Abstract
Objectives:
Differential expression of microRNAs (miRNAs) has been confirmed to promote hypertrophic scar (HS) formation. However, this mechanism remains to be further elucidated. This paper investigated the effects of microRNA26a on HS of rabbit ears or its mechanism.
Methods:
The rabbit HS models were established and randomly assigned to either the experimental group (20 rabbits with treatment through triamcinolone acetonide) or the scar group (20 rabbits without treatment). In addition, 10 unmodeled rabbits were served as control group. The expression of microRNA-26a in HS tissues was detected via a stem-loop real-time polymerase chain reaction (RT-PCR).
Results:
According to RT-PCR, we showed the decreased expression of microRNA-26a in the scar group compared with that in the experimental group, and in the experimental group compared with that in the control group (
Conclusions:
Our findings revealed that the increase of microRNA-26a expression might alleviate excessive inflammation during the HS formation, inhibit fibroblast proliferation and collagen deposition in HS, and promote the treatment of HS.
Introduction
HS is a pathological reaction of skin healing after trauma, which is characterized by abnormal proliferation of fibroblasts and excessive deposition of collagen fibers. 1 At present, there are many therapies for HS, including surgery, steroid injection, radiotherapy and application of laser technology, compression technology and silica gel preparation.2,3 Triamcinolone acetonide is a long-acting intradermal steroid, and the international scar management advisory committee recommends intradermal steroid injection for the treatment of keloid and HS. The intralesional injection of triamcinolone acetonide shows great prospects for the treatment of HS.4,5 At present, different treatment strategies have been tried in the treatment of HS, but there is no satisfactory treatment choice. 5 Therefore, a thorough understanding of the pathophysiological process of HS can provide a reasonable choice for the treatment of HS. MicroRNA is an endogenous noncoding RNA with a length of 18–25 nucleotides, which can regulate gene expression by inducing degradation or translation inhibition of target mRNA. As an epigenetic regulator, miRNA is closely related to the process of skin fibrosis. 6 miRNA-192-5p was found to reduce the fibrosis of hypertrophic scar tissue by targeting IL-17RA to regulate Smad signal transduction pathway in scar formation. 6 However, the effect of miRNA on scar through triamcinolone acetonide needs to be further explored. Previous studies have indicated that microRNA-26a may serve a key role in the growth and development of normal tissues, as well as in the pathogenesis of nonneoplastic diseases and tumorigenesis.7,8,9 It has been reported that microRNA-26a can inhibit the expression of Col I and Col III in HS and participate in the pathological process of HS. 10 Nevertheless, the functionary mechanism of microRNAs in HS formation remains to be further elucidated. 11 In this study, we examined the expression of miRNA-26a in rabbit HSs before and after treatment via triamcinolone acetonide in order to explore the correlation between the gene marker and HS formation, so as to provide a new target for HS treatment.
Methods
Animal models
