Abstract
Background and objective
The objective of this study was to investigate the potential of salidroside (SAL) (a major active compound in Rhodiola rosea L.) in regulating osteoclast differentiation and function by modulating the HIF-1α pathway and its downstream target genes.
Methods
The expression of HIF-1α and its downstream target genes was examined at both mRNA and protein levels in osteoclasts treated with SAL. Immunofluorescence analysis was performed to assess the nuclear translocation and transcriptional activity of HIF-1α in response to SAL. MTT, flow cytometry, qPCR, TRAP staining and bone resorption assays were used to evaluate the potential effect of salidroside on osteoclasts.
Results
SAL enhanced the expression of HIF-1α and its downstream target genes in osteoclasts. Immunofluorescence analysis confirmed the facilitation of HIF-1α nuclear translocation and transcriptional activity by SAL. In addition, SAL enhanced osteoclast viability, differentiation and bone resorption activity in an autocrine manner through HIF-1α/VEGF, IL-6 and ANGPTL4 pathways.
Conclusion
SAL promotes osteoclast proliferation, differentiation and bone resorption through HIF-1α/VEGF, IL-6 and ANGPTL4 pathways.
Keywords
Introduction
Bone remodeling is regulated by a dynamic balance between bone formation and bone resorption in order to maintain optimal bone health, with the involvement of osteoblasts, osteoclasts and bone marrow mesenchymal stem cells. 1 Among them, increased osteoclast activity induces erosion of trabecular and fragile bone. Hypoxia-inducible factor 1-alpha (HIF-1α), as an oxygen concentration-dependent transcriptional regulatory factor, plays a crucial role in osteoclast differentiation and physiological/pathological bone resorption.2–5 Several studies have demonstrated the correlation between HIF-1α and osteoclast differentiation. Hypoxia enhances osteoclastogenesis and increases bone resorption in inflammatory arthritis by activating HIF-1α. This suggests a potential link between hypoxia responses and osteoclastogenesis. 6 Furthermore, it was observed that the stable expression of HIF-1α resulted in impaired osteoclast differentiation of bone marrow-derived macrophages (BMMs). 7 This indicates that HIF-1α has a dual role in the regulation of osteoclasts. However, the bidirectional regulation of hypoxia on HIF-1α expression and the impact of HIF-1α on osteoclast differentiation have not been fully elucidated. 8
Vascular endothelial growth factor (VEGF) is a pro-angiogenic cytokine that is strongly induced by HIF-1α and drives angiogenesis. Interleukin 6 (IL-6) is a multifunctional cytokine that can affect osteoclast differentiation. Angiopoietin-like 4 (ANGPTL4) is an adipokine with pro-angiogenic functions that can enhance osteoclast proliferation and differentiation. Previous studies have shown that VEGF and ANGPTL4 are downstream target genes of HIF-1α.9,10 Additionally, our research group has demonstrated that IL-6 is also a downstream target gene of HIF-1α, which is involved in the regulation of bone resorption activity. 11 The activation of HIF-1α and its downstream pathways, including VEGF, IL-6, and ANGPTL4, play crucial roles in mediating the effects of SAL on osteoclasts.
Many plant-derived natural products have been used in traditional medicine for the treatment of bone loss-related diseases. Salidroside (SAL), the main active ingredient in Rhodiola rosea, has been studied extensively for its anti-inflammatory, 12 anti-hypoxia 13 and anti-oxidative 14 properties. Previous study revealed that SAL promoted angiogenesis within the callus and improves trabecular bone microarchitecture through the activation of HIF-1α signaling in vivo15,16 However, whether the effect of SAL on osteoclasts is related to HIF-1α activation has not been reported. In this study, we showed that SAL has the ability to enhance the expression, nuclear translocation, and transcriptional activity of HIF-1α in osteoclasts. This activation of HIF-1α leads to the promotion of osteoclastogenesis by activating the HIF-1α/VEGF, ANGPTL4 and IL-6 pathways in osteoclasts. This suggests that SAL could affect osteoclasts and enhance their formation. The activation of HIF-1α and its downstream pathways plays crucial roles in mediating the effects of SAL on osteoclasts.
