Abstract
Introduction
Abnormal activation of hypertrophic scar fibroblasts (HSF) plays an important role in the excessive fibrosis of hypertrophic scars (HS). However, the regulatory mechanism of HSF abnormal activation is not fully unclear. Early studies had shown that M2 macrophages were increased during scar formation. The aim of this study was to investigate the mechanism of M2 macrophage-derived exosomes (M2-EXOs) mediating HSF abnormal activation.
Methods
The blood samples of 20 normal people and 20 HS patients were collected from Xi’an Hospital of Traditional Chinese Medicine, and the level of M2 macrophages in the blood was measured by flow cytometry. Subsequently, HSFs were co-cultured with M2-THP-1 for 48 h to analyze the effect of M2 macrophages on the function of HSFs in vitro. HSFs were treated with exogenous chemokine (C-X-C motif) ligand 2 (CXCL2) or anti-CXCL2 to analyze the effect of CXCL2 on HSFs function and autophagy. HSFs were treated with exogenous CXCL2 and/or anti-CXCR7, and CXCL2 and/or 3MA to explore the molecular mechanism of CXCL2-mediated HS. Finally, a mouse HS model was constructed, and the effect of M2-Exos on the growth of HS was explored by subcutaneous injection of CXCL2 or M2-Exos in the scar site in vivo.
Results
We found that the proportion of M2 macrophages in the blood of HS patients increased. CXCL2-rich M2-EXOs promoted the abnormal proliferation, migration, and collagen deposition of HSFs in vitro. CXCL2 increased the level of p-mTOR in HSF and promoted the expression of autophagy proteins LC3II/I and Atg5 in vitro. Further results showed that CXCL2 activated autophagy through CXCR7/PI3K/mTOR signal transduction, thereby promoting collagen deposition and fibrosis in vitro. Autophagy inhibitor 3-MA reversed the effect of CXCL2 on HSFs in vitro. In addition, in the HS mouse model, after treatment with M2-EXOs or CXCL2 in vivo, the scar recovery time was significantly prolonged and the scar damage was aggravated.
Discussion
These results suggest that the CXCL2/CXCR7/mTOR pathway may be a promising target for the treatment of HS. Abnormal activation of hypertrophic scar fibroblasts (HSFs) plays an important role in the excessive fibrosis of hypertrophic scars (HS). However, the regulatory mechanism of HSFs abnormal activation is not fully unclear. Early studies had shown that M2 macrophages were increased during scar formation. The aim of this study was to investigate the mechanism of M2 macrophage-derived exosomes (M2-EXOs) mediating HSFs abnormal activation. Here, we analyzed the proportion of M2 macrophages in total macrophages in the HS patient’s blood, and we found that the proportion of M2 macrophages were elevated in the blood of HS patients. We found that C-X-C motif chemokine 2 (CXCL2)-rich M2-EXOs promoted abnormal proliferation, migration, and collagen deposition in HSFs in vitro. CXCL2 increased the phosphorylation level of mTOR protein and promoted the expression levels of autophagy related proteins LC3II/I and Atg5 in HSF in vitro. CXCL2 activated autophagy through chemokine (C-X-C motif) receptor 7(CXCR7)/PI3K/mTOR signal transduction, and promoted collagen deposition and fibrosis in vitro. The autophagy inhibitor 3-Methyladenine (3-MA) reversed the effect of CXCL2 on HSFs in vitro. Meanwhile, in the HS mouse model, the scar recovery time was significantly prolonged and the scar injury was aggravated after treatment with M2-EXOs or CXCL2 in vivo. These results suggest that the CXCL2/CXCR7/mTOR pathway may be a promising target for the treatment of HS.
Keywords
Introduction
Hypertrophic scar (HS) often forms after skin trauma and are often accompanied by itching, pain, and even loss of joint mobility at the wound site. 1 HS can greatly affect the life of patients. Current studies have shown that when the skin is injured, fibroblasts accumulate in large numbers in the injured area, with proliferation and differentiation into myofibroblasts that contribute the dermal fibrosis, thus promoting the formation of HS.2,3 α-smooth muscle actin positive (α-SMA+) fibroblasts promote collagen synthesis, including Collagen I (Col-1) and Collagen III (Col-3), which are key causes of nodular formation in HS. At present, it is mainly aimed at inhibiting the abnormal proliferation and differentiation to myofibroblasts of fibroblasts and inhibiting collagen synthesis at injured area. 3 However, due to unclear therapeutic targets, there is no effective method for the prevention and treatment of HS.