Fifty Japanese big ear rabbits with an average body weight of (2.6 ± 0.73) kg were selected as experimental animals. Both male and female rabbits were provided by Animal Center of Medical Department of Xi’an Jiaotong University. Fifty rabbits were randomly assigned to the scar group (20 rabbits), experimental group (20 rabbits), or the control group (10 rabbits). Rabbits in the experimental group and the scar group had the hair behind their ears shaved off, and were anesthetized by an injection of 3% pentobarbital sodium into the ear vein. In the ventral part of the rabbit ear, a round wound with the size of 1 cm * 1 cm was formed by a perforator. Then the whole skin, subcutaneous tissue, and perichondrium of the rabbits were cut. Finally, the hemostasis and fixing with tape for rabbit ears were implemented with an unsutured incision. The formation of HSs was observed for 28 days. Afterwards, the rabbits in scar group were not treated. Triamcinolone acetonide (Zhejiang Xianchen Pharmaceutical Co., Ltd., Zhejiang, China) was injected into HSs in the experimental group twice a week, 4 mg each time. On the 42nd day after the first treatment, HSs of 1 cm * 1 cm in size were collected with a local subcutaneous infiltration anesthesia, and the rabbits in the control group were shaved with a local subcutaneous anesthesia to collect the normal tissues of the size of 1 cm * 1 cm. HSs and normal tissues were washed with normal saline and divided into two parts. One was fixed in 10% formaldehyde and routinely embedded in paraffin for pathological examination, and the other was frozen in liquid nitrogen for the analysis of Enzyme linked immunosorbent assay (ELISA) and RT-PCR.
Six scars were created on one ear of each rabbit, which was shown in Figure 1. Hypertrophic scar on rabbit ear. Six scars were created on one ear of each rabbit.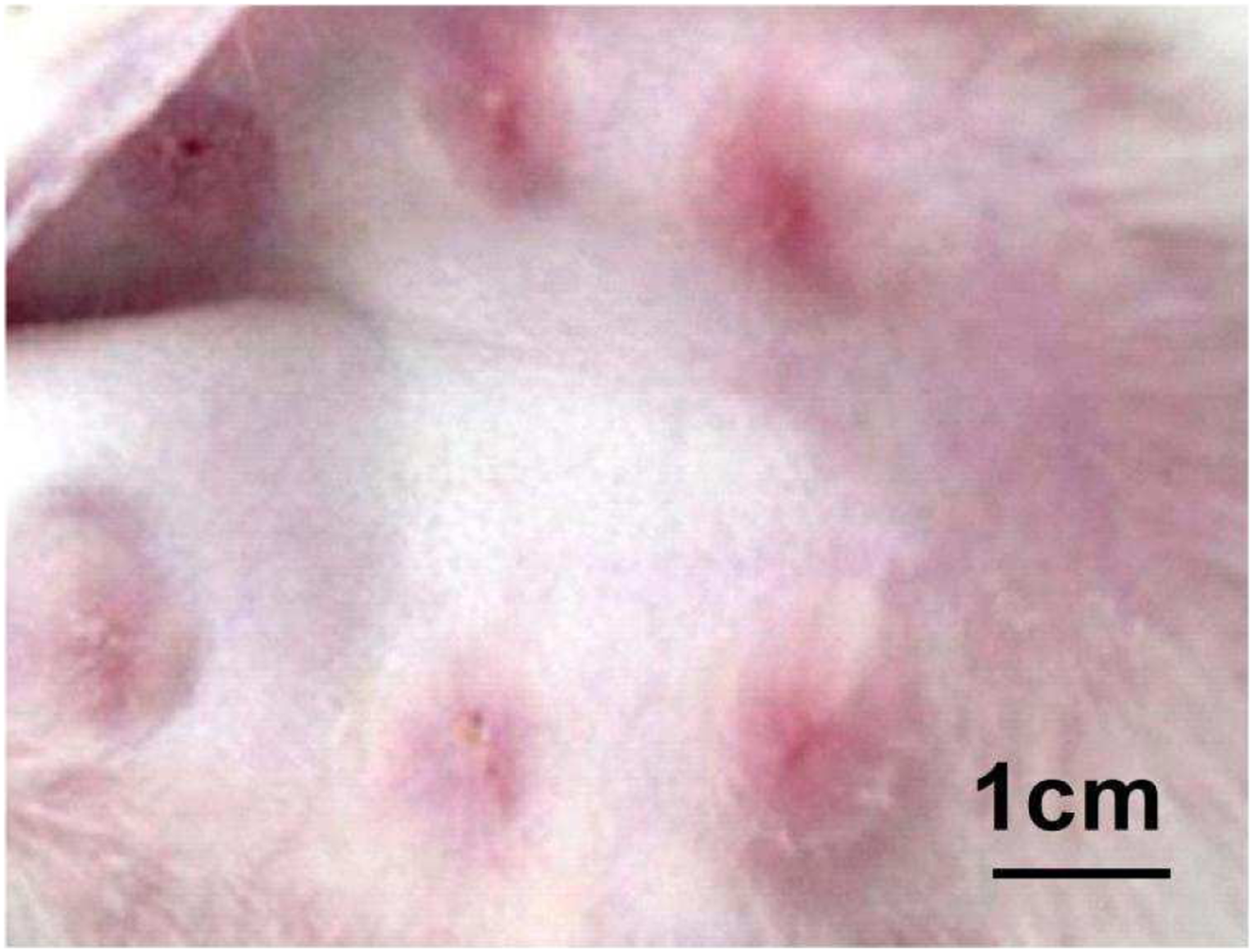
Experimental design
The STs of rabbit ears were measured by the color Doppler ultrasonic diagnostic instrument (GE company, USA; Figure 2). The HSs were stained by the staining of Van Gieson dye (Guangzhou Wiggs Biotechnology Co., Ltd., China) and Hematoxylin-eosin staining (Shanghai biyuntian Biotechnology Co., Ltd, China). Ultrasonic image of rabbit ear. (a-h) showed the ultrasonic image of rabbit ear on the 1st, 3rd, 5th, 7th, 11th, 16th, 20th and 28th day after operation. The oval area was the location of the scar.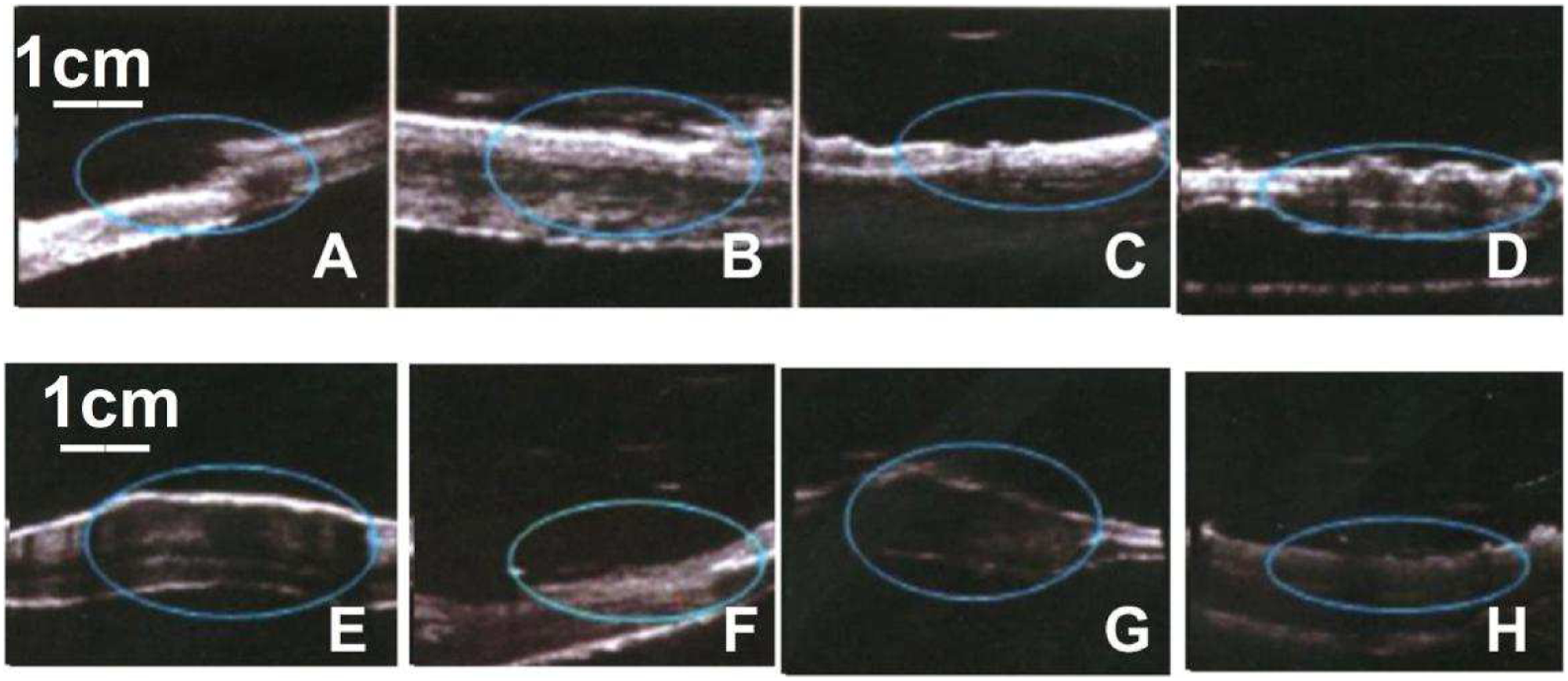
Hematoxylin-eosin staining: A 5-μm-thick paraffin tissue was cut and dewaxed to water, following a 20-min dye with Weigert’s hematoxylin. After rinsing with the running water for 10 min, the tissue was stained with eosin and finally installed under the cover glass, and observed under a 100x magnification optical microscope (Olympus Company, Japan). The number of cells in three different visual fields was recorded, and the mean value was taken as NFs.
Van Gieson staining: After dehydration and rehydration, the paraffin tissue was stained in Verhoeff solution for 1 h, washed, and then differentiated in 2% ferric chloride for 2 min.
The tissue should be completely blackened under the microscope, then treated with 5% sodium thiosulfate for 1 min, washed and counterstained in Van Gieson solution for 3–5 min. Finally, the tissue was dehydrated and sealed. Then the collagen distribution was observed under a 200x magnification optical microscope.
A HS of 2 mm * 2 mm in size was cut, and then grinded in a mortar with the appropriate amount of Tris buffer. The tissue homogenate was centrifuged at a speed of 2000 RPM for 20 min, and the supernatant was collected. The levels of Col I, Col III, IL-6, and TNF-α were detected by ELISA (R & D Company, USA). The operation was strictly carried out according to the kit instructions.
Treatment of rabbits after experiment
Sample evaluation of researchers
All the researchers did not know the grouping status and result evaluation of the experimental specimens in the process of the experiment.