Materials and methods
Materials
α-MEM (SH30265.01), DMEM (SH30021.01), FBS (SH30084.03), penicillin/streptomycin (Sv30010) were purchased from Hyclone, Logan, USA. Osteoclastogenesis‐inducing factors M-CSF (0815612), RANKL (0817245) were purchased from PeproTech, NJ, USA. Lipofectamine™ 2000 was purchased from Invitrogen, CA, USA. Salidroside (S25475, C14H20O7, ≥98%) was procured from YUANYE Bio-Technology, Shanghai, China. CoCl2 (232696, ≥97%), YC-1(Y102) were procured from Sigma, MO, USA. Avastin (H0201B05) was procured from Roche, IN, USA. HIF-1α (ab1), Anti-IL-6 (ab9324) were procured from Abcam, Cambridge, UK. Anti-β-actin (RM2001) was procured from Ray Antibody Biotech, Beijing, China. MBL (DF4152) and IgG (S0002) were procured from Affinity Biosciences, Ohio, USA. TRAP kit (G1492), MTT solution (M8180), Red Blood Cell Lysis (R1010), RIPA lysis buffer (R0010), Toluidine Blue O solution (G3663), 5% BSA (SW3015) and Annexin V-FITC/PI kit (CA1020) were procured from Solarbio, Beijing, China. Trizol reagent (DP424) was procured from Tiangen Biotech, Beijing, China. SYBR Green qPCR SuperMix (AQ601-03) was procured from TransGen Biotech, Beijing, China. HiFiScript cDNA Synthesis Kit (CW2569 M) and BCA kit (CW0014S) were procured from CWbio, Beijing, China. RANKL ELISA kit (EK1208) was procured from Multisciences Biotech, Shanghai, China. EasyBlot ECL kit (WBKLS0100) was procured from Millipore, MA, USA.
The extraction of primary osteoclasts
The femur and tibia of 6-week-old C57BL/6 mice were stripped, and the bone marrow cavity was flushed with α-MEM medium (containing 1% penicillin/streptomycin). The culture fluid from bone marrow cavity was collected and centrifuged to discard the supernatant. Subsequently, red blood cell lysis buffer was added for 5 min, followed by centrifugation to discard the supernatant. The cells were cultured in α-MEM complete medium (containing 12% FBS, 1% penicillin/streptomycin). After 24 h, the cell supernatant was discarded and combined with α-MEM complete medium supplemented with M-CSF (50 ng/mL) and RANKL (50 ng/mL). Mature osteoclasts derived from bone marrow macrophages (BMMs) could be obtained after 1 week of culture.
Cell culture and transfection
Mouse preosteoclast cell line RAW264.7 was obtained from the American Type Culture Collection (ATCC, USA) and cultured in DMEM complete medium (containing 10% FBS, 1% penicillin/streptomycin). RAW264.7 cells were induced to differentiate into mature osteoblasts by treatment with M-CSF (50 ng/mL) and RANKL (50 ng/mL). BMMs was cultured α-MEM complete medium (containing 12% FBS, 1% penicillin/streptomycin). RAW264.7 cells were transfected with the pGL3-4×HRE-luc plasmid using Lipofectamine™ 2000 (Invitrogen, San Diego, CA) and treated with SAL under hypoxic conditions.
Cell grouping and treatment
Cells were exposed to different concentrations of SAL solutions (1 nM, 10 nM, 100 nM, 1000 nM). The hypoxic groups were cultivated in a hypoxic incubator with 1% O2, or treated with CoCl2 (0.5 mM) for 24 h after 24 h treatment with SAL. In the blocking group, the following subgroups were added to the osteoblasts: HIF-1α specific blocking antibody YC-1 (10 μm) added 1 h in advance. VEGF inhibitor Avastin (4 μg/mL), Monoclonal anti-IL-6 antibody (0.3 μg/mL), ANGPTL4 inhibitor MBL (10 μg/mL), RANKL inhibitor Denosumab (10 μg/mL) and IgG (control antibody) were added with SAL simultaneously.
Luciferase (luc) reporter assay
The RAW264.7 was treated with SAL for 24 h and then subjected to different experimental conditions: incubation in a regular incubator or treatment with CoCl2 for 24 h. Following the treatment period, the cellular structures were subjected to lysis, subsequently enabling the quantification of luc activity through the utilization of the Luciferase Assay System provided by Promega. Luc activity was normalized to β-Galactosidase activity. The measurement of luc activity was carried out using a Thermo Fluoroskan Ascent FL microplate fluorometer and luminometer from Thermo Electron Corporation. In addition to the luc assay, β-galactosidase activity was also detected using the ONPG substrate from Sigma. The measurement of β-galactosidase activity was performed at 420 nm using a microplate reader from Thermo Fisher.
