Abstract
Lead is one of the heavy metals that is toxic and widely distributed in the environment, and children are more sensitive to the toxic effects of lead because the blood–brain barrier and immune system are not yet well developed. The objective of the study was to investigate the clinical characteristics of lead poisoning in children aged 0∼6 years in a hospital in Guangxi, and to provide scientific basis for the prevention and treatment of lead poisoning. We collected and analyzed the clinical data of 32 children with lead poisoning admitted to a hospital in Guangxi from 2010 to 2018. The results showed that most of the 32 cases presented with hyperactivity, irritability, poor appetite, abdominal pain, diarrhea, or constipation. The hemoglobin (HGB), mean corpusular volume (MCV), mean corpuscular hemoglobin (MCH), and hematocrit (HCT) of the lead-poisoned children were all decreased to different degrees and were below normal acceptable levels. Urinary β2-microglobulin was increased. Blood lead levels (BLL) decreased significantly after intravenous injection of the lead chelator, calcium disodium edetate (CaNa2-EDTA). In addition, HGB returned to normal levels, while MCV, MCH, and HCT increased but remained below normal levels. Urinary β2-microglobulin was reduced to normal levels. Therefore, in this cohort of children, the high-risk factors for lead poisoning are mainly Chinese medicines, such as baby powder. In conclusion, lead poisoning caused neurological damage and behavioral changes in children and decreased erythrocyte parameters, leading to digestive symptoms and renal impairment, which can be attenuated by CaNa2-EDTA treatment.
Introduction
Lead is a neurotoxic environmental contaminant that is widely found in air, soil, and water (Liu et al., 2022). It is also found in industrial products and household items, including gasoline, paint, ceramics, ceramic glazes, canned food, candy, cosmetics, jewelry, and toys and is a recognized global environmental health problem. Lead poisoning due to industrial pollution is one of the major causes (Bouftini et al., 2015). Lead can be absorbed through the respiratory tract, digestive tract, and skin and then transported through the systemic circulation, damaging several tissues and organs (nervous, digestive, and blood systems) and causing learning and memory dysfunctions (Byers et al., 2020; Orisakwe et al., 2020). Children at critical stages of growth and development are more sensitive to lead than adults because the excretory mechanisms for lead are not fully functional at early life stages and many developmental processes are still ongoing (Chehbani et al., 2020). Blood lead levels (BLL) in children are often used as one of the biomonitoring indicators of regional environmental lead contamination, which may cause long-term and irreversible damage to the organism, even involving learning behavior disorders, when blood lead concentration in children exceeds 50 μg/L (Wang et al., 2015). Childhood lead exposure is a global public health problem, especially in developing countries (Liu et al., 2022; Hoang et al., 2021; Ansari et al., 2020). In China, there has been no shortage of reported cases of lead poisoning (Yao, 2021; Zhang et al., 2020; Ren et al., 2019; Ying et al., 2016, 2018; Tang et al., 2017). In a recent nationwide study of children's blood lead survey, our children's blood lead level was 26.7 μg/L (Li et al., 2020), but it remains significantly higher than that of developed countries in Europe and America (Zhang et al., 2020; Ericson et al., 2021; Feinberg et al., 2018; S., 2021; Liu et al., 2021). Currently, clinically used calcium disodium edetate (CaNa2-EDTA) is a soluble complexing agent, which complexes with lead to form stable water-soluble complexes and excreted in the urine (Li et al., 2013).
Material and methods
Data on children with lead poisoning from a hospital in Guangxi from January 2010 to December 2018 were retrieved with the Lian Zhong Digital System. Inclusion criteria: ① According to the US Centers for Disease Control (CDC) lead poisoning classification (Ettinger et al., 2019), in the case of venous blood, lead is higher than 200 μg/L (more than this value requiring hospitalization to standardize lead treatment) in 0 to 6 years old children; ② Repeated hospitalization of children as one case data for analysis. Exclusion criteria: ① accompanied by other underlying diseases, such as congenital heart and brain diseases, hematopoietic system, digestive system, urinary system and other primary diseases, mental patients; ② During the hospitalization period, the treatment was not completed as required by the doctor, and the patient was out of hospital early; ③ Inpatient data lack relevant biochemical indicators.
