Abstract
Few preliminary reports studied the utility of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) for differentiation between infantile hemangiomas (IHs) and vascular malformations. The aim of this study was to investigate the role of serum VEGF and bFGF levels in differentiating IHs from vascular malformations and identifying the stage and clinical course of IHs. Serum levels of VEGF and bFGF were assessed in 60 infants and children with various cutaneous vascular anomalies defined in 3 groups: proliferating IHs (n = 25), involuting IHs (n = 23), and vascular malformations (n = 12), in comparison with their levels in 40 healthy matched control. Serum levels of VEGF and bFGF were significantly elevated in all groups as compared to control (
Keywords
Introduction
Infantile hemangioma (IH) is the most common benign vascular lesion occurring in infancy and childhood, with an incidence ranging from 1% to 12% depending on age and population studied. 1 Infantile hemangiomas usually develop soon after birth due to endothelial cell proliferation. They have a characteristic clinical life cycle consisting of progressive growth after birth followed by spontaneous involution over a course of 3 to 10 years. 2 In contrast, vascular malformations are congenital vascular defects resulting from errors in vascular development that appear at birth. Their histological appearance is of normal endothelium lacking cellular hyperplasia. They never undergo spontaneous involution. 3 Depending upon the size and location, significant functional and esthetic impairment can result from the growth of “problematic” IHs or vascular malformations. Bleeding, pain, and disability are also common. 4 Thus, an in-depth understanding of the natural history of vascular anomalies is critical for practitioners to diagnose and manage these lesions. 5
Vascular endothelial growth factors (VEGFs) are a family of 6 ligands. Vascular endothelial growth factor A is the predominant growth factor associated with endothelial proliferation, migration, and survival. 6 Fibroblast growth factors (FGFs) comprise a structurally related family of 22 molecules. Based on the new nomenclature, FGFs are numbered consecutively, basic fibroblastic growth factor (bFGF) being named FGF-2. 7 Vascular endothelial growth factor and bFGF appear to be essential for vasculogenesis and angiogenesis. 8 However, morphological differences between FGF- and VEGF-induced capillaries argue for differential roles of these growth factors in angiogenesis. 9 Vascular endothelial growth factor, being a potent inducer of vascular permeability, is known to cause edema and lead to formation of hemangiomas in high concentrations. 10 However, these deleterious effects are not reported with FGF over expression. 11 In contrast, FGF-induced, newly formed vessels are often characterized as functionally mature with increased mural cell investment. 12
Few preliminary reports studied the utility of these growth factors for differentiation between IHs and vascular malformations. 13,14 The aim of this study was to investigate the role of serum VEGF and bFGF levels in differentiating IHs from vascular malformations, as well as in assessment of the stage and the clinical course of IHs.
Participants and Methods
This is a cross-sectional, case–control study that included 48 children with IHs (3 males and 45 females) and 12 children with vascular malformations (7 males and 5 females), whose diagnosis was definite and typical based on history and physical examination, and they were attending the Vascular Anomaly Clinic of Ain Shams University hospitals. Forty healthy age- and sex-matched infants and children, free of vascular lesions, were enrolled as a control group between June 2013 and January 2015.
