Abstract
Lead (Pb) has become a major environmental contaminant. There are several ways in which lead can enter the human body and cause toxic effects on human health. This review focuses on the impact of lead toxicity at prenatal and early life stages and its effect on neurodevelopment. Lead exposure to the developing foetus targets foetal neural stem cells. Hence, it has detrimental effects on developing neural and glial cells, adversely influencing cognition and behaviour. Lead has a profound influence on the movement of calcium ions (Ca2+), which can be attributed to most of the mechanisms by which lead affects neurodevelopment. There is no known safe threshold of lead exposure for children. Lead can affect foetal neurodevelopment leading to various neurological disorders, and neurotoxic effects on behavioural and cognitive outcomes. In this review, we discuss prenatal and early-life lead exposure, its mechanism, and consequences for neurodegenerative disorders such as Alzheimer’s disease and Parkinson’s disease in later stages of life. This review further highlights the importance of lead exposure during pregnancy and lactation periods as well as early development of the child in understanding the extent of lead-induced neurological damage to the foetus/children and the associated future risks.
Introduction
Lead is one of the major causes of environmental toxicity and has been the cause of various developmental and growth-related disorders in children. Lead is a naturally occurring metal in the Earth’s crust and is a common environmental pollutant. 1 The heavy use of lead in day-to-day products is attributed to its malleability, inability to corrode easily, and ability to form alloys with various metals. 2 Lead exposure during the early period of pregnancy and the prenatal period is of major concern and may lead to serious health hazards later in life. 3 Lead acts as a neurotoxic agent and affects NSCs at a critical point in neurodevelopment by reducing brain plasticity. 4 Over the years, the emission of lead and its concentration in the environment have increased, leading to a decrease in the permissible levels of lead in the environment. Lead is transported freely across the placenta and the immature foetal blood–brain barrier (BBB). According to previous studies, adverse cognitive outcomes are directly associated with in utero lead exposure.2,5 It can affect brain development through various processes, such as interruption of the trimming and pruning of synapses, migration of neurons, and formation of neuron-glia interactions, leading to a variety of neurological disorders, including AD and PD. 4 Studies have also shown that prenatal lead exposure is associated with a lower mental development index (MDI) at 2 years of age. In another study, lead exposure in children under 7 years of age was associated with lower IQ scores at 11–13 years of age. 5 In laboratory research, lead has been demonstrated to affect NSC proliferation both in vivo and in vitro. Dendrites in NSCs that matured into neurons had changed shapes according to a study on rats exposed to lead. However, no influence on the fate of the cell type was observed. 6 Lead alters nitric oxide synthase activity, which affects the vasculature of the brain, leading to an alteration in the serotonergic system. This can cause increased aggressive behaviour along with depression. 4 Studies have shown the effect of lead on second messengers such as cyclic AMP (cAMP) and protein kinase C (PKC). 7 Lead can also affect posttranslational histone modifications, which are significant in the process of gene expression and therefore the development of the nervous system. 8
Methods
A literature search was performed using the “Science Direct” and “PubMed” search engines from 2013 to 2023 to investigate the correlations between prenatal, perinatal, and postnatal lead exposure and neurotoxicity in children. The search was conducted using the individual keywords “Alzheimer’s disease,” “Parkinson’s disease,” “ADHD,” “brain,” “IQ,” “Behavioural problems,” “Cognitive problems,” and “Lead neurotoxicity” in children under 12 years of age. A total of 15,032 papers were obtained based on the keywords, and 24 papers were shortlisted on the basis of the inclusion and exclusion criteria (Figure 1). The PRISMA guidelines were followed to construct the inclusion and exclusion criteria. The papers shortlisted on the basis of inclusion and exclusion criteria and the associated effects on the brain and neurotoxic effects are shown in Table 1. Outlines the different ways that lead exposure can occur during pregnancy, as well as the ways that lead affects different genetic and epigenetic processes. It provides a basic overview of the numerous consequences and illnesses brought on by lead. Neurobehavioural and neurodegenerative disorders caused due to lead exposure at different stages of development.

Inclusion criteria
All related studies evaluated the role of lead in neurological and behavioural changes based on the title, abstract and full text with keywords. The inclusion criteria were as follows:
This paper presents a study of prenatal and early exposure to lead in animal and human models causing neurological diseases in children under 12 years of age. Neurodevelopmental disorders are more common in children under the age of 12 years. 9 Model organisms such as rats, mice, zebra fish, and C. elegans.
Diseases
Effects of lead toxicity on brain development, AD, PD, ADHD (attention deficit hyperactivity disorder), ASD (autism spectrum disorder), dementia, and the effect of lead on the forebrain, cerebellum, and hippocampus.
Exclusion criteria
Lead neurotoxicity in adults and teenagers (age > 12 years).