Methylthiazolyldiphenyl-tetrazolium (MTT)
RAW264.7 and BMMs were exposed to various concentrations of SAL solutions (1 nM, 10 nM, 100 nM, 1000 nM) for 48 h. In the hypoxic groups, the cells were treated with 0.5 mM CoCl2 for 24 h after SAL treatment for 24 h in a conventional culture environment. Following 48 h incubation period, the medium was extracted, and 20 μL of 5 mg/mL MTT solution was introduced into each well and incubated at 37°C for 4 h. Absorbance at 490 nm was measured using a microplate reader, and cell proliferation was calculated according to the following formula: Relative cell viability = (experimental group/control) × 100%. All experiments were repeated at least three times.
RNA isolation and quantitative real-time PCR (qPCR)
Details of qPCR primer sequence.

Enzyme-linked immunosorbent assay (ELISA)
Following the treatment period, the supernatants were collected and subjected to clarification through centrifugation. The concentration of VEGF, ANGPTL4 and IL-6 were subsequently assessed utilizing ELISA kits in accordance with the guidelines. The ELISA experiments were repeated three times.
Western blot
The cells were subjected to RIPA lysis buffer (R0010, Solarbio) for duration of 30 min, and the resulting protein supernatant was quantified utilizing BCA kit. The proteins were separated by 10% SDS-PAGE gel and were transferred onto polyvinylidene fluoride (PVDF) membranes at the temperature of 4°C. Subsequently, the membranes were then blocked with 5% BSA and subjected to incubation with primary antibodies, including Anti-HIF-1α (1:800) and Anti-β-actin (1:1500). Afterward, the membranes were developed using the EasyBlot ECL kit and analyzed with the multifunctional imager. The results were detected by a Tanon 5500 chemiluminescent imager system (Tanon Science & Technology; Shanghai, China) and standardiz to β-actin protein. Western blotting experiments were repeated three times.
Immunofluorescence
For visualization of the cellular localization of HIF-1α, cells were fixed and permeabilized with 0.5% Triton X-100. To minimize non-specific binding, cells were then blocked with an appropriate blocking solution. Subsequently, cells were incubated with the HIF-1α antibody, followed by incubation with a secondary antibody conjugated with FITC. Finally, fluorescent signals were captured using light microscope for imaging. Immunofluorescence was performed with three independent experiments.
Flow cytometry assay
An assessment of the impact of SAL on apoptosis in osteoclasts was conducted using the Annexin V-FITC/PI kit through flow cytometry assay. The cells were incubated with 100 μL of Binding Buffer (1X) and 5 μL of Annexin V-FITC for 10 min in dark environment. Then, 10 μL of Propidium Iodide (PI) was introduced to the cells. Ultimately, the flow cytometer was utilized to analyze cell apoptosis. Flow cytometry assay was performed with three independent experiments.
Tartrate resistant acid phosphatase (TRAP) staining
To assess the formation of osteoclasts, TRAP staining was conducted using TRAP Stain Kit in accordance with the provided guidelines. We randomly selected three views under microscopy (Olympus IX71, USA). TRAP-positive multinucleated (number of nuclei ≥3) cells were scored as osteoclasts. The number and spread area of multinucleated (number of nuclei ≥3) osteoclasts were quantified under microscopy.
Bone resorption assay
Slices of bone were used in the bone resorption assay. Initially, the bone slices were polished to a thickness of 100 μm using a polishing machine. Subsequently, the bone pieces were cut into 5 mm × 5 mm size, rinsed twice with PBS, and subjected to ultrasonic cleaning for 10 min. The sterilized bone slices were then stored in DMEM medium supplemented with 1% Penicillin-Streptomycin. Following this, the bone slices were treated with SAL continuously. Hypoxic incubator and regular incubator were both used to incubate the cells. In co-culture experiments, RAW264.7 and BMMs were stimulated with RANKL and M-CSF until osteoclast formation was observed. These cells seeded onto the treated bone slices and cultured with CM derived from MC3T3-E1 and MOB. After 14 days of incubation, the bone slices were removed for staining with Toluidine Blue O solution. Subsequent to fixation, washing, dehydration, and staining, the bone lacunae were observed and measured in vitro. Image J software was used to analyze the percentage of resorption areas of bone slices. Bone resorption experiment was performed with three independent experiments.
Statistical analysis
The results of the statistical analysis were analyzed using SPSS Statistics 21.0 software (IBM, Armonk, NY, USA). One-way analysis of variance (ANOVA)was used to evaluate significant differences in normally distributed data. All data were presented as mean ± standard deviation (SD). Graphing and further analysis were conducted using GraphPad Prism seven software (GraphPad Software). The significance levels were denoted as *p < 0.05, indicating statistical significance.