Diagnostic criteria: Lead poisoning classification based on the US CDC (Ettinger et al., 2019). Treatment methods: The patient was given intravenous CaNa2-EDTA injection (body weight 25 mg/kg/d) for 5 days, and the liver and kidney function were determined after the first day of treatment to determine whether the children had toxic side effects. According to the BLL and the physical condition of the children after one course of treatment, clinical symptoms before and after treatment, blood routine, and urinary biochemical indexes, we decided whether to carry out the second course of treatment and analyzed the therapeutic effect of CaNa2-EDTA.
All statistical analyses were performed with the SPSS 22.0 software (SPSS Inc., Chicago, IL). The data were expressed as mean ± standard deviation. Correlation between the BLL before treatment and the pre-treatment red blood cell parameters was determined with Pearson correlation coefficient. BLL, red blood cell parameters, and liver and kidney function before and after treatment were compared by paired t-tests. The BLL, red blood cell parameters, and liver and kidney function and other indicators in different age groups were compared by one-way ANOVA, followed by Tukey’s posthoc test to correct for multiple comparisons. Significance was set at a p value < .05.
Results
General information
Classification of children with lead poisoning in different periods from 2010 to 2018. The data for hospitalized children diagnosed with lead poisoning in a hospital in Guangxi during the period from January 2010 to December 2018 were collected, and the patients were divided into three groups, Infant period, Toddler period, and Preschool period. Total sample number N = 32.
High risk factors in children with lead poisoning
Statistics on the number of high-risk factors in 32 children. The pathways of lead exposure for this group of children are categorized into the eight sections of the table. The highest exposure to lead in this group of children was the use of traditional Chinese medicine in 11 cases (34.4%) and the lowest was unknown in 1 case (3.1%).

Changes in BLLs before and after treatment in hospitalized children
BLL before and after treatment with CaNa2-EDTA in children of different ages
