Abstract
This study examined the impact of mercury (Hg) vapor exposure from amalgams among all American pregnant women. Amalgam-Hg vapor exposure among 1,665,890 weighted-pregnant women (
Introduction
Amalgams contain about 50% metallic mercury (Hg0) by weight. They are routinely utilized by dentists to restore a tooth damaged by decay. Each tooth has up to five surfaces, and a filling may cover one or more of these surfaces. 1 American dentists have utilized amalgams for more than 150 years, but the ongoing use of amalgams has been accompanied by significant controversy. 2 The United States (US) Food and Drug Administration (FDA) recognizes amalgams release amounts of Hg vapor dependent on the number of existing fillings and that such Hg vapor exposure may be harmful to certain patients. 3
We recently undertook a study of the 2015-2018 National Health and Nutrition Examination Survey (NHANES) data to examine the impact of amalgams on the American adult population. 4 At least one amalgam surface was present in about 58% of all American adults and significant correlations were observed between the number of amalgam surfaces and urinary Hg excretion. We also estimated daily Hg vapor doses from amalgams and observed that many American adults received daily Hg vapor doses from amalgams in excess of Hg vapor safety limits.
The purpose of the present study is to follow-up on our previous study of amalgams in American adults by examining newly available 2015-2020 NHANES data with a special focus on examining the sub-population of pregnant women. Pregnant women are an important sub-population to examine because developing fetuses are susceptible to many environmental toxins. 5 The aims of this study were to assess among pregnant women: urinary Hg excretion among those with and without amalgams; the correlation between the number of amalgam surfaces and urinary Hg excretion; estimate daily Hg vapor doses received from amalgams per Kilogram (Kg) bodyweight; and estimate the number receiving daily Hg vapor doses from amalgams in excess of Hg vapor safety limits.
Materials and methods
NAHNES data was examined using the Statistical Analysis System (SAS) system for Windows, Version 9.4 (Cary, NC, USA). 2015-2016 and 2017-2020 NHANES data were integrated to examine demographic survey questions, oral health examinations, clinical measurements, and lab test results.
The 2015-2016 NHANES data collection methods were approved by the National Center for Health Statistics (NCHS) Research Ethics Review Board (ERB) (Protocol#2011-17). The 2017-2020 NHANES data collection methods were approved by the NCHS ERB (Protocol#2011-17 and Protocol#2018-01). Each study subject provided informed consent to participate in the NHANES program. The health information collected in the NHANES program is kept in strictest confidence, and is only used for stated purposes.
Study participants
Figure 1 shows a schematic flowchart documenting the selection criteria utilized to assemble the group of pregnant women examined in the NHANES data. An overall population of 319, 402, 608 weighted-persons was examined. The overall population was reduced by including only persons with measured dental filling surface status, urinary Hg levels, bodyweight, urine flow rate, and urine pregnancy test results. In addition, each person included in the present study was required to have a documented demographic variable for gender (male or female), age in years at examination, race (non-Hispanic White, non-Hispanic Black, non-Hispanic Asian, Hispanic, or other – including multi-racial), and country of birth (born in the US or born outside of the US). A flowchart of the persons examined in NHANES.
The weighted number of persons was derived by applying the subsample A weight to each person examined in this study. The subsample A weight was created by the NHANES program, and assigned because urinary Hg levels were only measured from a one-third subsample of NHANES participants 6 years-old or older. The subsample A weight is a measure of the number of persons in the general population that a sampled individual represents and is needed to obtain unbiased estimates of population parameters when sample participants are chosen with unequal probabilities. Since, the NHANES data examined were collected from two separate samples (2015-2016 and 2017-2020), the subsample A weight value for each person was divided by 2, to provide an accurate overall weighted-person value for each person examined. After all of the inclusion study criteria were applied (see below), a total of 43,312,070 weighted-women were assessed for their pregnancy status, of which, a total of 1,665,890 weighted-pregnant women (3.85%) were examined in this study.
Pregnancy status
The pregnancy status of participants was determined by exanimating the urine pregnancy lab test NHANES dataset. The publically available dataset provides pregnancy test results for women between the ages of 22 to 44 years-old. To determine pregnancy status, an Icon 25 human chorionic gonadotropin (hCG) test kit (Beckman Coulter) was utilized. Urine samples were analyzed using a rapid chromatographic immunoassay for the qualitative detection of hCG in urine to aid in the detection of pregnancy. The test utilizes a combination of monoclonal and polyclonal antibodies to selectively detect elevated levels of hCG in urine. There were no changes to measurements during 2015-2020 NHANES data collection.
Amalgam surfaces
The coronal carries assessment was conducted on each pregnant woman by dental examiners, who were dentists licensed in at least one US state, and were examined in the NHANES oral health – dentition dataset. For those pregnant women with filled surfaces, the restoration type of the filled surfaced (amalgam or other) was specified. The total number of dental amalgam filling surfaces or other dental filling surfaces was computed for each pregnant women examined.