Results
SAL up-regulated the expression of HIF-1α pathway in osteoclasts
To investigate the impact of SAL on the HIF-1α pathway, the gene and protein expression levels of HIF-1α, VEGF, ANGPTL4 and IL-6 in osteoclasts were assessed using qPCR, Western blotting, and ELISA, respectively. Hypoxia was found to enhance the expression of HIF-1α in both RAW264.7 and BMMs, and SAL further augmented this effect (Figure 1(a) and (b)). Additionally, upon the induction of RAW264.7 to mature into osteoclasts using M-CSF and RANKL, the expression of each molecule in the HIF-1α pathway was upregulated, and consistent with previous findings (Figure 2(a)). However, the effect of SAL was nullified after the addition of the HIF-1α-specific blocking antibody YC-1 (Figure 2(b)). The findings of this study suggest that SAL has the potential to enhance the expression of HIF-1α and downstream target genes, including VEGF, ANGPTL4 and IL-6, under hypoxic conditions in osteoclasts. The effect of SAL on gene and protein expression of HIF-1α, VEGF, ANGPTL4 and IL-6 in osteoclasts. (a) SAL promoted expression of HIF-1α, VEGF, ANGPTL4 and IL-6 in RAW264.7. (b) SAL enhanced expression of HIF-1α, VEGF, ANGPTL4 and IL-6 in RAW264.7. The results are presented as the mean ± SD, and the data shown are representative of three independent experiments. *p < 0.05. The effect of SAL on gene and protein expression of HIF-1α, VEGF, ANGPTL4 and IL-6 in osteoclasts. (a) SAL promoted expression of HIF-1α, VEGF, ANGPTL4 and IL-6 in RAW264.7 after stimulated with RANKL and M-CSF. (b) The effect of SAL on RAW264.7 were blocked with HIF-1α-specific blocking antibody YC-1. The results are presented as the mean ± SD, and the data shown are representative of three independent experiments. *p < 0.05.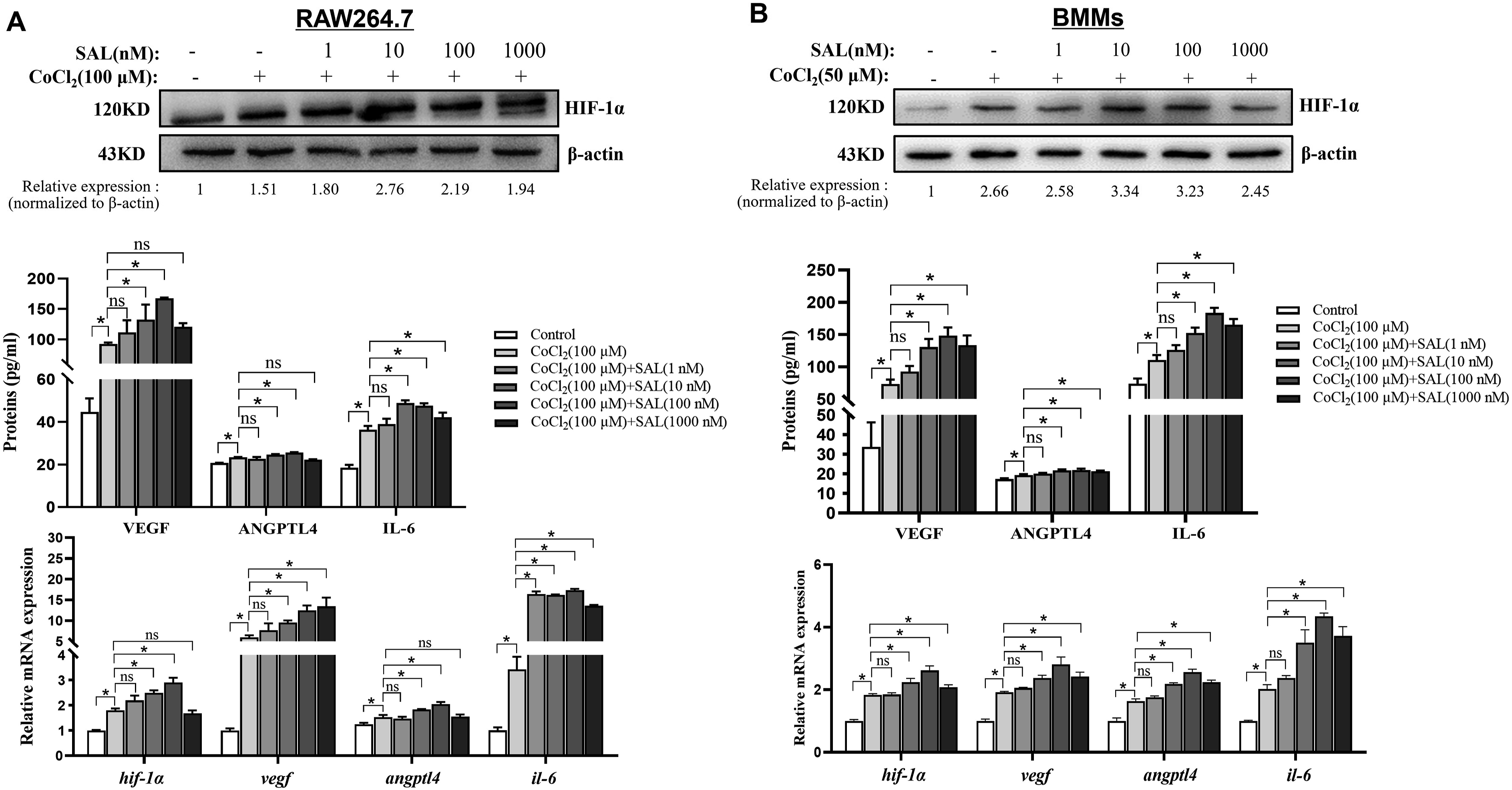