Twenty-nine cases (90.6%) were diagnosed as moderate, with 3 cases of severe lead poisoning accounting for 9.4% of the children. The lowest blood lead concentration in this group before treatment was 231.1 μg/L and the highest was 620.6 μg/L, with an average concentration of 357.3 ± 97.6 μg/L. After treatment, the lowest blood lead concentration was 42.1 μg/L, the highest 276.9 μg/L, and the average concentration 169.0 ± 66.2 μg/L, as shown in Figure 1. The difference between the before and after treatment means was statistically significant (p < .05). There was no difference in BLL between groups before or after treatment (p > .05). BLLs were statistically different within each group before and after treatment (p < .05). BLLs before and after treatment with CaNa2-EDTA in different age groups. The children were given CaNa2-EDTA injection (25 mg/kg/d) intravenously for 5 days, and then the second course of treatment was decided according to the blood lead level after the first course of treatment and the children's physical condition. Comparisons were made to determine whether the differences in BLLs before and after the treatment of lead expulsion were statistically significant in children of different ages. Total sample (a), infant group (b), toddler group (c), preschool group (d). BLL: blood lead level.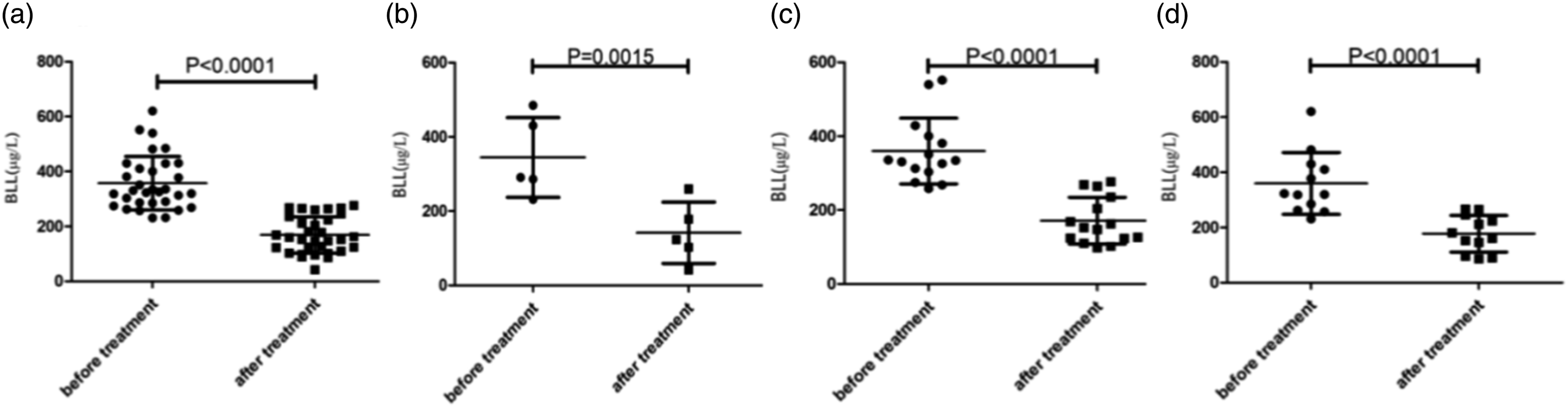
BLLs before and after treatment with CaNa2-EDTA in children of different genders
No difference in BLLs was noted before and after treatment in children of different genders, as shown in Figure 2. BLLs before and after treatment of different sexes. The children were given CaNa2-EDTA injection (25 mg/kg/d) intravenously for 5 days, and then the second course of treatment was decided according to the blood lead level after the first course of treatment and the children's physical condition. Compared with before and after treatment in total example, p < .0001, compared with before and after treatment in male group, p < .0001, and compared with before and after treatment in female group, p < .0001. Total example (a), male group (b), female group (c). BLL: blood lead level.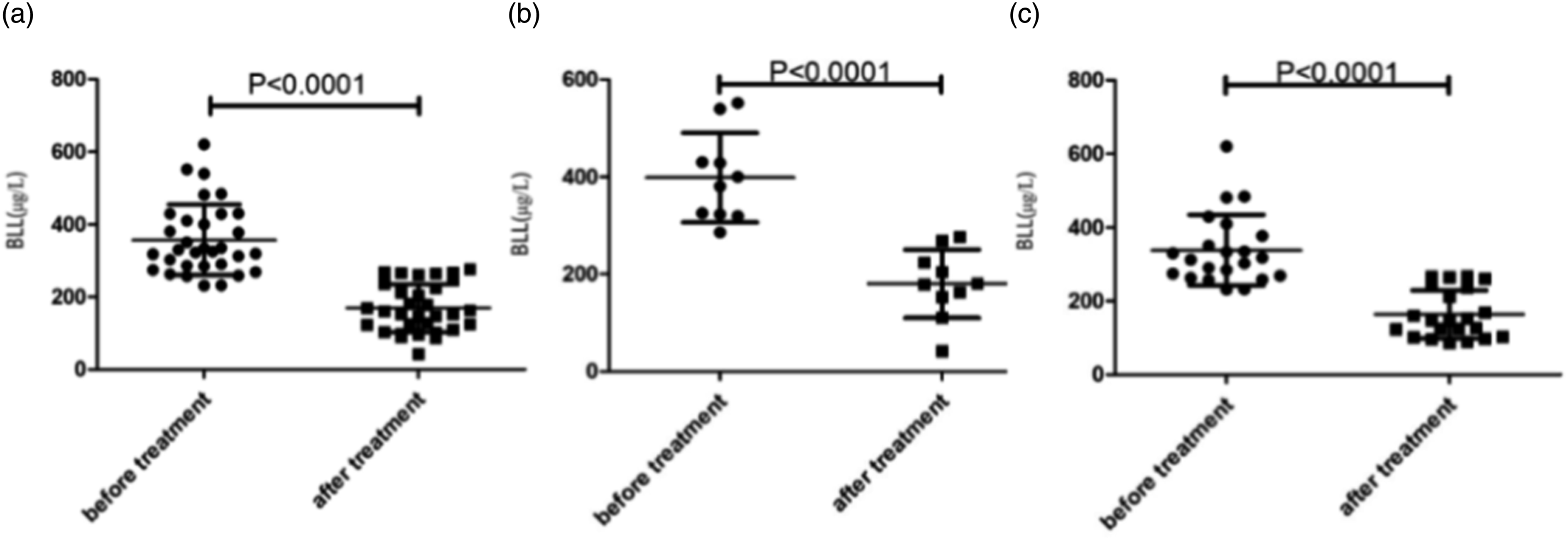
Changes in BLLs before and after treatment with CaNa2-EDTA in symptomatic and asymptomatic children
Comparison of BLLs between symptomatic and asymptomatic patients before and after treatment with CaNa2-EDTA in children. Data are means ± SD, total sample number N = 32; **compared with the symptomatic group before treatment, p < .01, ## compared with the asymptomatic group before treatment, p < .01, BLL: blood lead level.