Neurological changes caused by the co-occurrence of lead and other metals/chemicals. Metals other than lead causing AD, PD, ADHD, ASD, dementia, and neurological damage.
Duplicate and review papers were excluded.
Sources of lead in the environment
Lead is a toxic metal that is present naturally in the Earth’s crust in significant amounts. It is introduced into the environment by natural means such as volcanic eruptions, geochemical and soil weathering, and mixing of soil with river water. Furthermore, mining and smelting can cause the release of lead in the form of dust that can contaminate the surroundings. 10 Lead is also used in various products, such as petrol, batteries, paints, cosmetics, pesticides, children’s jewellery and toys, lead-glazed ceramics, and pottery. The presence of lead in the air, dust, or soil can make it easy to inhale or ingest through water or food. 11 Soil acts as a major reservoir of this heavy metal, where it can accumulate through various natural and anthropogenic activities, such as battery manufacturing and recycling, soldering, ceramic manufacturing, burning or spillage of petrol, paint dust, use of pesticides and burning of coal. 12 Heavy metals such as lead are not degraded in soil by bacteria; hence, their retention in the soil is increased. This can cause the absorption of lead by crops growing in such soil, through which it can then be ingested via food. 13 Moreover, the accumulation of lead in soil affects the soil microflora. According to one study, secondary lead smelting involved in dismantling lead-acid batteries led to soil acidification due to the discharge of acidic wastewater. 14 This can further cause changes in the entire ecosystem of the region.
Another source of lead pollution is petrol/gasoline. A comparison of the exhaust emissions of leaded gasoline and unleaded gasoline revealed differences in the increased concentrations of hydrocarbons and carbon monoxide in the exhaust emissions, suggesting that lead promotes the release of other harmful contaminants.
15
The use of leaded petrol/gasoline increases exposure to toxic metals through the air in the form of vapour, aerosols, and particulate matter. This increases the lead content in the soil and dust, which further contaminates areas in proximity to roads or highways. Lead can also be deposited on nearby vegetation and in water bodies, which increases the risk of exposure through food and water.
16
According to one study, 75% of the lead in petrol or gasoline is emitted as exhaust.
17
Aerosol particles of lead larger than 10 μm settle in areas surrounding the source, such as roadside soil, trees, and pavements, and studies have shown that lead levels locally increase and decrease as the distance from the road increases.
18
Particles less than 10 μm in size can be collected from buildings, soil, and other surfaces. Furthermore, the lead used in pipes can also contribute to the presence of lead in water. Figure 2 shows the possible sources of lead poisoning, and the neurological effects caused in the infants. Provides a thorough explanation of the quantity of papers retrieved from databases, the criteria used to include and exclude papers based on keywords, and the research models.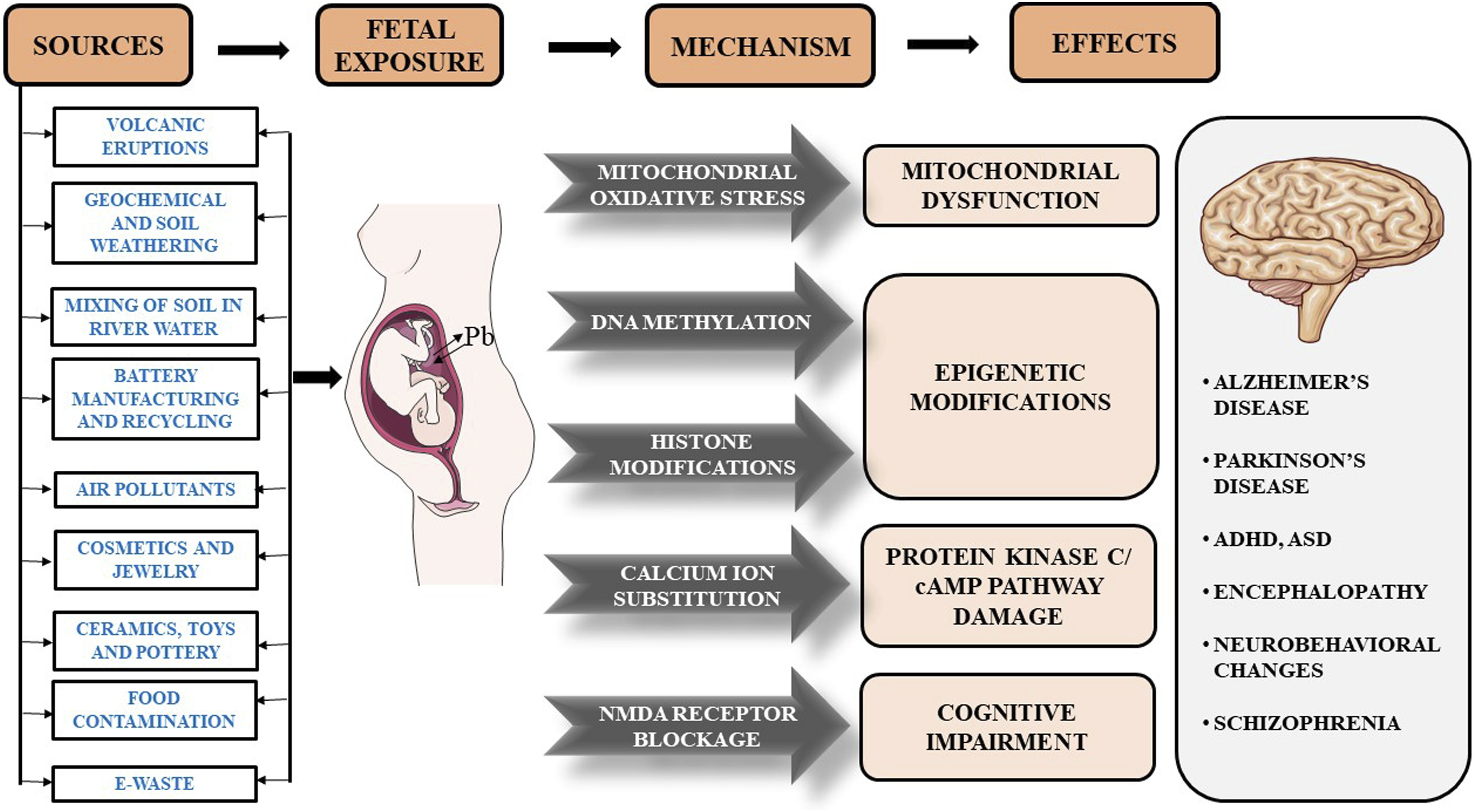
As electronics have become a major part of our lives, the generated electronic waste (e-waste) is difficult to dispose of and acts as a source of toxic metals. Electronic waste is usually dumped on empty ground. Global e-waste generation was estimated to be approximately 80.1 million tons by 2019, accounting for 11% of the annual increase. 19 Only 5% of this total e-waste generated is properly recycled. Dumping of this waste results in storage in the environment with a slow release of toxic metals such as lead in dust and soil. People living in nearby areas are constantly exposed to these metals through air, soil, water, and dust. Lead is commonly used in batteries, circuit boards, cathode-ray tubes, and solders. 20 Lead exposure through e-waste is due to informal recycling processes such as burning, acid leaching, roasting, and dismantling. 21 In a study conducted in Uruguay, South America, where informal lead processing was conducted, the lead level in the soil ranged from approximately 650 to 19000 mg/kg, which is far above the EPA standard level of 400 mg/kg. 16