Real-time polymerase chain reaction
Hypertrophic scars of 100 mg were cut and added to 1 mL of Trizol solution with a grind and homogenization on ice, where RNA extraction was performed according to the instructions (Takara, Dalian, China). Ultraviolet spectrophotometer (Beckman Company, USA) was used to detect the absorbance vales of RNA extract at 260 nm and 280 nm in wavelength. If the ratio of two absorbances was between 1.8 and 2.0, the extraction could be considered without impurities for amplification experiment. PCR primers were designed by Premer Primer 5 software and synthesized by Shanghai Yinghuaijie Company. U6 was used as internal reference, its sequences of primers were as follows: Forward 5′-TCACCATCTTCCAGGAGCGA-3′, Reverse: 5′-CAATGCCGAAGTGGTCGT-3′. The primer sequences of microRNA-26a: Forward 5′-CAAGUAAUCCAGGAUAGG-3′, Reverse 5′-GGCCAACCGGAAGATG-3′. PCR was carried out according to the standard protocol on a real-time PCR-system (Applied BioSystems). PCR reaction system: ddH2O 12 μL, buffer solution 2 μL, dNTP 1 μL, upstream and downstream primers 1 μL, cDNA 3 μL, Taq enzyme 1 μL, total reaction system was 20 μL. After an initial incubation of 10 μL reaction mixtures for 1 min at 95°C, 40 cycles (95°C for 20 s, 58°C for 25 s per cycle) were performed for amplification. The amplified products were performed electrophoresis in 3% agarose gel and analyzed via the gel imaging system. Referring to the cyclic threshold (CT) of U6, the CT changes (△CT) of miRNA-26a was calculated, and the expression of miRNA-26a was obtained by 2-△ct.