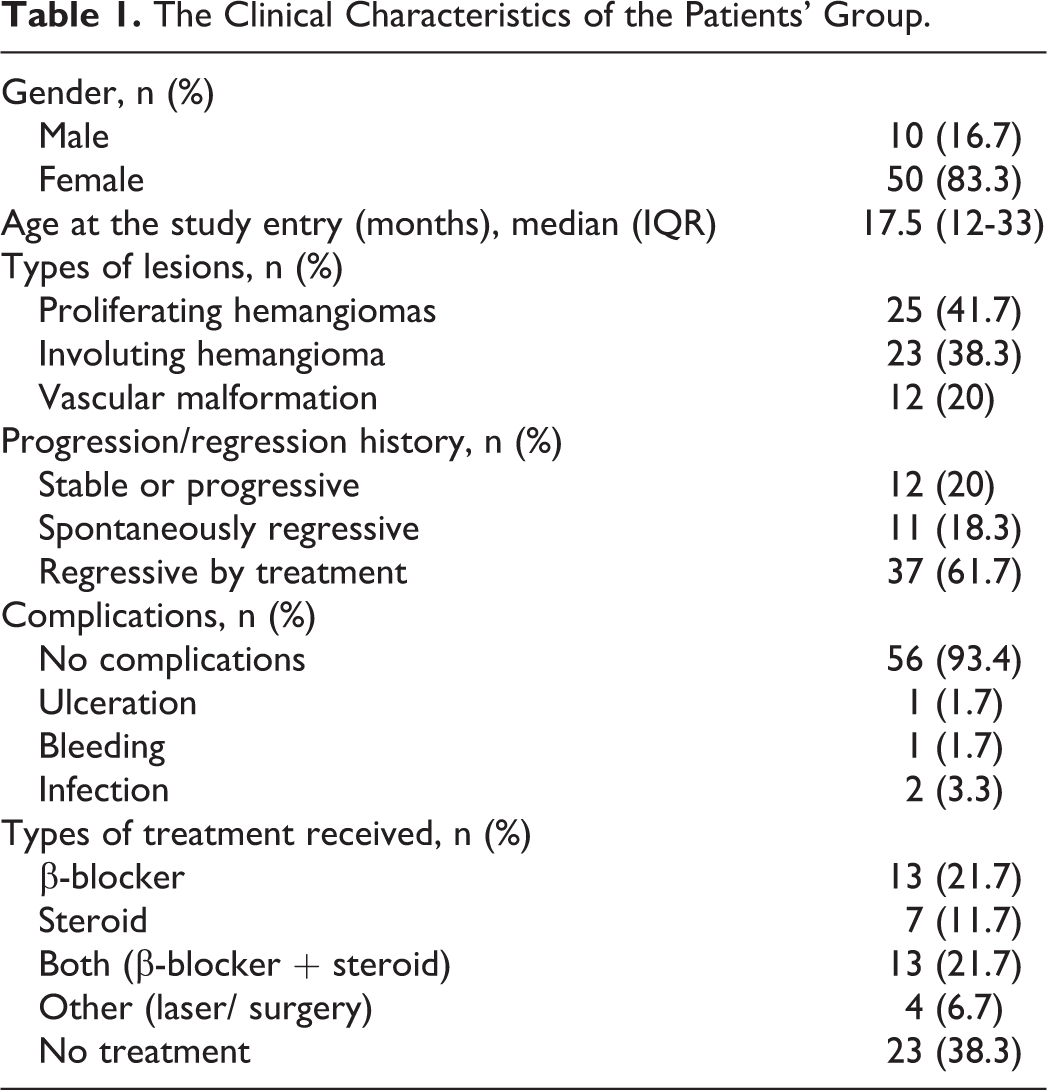
Twenty-five cases with IHs were first reported in children who were less than 12 months of age, and their tumors were enlarging, red in color, and hard on palpation; the lesions were categorized as proliferative hemangiomas. The remaining 23 cases with IH were reported in children more than 12 months of age and had tumors that were regressing gradually or stable; the center surface of the tumors was pale (giving the tumors a white and red appearance), and the tumors were soft on palpation and were categorized as involuting hemangiomas. None of the patients had completely involuted lesions. Vascular malformations included venous malformations and capillary malformations; most of the lesions had been present since birth and had expanded with the child’s development appropriately, with no history of either rapid progression or slow regression (Table 1).
The Clinical Characteristics of the Patients’ Group.
The study was conducted according to the Declaration of Helsinki principles and was approved by the ethical committee of Faculty of Medicine, Ain Shams University. Patients and controls’ guardians signed an informed consent form before inclusion in the study.