The presence of lead in food and water bodies can also be attributed to the pesticides used in the fields. The presence of heavy metals in pesticides has been studied, and elevated levels of lead crossing permissible limits have been detected in various pesticide formulations. This could have been attributed to petroleum distillates, POEA (polyoxyethylene tallow amine), a surfactant used in herbicides and pesticides, or may have been added to the pesticides manually in small quantities. 22 The permissible lead concentration in paints in India was 1000 mg/kg, 23 which decreased to 300 mg/kg in 2011. 24 On 1st November 2017, the regulation of lead contents in household and decorative paint rules came into effect, which further reduced the permissible lead levels in paints to less than 90 ppm. 25 However, the concentrations of lead in enamel home paints have been reported to exceed acceptable levels.19,20 Lead-based paints in houses over time become old and form chips, flakes, and peels off, adding to household dust or can combine with surface runoff and rain, thereby contaminating water bodies. 26
Prenatal and early-life lead exposure
Studies have reported detrimental effects caused by prenatal and early-life lead exposure in later years of life.27,28 Children are easily exposed to lead through dust, soil, paint, petrol/gasoline aerosols, and pesticides seeping into water. 11 The presence of lead in dust makes it easy for children to be inhaled, as they are the most exposed to the ground. Most of the lead dust in houses comes from paint used on walls. According to a previous study, lead-based paint dust is the primary source of lead exposure to children, 29 with 70% of elevated blood lead levels (BLLs) corresponding to lead paint exposure. 30 The direct implications of this high amount of lead in the soil were observed in the BLLs of the children living around the region of the electronic waste disposal site. 31 Young children are vulnerable to lead because of their hand-to-mouth and pica (persistent and compulsive cravings to eat nonfood items) habits. Lead is a major concern in the paints used in toys where children are frequently touching and mouthing. Air pollutants such as nitrogen dioxide (NO2) and ozone (O3) can react with the polymeric binders of this lead paint and can cause lead particles to easily dissociate and deposit on hands and skin upon contact. 32 Cosmetics and medicines can also be sources of lead for women and children alike. Studies have confirmed the presence of lead in cosmetics such as lipsticks, eye shadows, henna, creams, cleansing milk, shampoos, hair dyes, and kohl (surma/kajal) at extreme concentrations.33–35 These compounds are directly applied to the skin and are therefore are at increased risk, as they can easily enter the body through absorption or ingestion. 36 The use of lead-contaminated cosmetics by pregnant or lactating women can act as a route of direct exposure to lead to the foetus or the infant. The lead that is ingested, inhaled, or absorbed by the mother is gradually deposited in the skeletal system due to the possible influence of lead on Ca2+ channels and pumps. The lead thus deposited acts as a reservoir, and during pregnancy and lactation, increased bone turnover causes the release of lead, which becomes a major source of exposure to the growing foetus. There is no placental barrier for lead; hence, the developing foetus can be easily exposed to lead. 37 The ability of lead to substitute for Ca2+ causes it to accumulate in bones and be released during Ca2+ turnover, which is observed in pregnancy and lactation. 38