Exosomes, as the nanoparticles participate in intercellular communication, had shown strongly association with several progress, such as immune responses, viral pathogenicity, cancer progression. 4 Exosomes are membrane vesicles that released by most cells, containing different cargo to regulate the biological activity of recipient cells. 5 In the process of HS formation, different kinds of cells coordinate to regulate the fibrosis process. Macrophages, arrive at the injured area in the early stage of skin injury to participate in the inflammatory response and the damage repair mediated by fibroblasts. 6 According to different phenotype and secretory cytokine, macrophages are major divided into two polarization states, M1 macrophages (classically activated type 1) and M2 macrophages (alternatively activated type 2). 7 Existing studies have shown that M2 macrophages are significantly increased in pathological HS tissues, and the cytokine transcription levels of TGF-β, iNOS, IL-10 and other cytokines are significantly increased. 8 This suggests that M2 macrophages are closely related to HS formation. Chen et al. demonstrated that removal of M2 macrophage-derived exosomes could inhibit the activation of scar fibroblasts by reducing lncRNA-ASLNCS5088. 9 In the study, we mainly focused on the roles of M2 macrophage-derived exosomes (M2-Exos) in HS fibrosis.
C-X-C motif chemokine 2 (CXCL2) is a chemokine involved in a variety of physiological processes, such as liver fibrosis and osteoblast differentiation.6,10 The role of CXCL2 in fibrosis has been reported, such as the activation of dental ligament fibroblasts by increasing CXCL2 in diabetes, 11 and the inhibition of keratinocyte migration and the activation of dermal fibroblasts by CXCL2. 12 These results suggest that CXCL2 related to fibrotic and activating fibroblasts, so we continue to explore the role of CXCL2 in HS formation.
In this study, we identified M2-type macrophage-derived exosomes (M2-Exos). The effect of M2-Exos on HS fibrosis was investigated in vitro and in vivo, and the possible mechanism of M2-Exos anti-fibrotic properties was explored. Meanwhile, we also analyzed that M2-Exos influenced scar formation in BALB/c mouse excision models, contributed to collagen deposition and myofibroblast trans differentiation in vivo. In conclusion, our work may provide an idea for targeting HS therapy.
Materials and methods
Human tissue samples
In this study, blood samples from 20 normal subjects and 20 HS patients were obtained from Xi’an Hospital of Traditional Chinese Medicine. The obtained blood cells were centrifuged at once and divided into two parts for flow cytometry and western blotting. All patients with HS used in this study signed pre-study informed consent, and the study was approved by Xi’an Hospital of Traditional Chinese Medicine.
Animal
Fifteen 8-week-old female BALB/c nude mice (Xi’an Air Force Medical University) were selected and divided into three groups. They were anesthetized with 1% pentobarbital sodium (50 mg/kg). After the skin of the mice was disinfected, and 2 cm incision was made on the dorsal midline of the mouse back and reapproximated with 6-0 nylon sutures. On the fourth day after the incision, the nylon sutures were removed and a biomechanical loading device was attached on the scars. On subsequent days, spread the expansion screws of the device by 4 mm until 2 weeks. The model of load-induced hypertrophic scar was established. The mice were randomly divided into three groups: PBS, CXCL2 groups. In order to explore the effect of M2-Exos on the growth of HS in vivo through the CXCR7/PI3K/mTOR pathway, 200 μL of CXCL2 (10 μg/mL) or 200 μL of M2-Exos (20 μg/mL) were subcutaneously injected into the scar site, and the scar area was analyzed after 14 days.
Cell isolation
The blood samples of the volunteers were centrifuged at room temperature, 2000 r/min, 15 min. A narrow band of a white cloud layer (i.e., the mononuclear cell layer) at the upper and intermediate interfaces is collected by capillary tubes into another centrifugal tube, centrifuge (1500 r/min, 15 min). The supernatant was discarded, and the cells were precipitated and then inoculated in a petri dish. After standing culture at 37°C for 3 days, the adherent cells were removed, and the non-adherent cells obtained were macrophages.
The hypertrophic scar tissue was isolated and collected, soaked in 0.25% pancreatic enzyme, and digested overnight. The dermal tissue was removed and the tissue was divided into small pieces less than 1 mm in diameter with a blade. The separated tissue was placed in a petri dish, type 1 collagenase was added, digested in a cell incubator at 37°C for 2 h, cells were collected after centrifugation for culture, and fibroblasts were used for experiment after 3-5 generations.
For M2-polarized macrophages, 5 ng/mL PMA (Sigma, USA) treated THP-1 cells for 24 h and then incubated with 20 ng/mL IL-4 and 20 ng/mL IL-13 (PeproTech, USA) for 48 h. They were then identified by Western blotting.
Exosome isolation and identify
After 48 h culture of M2-like THP-1, the medium was collected and centrifuged at 1000 g at room temperature for 5 min, followed by 15 000 g at 4°C for 15 min. Then, the supernatant 110,000 g was centrifuged for 70 min using an ultracentrifugation. The supernatant was then discarded and the sediment was re-suspended in PBS. Exosomes were identified by Western blotting.
Cell culture and treatment
The isolated HSFs were cultured in RPMI-1640 medium containing 10 % high-quality fetal bovine serum and placed in a 5% humidity incubator at 37°C CO2 overnight.