SAL promoted HIF-1α nuclear translocation and transcription activity in osteoclasts
The fluorescence intensity and protein expression of HIF-1α in RAW264.7 under hypoxia were assessed using immunofluorescence and Western blotting, respectively. The results depicted in Figure 3(a) demonstrate that SAL significantly enhanced the fluorescence intensity and protein expression of HIF-1α in the nucleus of RAW264.7 under hypoxia. Furthermore, the protein expression of HIF-1α in the nucleus was found to be 2-3 times higher than in the cytoplasm, indicating that SAL, under hypoxic conditions, facilitated the nuclear translocation of HIF-1α. This observation aligns with the findings from immunofluorescence analysis. Moreover, when RAW264.7 were transfected with pGL3-4×HRE-luc and treated with SAL under hypoxia, there was a notable increase in the relative activity of HRE-luc, as depicted in Figure 3(b). However, the effects of SAL were abolished upon the addition of YC-1 (Figure 3(c) and (d)). These results suggest that the stimulation of SAL facilitated the translocation of HIF-1α into the nucleus and enhanced the transcriptional activity within osteoclasts, subsequently regulating the expression of downstream target genes. The effect of SAL on HIF-1α nuclear translocation and transcription activity in osteoclasts. (a) SAL enhanced the fluorescence intensity and protein expression of HIF-1α in the nucleus of RAW264.7. (b) SAL promoted transcriptional activity of HIF-1α in RAW264.7. (c, d) The effect of SAL on HIF-1α nuclear translocation and transcription activity of RAW264.7 were blocked with HIF-1α-specific blocking antibody YC-1. The results are presented as the mean ± SD, and the data shown are representative of three independent experiments. *p < 0.05.