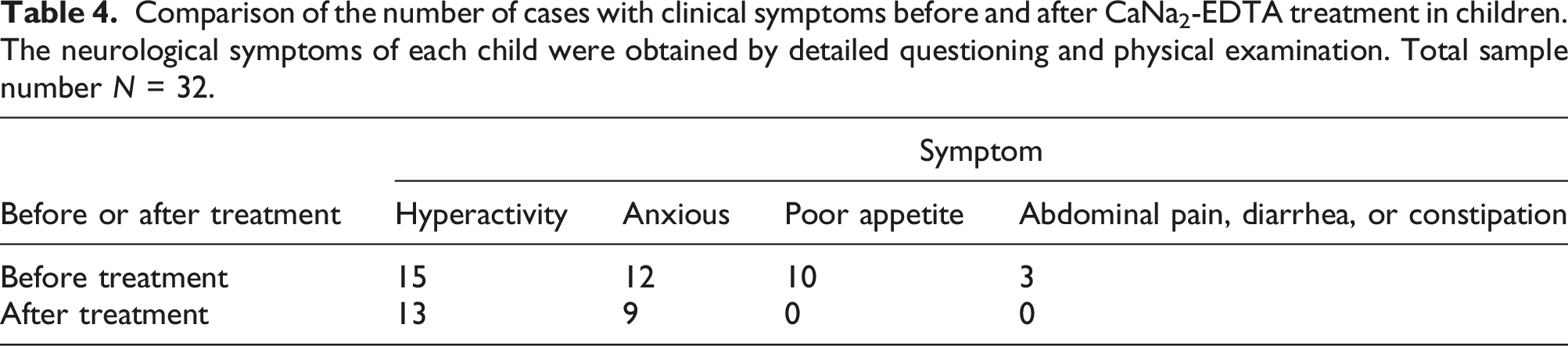
Comparison of clinical symptoms before and after treatment with CaNa2-EDTA in children
Comparison of the number of cases with clinical symptoms before and after CaNa2-EDTA treatment in children. The neurological symptoms of each child were obtained by detailed questioning and physical examination. Total sample number N = 32.
Analysis of red blood cell parameters in children
Correlation between blood lead value and red blood cell parameters before treatment
Correlation analysis between pre-treatment blood lead values and pre-treatment red blood cell parameters. Total sample number N = 32. 2 mL of blood was collected from patients through the median vein of the elbow, placed in EDTA tubes (Jiangxi Evergreen Medical Technology Co., Ltd.), and the blood routine was determined using a fully automated hematology analyzer (HysonMedicom, Inc.). HGB: Hemoglobin, MCV: Mean Corpusular Volume, MCH: Mean Corpuscular Hemoglobin, HCT: Hematocrit.
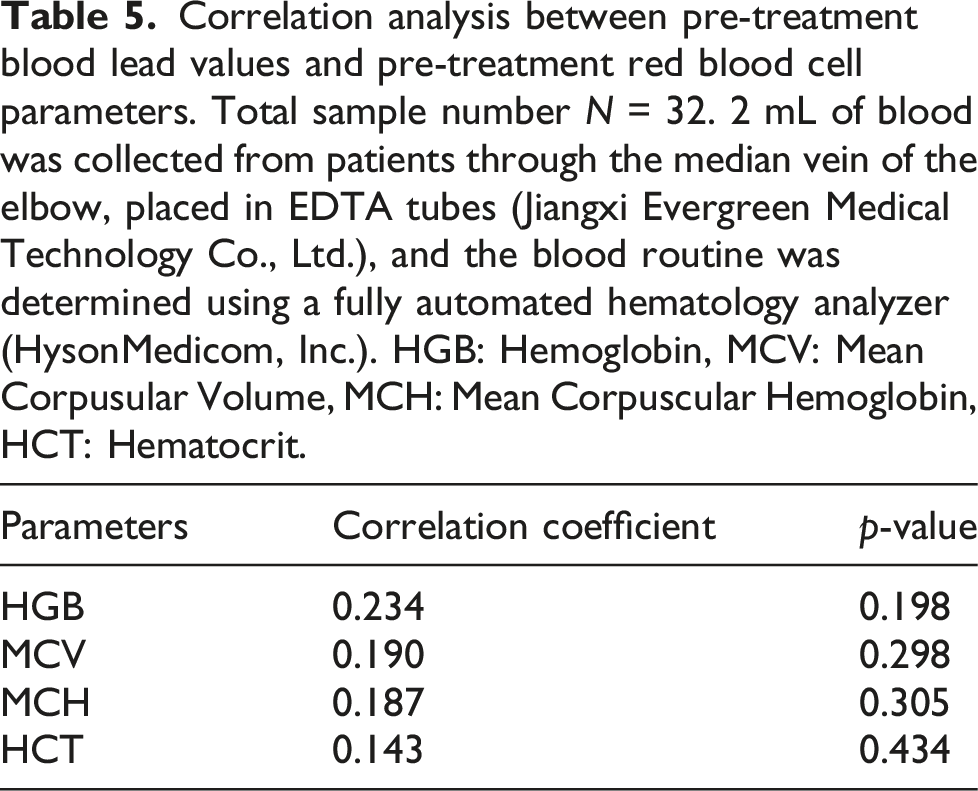
Comparison of red blood cell parameters before and after treatment with CaNa2-EDTA in different age groups
Comparison of red blood cell parameters before and after with CaNa2-EDTA treatment in children. Data are means ± SD, total sample number N = 32; * Compared with HGB and HCT in infants, p < .05, # compared with MCV before treatment, p < .05, ** compared with HGB before treatment, p < .01, ▲▲compared with pre-treatment HCT, p < .01, HGB: Hemoglobin, MCV: Mean Corpusular Volume, MCH: Mean Corpuscular Hemoglobin, HCT: Hematocrit.