In order to explore the effect of M2 macrophages on the function of HSFs in vitro, HSFs were co-cultured with 20 μg/mL M2-THP-1 for 48 h. In order to explore the effect of M2-Exos on the function of HSFs, HSFs were treated with 20 μg/mL exosomes for 48 h. In order to explore the effect of CXCL2 on the function of HSFs, HSFs were treated with 1 ng/mL and 10 ng/mL exogenous CXCL2 for 48 h, and then cells were collected for experiments. In order to explore the effect of CXCL2 on autophagy of HSFs, HSFs were treated with 1 μg/mL anti-CXCL2 for 48 h, and then cells were collected for experiments. In order to explore the molecular mechanism of CXCL2-mediated HS, cells were treated with 10 ng/mL CXCL2 and/or 1 μg/mL anti-CXCR7, as well as 10 ng/mL CXCL2 and/or 5 mM 3MA for 48 h.
Western blotting
The cells were treated with PMSF-added RIPA protein lysate (Beyotime, P0013 B) for 30 min, and the supernatant was collected by centrifugation. The lysate was collected and centrifuged at 4°C, 16,000 g for 10 min. The supernatant was collected for protein analysis. The protein concentration in supernatant was determined by bischondroitin acid (BCA) protein method. 12% protein was isolated SDS-PAGE and blot was performed with PVDF membrane (Beyotime, FFP39). Antibodies were incubated overnight using 5% skim milk at room temperature. The next day, PVDF membrane was washed with TBS and incubated with secondary antibody. The results were recorded by chemiluminescence apparatus (BIO-RAD, 1708280). The primary antibodies used in this study and their concentrations are as follows: anti-Col1a1 (CST, #72026, 1:1000), anti-Col3a1 (NOVUS, NBP1-05,119, 1:500), anti-α-SMA (Abcam, ab5694, 1:1000), CD9 (Proteintech, 20,597-1-AP, 1:1000), CD63 (Proteintech, 67,605-1-Ig, 1:1000), β-actin (Cell Signaling Technology, # 4967S, 1:1000), anti-Alix (Abcam, ab76608, 1:1000).
Transwell assay
The experiment was carried out using matrix-coated transwell insert 24 well cell culture plates (Corning, 3464). The cells were seeded in the upper compartment at 1 × 105/well, and the medium was added in the lower compartment. After incubation for 24 h, the lower layer was fixed with 4% paraformaldehyde (Beyotime, P0099) and dyed with crystal violet for 10 min. Gently wiped with a cotton ball to remove the upper layer of non-migrated cells and non-specific binding crystal violet. The number of migrating cells was observed under microscopes (Nikon).
EdU proliferation assay
The cells were inoculated in a 24-well plate with coverslip and incubated in a 5% CO2 incubator (37°C) for 24 h. Cell proliferation assay was performed according to EdU Cell Proliferation Kit with Alexa Fluor 488 (Beyotime, C0071S) instructions. The test results were imaged using fluorescence microscopy and the data results were analyzed using ImageJ software.
Sirius red staining, H&E and IHC
The isolated mouse tissues were soaked and fixed with 4% paraformaldehyde, embedded, and sliced with paraffin wax. Sections were stained with specific dyes (Hematoxylin and Eosin, Sirian Red).
The fixed tissues soaked with 4% paraformaldehyde were treated with 30% sucrose solution and then sliced after OTC embedding. The sections were treated with acetone and washed with PBS. Then, the sections were incubated overnight with primary antibody. The next day, after PBS cleaning, the second antibody was incubated for 2 h. Fluorescence imaging uses fluorescence microscope. The results were analyzed using the imageJ software.
Enzyme linked immunosorbent assay
After collecting HSFs with different treatments, the levels of TGF-β1, IL-1β, IL-6 and TNF-α were detected by TGF-β1 ELISA kit (Abcam, ab100647), Mouse IL-1β ELISA Kit (Biyuntian, PI301), Mouse IL-6 ELISA Kit (Biyuntian, PI326) and Mouse TNF-α ELISA Kit (Biyuntian, PT512).
Statistical analysis
Statistical analysis was performed using Graphpad. Student’s t test was used to analyze differences between two groups. One-way analysis of variance (ANOVA) with post hoc test (Tukey test) was used to compare in multiple comparisons. Normally distributed data are expressed as mean ± standard (SD). In the significance analysis of this paper, p < 0.05 was statistically significant. All experiments were repeated at least 3 times.
Results
The number of M2 macrophages was increased in the HS patients
Results from the previous study had shown that the number of macrophages in the HS patients, was significantly increased compared to normal people.
13
In order to verify the role of macrophages in HS formation, we separated the blood of HS patients and normal people and made a comparison. Flow cytometry was used to study the isolated blood cells, and the results showed that the number and proportion of CD11b+ cells in the blood of HS patients were significantly higher than that of normal people, and the proportion of CD11b+CD86+ cells were significantly lower than that of CD11b+CD206+ cells (Figure 1(A)). Then, we compared the expression levels of collagen deposition-related genes in normal skin tissues and HS tissues, and the results showed that the expression levels of Col-1 and α-SMA in HS tissue were significantly increased (Figure 1(B)). Transforming growth factor (TGF) is a proven cytokine that stimulates fibroblasts to differentiate into myofibroblasts.