Patients were subjected to detailed history taking with special emphasis on maternal history of medical diseases, infections or drug intake during pregnancy, age at first appearance of the lesions, history of progression or regression or stability of the lesion, and current or previous treatments. Thorough examination included lesion location, size, type, color, consistency, compressibility, local temperature, and complications (eg, ulceration, bleeding, pain, infection, high-output cardiac failure, postural or functional difficulties, airway compromise, scarring, eating difficulty, auditory canal occlusion, and ophthalmologic problems), in addition to Doppler ultrasonography.
Patients with other associated deep vascular abnormalities, based on imaging studies, such as visceral hemangiomas, eye hemangiomas, or spinal hemangiomas, were excluded. Patients with current or previous treatment, likely to influence the study outcome, were also excluded.
Patients with IHs were assigned to either regimen of management: conservative or interventional treatment as previously described, 15,16 including systemic therapies such as oral prednisone and subcutaneous interferon (IFN) as well as localized therapies such as intralesional steroids, topical steroids, topical medicaments other than steroids (eg, topical antibacterial agents and colloidal dressings), eye patching, laser treatments, and embolization. Beta-blockers alone or in combination with steroids were the most frequently applied therapeutic interventions in the present study.
The clinical course of the cutaneous vascular anomalies and the response to the given treatment were reported. Patients were categorized into 12 with nonregressive lesions (stationary or increases), 11 with spontaneously regressive lesions, and 37 regressive by treatment (Table 1).
Venous blood samples were withdrawn from all study participants at study entry for assessment of serum levels of VEGF and bFGF by enzyme-linked immunosorbent assay (ELISA) technique. Blood was collected in sterile test tubes and centrifuged for 15 minutes at 50 g. Serum was separated and kept at −20°C until used for ELISA estimation. Vascular endothelial growth factor and bFGF were quantitatively estimated by RayBio human VEGF ELISA kit (ELH-VEGF-001, Norcross, GA) and RayBio human bFGF ELISA kit (ELH-bFGF-001, Norcross, GA), respectively, according to manufacturer instructions. The minimum detectable level of VEGF was 20 pg/mL and for bFGF was 50 pg/mL, with intra-assay and inter-assay coefficient of variation of <10% and <12%, respectively.
Statistical Analysis
The collected data were organized, tabulated, and statistically analyzed, using SPSS version 20 (SPSS Inc, Chicago, Illinois), running on IBM compatible computer with Microsoft Windows 7 Operating System. Quantitative variables were described in the form of mean, standard deviation or median, and interquartile range, and qualitative variables were described as number and percentage. Kolmogorov-Smirnov test was used for testing the distribution of normality. For quantitative data, Student
Results
This study included 60 patients: 10 (16.7%) males and 50 (83.3%) females, with their age at the study entry ranging from 1 month to 13 years (median of 17.5 months), and 40 controls: 11 (27.5%) males and 29 (72.5%) females, with their age ranging from 8 months to 13 years (median of 22 months) with significant female predominance among patients with IH (
Patients with proliferating IH had significantly younger ages and larger surface area of their lesions than those with involuting IH or vascular malformations (
Serum VEGF and bFGF Levels in the Studied Patients Versus Controls.