Frequent washing and cleaning of toys and house floors can reduce lead exposure in young children. A Ca2+-rich diet during pregnancy can inhibit the mobilization of maternal bone lead and/or intestinal absorption of ingested lead, attenuating foetal lead exposure. 39 Iron deficiency during pregnancy can increase maternal BLLs, which further exposes the growing foetus. 40 Adequate intake of minerals can minimize the absorption of lead, thus reducing the risk of lead-induced toxic effects. 41
Mechanism of lead entry through the placenta
The placenta helps to protect the foetus from a variety of toxicants and unnecessary elements. It acts as the organ that facilitates transfer between the mother and the foetus. It provides passage to all the essential nutrients and oxygen for the developing foetus and acts as a barrier to toxic elements and harmful substances. The cells of the placenta contain many proteins that help in the detoxification of incoming substances. However, studies have been conducted on maternal and foetal blood samples for the presence of toxic elements such as lead, mercury, and cadmium, which have made it evident that the placenta cannot completely prevent the passage of these toxicants. 42 The human placenta consists of syncytiotrophoblasts, cytotrophoblasts, extraembryonic mesoderm, and foetal endothelial cells. 43 The syncytiotrophoblast lines the intervillous space where maternal blood is in contact with the placenta. The lead from the mother’s blood must pass through these layers of cells to reach the foetus. Although the exact mechanism of this transport is not clearly understood, a few studies have indicated some ways through which lead can pass through the placenta freely. 44 It has been assumed that lead undergoes passive diffusion through placental cells. Lead intake by the placenta is also thought to be mediated by divalent metal transporter 1 (DMT1), which also mediates the uptake of lead in the intestine. 45 Evidence suggests that DMT1 is also capable of transporting lead across the membrane depending on the availability of the metal. 46 Furthermore, studies have investigated the expression and distribution of both isoforms of this transporter, DMT1-iron responsive element (DMT1-IRE), and DMT1-non-iron responsive element (DMT1-nonIRE), in placental tissues throughout gestation. 45
Another protein that is assumed to play a significant role in lead transport is metallothionein (MT). Owning to its relatively high cysteine content, MT can readily bind to lead present in the intervillous space even at lower concentrations. 47 This scavenging effect leads to accumulation in the placenta and eventually transported to the foetus. Evidence also suggests that heavy metals such as lead, and mercury can induce the activation and synthesis of MT in the placenta. 47
Ca2+ is also assumed to play a role in lead transport through the placenta. A histological study revealed that Ca2+ acts as a carrier of lead.
48
Moreover, lead and Ca2+ movements across the placenta are closely related. Ca2+ homeostasis occurs when the Ca2+ level in foetal blood is greater than that in maternal blood. This active transport of Ca2+ is facilitated by transplacental proteins that consist of four plasma membrane ATPase isoforms located in the syncytiotrophoblast.
38
This movement is hindered by lead disturbing Ca2+ channels and pumps. Lead also affects the function of PKC and Ca2+-binding proteins, thereby modifying Ca2+ movement and promoting its movement. It was also observed that lower Ca2+ levels increase the uptake of lead. Studies suggest that as pregnancy progresses and organs develop, lead uptake and accumulation in the brain and other tissues of the foetus increase as the BBB and blood-tissue barrier are not completely mature. According to a study by Neda et al., the lead concentration in the foetus depends on maternal exposure to lead from the environment and its transport through the umbilical cord.
49
The different methods of lead entry through the placenta are described in Figure 3. Various routes of entry of lead through placenta and syncitotrophoblast crossing via divalent metal transporter 1 transporters, metallothionein proteins binding to lead, simple diffusion, passive diffusion, and calcium channels to the foetal blood. The image was created using a biorender.