14
Therefore, we detected the level of TGF-α in serum of normal subjects and HS patients, and the results showed that the concentration of TGF-α was significantly higher in serum of HS patients. (Figure 1(C)). These results suggest that M2 macrophages highly related to myofibroblast transformation and collagen generation in HS. M2 macrophages increased in the HS area. (A) The statistics of CD11b+, CD11b+CD86+, CD11b+CD206+ cell proportion. (B) The expression of col1 and α-SMA was measured using Western blotting and the results were analyzed using Image J. (C) The concentration of TGF-β in blood were tested by Elisa. Similar results were obtained in three independent experiments. Compared with normal group *p < 0.05.
M2 macrophages affect HSF function
In order to further explore the influence of M2 macrophages on fibroblasts, we co-cultured M2 macrophages with fibroblasts in vitro. THP-1 is a simplified model for studying the polarization of macrophages.
15
Excessive proliferation of fibroblasts in healing wound has been reported to cause the increase of collagen synthesis and the change of collagen morphology.
16
The expression of M2 THP-1 surface marker proteins Arg-1 and CD206 in M2 macrophages was detected by immunofluorescence. The results showed that the dual fluorescence intensity of Arg-1 and CD206 in M2 macrophages was strong (Figure 2(A)). After co-culture of M2-like TPH-1 and HSFs, we detected the EdU+ and migrated cells to examine the proliferation and migration capacity of HSFs. The results showed that compared with HSFs cultured alone in the control group, the proliferation capacity of HSFs co-cultured with M2-like THP-1 group was significantly increased, and their migration capacity was also significantly enhanced (Figures 2(B) and (C)). ELISA results showed that hydroxylysylpyridinoline (HP) and lysylpyridinoline (LP) content were increased in M2-like THP-1 group (Figure 2(D)). Then, we continued to explore the expression levels of Col-1, Col-3, and α-SMA in HSFs. The results showed that the expression of Col-1, Col-3 and α-SMA were significantly increased after co-culture with M0-like THP-1 or M2-like THP-1 (Figure 2(E)). These results suggest that M2 macrophages may promote scar formation by influencing HSFs function. M2 macrophages contributed the proliferation and migration of HSFs in vitro. HSFs were co-cultured with 20 μg/mL M2-THP-1, or 5 × 103 cells/well THP-1 for 48 h. (A) Double immunofluorescence of Arg-1 and CD206 was used to detect the surface marker protein of M2 type THP-1 in M2 macrophages. (B) EdU+ cells were determined by fluorescence microscope. (C) The migration capability of HSFs was evaluated using Transwell assay. (D) ELISA assay was used to measure the HP and LP content in the supernatant of the HSFs from each group. (E)The expression of Col1, Col3 and α-SMA was measured using Western blotting and the results were analyzed using Image J. Data were shown as mean ± SD of one representative experiment. Similar results were obtained in three independent experiments. Compared with control or co-M0 group *p < 0.05.
M2 macrophage-derived exosomes affected HSF function