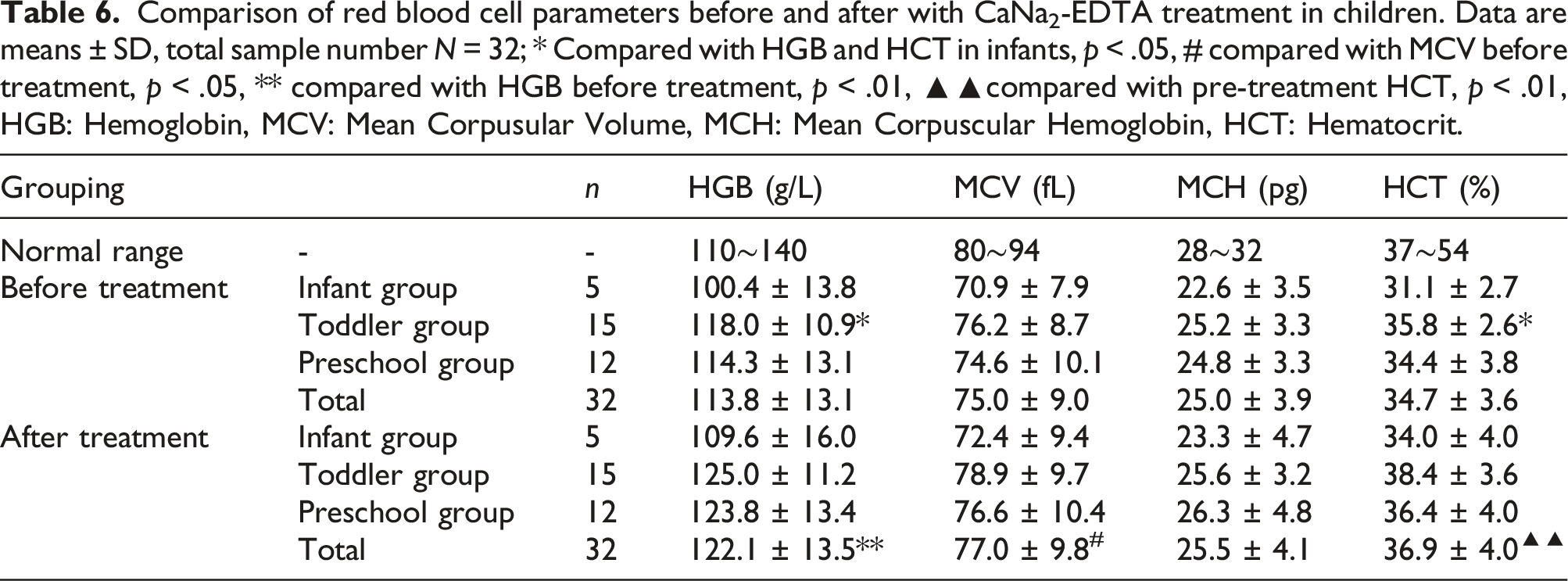
Analysis of biochemical indicators of liver function before and after treatment with CaNa2-EDTA in children
Comparison of main liver function indexes before and after treatment with CaNa2-EDTA in children. 2 mL of blood was collected from patients via the median elbow vein, placed in EDTA tubes (Jiangxi Evergreen Medical Technology Co., Ltd.), and the liver function was measured using a Hitachi 7600 series biochemical analyzer (Hitachi High-Technologies Co., Ltd., Japan). Data are means ± SD, total sample number N = 32. ALP: Alkaline Phosphatase, DBiL: Direct Bilirubin, IBiL: Indirect Bilirubin.