Statistical analysis
Data were analyzed by Graphpad 6 software (USA). Continuity data are expressed as mean ± standard deviation (
Results
Formation of HSs on rabbit ears
After 28 days, 240 successfully formed HSs were selected on rabbit ears (Scar elevation index > 1 12 ). HSs were red, elevated from surrounding tissues and stiff on palpation (Figure 1). Subsequently, HSs were assigned to two groups: the scar group (120 HSs) and experimental group (120 HSs).
Triamcinolone acetonide improves appearance of scar
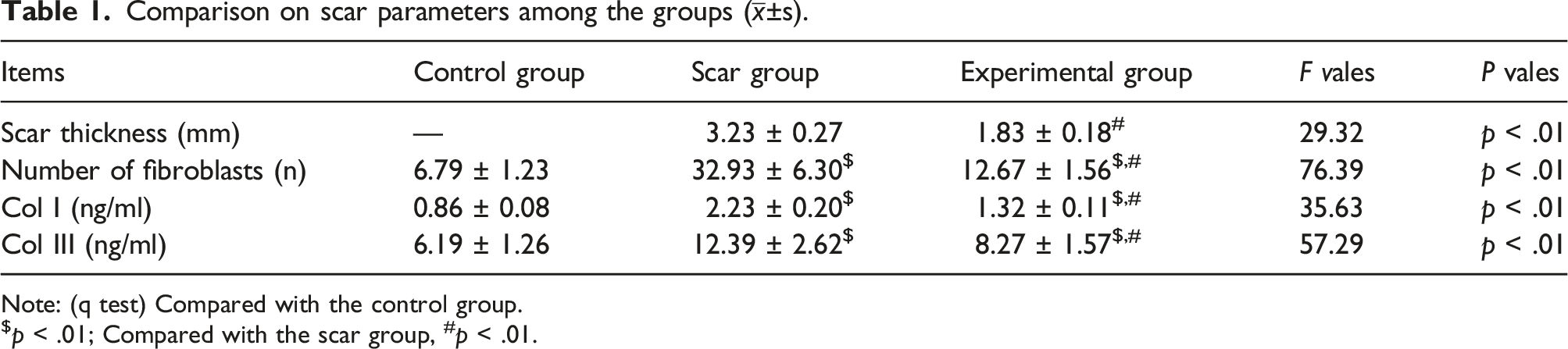
As shown in Figure 3 and Table 1. The experimental group (Figure 3(b)) had softer, shallower and thinner HS than the scar group (Figure 3(a)). The appearances of HS. (a) showed the appearance of HS in the scar group on the day prior to treatment, (b) showed the appearance of HS in the experimental group on the 42nd day after the first treatment. HS = hypertrophic scar. Comparison on scar parameters among the groups ( Note: (q test) Compared with the control group. $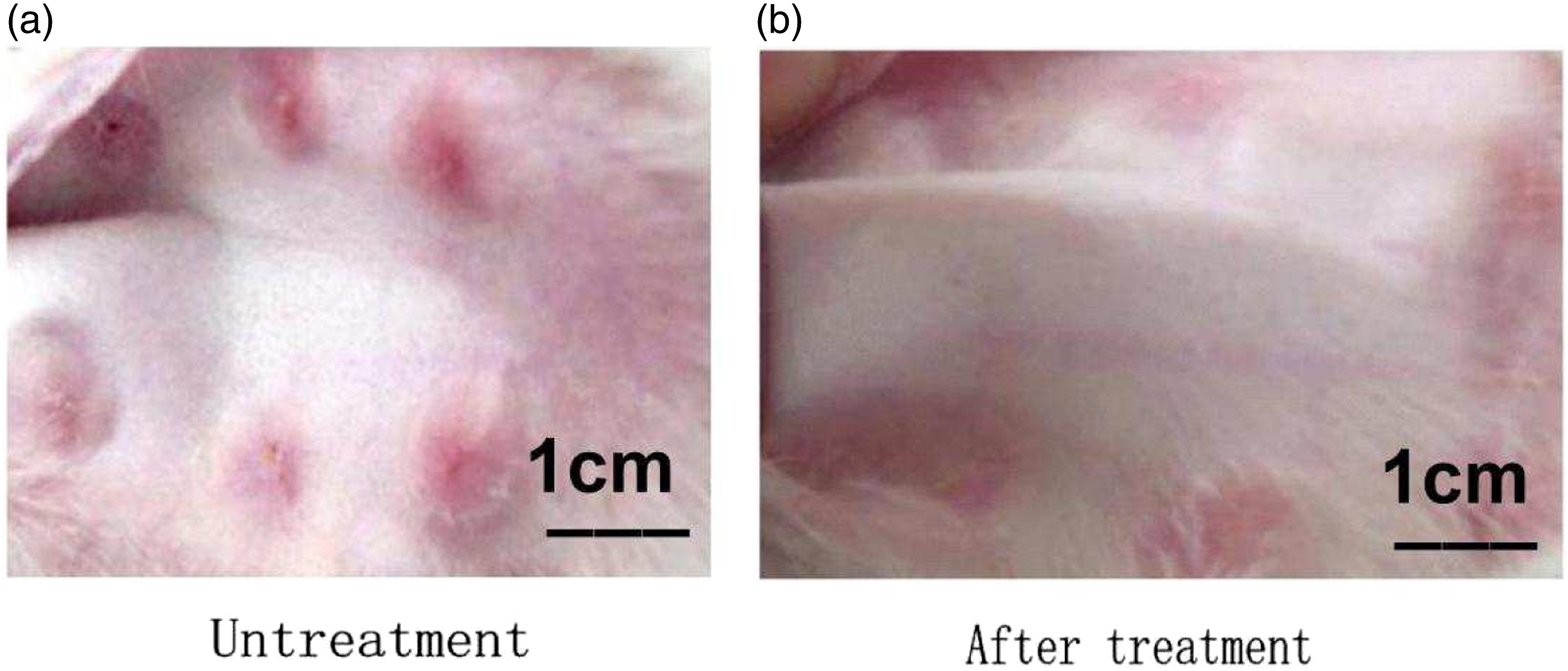
Scar parameters
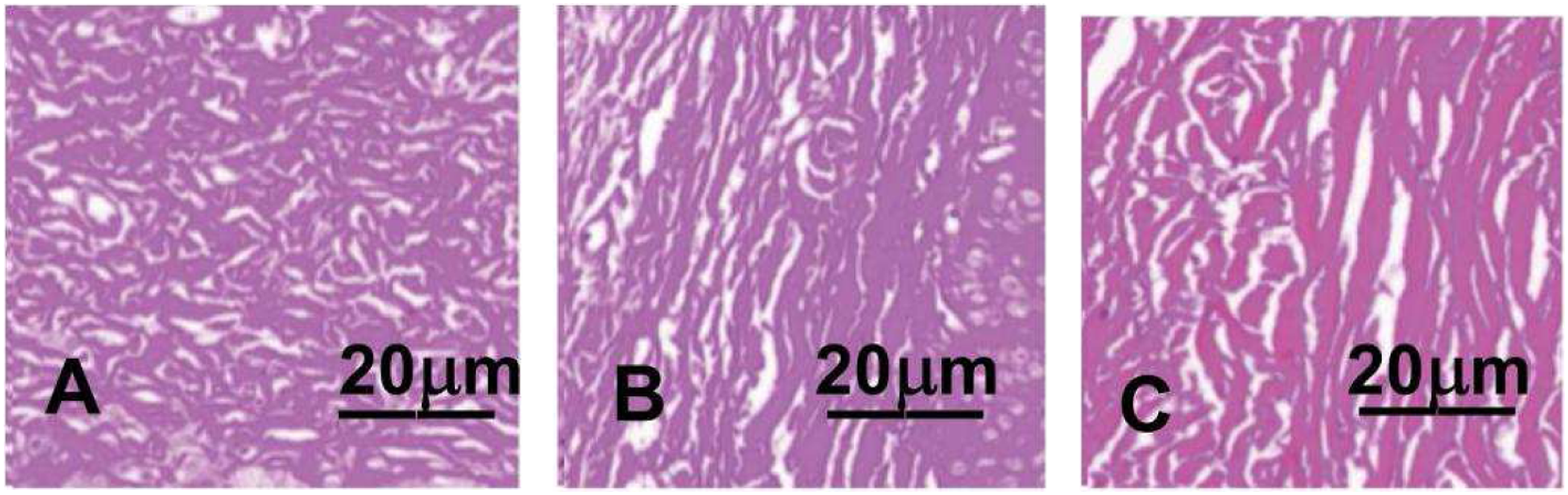
To investigate whether treatment improved HSs, Van Gieson staining was conducted. As shown in Figures 4 and 5 and Table 1. In the scar group, scar structure was compact, collagen was abundant and arranged disorderly, and fibroblasts were increased. The collagen and fibroblasts were less in the experimental