To further investigate the effect of M2 macrophages on HSFs, M2-Exos were isolated from the supernatant of M2-like THP-1 cultured in vitro. Firstly, we examined the effect of M2-Exos on the viability of HSFs, and showed that the viability of HSFs was increased after M2-Exos treatment (Figure 3(A)). Since M2-Exos is an important way in the exchange of information between macrophages and other cells, we hypothesized that M2-Exos plays a role in the abnormal HSFs function. In the proliferation analysis, M2 Exos treatment significantly increased EdU+ cells in HSFs (Figure 3(B)). Transwell results showed that the migration ability of HSFs was significantly increased after M2-Exos treatment (Figure 3(C)). In addition, the protein levels of Col-1, Col-3 and α-SMA were significantly increased in HSFs treated with M2-Exos (Figure 3(D)). These results suggested that M2-Exos promoted the abnormal HSFs function in vitro. M2 macrophages derived exosome accelerated the autophagy of HSFs in vitro. Exosome were isolated from M2-THP-1, and treated HSFs with 20 μg/mL exosome. (A) The cell viability of HSFs was evaluated using CCK-8 assay after exosome addition. (B) EdU+ cells were determined by fluorescence microscope. (C) The migration capability of HSFs was evaluated using Transwell assay. (D) The expression of Col1, Col3 and α-SMA was measured using Western blotting and the results were analyzed using Image J. Data were shown as means ± SD of one representative experiment. Similar results were obtained in three independent experiments. Compared with Vehicle group *p < 0.05.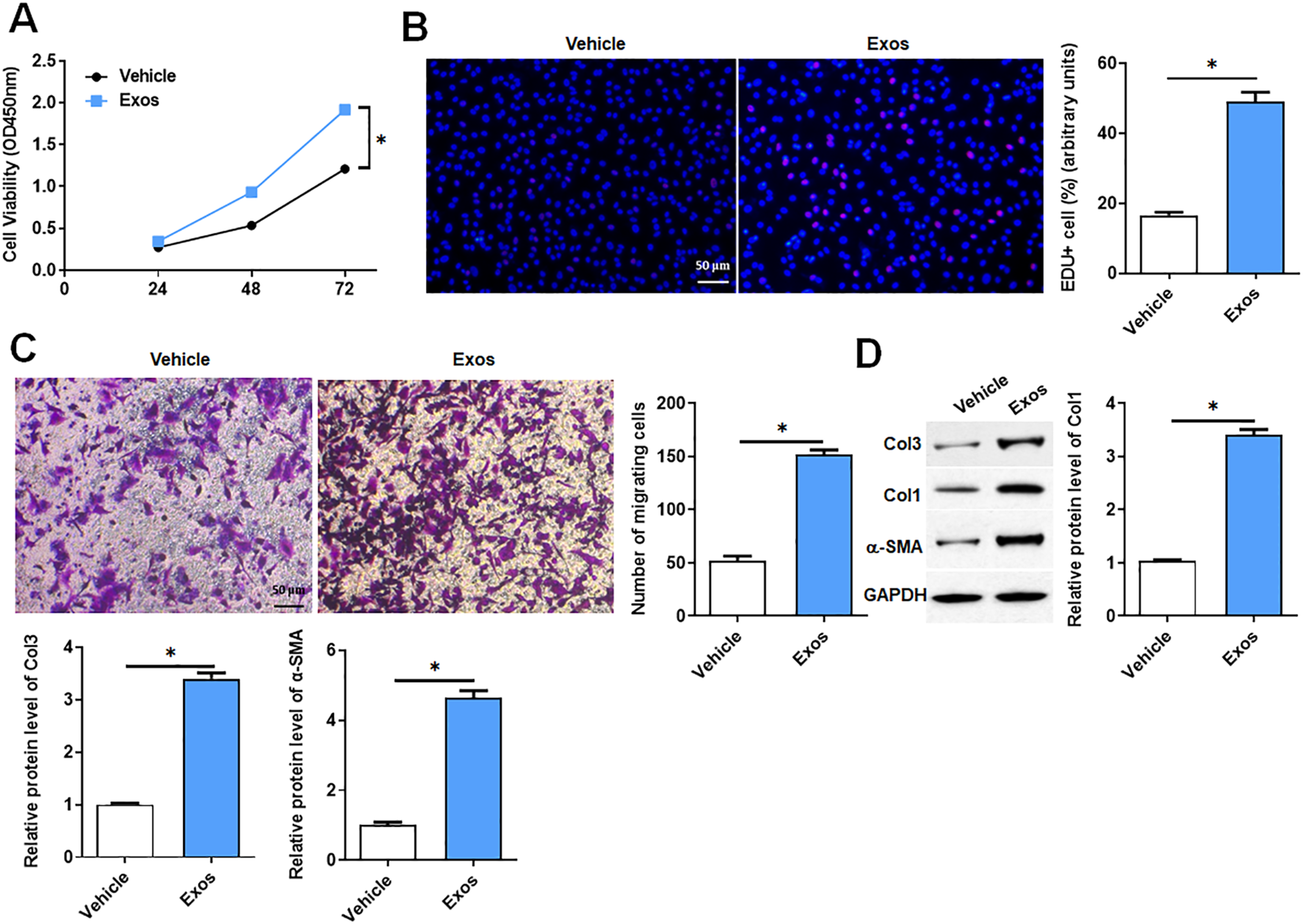
Exogenous addition of CXCL2 promoted HSF proliferation and migration
Next, we explored the reason of profibrotic properties in M2-Exos. Previous study has reported that the mRNA and protein level of CXCL2 was significantly up-regulated in M2 macrophage in vivo and THP-1
17
Since we had shown that M2-Exos promoted HSFs proliferation and migration, as an important component of M2-Exos, we hypothesized that CXCL2 takes an essential part in HSFs hyperproliferation. We first examined the expression of CXCL2 in M2-Exos. As shown in Figure 4(A), the expression of CXCL2 was increased in M2-Exos secreted. When the HSFs were subsequently treated with 1 ng/mL or 10 ng/mL CXCL2, the proliferation and migration capacity of HSFs were significantly increased, shown as more EdU+ cell and migrated cells (Figure 4(B) and (C)). We further found that the expression of Col-1, Col-3 and α-SMA were increased in HSFs treated with CXCL2 (Figure 4(D)). Moreover, exogenous CXCL2 increased the concentrations of IL-1β, IL-6 and TNF-α in the HSFs (Figure 4(E)). These results suggest that CXCL2 regulated the function of HSFs. CXCL2 accelerated the activation of HSFs in vitro. Exogenous CXCL2 was addicted to HSFs, with 1 and 10 ng/mL for 48 h. (A) The expression of CXCL2 in M2-Exos was measured using Western blotting and the results were analyzed using Image J. (B) EdU+ cells were determined by fluorescence microscope, and the statistical results were shown in right. (C) The migration capability of HSFs was evaluated using Transwell assay, and the statistical results were shown in right. (D)The expression of Col1, Col3 and α-SMA was measured using Western blotting and the analyzed results. (E) The concentration of Inflammatory factor (IL-1β, IL-6, TNF-α) in HSFs supernatant were tested by Elisa. Data were shown as mean ± SD of one representative experiment. Similar results were obtained in three independent experiments. Compared with control group *p < 0.05.