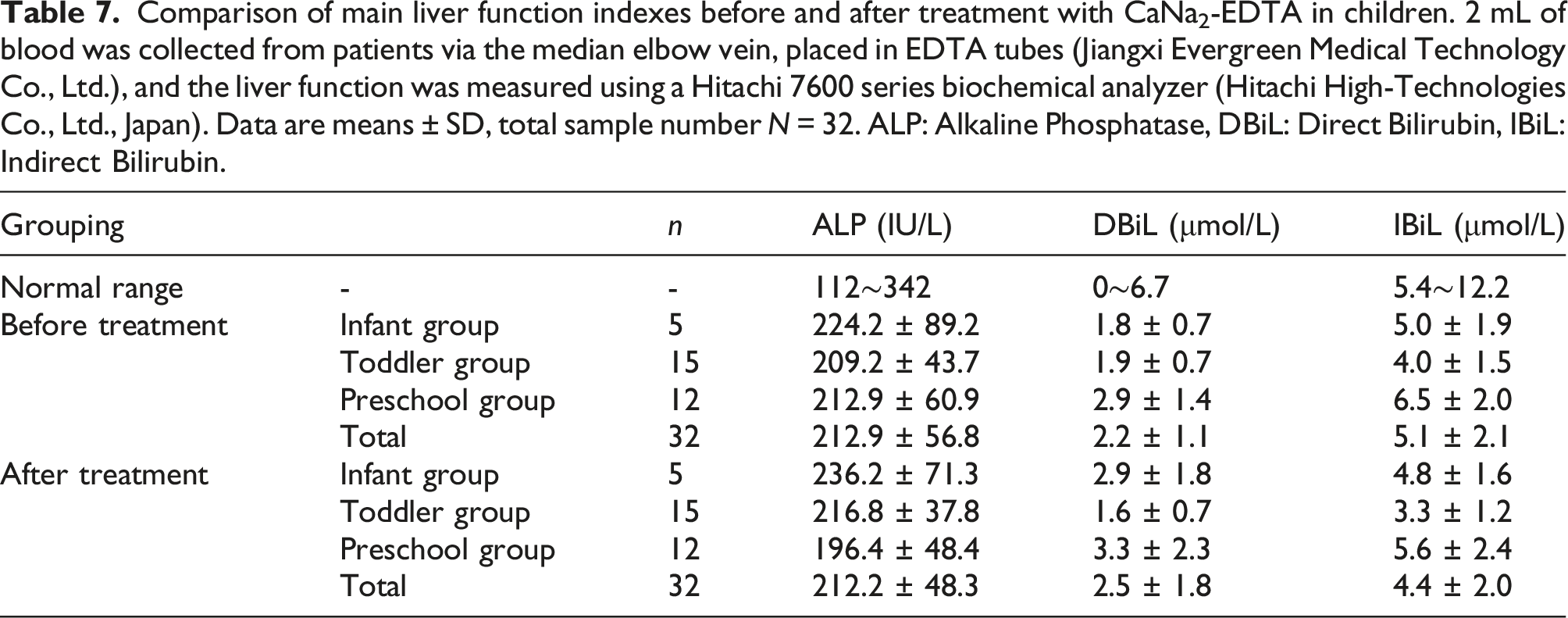
Analysis of renal function biochemical indicators before and after treatment with CaNa2-EDTA in children
Comparison of main renal function indicators before and after treatment with CaNa2-EDTA in children. Scr and BUN values of patients before and after treatment were within the normal range. Data are means ± SD, total sample number N = 32. Scr: serum creatinine, BUN: blood urea nitrogen.
Discussion
Elevated blood lead levels can damage the liver, kidneys, neurologic and reproductive systems in children (Mitra et al., 2017). Previously, 10 mg/dl of lead was considered as a safe level, but it is now known to cause chronic health effects in children, including decreased IQ, neurodevelopmental effects, reduced aminolevulinic acid dehydrase(ALAD) activity, and attention deficits (Wang et al., 2008; Jusko et al., 2008). Based on contemporary understanding of lead toxicity, damage induced by this metal is considered irreversible, and the younger the age and the more critical the developmental period, the more severe the lead-induced neurological damage. Therefore, early detection of abnormalities in the organism and timely targeted treatment of childhood lead poisoning may have a more important impact on its outcome and prognosis.