group than those in the scar groups and more than those in the control group. The appearance of Hematoxylin-eosin staining for HS. (a, b) and (c)showed the appearance of Hematoxylin-eosin staining at a 100x magnification for HS on rabbit’s ear in the scar group, the experimental group and the control group, respectively. Scar group (a) showed irregular and abundant fibroblasts; Fibroblasts decreased in experimental group (b) and decreased significantly in control group (c). HS = hypertrophic scar. The appearance of Hematoxylin-eosin staining for HS. (a, b) and (c)showed the appearance of Van Gieson staining at a 200x magnification for HS on rabbit’s ear in the scar group, the experimental group and the control group, respectively. The collagen fibers in scar group (a)were denser, thicker and more disordered. Typical fascicular collagen fibers appeared in the experimental group (b), while the collagen fibers in the control group (c) were orderly and less dense. HS = hypertrophic scar.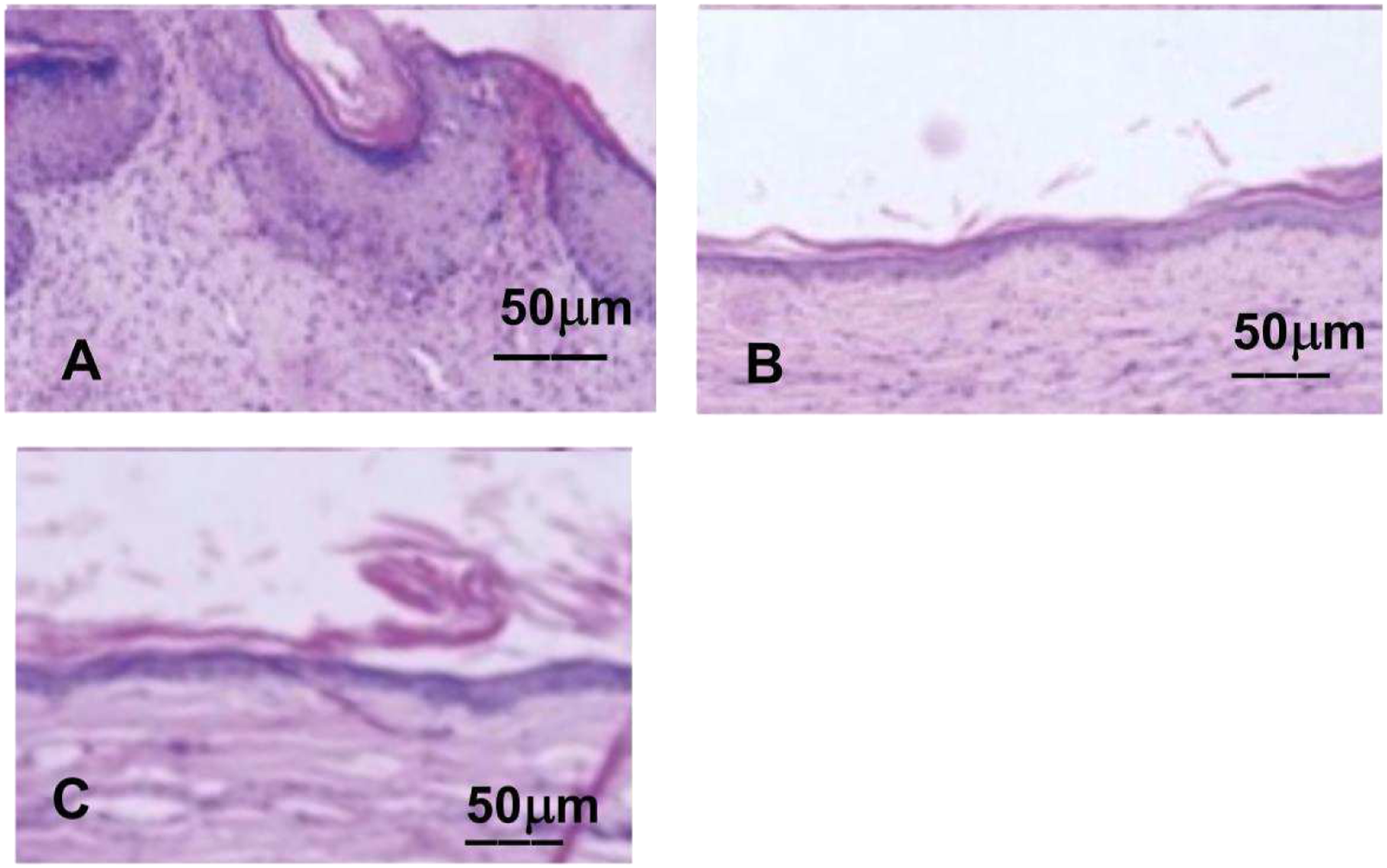
Our results showed that the levels of STs, NFs, Col I, and Col III in the experimental group were significantly lower than those in the scar group (all The scar parameters among the groups were presented. b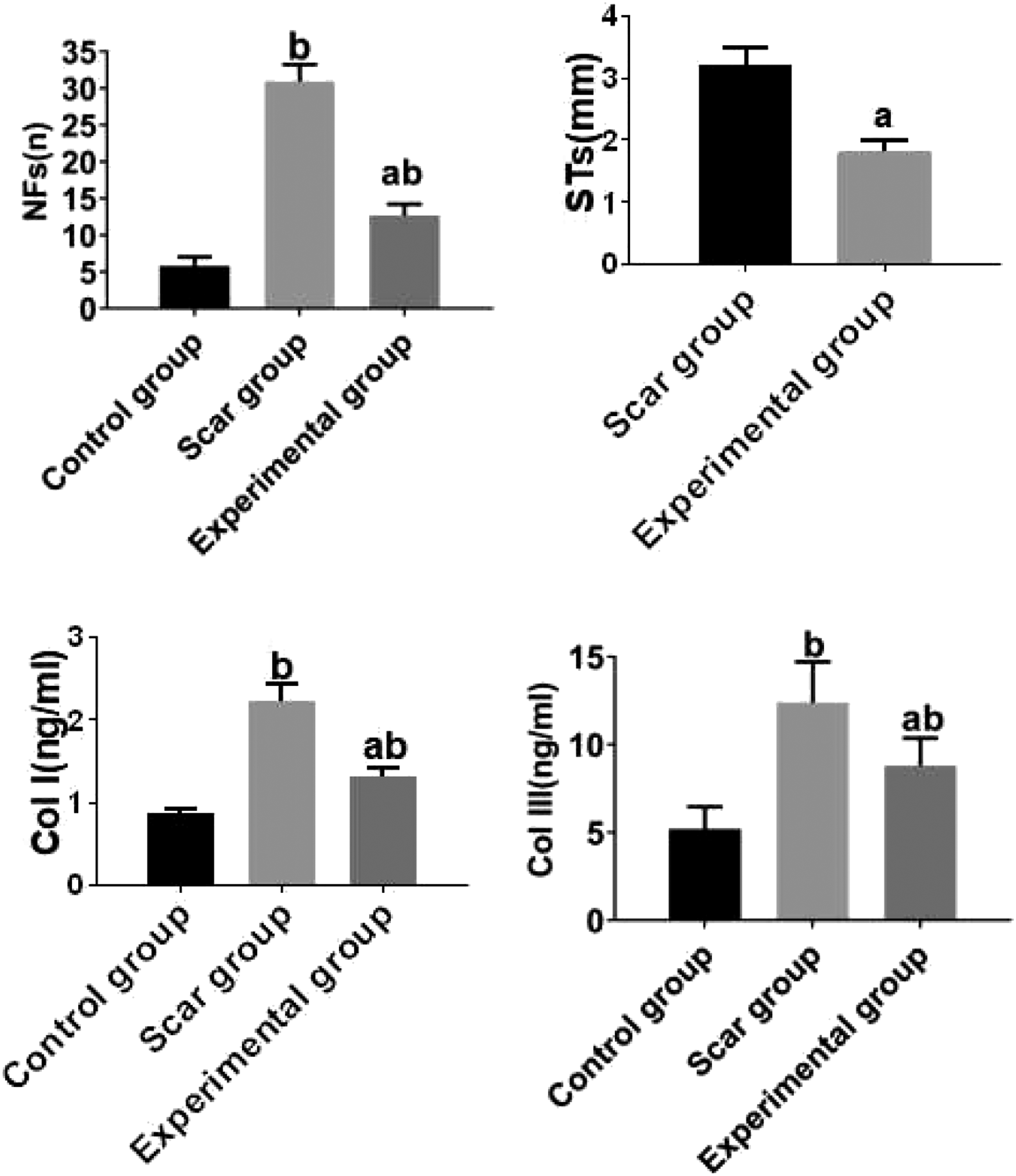
Inflammatory factors
Comparison on the levels of IL-6 and TNF-α as well as expression of microRNA-26a among the groups (
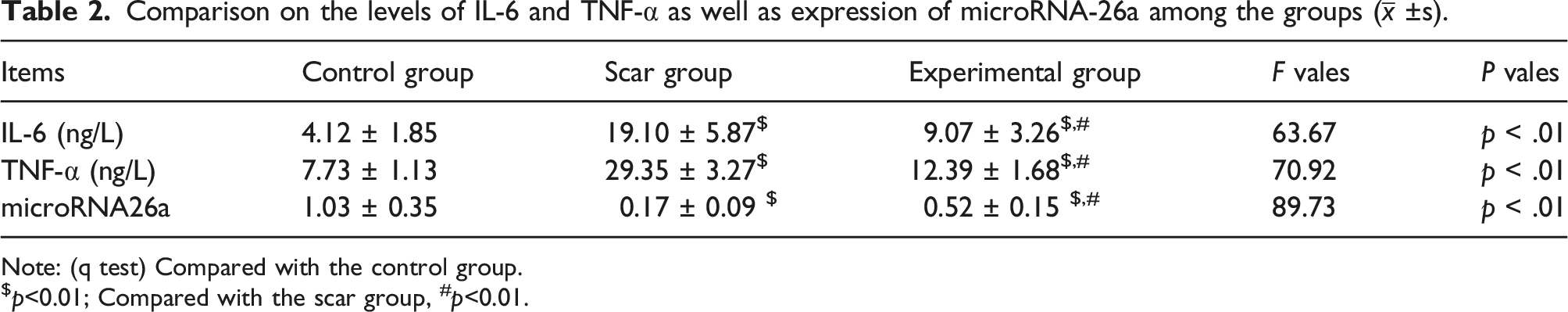
Note: (q test) Compared with the control group.