CXCL2-enrich M2 macrophage-derived exosomes affected HSF autophagy
We used CXCL2-neutralizing antibodies (anti-CXCL2) to examine whether CXCL2 is the key cytokines that M2-Exos influence HSFs function. M2-Exos and anti-CXCL2 were added to HSFs alone or in combination, and CXCL2 protein level was detected by Western blotting. The results showed that M2-Exos significantly promoted the expression of CXCL2, whereas anti-CXCL2 reversed this effect (Figure 5(A)).As the result in Figure 5(B), the anti-CXCL2 significantly attenuated the promotion of M2-Exos on proliferation of HSFs. In the cell migration experiment, the anti-CXCL2 also reduced the migration of HSFs influenced by M2-Exos (Figure 5(C)). Subsequently, we also detected the expression of Col-1, Col-3 and α-SMA. The results showed that anti-CXCL2 reversed the promotion of M2-Exos on the expression levels of Col-1, Col-3 and α-SMA in HSFs (Figure 5(D)). CXCL2 promoted the autophagy of HSFs. HSFs were treated with 1 μg/mL CXCL2 antibody, M2-Exos for 48 h. (A) Western blotting measured the expression of CXCL2 in M2-Exos and the analysis results. (B) EdU+ cells showed with fluorescence microscope and the statistical results. (C) Transwell assay of HSFs and the statistical results. (D)The expression of Col1, Col3 and α-SMA was measured using Western blotting and the analyzed results. (E) The relative expression of LC3 II/I and Atg5 was measured using Western blotting and the analyzed results. Data were shown as mean ± SD of one representative experiment. Similar results were obtained in three independent experiments. Compared with Exo or control group *p < 0.05.
Autophagy is a process in which the body engulfs and degrades its own organelles, and is an important biological process in cell metabolism and renewal. 18 It has been reported that autophagy can regulate the function of fibroblasts during HS. 19 We detected the expression of LC3 II/I and Atg5 in HSFs when treated with M2-Exos and anti-CXCL2. The results showed that the levels of LC3 II/I and Atg5 in HSFs were significantly increased after treatment with M2-Exos, whereas anti-CXCL2 reversed the effect of M2-Exos (Figure 5(E)). This result once again proved that CXCL2 was a key cytokine in the effect of M2-Exos on HSFs, and that M2-Exos regulated HSFs autophagy through CXCL2.
CXCL2 regulated fibroblast autophagy by up-regulating CXCR7
To understand the mechanism of HS mediated by CXCL2, we evaluated the effect of CXCL2 on the chemokine receptor CXCR7. First, we treated HSFs with anti-CXCR7 and detected the transcription level of CXCR7. The results showed that anti-CXCR7 significantly inhibited the level of CXCR7. (Figure 6(A)). We found that the proliferation and migration ability of HSFs were significantly increased when CXCL2 was added alone, whereas anti-CXCR7 reversed the effect of CXCL2 (Figure 6(B) and (C)). In addition, we evaluated the involvement of CXCR7 in the regulation of CXCL2-induced HSFs differentiation and collagen synthesis. Western blotting showed that CXCL2 increased the expression of Col-1, Col-3 and α-SMA, and anti-CXCR7 restored the effect of CXCL2 (Figure 6(D)). These results suggest that CXCR7 was critical in CXCL2-mediated HSFs differentiation and collagen synthesis. Upregulation of CXCR7 was key to autophagy of HSFs. HSFs were treated with 10 ng/mL CXCL2, 1 μg/mL anti-CXCR7, both CXCL2 and anti-CXCR7 for 48 h. (A) The expression of CXCR7 in HSFs was measured using qPCR. (B) EdU+ cells were determined by fluorescence microscope, and the statistical results were shown in right. (C) The migration capability of HSFs was evaluated using Transwell assay, and the statistical results were shown in right. (D) The expression of Col1, Col3 and α-SMA was measured using Western blotting and the results were analyzed using Image J. (E) The relative expression of LC3 II/I and Atg5 was measured using Western blotting and the analyzed results. Data were shown as mean ± SD of one representative experiment. Similar results were obtained in three independent experiments. Compared with CXCL2 or control group *p < 0.05.
Since the mechanism of CXCR7 on HSFs differentiation and collagen synthesis is still unclear, we hypothesized that CXCR7-mediated HSFs differentiation and collagen production may be related to autophagy. The results showed that CXCL2 promoted the expression of autophagy-related proteins LC3 II/I and Atg5 in HSFs, while anti-CXCR7 reversed this effect of CXCL2 (Figure 6(E)).Together, these results suggest that autophagy was regulated by the CXCL2/CXCR7 axis in HSFs.
CXCL2 affected fibroblast autophagy through the mTOR pathway
mTOR is composed of two components, mTORC1 and mTORC2.
20
Previous study has shown that the PI3K/Akt/mTOR pathway is closely related to autophagy in the development of many diseases.