Mechanisms of lead-induced neurotoxicity
Prenatal exposure to lead is possible because lead can freely cross the placenta as described above. Neural stem cells (NSCs) are the main targets of lead toxicity. 6 These are multipotent cells of the nervous system that differentiate to give rise to various cell types, such as neurons and glial cells (microglia, astrocytes, and oligodendrocytes). These arise from the neural tube, which has an ectodermal origin, during early embryogenesis. Lead affects the growth and differentiation of NSCs and the functions of differentiated cells. It affects signal transmission in neurons by influencing neurotransmitter release and storage. 50 Many of these effects can be directly associated with the competitive relationship between lead and Ca2+. One of the major ways by which lead can cross the BBB is because it can substitute for Ca2+. As discussed above, the perturbation of Ca2+ movement through Ca2+ channels by lead causes a serious imbalance of the signal transduction system. Ca2+ also acts as a cofactor for many enzymes, affecting multiple cellular processes. Moreover, an increase in Ca2+ levels has been shown to inhibit the effects of lead by causing lead efflux via the Ca-ATPase pump. 51 Ca2+ plays a key role in the release of neurotransmitters in response to incoming action potentials at synaptic terminals. This process also involves the activation of calmodulin, which leads to the release of neurotransmitters via the CaM kinase pathway. 52 In the presence of lead, the Ca2+ channels are blocked, and further signal transmission does not occur. Other Ca2+-activated processes, such as the phospholipase C pathway and synaptic vesicle endocytosis, are also blocked, which overall hinders neuronal functions and the transmission of information. This can lead to the accumulation of neurotransmitters that cause neurotoxicity and neurodegenerative disorders. 53
Role of lead in apoptosis and neurodegeneration
Lead is also known to cause cell death through mechanisms such as oxidative stress, excitotoxicity, and the induction of apoptosis. Apoptosis is the response to external or internal stimuli that leads to cell-activated programmed death. One of the major pathways of apoptosis is mitochondrial dysfunction. Ca2+ is necessary for maintaining a stable redox potential across the mitochondrial membrane. 54 The perturbation of Ca2+ homeostasis by lead disrupts the redox potential and accumulation of Ca2+ inside cells. This causes the binding of Ca2+ and leads to the metal-binding domain of the mitochondrial transition pore, which then leads to its opening, which causes depolarization of the membrane and activation of death effector proteins and cytochrome C-caspases that eventually cause apoptosis of the cell. 55 The presence of lead at very minor concentrations can also block Ca2+ movement and disrupt the redox potential. This neuronal degeneration is related to PD, where preferential degeneration of dopaminergic neurons in the substantia nigra pars compacta occurs. 56
Another mechanism of lead neurotoxicity is its effect on the N-methyl-D-aspartate (NMDA) receptor. Lead acts as a noncompetitive antagonist for this receptor, which participates in a process called long-term potentiation (LTP), which is a long-lasting postsynaptic response enhancement to high-frequency stimulation. The release of the postsynaptic response is facilitated by Ca2+, whose movement can be restricted by the blocking action of lead. Furthermore, in postsynaptic neurons, NMDA receptor channels are blocked by Mg2+, which opens due to the signals received from neurotransmitters. 57 This affects cognitive ability and related disorders.
Lead interference in Ca2+ activity also influences second messengers, such as PKC and cAMP, which are necessary for a variety of pathways. PKC participates in the regulation of genes important for the generation of astrocytes. Higher levels of lead can disrupt this BBB because of its ability to substitute for Ca2+, causing acute lead encephalopathy leading to edema, increased cranial pressure, and lymphatic drainage. Astrocytes also store lead to protect other neurons from exposure. However, this can inversely function as a reservoir of lead from which it is released in small amounts enough to damage nearby neurons. 58 Excessive release of glutamate in the synaptic region leads to dysregulation of Ca2+ homeostasis, causing excitotoxicity and eventually death of the cell. 59
Lead-induced neurological effects
The above-mentioned mechanisms by which lead affects neurodevelopment suggest that lead plays a significant role in behavioural and cognitive outcomes, and the intensity of lead-induced damage also represents the possibility of neurological disorders such as encephalopathy, edema, AD, PD, intellectual disabilities, schizophrenia, dementia, ADHD, ASD, and hypertension. 59 Exposure to lead at an early age correlates with intellectual and coordinative deficits, and studies have reported a relative loss of attention, muscular coordination, decreased scores in intellectual tasks, motor function, and reaction time. Reduced cognitive ability can be attributed to the effect of lead on the function of neurons and can be determined by IQ tests. Evidence suggests that even low to moderate levels of lead exposure can adversely affect IQ, resulting in extensive negative effects in adulthood. 