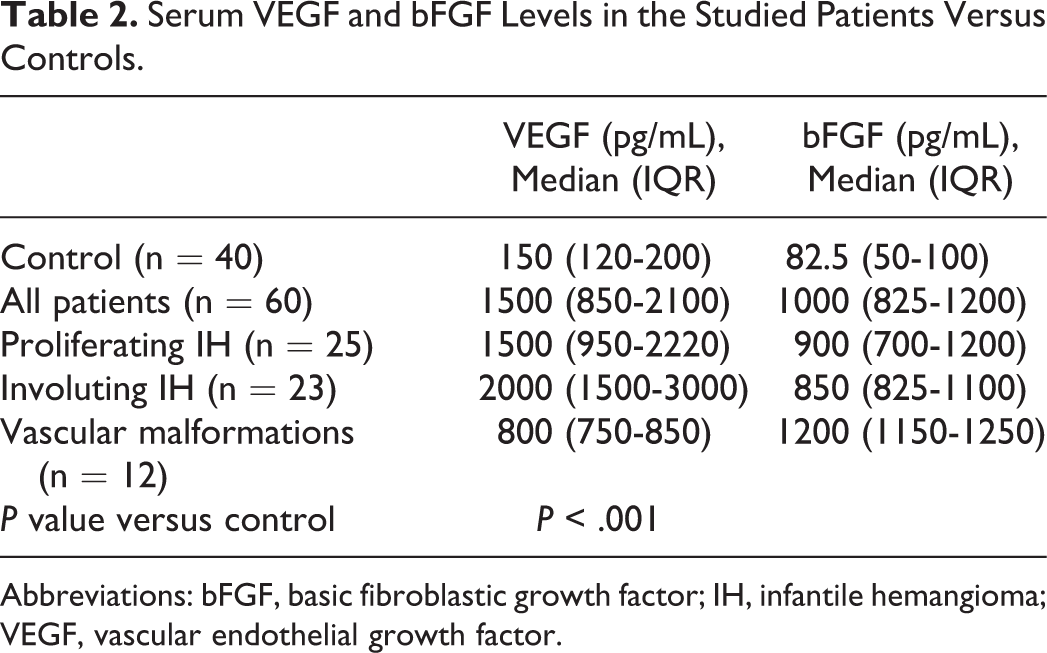
Abbreviations: bFGF, basic fibroblastic growth factor; IH, infantile hemangioma; VEGF, vascular endothelial growth factor.
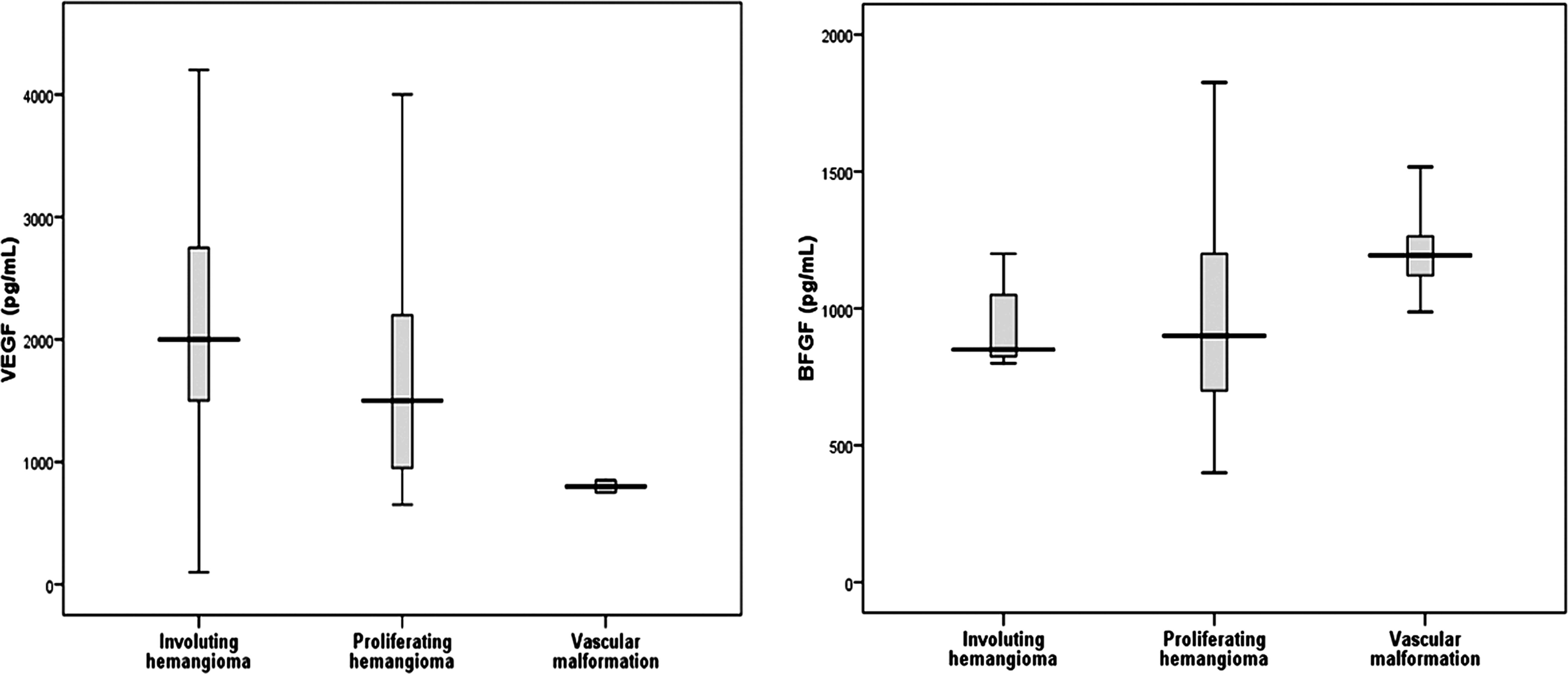
Serum levels of bFGF and VEGF (pg/mL) in the patients’ subgroups. bFGF indicates basic fibroblast growth factor; VEGF, vascular endothelial growth factor.
Upon comparing patients with stable/progressive course and patients with regressive course whether those regressed spontaneously or by treatment using Kruskal-Wallis test, serum VEGF showed significant difference between the 3 groups with overall
Serum VEGF and bFGF Levels in Patients With Stable/Progressive Course and Patients With Regressive Course Whether Spontaneously or by Treatment.
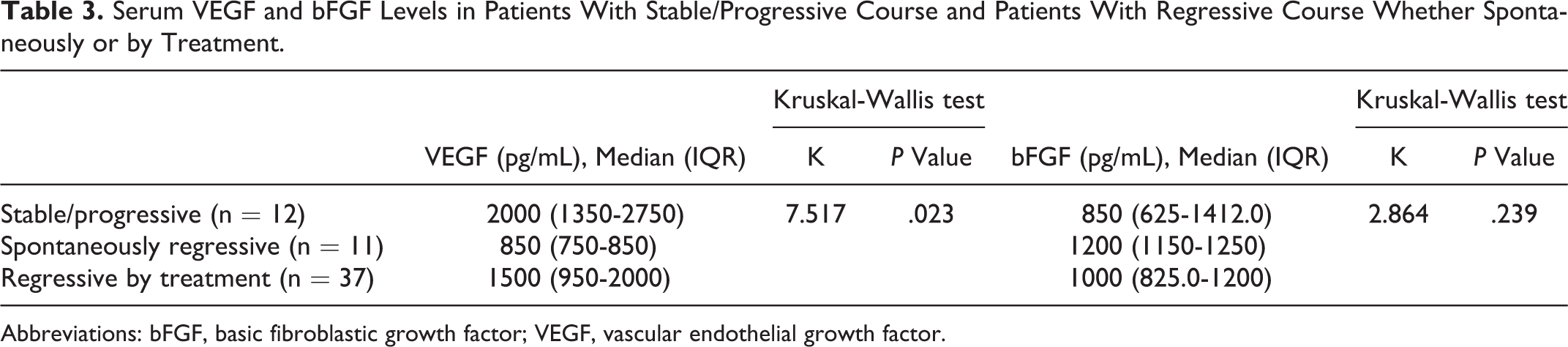
Abbreviations: bFGF, basic fibroblastic growth factor; VEGF, vascular endothelial growth factor.
Correlation studies revealed that VEGF serum levels had a weak negative correlation with the age at diagnosis (
Using ROC curves at multiple cutoff values, a cutoff value of >850 pg/mL for VEGF was the best to differentiate IHs from vascular malformation with a sensitivity of 89.58% and a specificity of 100%, whereas a cutoff value of ≤1100 pg/mL for bFGF has a sensitivity of 72.92% and a specificity of 100%, and measurement of both markers together has a sensitivity of 85.42% and a specificity of 100% to differentiate IHs from vascular malformation. Combined use of both markers together yielded a sensitivity of 78.26% and a specificity of 43.24% to predict the progression of IHs and a sensitivity of 86.96% and a specificity of 29.73% to predict the response to treatment.