21
In our results, CXCL2 increased the phosphorylation level of mTOR. When CXCL2 treated combine with Autophagy inhibitor (3 MA), the phosphorylation level of mTOR decreased comparing to CXCL2 treated group (Figure 7(A)). In the study of fibroblast proliferation, the addition of 3 MA reversed the abnormal proliferation of HSFs which caused by CXCL2, reducing the proportion of EdU+ cells (Figure 7(B)). In the cell migration experiment, 3 MA reduced the number of migrating cells of HSFs in CXCL2 group (Figure 7(C)). In addition, 3 MA also reduced the increased of the expression of Col-1, Col-3 and α-SMA in HSFs induced by CXCL2, (Figure 7(D)). These results suggest that CXCL2-mediated mTOR activation played an important role in fibroblast autophagy. CXCL2 promoted the activation of HSFs via PI3K/Akt/mTOR pathway. HSFs were treated with 10 ng/mL CXCL2, 5 mM 3MA, both CXCL2 and 3MA for 48 h. (A) The total and phosphorylated mTOR protein expression was analyzed by Western blotting. (B) EdU+ cells were determined by fluorescence microscope, and the statistical results were shown in right. (C) The migration capability of HSFs was evaluated using Transwell assay, and the statistical results were shown in right. (D)The expression of Col1, Col3 and α-SMA was measured using Western blotting and the results were analyzed using Image J. Data were shown as mean ± SD of one representative experiment. Similar results were obtained in three independent experiments. Compared with CXCL2 or control group *p < 0.05.
CXCL2 promoted HS grows in HS model
In above, CXCL2 had been shown to promote HSFs differentiation and collagen generation in vitro. Therefore, we will continue to explore the role of CXCL2 in the HS model mice. Since CXCL2 was released by M2-Exos, we evaluated the M2-Exos by subcutaneous injection into scar sites in mice. As shown in the figure, in the CXCL2 and M2-Exos significantly increased the scar area, collagen density and cross section size of mice (Figure 8(A)–(C)). Moreover, CXCL2 and M2-Exos significantly increased the number of α-SMA positive fibroblasts in scar tissue sections compared with the control group (Figure 8(D)). These results demonstrate that CXCL2 is involved in the deterioration of HS and may be a promising target for HS treatment. M2-Exos promotes experimental HS growth of mice in vivo via CXCR7/PI3K/mTOR pathway. (A) The size of mouse scars was Measured. (B) Collagen density of scar in mice was measured. (C) H&E staining. (D) The proportion of α-SMA cells was detected by immunofluorescence staining. (E) The CXCR7, total and phosphorylated PI3K, Akt and mTOR protein expression were analyzed by Western blotting, then the result was quantified by ImageJ. Data were shown as mean ± SD of one representative experiment. Similar results were obtained in three independent experiments. Compared with PBS group *p < 0.05.
The expression of PI3K and Akt were enhanced in scar tissue of CXCL2 and M2-Exos treated mice comparing with control group. The proportion of phosphorylated mTOR in total mTOR protein also increased significantly in CXCL2 and M2-Exos groups (Figure 8(E)). These results are consistent with those found on HSFs in vitro. It is suggested that mTOR pathway affects fibroblast transformation and collagen synthesis by influencing fibroblast autophagy in vitro and in vivo.
Discussion
Compared with normal healing skin, the number of M2 macrophages increased and M1 macrophages decreased in pathological scar tissue. The increase of early M2 macrophages resulted in deterioration of scar formation, suggesting that M2 macrophages promoted scar collagen generation and fibrosis in a certain way. Compared to other secreted products of macrophages, including growth factors, cytokines, chemokines, signaling molecules, and enzymes, is little known about the composition and function of macrophage derived exosomes. Existing reports have shown that M2-Exos play a role in tissue inflammatory response and fibrotic pathology. For example, M2 macrophages derived exosomes could activate CCL1/CCR8 pathway and stimulate the secretion of IL-4, thus alleviating colitis induced by dextran sulfate sodium. 22 In the past study on HS, the research from the perspective of exosome mainly focused on exosome contents. Li et al reported that exosomes derived from human adipose mesenchymal stem cells attenuated HS fibrosis by miR-192-5p. 23 Chemokines are also a key cytokine secreted by macrophages through exosomes. Here, we found that the number of M2 macrophages in HS patients increased, and M2 macrophage-derived exosomes promoted the proliferation and migration of HSFs. In addition, M2-Exos promoted the expression levels of Col-1 and α-SMA in HS tissues and HSFs. It is suggested that M2-Exos may promote scar formation by affecting the function of HSFs. Our study provided new theoretical support for targeting chemokines CXCL2 to regulate HS. Focusing on M2-Exo, this project studied the mechanism of M2-Exo rich in CXCL2 to regulate the differentiation of HSFs and autophagy in vitro, and explored the effect of CXCL2/CXCR7 axis that regulating autophagy in the treatment of HS. It is a relatively novel idea and attempt.