2 Chronic lead exposure can lead to excessive nerve damage, convulsions, and coma. Lead exposure during childhood is a matter of concern because lead is a toxic metal that is not easily processed by the body and remains inside for a long time, providing it time to cause toxic effects on various organs. Inside the body, lead has a half-life of approximately 30 days and is excreted via urine and bile, with an average clearance rate of 2 mL/min. 60 Bone is a major reservoir and an endogenous source of lead, and its slow release during bone turnover, results in a half-life of approximately 20 years. 61 This is because of the influence of lead on Ca2+ movement and Ca2+ channels and pumps. The skeleton acts as a reservoir of lead and releases it slowly during bone turnover. This process is increased during pregnancy and lactation because the developing foetus and infants require more Ca2+, thereby exposing them to lead. 42 In several studies, umbilical cord blood levels indicated the slow development of sensory and visual-motor domains with the help of the Bayley Scales of Infant Development (BSID). 60 The effects on intellectual ability, as studied, suggest a dose-dependent decrease in IQ. As estimated by the WHO, an increase in the BLLs of 1 mg/dl results in a decrease of 0.25 IQ points. 2 Research has also shown a decrease in memory and learning ability. This can be attributed to the inhibition of neurotransmission through neurons and the prevention of long-term potentiation, which plays a role in the learning and memory storage process. Decreased memory scores lead to a decrease in recall and recognition ability. 62 Lead has also been demonstrated to affect motor skills, particularly visual-motor coordination, and dexterity. 63
Exposure to lead during development was linked to an increase in kinases and activators, which was deemed a factor in the phosphorylation of tau. 64 A decrease in microglia and hippocampal volume was observed due to prenatal low-level lead exposure. 65 Downregulation of cAMP-response element binding protein (CREB) phosphorylation and silent information regulator 2 homologue 1 (SIRT1) mRNA and protein was observed in the cerebral hippocampus of rats due to prenatal exposure. 66 Latronico et al., 2014 reported high lead levels in the myelin sheath due to prenatal and postnatal lead exposure. 67 A study conducted by Gąssowska et al., 2016 observed swelling in nerve endings and synaptic changes due to prenatal and neonatal lead exposure; furthermore, they reported elongated, swollen, or shrunken mitochondria with fused mitochondrial cristae and membranes. 68 The studies describing the effects of lead on neurodevelopmental and neurodegenerative diseases are listed in Table 1.
Role of lead in neurodegenerative disorders
Studies on the effects of lead on the brain resulting in the early onset of AD have shown increasing atrophy of the cortical, hippocampal, and temporal regions of the brain, leading to neurofibrillary tangles. This is one of the major hallmarks of AD. Another hallmark of AD is altered circulating levels of S-adenosyl methionine (SAM), homocysteine, vitamin B12, and folate. Studies have shown that these proteins are perturbed by lead through reactions with their sulfhydryl groups. 69 The association of lead with PD may be due to its involvement in the disruption of the dopaminergic system. Alternatively, lead can cause PD by inducing excessive oxidative stress in cells through the depletion of antioxidants or the inhibition of δ-aminolevulinic acid dehydratase (δ-ALAD). 70
A study conducted in animal models revealed a significant change in tau protein levels and phosphorylation due to prenatal, perinatal, and neonatal exposure to lead. Perinatal exposure to lead increased tau protein levels in the forebrain cortex and cerebellum, but no significant changes were detected in the hippocampus. The levels of tau mRNA was not affected in any region. A significant increase in tau phosphorylation at serine (Ser)-396 was observed in the forebrain cortex and cerebellum due to prenatal and neonatal exposure to lead. 71 Compared with the control group, perinatal lead exposure caused a significant increase in the level of phosphorylated glycogen synthase kinase 3β (GSK-3β) mRNA and its protein at tyrosine (Tyr)-216 in the forebrain, cortex and cerebellum in rat models. 71 Bihaqi et al., 2014 reported an increase in tau protein levels and tau phosphorylation at Ser-396, threonine (Thr)-181, and Ser-235 in late life due to infantile lead exposure in mice compared with the control group. 64 Population studies have shown that greater bone and BLLs are associated with an increased risk of PD. Animal studies have shown that exposure to low amount of lead during the very early stages of life enhances the formation of the protein alpha-synuclein. Although the exact processes underlying this correlation have not yet been thoroughly examined, oxidative stress and mitochondrial malfunction are likely involved and could account for the harmful consequences linked to lead exposure in parkinsonism. 72
Role of lead in neurodevelopmental disorders
High levels of lead in milk can either activate or suppress critical immune modulators, which can have an indirect impact on a child’s health and lead to conditions including allergy development, endocrine system problems, and even neurodevelopmental delay. 73 Research regarding the association of lead with ADHD suggests that lead accumulation in myelinated neurons in the striatum might be the reason for this disorder. 59 Lead exposure can also alter glucocorticoid deregulation and the hypothalamic-pituitary axis and change GABAergic (γ-aminobutyric acid-containing) systems, which causes an increase in anxiety and therefore a decrease in social activity. 74 These changes are also associated with other alterations that play a role in the schizophrenic behaviour observed in some patients. Studies have connected exposure to lead to the incidence of schizophrenia and bipolar disorder. 75