$
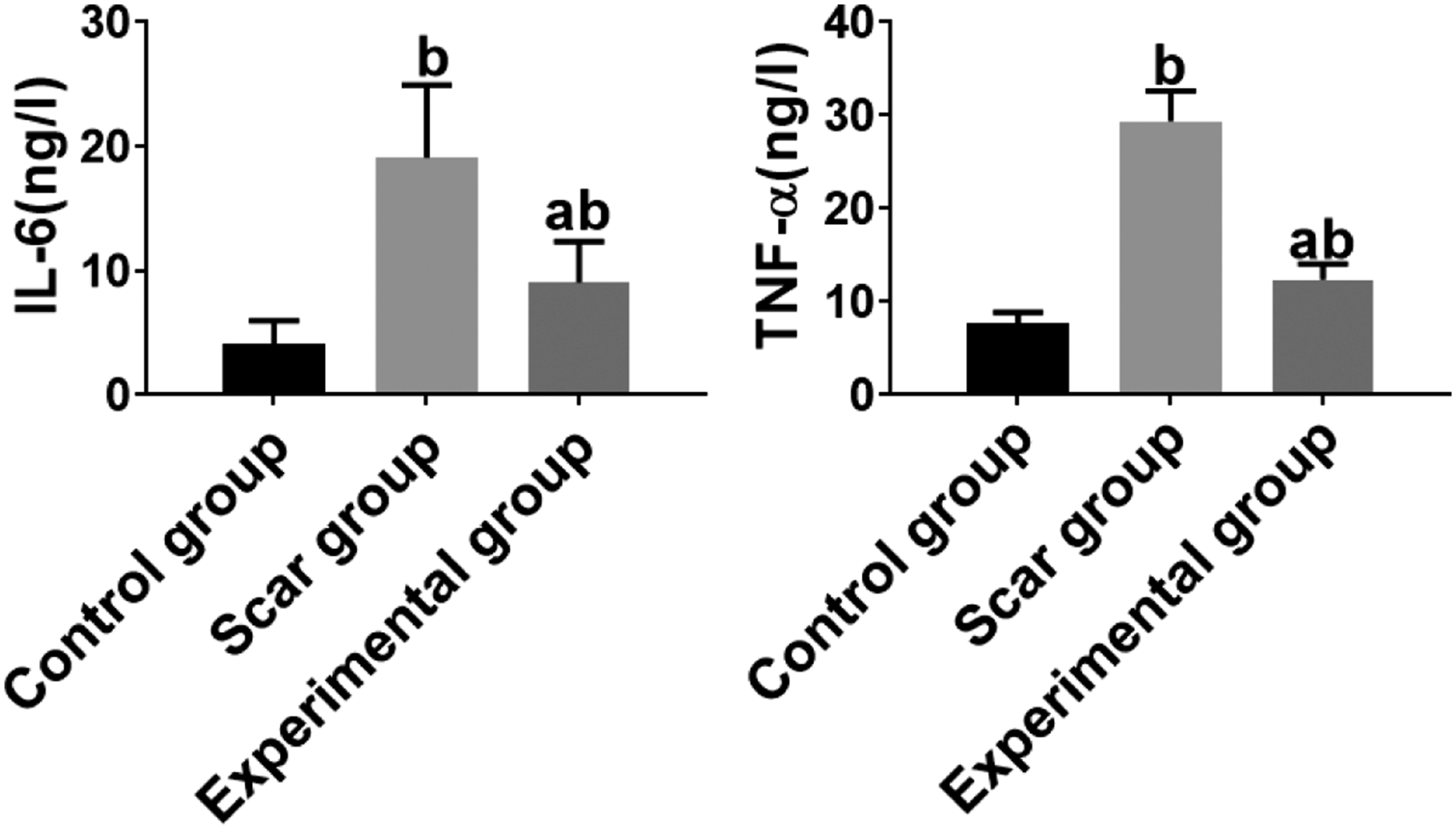
The inflammatory factors among the groups were presented. b
Expression of microRNA-26a
To verify the therapeutic effect of Triamcinolone acetonide on HSs, the expression of microRNA-26a was detected by RT-PCR. These results suggested that Triamcinolone acetonide significantly increased the expression of microRNA26a.However, the expression of microRNA-26a in the untreated scar group was significantly lower than that in the control group (all Comparison of the expression of microRNA-26a among the groups. (a) showed the gene expression of microRNA-26a and (b) showed the electrophoresis status of the amplified products of microRNA 26a by real-time polymerase chain reaction. b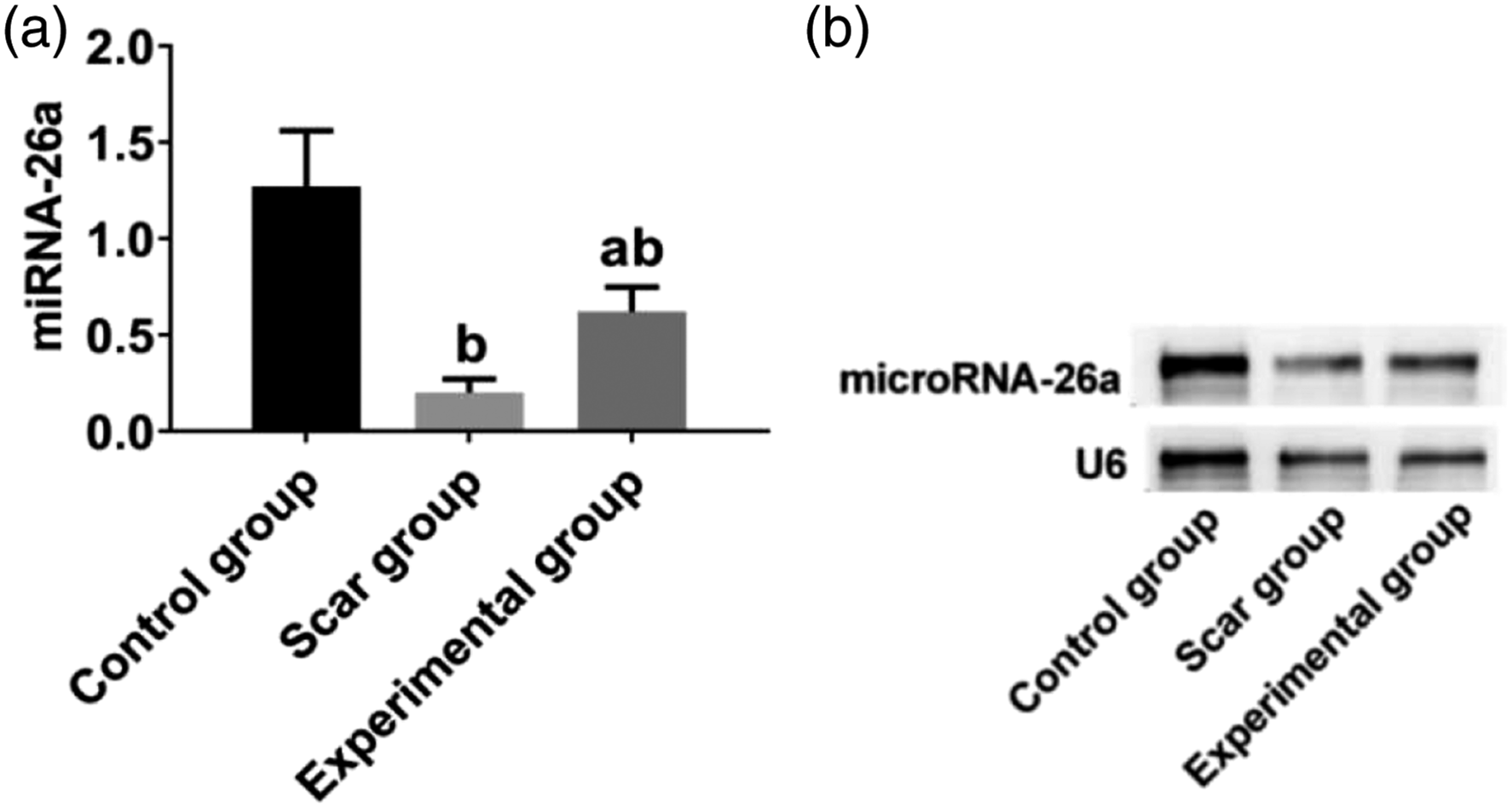
Correlation analysis
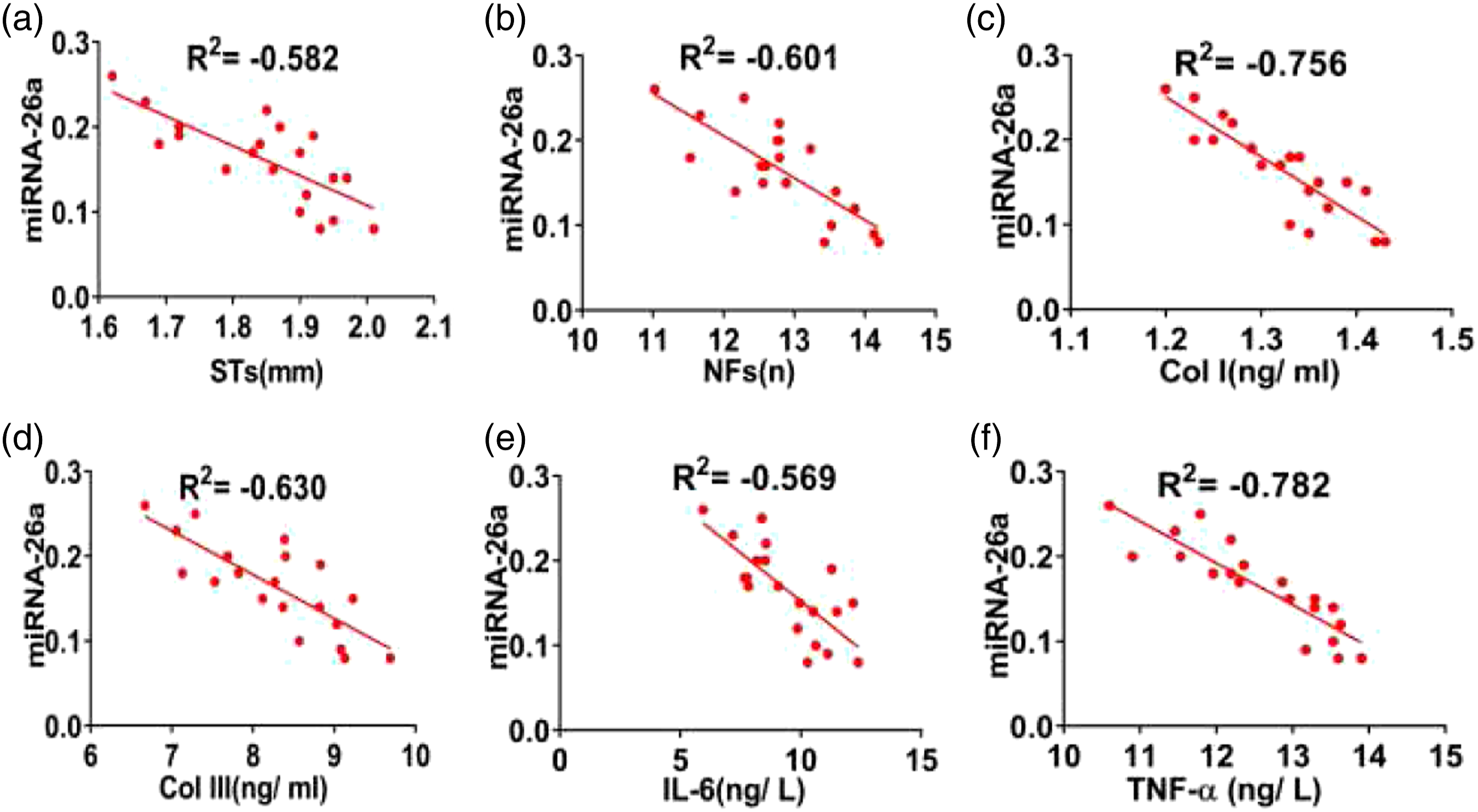