CXCL2 is involved in a variety of fibrotic diseases, including lung, liver, and skin fibrosis.11,24–26 However, the role of CXCL2 in the effect of macrophages on skin fibroblasts has been unclear. In this study, we reported the effect of elevated CXCL2 levels on HSFs. We found that CXCL2 promoted the proliferation and migration of HSFs and increased the expression levels of Col-1, Col-3 and α-SMA in HSFs. It is suggested that CXCL2 may induce HSFs differentiation to promote scar formation in mice, which provides new insights into HSFs and other fibrotic diseases. Previous study has shown that CXCR7 is closely related to the process of fibrosis. CXCR7 could stimulate the proliferation and migration of prostate fibroblasts. 27 At the same time, CXCR7 has also been found to reduce endothelial damage and improve fibrosis in lung endothelial cells, which is related to the inhibition of Jag1-Notch signaling pathway. 28 Our study filled the gap of CXCR7 in the direction of skin fibrosis and proposed the mechanism of CXCL2/CXCR7 synergistic regulation of HSFs differentiation. Here, we found that anti-CXCR7 reversed the induction of CXCL2 on the proliferation and migration of HSFs and the expression levels of Col-1, Col-3 and α-SMA in HSFs, indicating that CXCR7 plays a key role in CXCL2-mediated HSFs differentiation and collagen synthesis. In addition M2-Exos also influenced the level of inflammation in HSFs, and whether CXCL2 played a role in this process still needs to be further studied.
Autophagy regulates the process of wound healing and tissue repair, regulating the survival, differentiation, and apoptosis of fibroblasts, thus leading to the occurrence of HS and keloids. Shi et al. showed that decreasing the ratio of autophagy marker protein LC3 II/I by inhibiting Akt/mTOR signaling pathway contributed to inhibit pathological scar t issue and scar fibroblasts. 29 Another important study showed that the hypoglycemic drug lipliptin inhibited scar fibroblast differentiation and fibrosis through the mTOR signaling pathway, along with inhibition of cell proliferation and migration. 22 There is evidence that showed high relation between CXCR7 and several activated cells signaling pathways, including Akt. 30 It has been reported that the increased release of CXCL2 can activate downstream Akt signaling in microglia. 31 Various evidences had shown that CXCL2 and its ligand CXCR7 may be related to the program of cell autophagy, which may be realized through the Akt pathway. In our study, we found that M2-Exos promoted the expression levels of autophagy-relative proteins (LCII/LCI and Atg5), whereas anti-CXCL2 reversed this effect, suggesting that CXCL2 is a key cytokine of M2-Exos acting on HSFs, and M2-Exos regulates HSFs autophagy through CXCL2. CXCL2 promoted the expression of LCII/LCI and Atg5, whereas anti-CXCR7 reversed this effect, suggesting that CXCR7 may induce autophagy on downstream of CXCL2 in HSFs. In addition, inhibition of PI3K/Akt/mTOR signaling pathway could reduce the level of autophagy and inhibited the proliferation and collagen synthesis of fibroblasts in pathological states. Blocking autophagy could be an effective means to inhibit the proliferation and collagen synthesis of fibroblasts.
Exosomes have the advantages of higher safety, easier storage, and transportation, rapid and efficient, no ethical restrictions, and wide sources. 32 In recent years, exosomes have become promising diagnostic biomarkers in dermatology, which have clinical efficacy in wound healing, skin aging and treatment of scars. Clinical study has confirmed that exosomes have a significant improvement effect on skin wrinkles, elasticity, and pigmentation. 33 Exosomes from different sources also have a significant improvement in wound healing and acne scars, and have no adverse reactions.34,35 The results of this study demonstrated that M2-Exos further induced scar formation by promoting collagen deposition in HSFs, and this effect of M2-Exos is mainly achieved by regulating the downstream CXCL2/CXCR7/mTOR pathway. It can be seen that targeting the M2-Exos/CXCL2/CXCR7/mTOR pathway may be an effective way to treat skin scars. The above mechanism provides a new method for the realization of cell-free treatment of pathological scars, and provides a new treatment strategy for M2-Exos clinical treatment of hypertrophic scars. Although many studies have shown that exosomes are crucial in the pathogenesis of skin diseases, effective and large-scale production of exosomes for the treatment of skin diseases has not yet been explored. Therefore, in the future, we can continue to explore new targets for the treatment of skin diseases and improve the repeatability of exosome-related treatment. In addition, due to the heterogeneity of exosomes, it is challenging to interpret its special role in skin diseases. Therefore, exosome modification can also be carried out in the future to improve the efficacy of exosomes in the treatment of skin diseases.
In this study, M2-Exo was shown to promote HSFs progress, collagen deposition, and fibroblasts differentiation, and then in HS mouse model, M2-Exo also increased collagen synthesis. In addition, we demonstrated that CXCL2 in M2-Exo can aggravate fibrosis in HSFs and directly target CXCR7 in regulating the mTOR pathway in HS formation. Our study provided a new therapeutic strategy and clarifies a specific mechanism for the clinical treatment of HS.
Footnotes
Author contributions
Min Shi: Conceptualization, Investigation, Methodology, Writing-original draft. Lu Zhang: Methodology, Visualization, Software. Fangfang Bi: Methodology, Resources, Validation, Data curation. Xiaohong Ma: Conceptualization, Supervision, Writing-Review & Editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by Natural science Basic research project of Shaanxi Province (2022JM-558).