Pathway of exposure to lead poisoning in 32 children
Lead can enter the body through the respiratory tract, digestive tract, and skin. This reported survey showed that the number of lead poisoning cases in boys was 22, accounting for 66.3% of the total number of cases. The higher number of lead poisoning in boys than girls may be due to the fact that boys spend more time outdoors, which increases their exposure to lead (Du et al., 2022; Guo et al., 2022). In addition, boys tend to have more frequent behavioral patterns, such as the unhealthy practice of hand-to-mouth, that may promote lead absorption (Tang and Yang, 2019). In the present investigation, there were 4 cases (12.5%) of children with lead poisoning induced by air exposure; 5 cases (15.6%) of children whose parents were engaged in lead-exposed occupations, and the parents of children who worked with lead did not change their clothes or shoes after work and brought the lead home, leading to lead poisoning in children (OON Us Epa, 2000). Three cases (9.4%) of children had lead poisoning due to living in a nearby electronic scrapyard (Zeng et al., 2018; Xu et al., 2018; Kim and Williams, 2017; Zhang et al., 2020).
The effect of lead poisoning on various systems
Children in this investigation showed abnormal neurological symptoms in 16 cases (50%), including 15 cases of hyperactivity (46.9%) and 12 cases of irritability (37.5%). In the infant group, there were 2 cases (12.5%) with neurological symptoms; in the toddler group, there were 8 cases (50.0%) with neurological symptoms; in the preschool group, there were 6 cases (37.5%) with neurological symptoms; in the infant group, there were 62.5%; the younger the age, the higher the proportion of children with neurological symptoms. As children's brains are in a period of growth and development, the blood-brain barrier that protects the brain is not well developed. The younger the child, the more imperfect the protective mechanism of the brain, resulting in tissue cells susceptible to the influence of external factors (Zeng et al., 2020; Chan et al., 2013). Pediatric nervous system damage is more serious in comparison to lead poisoning in adulthood (Chehbani et al., 2020). In patients from 0 to 6 years of age, a total of 13 cases with hemoglobin concentration had lower than the normal levels, accounting for 40.6% of cases. And most of the children's blood routine red blood cell parameters, such as MCV, MCH, and HCT (see Appendix for full names)
HGB and HCT (see Appendix for full names) changes in the infant group were more pronounced than those in the toddler and the preschool groups, which was consistent with Tao's findings in Pany district that there was a positive correlation between HGB and BLLs in children aged 0–6 years (Tao, 2013). BLLs in children at different growth stages were proportional to the hemoglobin concentration. The younger the age, the higher the incidence of lead poisoning and anemia, and it was most obvious in early childhood. The results showed that children with lead poisoning are more likely to develop anemia than normal children. Indeed, during school age, the incidence of lead poisoning and anemia decreases with age (Chen and An, 2012). The main reason for this effect is that the children's body is more susceptible to lead toxicity, or alternatively it may be related to inadequate health education in children commencing their schooling (Hegazy et al., 2010).
By analyzing the correlation between pre-treatment BLL and pre-treatment red blood cell parameters, we found that lead caused hypoallergenic anemia, but lead concentration was not proportional to the damage in the hematopoietic system. There are similar conclusions in the literature corroborating the absence of correlation between BLL and HGB (Zhang et al., 2020). There are also reports that there is a negative correlation between blood lead and hemoglobin (Mo et al., 2004; Zhang et al., 2011). The conclusions reported in this paper may be biased by the small number of samples, requiring expansion of the number of cases in future analyses. Therefore, whether there is a correlation between blood lead and hemoglobin remains to be determined. The gastrointestinal tract is the main route for the lead absorption. In addition to encephalopathy, upon exposure to high concentrations of lead, abdominal pain, constipation, and intestinal paralysis also represent early symptoms of lead poisoning (Shabani et al., 2021). About 34.4% of the children in this survey had taken traditional Chinese medicine containing lead, which led to symptoms such as abdominal pain, diarrhea, or constipation.
Lead can also cause liver damage, which accounts for 33% of the body’s soft tissue lead accumulation (Mudipalli, 2007). Lead inhibits cytochrome P450 function, induces changes in hepatic cholesterol metabolism, and causes elevated levels of total cholesterol in the liver and serum, suggesting that lead induces changes in liver physiological function (Mudipalli, 2007; Haag et al., 1973; Pillai and Gupta, 2005).