In the scar group, the expression of microRNA-26a was negatively correlated with STs, NFs, Col I, Col III, IL-6, and TNF-α (all Correlation of microRNA-26 in the scar group. (a–f) showed the correlation between microRNA-26a and STs, NFs, Col I, Col III, IL-6, and TNF-α in the scar group (respectively, all Correlation of microRNA-26a in the experimental group. (a–f) showed the correlation between microRNA-26a and STs, NFs, Col I, Col III, IL-6, and TNF-α in the experimental group (respectively, all 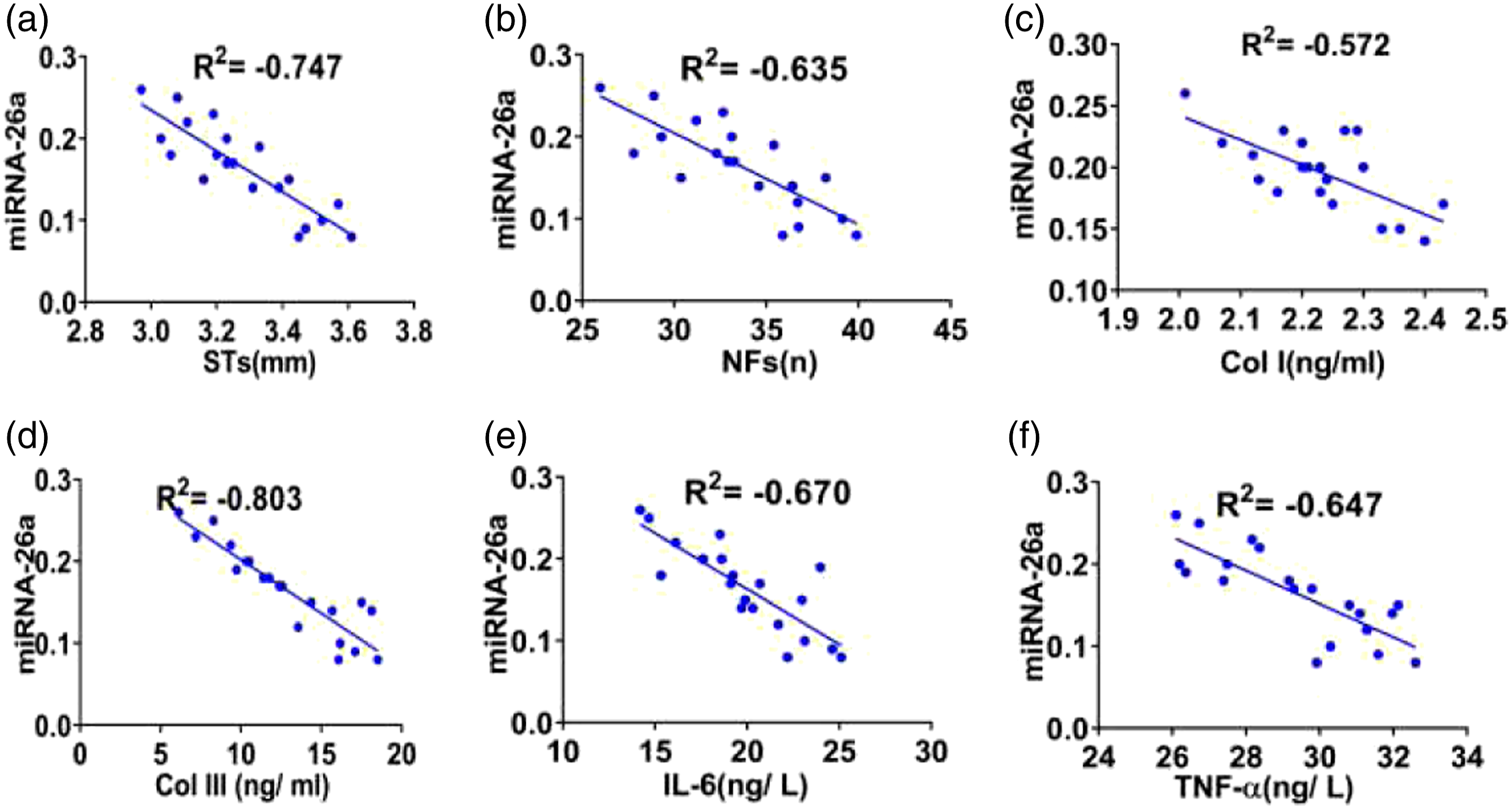
Discussion
Hypertrophic scar is a pathological reaction produced by the body to repair damaged skin tissue. It is characterized by excessive proliferation of fibroblasts, excessive synthesis, deposition of collagen fibers, and neovascularization. These pathological conditions respond to overproliferation of fibroblasts with a secretion of a large number of extracellular matrix where there are rich Col I and Col III.