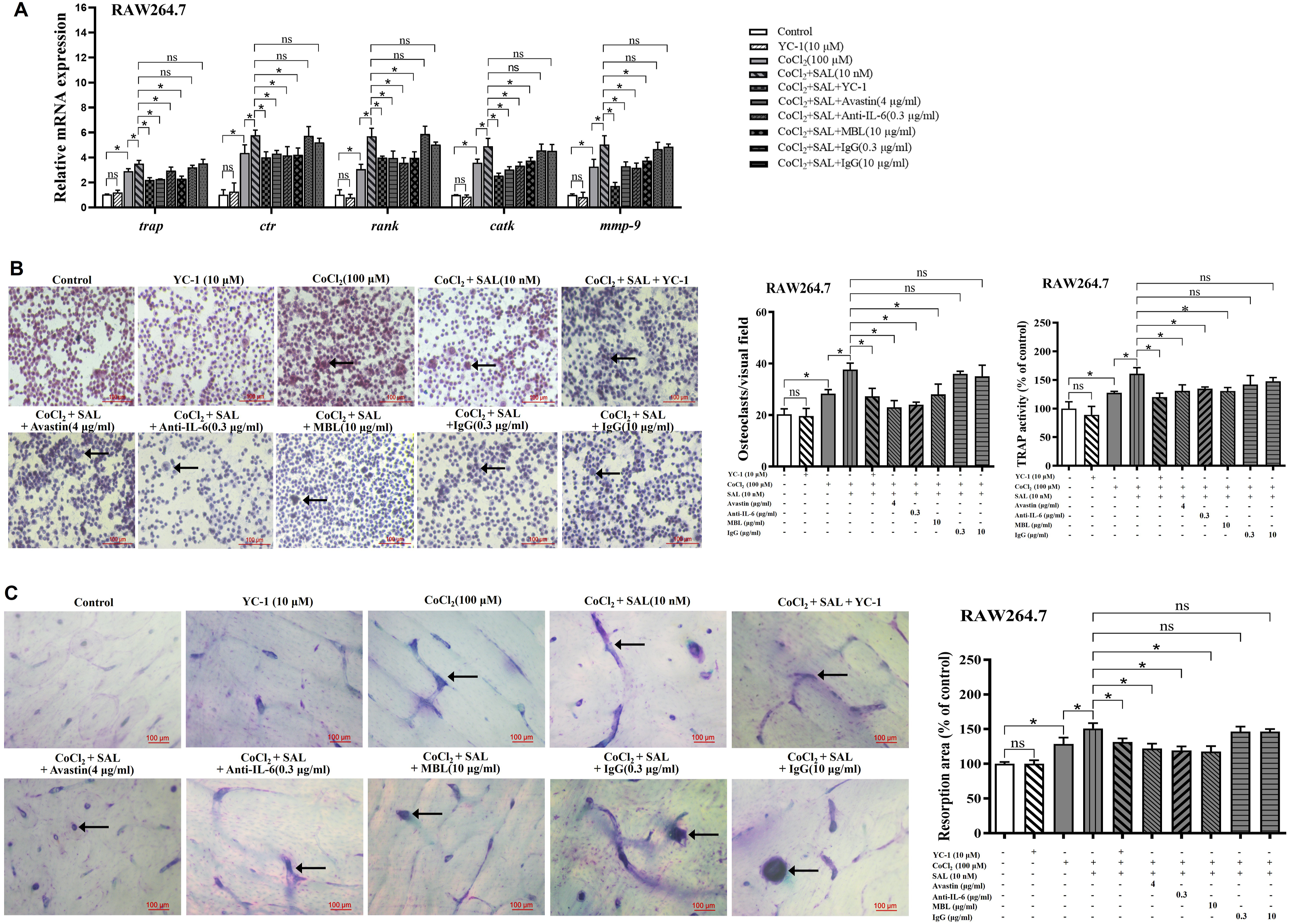
SAL promoted osteoclastogenesis via HIF-1α pathway in an autocrine manner
To investigate the effect of SAL on osteoclasts, various assays were employed, including MTT, flow cytometry, qPCR, TRAP staining, and bone resorption assays. It was observed that hypoxia promoted cell viability in RAW264.7 and BMMs, and osteoclast viability was significantly increased at SAL concentrations of 10 nM and 100 nM (Figure 4(a)). Thus, subsequent experiments utilized these concentrations as the effective SAL concentrations. Flow cytometric analysis revealed no significant changes in the apoptosis rate of RAW264.7 at SAL concentrations of 10 nM and 100 nM, with or without the addition of RANKL and M-CSF (Figure 4(b)). Based on these findings, further investigations were conducted on osteoclast differentiation and function. Trap, ctr, rank, catk and mmp-9 are crucial genes involved in osteoclast differentiation and function. The expression of these genes in osteoclasts was promoted to varying levels by SAL under normoxic or hypoxic conditions (Figure 4(c) and (d)). The effects of SAL on cell viability and differentiation related genes expression of osteoclasts. (a) MTT assay was used to determine the effective SAL concentrations. (b) SAL did not affect the apoptosis rate of RAW264.7. (c) SAL promoted the gene expression of trap, ctr, rank, catk and mmp-9 in RAW264.7. (d) SAL promoted the gene expression of trap, ctr, rank, catk and mmp-9 in BMMs. The results are presented as the mean ± SD, and the data shown are representative of three independent experiments. *p < 0.05.