Healthy alkaline phosphatase (ALP) value of children refers to the reference interval of serum ALP in healthy children aged 0∼12 years old from a hospital in Beijing (2016) (Zhang et al., 2016). In this article, the mean ALP before treatment was 212.9 ± 56.8 u/L and 212.2 ± 48.3 u/L after treatment, all within the normal range, and there was no difference before and after treatment (p > .05). However, it is higher than in adults, which is mostly due to the physiological increase caused by bone metabolism in children during growth and development (Akbal et al., 2014). Therefore, it is not listed as an indicator of liver damage. In addition, the normal ranges of children’s DBiL and IBiL refer to the reference ranges for ethnic Han Chinese aged 1 month to 14 years in Changchun City. (Li et al., 2016). The DBiL and IBiL values in this group were 2.2 ± 1.1 μmol/L, 2.5 ± 1.8 μmol/L, 5.1 ± 2.1 μmol/L, and 4.4 ± 2.0 μmol/L before and after treatment. Both were within the normal range. Children studied herein did not have B-ultrasound, and there were no reports on abnormalities in direct bilirubin and indirect bilirubin. Nonetheless, further confirmation is needed to determine whether there is liver damage upon lead exposure in children in these age groups.
The normal range of renal function indicators such as Scr and BUN (see Appendix for full names) of children refers to the reference interval of the “children’s renal function biochemical project” in Shanghai in 2018 (Chen et al., 2018). The Cr and BUN values of the patients in the present study were 35.0 ± 21.0 μmol/L, 32.3 ± 11.4 μmol/L, 3.7 ± 1.0 mmol/L, and 4.9 ± 1.4 mmol/L, respectively, before and after treatment, which were all within the normal range. Therefore, BUN and Scr cannot be used as markers of kidney damage.
It is noteworthy that the concentration of urinary β2-microglobulin in the renal function index of children in our cohort was increased, indicating that there is a certain degree of pathological changes in the glomeruli and renal tubules in these children. After 1∼2 courses of lead-extraction treatment, the concentration of urinary β2-microglobulin decreased to normal levels.
Analysis of treatment effects
Here, patients were treated with intravenous CaNa2-EDTA injection (body weight 25 mg/kg/d) for 5 consecutive days, concomitant with vitamins, trace elements, and nutrients as adjuvant treatment. The liver and kidney functions were measured after the first day of treatment to determine whether CaNa2-EDTA had toxic side effects. According to the blood lead level after the first course of treatment and the physical condition of the children, it was decided whether to perform a second course of treatment. All patients completed the standardized treatment of 1 or 2 courses during hospitalization. Next, the clinical symptoms, BLLs, and urinary biochemical indicators were determined before and after treatment. The average blood lead concentration in children decreased to 169.0.3 ± 66.2 μg/L (p < .05). Twenty-three children with clinical symptoms improved after treatment, including some of the children’s neurological symptoms. Children’s HGB after treatment were significantly higher than before, and the difference was statistically significant (p < .05). Although MCV, MCH, and HCT all improved compared with before treatment, they still did not reach normal levels. This may be due to the short time interval before and after treatment. Given the life cycle of hematopoietic cells, the hemoglobin concentration of children after treatment increased in 1 to 2 weeks, while MCV, MCH, and HCT (see Appendix for full names) may require longer period of recovery. The concentration of urinary β2-microglobulin in 7 children was higher than normal before treatment. After treatment, the concentration of urinary β2-microglobulin decreased and returned to normal level, suggesting its pathological changes are reversible.
Conclusion
Lead poisoning may damage the nervous system in children with ensuing behavioral changes. Lead poisoning also causes reduction in erythrocyte parameters in children. Here, we showed that HGB returned to normal levels shortly after CaNa2-EDTA treatment, and that the levels of MCH, MCV, and HCT trended towards normal values, although they remained below normal levels. Lead poisoning can affect the digestive system and increase urinary β2-microglobulin as a manifestation of renal functional damage in children, yet, after CaNa2-EDTA treatment, renal function was restored to normal values.
Footnotes
Acknowledgements
The authors gratefully acknowledge the financial supports by the National Natural Science Foundation of China and Guangxi Natural Science Foundation.
Authors’ contributions
YW, CG, and FX collected patient’s information, performed the statistical analysis, and drafted the manuscript. YW, BZ, and WL were responsible for the clinical investigation. MA was responsible for reviewing and polishing papers. KN assisted in the design and implementation of the study. YJ designed and led the study. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by grants from the National Natural Science Foundation of China (NSFC 81773476).
Correction (March 2025):
Article updated; for further details please see 10.1177/07482337251332187.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