A study comparing the BLLs of mothers in the first, second, and third trimesters suggested that foetal lead exposure in the first trimester was indicative of adverse neurodevelopmental changes later in life. 76 Sex-related differences in the effects of lead were also studied. The results showed that in children less than 8 years of age, the scores for IQ tests were lower for girls than for boys. 2 These differences are assumed to be the result of the reaction of lead to different hormones resulting in epigenetic and genetic differences. 77 Albores-Garcia et al., 2021, reported that early lead exposure causes neuropsychiatric problems such as schizophrenia and substance misuse, as well as impacts neurodevelopment. The study also noted the effects of childhood lead exposure on mental health, such as an increased risk of psychopathology and altered personality traits in adulthood. Prolonged developmental lead exposure has also been shown to interfere with the brain ontology of the μ opioid receptor, which may have an effect on teenage drug use and misuse. 78 A literature review on early-life lead exposure is linked to neurodevelopmental and neurobehavioral issues, whereas early-life exposure can result in neurodegenerative consequences later in life owing to altered epigenetic imprints and ongoing internal lead exposure. Research also shows that antioxidants, macro- and micronutrients can ameliorate the effects lead exposure. 79
According to previous studies, children under the age of 12 with high blood or urine lead levels are more likely to have ASD, ADHD, low IQ, and cognitive impairment.50,80 Owing to the vulnerability of the brain, children are likely to be more prone to developing toxicity, which can cause dyslexia by damaging the nervous system. 80 An investigation conducted in 2021 by Baj et al. revealed that children exposed to lead had lower amounts of vital trace metals such as zinc, which could lead to neurodevelopmental issues such as ASD. 81 Persistent exposure to lead in children significantly decreased auditory speech processing and IQ test scores and was associated with behavioural changes such as hyperactivity and impulsiveness. Early studies of the neurotoxic effect of lead in animal models revealed nonlinearity in locomotor activity, multiple physiological functions, and cerebral cortical growth. 82 A study conducted by Sioen et al., 2013 reported that prenatal exposure to lead has a significant effect on hyperactivity and mental and psychomotor development. 83 Maternal exposure to lead was observed to decrease BDNF density in the hippocampus and frontal cortex, and drastically increase serotonin transporter (SERT) levels in the cortex of rats. N-methyl-D-aspartate receptor 2A (NMDAR2A) levels were low in the nucleus accumbens and hypothalamus. The levels of serotonin/5-hydroxytryptamine (5-HT) were significantly decreased in the striatum and nucleus accumbens of male rats. 84 Schneider et al., 2013 observed variations in DNMT1 levels and their expression at various stages of lead exposure during pregnancy in animal models. A notable change in DNMT1 expression in male progeny due to early postnatal exposure to lead, and a notable change in DNMT1 levels in female progeny due to perinatal lead exposure have been reported. However, DNMT3a expression was high due to perinatal exposure in male progeny. There was no significant effect of perinatal or postnatal lead exposure on methyl-CpG binding protein 2 (MeCP2) levels in male progeny, but early postnatal exposure and perinatal exposure had significant effects on female progeny. 85 Despite progress in preventive measures for lead exposure, lead still poses a great threat to children, especially in developing countries. Therefore, preventive measures should be taken to minimize lead exposure in children. One study used an experimental design to examine lead exposure from handcrafted glazed pottery in Mexico. Pregnant rats were given lemonade preserved in glazed pottery, which led to elevated levels of lead in their blood and the brains of their offspring. Exposure to these compounds resulted in increased caspase-3 activity in the hippocampus of neonates, indicating probable brain injury. 86 A study on children in southern Sweden revealed that lead exposure decreased dramatically over time. These findings demonstrated a link between blood-lead concentrations and teenagers’ academic performance and cognitive ability. This study underscores the need to consider very low levels of lead exposure into account, as well as the long-term consequences for children. 87 One study demonstrated that exposure to lead during pregnancy causes spatial learning and memory problems in children and revealed the impact of lead exposure on hippocampal synaptic plasticity and glucose metabolism. By influencing the membrane translocation of the glucose transporter type 4 (GLUT4) protein, lead exposure was shown to decrease the hippocampal uptake of glucose. Additionally, by increasing glucose absorption and reversing the loss of synaptic plasticity, the overexpression of GLUT4 improved the cognitive deficiencies caused by lead exposure. 