TRAP staining and activity assays were performed to explore the impact of SAL on RANKL-induced osteoclast differentiation. Osteoclasts were identified as TRAP-positive cells exhibiting the presence of three or more nuclei, as indicated by arrows. The findings from the TRAP staining and activity assays indicated that SAL significantly increased the number of multinucleated osteoclasts and enhanced TRAP activity under hypoxia (Figure 5(a) and (b)). Furthermore, mature osteoclasts possess the ability to degrade the bone matrix and create cavities which aid in the process of bone resorption. To validate the effects of SAL on the bone resorptive activity of osteoclasts under hypoxic conditions, bone resorption tests were performed using bovine bone slices. As shown in Figure 5(c) and (d), SAL effectively facilitated bone resorption in both RAW264.7 and BMMs under hypoxia. The effects of SAL on osteoclasts were significantly reduced when the HIF-1α specific blocking antibody YC-1 and neutralizing antibodies against VEGF, IL-6 and ANGPTL4 were added (Figure 6). This suggests that SAL promotes osteoclastogenesis and initiates an autocrine positive feedback loop through the HIF-1α pathway. In summary, SAL has the ability to promote osteoclast function and regulate osteoclastogenesis through the HIF-1α pathway. The effects of SAL on differentiation and bone resorption activity of osteoclasts. (a, b) SAL increased the number of multinucleated osteoclasts and enhanced TRAP activity of osteoclasts. (c, d) SAL facilitated bone resorption of osteoclasts. The results are presented as the mean ± SD, and the data shown are representative of three independent experiments. *p < 0.05. The effects of the HIF-1α specific blocker YC-1 and neutralizing antibodies against VEGF, IL-6 and ANGPTL4 on SAL-treated osteoclasts. (a) The effect of SAL on differentiation related genes expression of osteoclasts were blocked with blocking antibodies. (b) The effect of SAL on TRAP staining and activity assays of osteoclasts were blocked with blocking antibodies. (c) The effect of SAL on bone resorption of osteoclasts were blocked with blocking antibodies. The results are presented as the mean ± SD, and the data shown are representative of three independent experiments. *p < 0.05.
Discussion
Hypoxia, a condition of low oxygen levels, has been shown to stimulate bone regeneration through the HIF-1α pathway. 19 The activation of HIF-1α is intricately linked with neoangiogenesis and serves as a critical factor in both skeletal development and the process of bone fracture healing.10,20 VEGF is a growth factor that assumes a crucial role in angiogenesis, particularly during the process of endochondral ossification in bone repair. 21 HIF-1α is responsible for inducing VEGF expression, thereby promoting angiogenesis and aiding in fracture repair. 22 Our previous study found that SAL prevents bone loss via HIF-1α/VEGF pathway-induced angiogenesis. 15 Moreover, the nuclear translocation and transcriptional activity of HIF-1α, as well as the expression of the HIF-1α-responsive gene VEGF, are increased following treatment with SAL. 16 These findings highlight the crucial role of VEGF and the HIF-1α pathway in bone regeneration. ANGPTL4 is a secreted adipokine that has the potential to promote angiogenesis independently of VEGF. 23 It has been shown that ANGPTL4 is a downstream target gene of HIF-1α, as demonstrated by DNA chip technologies. 9 Additionally, ANGPTL4 has been found to promote monocyte proliferation and osteoclast bone resorption. Further studies have revealed that ANGPTL4 can compensate for the deficiency of HIF-1α in stimulating osteoclast activity and promoting osteoclast differentiation. Research by Inoue using ChIP-seq and microarray analysis has confirmed that ANGPTL4 can be regulated as a target gene. 24 IL-6 is a cytokine produced by various cell types. Chromatin immunoprecipitation experiments have shown that hypoxia increases the DNA binding of HIF-1α to the IL-6 promoter. 11 Both ANGPTL4 and IL-6 are downstream target genes of HIF-1α. These genes are involved in the regulation of osteoclast differentiation and function. In this study, we examined the effects of hypoxia on the activity and differentiation of osteoclasts by using RAW264.7 and primary BMMs as the research subjects. We found that hypoxia acts on HIF-1α nuclear translocation and transcription, promoting the expression of HIF-1α and its downstream target genes such as VEGF, ANGPTL4, and IL-6, thereby regulating osteoclast activity and differentiation. In this study, we demonstrated that SAL has the potential to activate osteoclast differentiation through the HIF-1α/VEGF, ANGPTL4, and IL-6 pathways.