Daily Hg vapor exposure from Amalgams
Inductively coupled plasma mass spectrometry (ICP-MS) was used to measure urinary Hg excretion in random spot urine samples. Urinary Hg excretion was determined as micrograms (µg) of Hg/liter (L) of urine. The derived lower limit of detection for urinary Hg excretion was 0.13 µg of Hg/L. There were no changes in urinary Hg testing procedures in the data examined.
Since random spot urine samples, as previously described, can vary depending on the level of hydration and other factors, urine flow rates are collected for each NHANES participant.4,6 Furthermore, given that Hg exposure occurs over a sustained period, it is possible to estimate for each person their daily urinary Hg excretion rate by integrating their urine flow rate and urinary Hg excretion in a random spot urine sample. 4
The urine flow rate for each NHANES participant was determined as the number of milliliters (mL) of urine produced per minute (min) of time. The urine flow rate for each person was then multiplied by 60 (to convert minute to hours) then by 24 (to convert hours to 1 day) and finally by 0.001 (to convert mL to L), to yield the total number of L of urine per 24 h (or per day).
The total daily urinary flow rate for each pregnant woman (L of urine per day) was then multiplied by their random spot urine Hg excretion sample (µg of Hg/L) to determine daily urinary µg of Hg. 4 Once determined, the data were analyzed for all pregnant women to calculate a beta (β)-coefficient between the number of amalgam surfaces and daily urinary Hg excretion using survey regression modeling in SAS. 4 The β-coefficient value reflects the contribution of amalgam surfaces to urinary Hg excretion (i.e., the greater the value, the more contribution, whereas the lower the value, the less contribution).
The daily estimated µg of Hg vapor exposure from amalgams per kilogram (Kg) bodyweight for each pregnant woman was determined by a series of additional calculations. 4 First, for each pregnant woman, their number of amalgam surfaces was multiplied by the β-coefficient derived above (the correlation term describes the relationship between the number of amalgam surfaces and daily urinary Hg excretion). The resulting value for each pregnant woman was the estimated daily urinary Hg excretion from their amalgam surfaces. Second, since this value is only a reflection of urinary Hg excretion from amalgams, it was necessary to determine what fraction of the total Hg vapor dose from amalgams was excreted in the urine. Previous controlled human clinical studies examined the relationship between known amounts of Hg vapor exposure and urinary Hg excretion. These studies revealed that the estimated fraction of urinary Hg excretion from amalgams as compared to total Hg vapor dose from amalgams ranged from 0.10 for one amalgam surface to 0.40 for 128 amalgam surfaces. The following equation expresses the fraction of the total Hg vapor dose received from amalgams excreted in the urine: 0.1 + [(number of amalgam surfaces – 1) x 0.00236].4,7 This equation was applied to each pregnant woman examined by applying their appropriate number of amalgam surfaces, so as to determine the proportion of the total Hg vapor dose from amalgams excreted in the urine. Then, for each pregnant woman, the inverse of their estimated proportion of total urinary Hg excretion from amalgam surfaces was multiplied by their estimated daily amount of urinary µg of Hg from amalgam surfaces. The resultant value for each pregnant woman was their daily total µg of Hg vapor from amalgams. Third, utilizing the weight collected by NHANES for each pregnant woman, the estimated daily Hg vapor from amalgams was divided by each pregnant woman’s weight in Kg. The resultant value for each pregnant woman was their µg of Hg vapor exposure from amalgams/Kg bodyweight/day. 4 It should be noted that those pregnant women with no amalgams would have 0 µg of Hg vapor exposure from their amalgams/Kg bodyweight/day.
Daily Hg vapor exposure from Amalgams versus Hg vapor safety limits
Daily Hg vapor exposure safety limits have been established by governmental health agencies and researchers. The Hg vapor safety limits are: the US Environmental Protection Agency (EPA) at 0.048 µg of Hg/Kg bodyweight/day, the US Agency for Toxic Substances and Disease Registry (ATSDR) at 0.032 µg of Hg/Kg bodyweight/day, Health Canada at 0.011 µg of Hg/Kg bodyweight/day, Richardson et al. (2011) at 0.010 µg of Hg/Kg bodyweight/day, and California’s EPA at 0.005 µg of Hg/Kg bodyweight/day.4,7,8 For each pregnant woman, their estimated µg of Hg from amalgams/Kg bodyweight/day was divided by each of the aforementioned Hg vapor safety limits (also expressed as µg of Hg/Kg bodyweight/day). Pregnant women with computed values >1 were in excess of the safety limit, whereas those with computed values ≤1 were not.