88 According to a study on prenatal lead exposure, lead exposure during brain development can result in long-lasting changes in gene expression, which may play a role in the development of adult neurological disorders. The long-term consequences of early-life lead exposure were further highlighted in the study by the negative effects of lead exposure on behaviour, cognitive processes, and neural networks. Additionally, the study addressed the role of ionotropic receptors for glutamate and the neurotrophin BDNF in mediating the effects of lead on brain function. 89 Researchers have studied the impact of lead exposure in early life on the CNS and future neurobehavior in zebrafish. The exposure of zebrafish embryos to lead concentrations comparable to those in the environment resulted in neurodevelopmental abnormalities and aberrant neurobehavior in both juvenile and adult fish. The lead-exposed fish showed symptoms akin to ADHD and anxiety, as well as impaired learning and memory capacities. This study also revealed alterations in the expression of genes involved in neurodevelopment and neurotransmitter systems, as well as changes in neuronal density and architecture. 90 A study revealed that lead exposure increases the number of μ-opioid receptors (MORs) in the rat brain during preadolescence and early adolescence, with sex-specific effects on specific brain areas. The study emphasized the possible effects of childhood lead exposure on opioid drug use and abuse, especially during periods of increased reward and drug-seeking behaviour. Research has further clarified the link between developmental lead exposure and psychological conditions such as severe depressive disorder and drug addiction. 78 Pregnant participants in Japan’s statewide birth cohort research, the Japan Environment and Children’s Research (JECS), were registered between 2011 and 2014, blood samples were obtained from mothers and umbilical cords, and information was gathered from medical records and questionnaires. The neurodevelopment of 80,759 children was evaluated in the study using the Ages and Stages Questionnaire (ASQ). Regression model analysis, revealed no significant correlation between maternal-umbilical cord BLLs and neurodevelopmental delays. 91
Discussion
There is no safe threshold for lead exposure, and a safe BLLs have not yet been defined. 92 Children’s behavioural problems, learning challenges, and decreased intellect have been connected to BLLs as low as 3.5 µg/dL. A vital issue to grasp is the critical phase of a child’s development that is influenced by lead, which can result in increased cognitive and behavioural deficiencies in the future. Elevated peripheral BLLs in newborns due to maternal exposure to lead cause different types of DNA methylation. 3 Toxic metals (lead, cadmium, mercury, and arsenic) are widely found in our environment. Humans are exposed to these metals from numerous sources, including contaminated air, water, soil, and food. Recent studies have indicated that transition metals act as catalysts in the oxidative reactions of biological macromolecules; therefore, the toxicity associated with these metals might be due to oxidative tissue damage. Redox-active metals, such as iron, copper, and chromium, undergo redox cycling, whereas redox-inactive metals, such as lead, cadmium, mercury, and others, deplete major antioxidants, particularly thiol-containing antioxidants and enzymes. Both redox-active and redox-inactive metals may cause an increase in the production of reactive oxygen species (ROS), such as hydroxyl radicals (HO.), superoxide radical (O2-) or hydrogen peroxide (H2O2). Enhanced generation of ROS can overwhelm intrinsic antioxidant defenses in cells and result in a condition known as “oxidative stress”. Cells under oxidative stress display various dysfunctions due to lesions caused by ROS to lipids, proteins, and DNA. Consequently, metal-induced oxidative stress in cells may be partially responsible for the toxic effects of heavy metals. Several studies are underway to determine the effect of antioxidant supplementation following heavy metal exposure. These data suggest that antioxidants may play an important role in abating some heavy metal hazards. To prove the importance of using antioxidants in heavy metal poisoning, pertinent biochemical mechanisms for metal-induced oxidative stress should be reviewed. 70 Various studies have reported the role of early-life lead exposure in causing neurodevelopmental and neuropsychiatric disorders.78,79 However, this review mainly focuses on the role of prenatal and early-life lead exposure in causing neurodevelopmental and neurodegenerative diseases.
Conclusion
Environmental lead pollution has a severe effect on the neurodevelopment of children and neurodegeneration. Importantly, during the essential period of neurodevelopment, such as pregnancy and lactation, care needs to be taken so that the mother, foetus, or infant is not exposed to lead. Prevention and detection of prenatal and early-life lead exposure as well as any therapeutic interventions during the early stage of brain development are more effective in preventing future risks and the extent of neurodevelopmental disorders. The mechanisms discussed in this review might aid in clinical evaluation, early diagnosis, management, intervention and the development of any putative therapeutic strategies.
Footnotes
Acknowledgments
The authors would like to thank the Manipal School of Life Sciences and the Manipal Academy of Higher Education (MAHE) for their infrastructure and facilities. PA would like to thank MAHE, Dr TMA Pai Structured PhD. Fellowship, for financial assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the author(s) used the Rayyan tool for the inclusion and exclusion of papers. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