Rhodiola crenulata is a medicinal herb that is commonly found in high-altitude regions of Tibet. It has been known to improve tissue oxygenation, helping individuals adapt to high-altitude environments and alleviating symptoms associated with high altitude.25–27 The main active ingredient of R. crenulata, SAL, has recently been confirmed to up-regulate the expression level of HIF-1α in various diseases.28,29 In this study, the dosage of SAL was determined by referencing our previously used concentration.15,16 SAL activated HIF-1α pathway to promote the expression of downstream target genes in osteoclasts, which in turn promoted proliferation, differentiation and bone resorption of osteoclasts. However, this is contrary to the proven therapeutic role of SAL in models of osteolytic disease and osteoporosis. This phenomenon could potentially be attributed to the ongoing process of bone remodeling, resulting in the differentiation of osteoclasts and subsequent activation of bone resorption activity. The process of bone tissue remodeling is characterized by the synchronized activities of various cells, wherein osteoclasts are responsible for bone resorption, while osteoblasts are responsible for bone formation. The timeframes for these processes are approximately 7 to 10 days for bone resorption and 2 to 3 months for bone formation. 30 The regulation of bone resorption and formation entails a intricate interplay of diverse factors, including cytokines, hormones, and external biomechanical stimuli.31,32 These factors tightly control the balance between bone resorption and formation. The occurrence of any disturbance within this intricate balance has the potential to result in the skeletal system disorders.33–35 Additionally, hypoxic conditions have been shown to promote osteoclastogenesis, leading to increased osteoclastic function and bone resorption through activating the HIF-1α pathway, increasing glycolysis, inhibiting ferroptosis.36–39 Given the significance of HIF-1α in bone regeneration, it represents a potential target for inhibiting bone resorption and treating osteolytic bone diseases.
Based on the aforementioned findings, we are not able to exclude the presence of other correlated factors. Hypoxia is a prevalent condition within the microenvironment of the skeletal system. The bone lining cells, which include osteoblasts, osteoclasts, and periosteal and endosteal cells, collectively participate in the repair process. Therefore, the anti-anoxic mechanisms of SAL in this model may exhibit variations compared to its impact on cellular functions. It is necessary to gain a comprehensive understanding of the mechanisms that govern the cell-cell interactions, as well as to ascertain efficacious therapeutic approaches. Other studies have shown the existence of a bidirectional regulatory mechanism of diseases due to differences in drug concentrations. For example, pretreatment with low concentrations of SNP (0.3 mM) can inhibit subsequent cell apoptosis induced by high concentrations of SNP (1.5 mM). 40 High concentrations of hydroxychloroquine can completely inhibit DNA-induced stimulation. However, it has been found that low concentrations of hydroxychloroquine have the opposite effect, including increased expression of the co-stimulatory molecules CD40 and HLA-DR molecules by B cells, increased B cell proliferation, and antibody production. The study explains the reasons for the poor clinical treatment outcomes in systemic lupus erythematosus (SLE) patients with poor compliance. 41 Many studies have shown that alcohol consumption is closely associated with the occurrence of cardiovascular diseases, significantly increasing the incidence of ischemic stroke and angina.42–44 However, low-level alcohol consumption can inhibit high-fructose-related myocardial hypertrophy, cardiac mitochondrial damage, and glucose-lipid metabolism disorders. 45 In this study, it was found that SAL has the potential to promote osteoclast differentiation and function. However, by comparing the effects of concentrations of 100 nM and 1000 nM on osteoclasts, it was found that the promoting effect of 1000 nM on osteoclasts was weakened. This suggests that the effect of SAL on osteoclasts may be concentration-dependent. Based on the above, future studies will use higher concentrations of SAL and combine them with multicellular models to comprehensively assess the mechanism of action of SAL on osteoclast differentiation, in order to provide a molecular biology basis for the clinical use of SAL.
Conclusions
This research confirms that SAL can promote osteoclast proliferation, differentiation and bone resorption through HIF-1α/VEGF, IL-6 and ANGPTL4 pathways.
Footnotes
Author contributions
The authors of this study made the following contributions: Yue Wang and Zhengyang Li designed the experiments, Yutong Jin and Lingling Zhang analyzed the data and wrote the manuscript, Dandan Gao, Lin Qi, Chenchen Tian and Haizhao Liu performed the experiments, Qingwen Cao prepared the figures. Yao Wang and Qun Xia reviewed and revised the article. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Nos. 81572852, 82104671), the Great Program of the Science Foundation of Tianjin (Nos. 18JCZDJC33200), Heilongjiang Province Fund (Nos. LH2020H102), Tianjin University of Traditional Chinese Medicine Talented Youth Program (Nos. XJS2023108), Ministry of Education “Chunhui plan” cooperative scientific research project (HLJ2019034).
Ethical statement
Data availability statement
The data that support the findings of this study are not publicly available due to confidentiality but are available from the corresponding author.