Discussion
There is hardly a convenient, noninvasive, and objective method to differentiate IHs from vascular malformations, to determine whether a hemangioma is in the proliferating or involuting phase, or to detect the efficacy of therapeutic intervention or follow up the course of IHs. 1 The present study is designed to investigate an easy noninvasive method to objectively distinguish IHs and vascular malformations, identifying the stage, and the clinical course of IHs.
Most of our hemangioma cases were females (45 of 48 cases). This goes in concordance with epidemiological observations, which revealed that patients with IHs are more likely to be females rather than males. 17,18
In the current analysis, serum VEGF was significantly higher in patients with IHs or vascular malformations, compared with controls. Moreover, it was significantly elevated in patients with IHs, whether proliferating or involuting, compared with vascular malformation cases. This observation may provide an objective basis for distinguishing IHs from vascular malformations.
These findings are consistent with the initial hypothesis of increased activity of this cytokine in IHs and are comparable to the results reported by Zhang et al, 19 Przewratil et al, 13 and Zhang et al. 20
Nevertheless, it was not helpful in differentiating different phases of the tumor, since the difference between proliferating and involuting IHs was nonsignificant. However, several reports have demonstrated a strong association between elevated tissue expression of VEGF in proliferating versus involuting IHs and vascular malformations. 3,21 These reports support the pivotal role of VEGF in the regulation of hemangioma angiogenesis.
Moreover, it is worth mentioning that VEGF could be used to predict lesions that needs therapeutic intervention, since we found a significantly lower VEGF levels in patients whose lesions had spontaneously regressed when compared with those regressed by treatment. This might indicate that VEGF, as an angiogenic cytokine, is helpful in following the clinical course of the lesion and predicting the onset of regression and to decide whether to treat these lesions or not.
The results of our study also demonstrated that serum bFGF levels were significantly elevated in patients with IHs or vascular malformations compared to controls and in patients with vascular malformations compared to those with IHs.
In agreement with our findings, elevated levels of bFGF were found in the urine of patients with IHs, as well as vascular malformations. 20,22 –24 Moreover, bFGF, like VEGF, was reported to be overexpressed in IH tissue, and its expression remains elevated during the early involuting phase. 3,21,22
Although there are few controlled studies, current treatments include complete surgical excision, volume reduction surgery, embolization therapy, sclerotherapy, pulsed dye laser treatment, cryosurgery, topical steroid application, intralesional corticosteroid therapy, high-dose systemic corticosteroid therapy, eosin (a triphenylmethane dye), imiquimod, IFN-α (an angiostatic factor), becaplermin (recombinant platelet-derived growth factor), cyclophosphamide, bleomycin or vincristine, the β-adrenergic receptor antagonist propranolol, and bisphosphonates. 25,26 It has been claimed that the new treatment modalities, such as oral propranolol, would provide the caregiver with a better therapeutic option, which can prevent or minimize medical risk or scarring and long-term squeal. 18,27 Holmes et al 28 considered early treatment with β-blockers as the first-line for rapid treatment for proliferating IHs. In the present study, β-blockers alone or in combination with steroids were the most frequently applied therapeutic interventions.
In conclusion, serum VEGF and/or bFGF levels are increased in cutaneous vascular anomalies and can help differentiate IHs from vascular malformations. None of these markers could help in identifying the stage of IHs. Low VEGF is associated with spontaneous regression of IHs, thus it might be used as a potential marker to monitor the response to therapy.
Footnotes
Authors’ Note
All authors contributed extensively to the work presented in this paper. All authors critically read the manuscript for intellectual content and approved the final version. Informed consent was obtained from all parents before participation in the study. All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ethical Committee of Human Experimentation of Ain Shams University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