1
The recent study suggested that the contents of Col I and Col III in tissues increased significantly during HS formation, which were directly proportional to the degree of scar hyperplasia.
13
In addition, there was a similar evidence that the sign of HS formation was the increase of Col I and Col III, as well as the disorder of collagen fibers.
2
In this study, the contents of Col I and Col III in HSs were significantly higher than those in normal skin tissues (
The formation of HS is a normal physiological manifestation, which is accompanied by the healing and repair of wounds after skin damage. If accompanied by infection in this process, excessive or abnormal repair will occur as a consequence of forming pathological scars (including hypertrophic scars, keloids). At present, there are many ways to treat scars, but they cannot fundamentally achieve the purpose of preventing and treating pathological scars. Although its pathogenesis is not yet clear, the study of scar formation genes has gradually become an important topic with the deepening of gene research.14,15,16
Previous studies have shown that microRNA-26a plays a key role in the growth and development of normal tissues, non-neoplastic diseases, and tumorigenesis.8,9 It has been reported that microRNA-26a can inhibit the expression of Col I and Col III in HSs by acting on Smad2, the target gene of microRNA-26a, and reduce the excessive growth of fibrous tissues. 10 However, the opposite conclusion has also been reported. The expression of microRNA-26a in HSs was proved to be significantly down-regulated, which was different from that in normal skin tissues. 10 Overexpression of microRNA-26a has been shown to inhibit the proliferation of extracellular matrix fibroblasts in HSs and to down regulate the expression of Col I and Col III. A recent study showed that expression of microRNA-26a was significantly down regulated in the HSs after glaucoma filtration surgery. 17 Overexpression of microRNA-26a has been proved to reduce the expression of collagen fibers in keloid and to restrain the occurrence of skin fibrosis. 18 In this study, the expression of microRNA-26a in HSs was significantly up- regulated after the treatment via triamcinolone acetonide, suggesting that microRNA-26a might be involved in the development of HS and its pathological process. There were few reports on the relationship between microRNA-26a and HS formation in China, and the deeper mechanism needs to be further explored.
Inflammatory reaction may occur during wound healing and scar formation. It further leads to a large number of inflammatory cell infiltration and release of inflammatory factors, promotes the synthesis of Col I and col III in extracellular matrix, promotes the proliferation of fibroblasts, and thus leads to the formation of HS.2,19,20 The formation trend of HS may also be related to systemic inflammation, which is characterized by excessive secretion of IL-6 and TNF-α in HS.
21
In this study, the levels of IL-6 and TNF-α in HSs were shown to significantly increase, and to decrease significantly after treatment of triamcinolone acetonide. A evidence has also been showed by Lee, et al., where activation of TGF-β involves in inflammatory reaction, and long term over inflammation can increase the activity of fibroblasts and lead to excessive extracellular matrix. At this stage, platelet degranulation releases and activates TGF-β, especially TGF-β1, TGF-β2, platelet-derived growth factor, et al.
10
In 18 HS patients, Ginsenoside Rg3 inhibited the proliferation and collagen synthesis of HS fibroblasts, the TGF-β/Smad pathway, and the activation of IL-6 and TNF-α.
22
Recent research showed that microRNAs was related to TGF-β signaling and the inflammatory microenvironment in cancer.
23
Our correlation analysis demonstrated that the expression of microRNA-26a in HS for pretherapy and posttreatment were correlated with the levels of IL-6 and TNF-α (
Conclusions
In this study, our findings showed that the increased expression of microRNA-26a could reduce the recruitment of pro-inflammatory cells, alleviate the excessive inflammatory response during the formation of HS, inhibit the proliferation of fibroblasts and collagen deposition, and promote the treatment of HS.
Limitations
The current research is only a preliminary study on the effect of microRNA-26a on HSs, and further research is needed. For example, the effect of microRNA-26a on collagen and fibroblasts need to be further studied. In addition, the correlation between the expression of microRNA-26a and the clinicopathological parameters of HS patients need to be further studied. Because our research is based on the rabbit ear model, and the scar on the rabbit ear tissue also has limitations. Because the location and way between human scar and rabbit ear scar were different, the proliferative stage and healing stage are also different. In addition, all the results of this study were obtained in the laboratory, so the clinical application needs further study. At present, the first-line treatment of scar is triamcinolone acetonide injection, but there are many local adverse reactions. Among them, the incidence of skin atrophy caused by triamcinolone acetonide injection was 44%, and the incidence of local telangiectasia was 50%. 24 In recent years, intralesional injection of interferon alpha was often used to treat keloid. Although the recurrence rate is unclear, in most cases, more than 80% of the cases have improved. 25 In addition, this study did not evaluate the selected sample size through the power calculation. Although previous studies 10 has shown that microRNA-26a inhibits the formation of HS by targeting Smad2 and inhibiting the expression of extracellular matrix related proteins. In this study, we additionally emphasized the relationship between inflammation and microRNA-26a, and the role of inflammation in the formation of HS.
ORCID iD
Xiong Zhang https://orcid.org/0000-0002-1014-4126
Footnotes
Acknowledgements
Yi Liu, our colleague, master graduate students, offered writing assistance.
Authors’ Contributions
XZ, and LG implemented the experiment and written. XZ, YQ-L and LG performed design topic, and modifyed in reviewing. XZ and LG were responsible for the article. All the authors final approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study has been approved by the Ethics Committee from the Affiliated Hospital of Traditional Chinese Medicine University of Shaanxi Province and First people’s Hospital of Xianyang City (approval number 13/2019).
Consent to publish
All authors approved to submit this original article.
Animal welfare
All animal experiments were performed in compliance with the NIH Guide for the Care and Use of Laboratory Animals.
Protocol registration
This study has been carefully prepared (including research questions, main design features and analysis plan) before the experiment and filed with the ethics committee of our institution.
Availability of data and materials
All data generated or analyzed during this study are included in this published article. The raw data sets used and/or analyzed during the current study are available from the corresponding author and XZ on reasonable request.
Data access
The data of this study were obtained in the laboratory, and the clinical application needs further study.
