge the members of the trace elements laboratory.
Authors’ contributions
MEH designed and supervised the studies, performed experiments, analyzed data, participated in manuscript preparation, wrote the manuscript, and reviewed the manuscript. FYA performed experiments and analyzed data, wrote and reviewed the manuscript. MNG assisted with studies, wrote the manuscript, and reviewed the manuscript. DME provided technological support and reviewed the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and no paper mill was used.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