Statistical analyses
The statistical package in SAS was used in all statistical analyses. A two-sided
The survey regression modeling procedure in SAS performs regression analysis for sample survey data. This procedure can handle complex survey sample designs, including designs with stratification, clustering, and unequal weighting. The procedure fits linear models for survey data and computes regression coefficients and their variance-covariance matrix. It also provides significance tests for the model effects and for specified estimable linear functions of the model parameters.
The survey regression modeling procedure in SAS uses elementwise regression to compute the regression coefficient estimators by generalized least squares estimation. The procedure assumes that the regression coefficients are the same across strata and primary sampling units. To estimate the variance-covariance matrix for regression coefficients, this procedure uses either the Taylor series (linearization) method or replication (resampling) methods to estimate sampling errors of estimators, based on complex sample designs.
In the survey regression statistical models constructed for this study (regression models require continuous variables), the categorical variables of race and country of birth were converted to continuous variables as follows: race (Hispanic = 1, non-Hispanic white = 2, non-Hispanic black = 3, non-Hispanic Asian = 4, and other – including multi-racial = 5), and country of birth (born in the US = 1 and born outside of the US = 2). The variables of total amalgam surfaces, body mass index, and age in years were examined as continuous variables. In addition, variables for stratum (NHANES variable: sdmvstra), cluster (NHANES variable: sdmvpsu), and weight (NHANES variable: subsample A weight/2) were employed in the survey statistical models constructed in this study.
Results
Demographic and physical measurements of the pregnant women examined in NHANES.

Kg = kilogram, min = minute, mL = milliliter.
Amalgam exposure and urinary Hg excretion among the pregnant women examined in NHANES

Hg = mercury, L = liter, µg = microgram.
aThe total daily urinary flow rate for each pregnant woman (L of urine per day) was then multiplied by their urinary µg of Hg/L to determine daily urinary µg of Hg.
A summary of the correlation between daily urinary µg of Hg excreted/day and the number of amalgam surfaces in pregnant women.
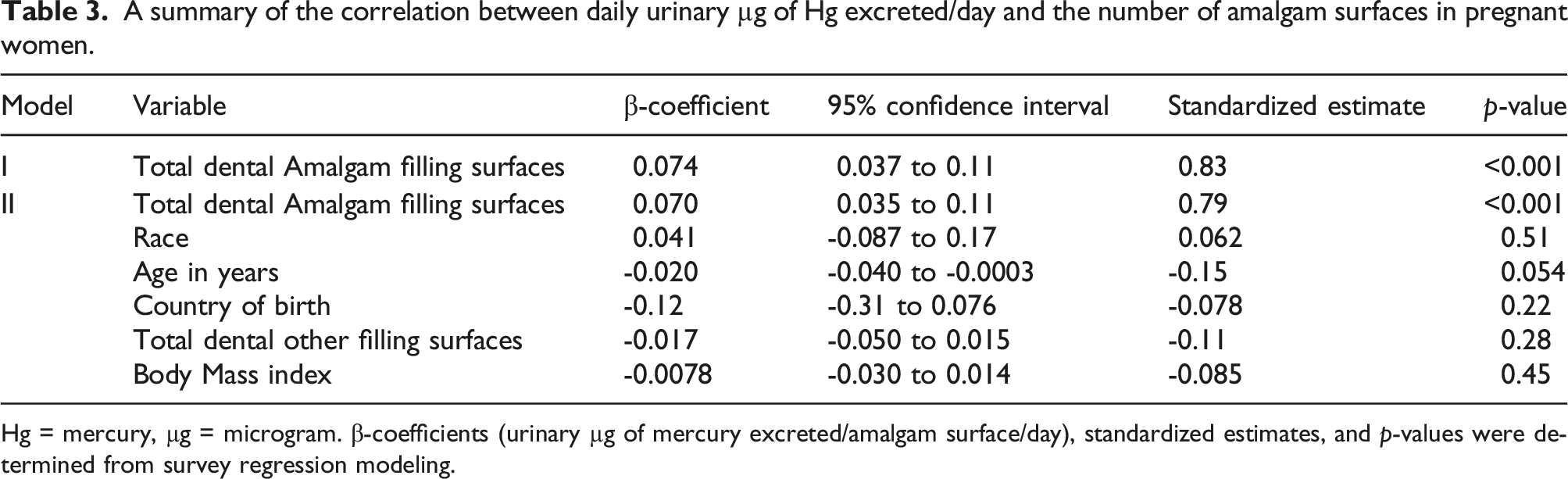
Hg = mercury, µg = microgram. β-coefficients (urinary µg of mercury excreted/amalgam surface/day), standardized estimates, and
A summary of estimated daily Hg vapor exposure from amalgams. a

Kg = kilogram, Hg = mercury, µg = microgram.
aThe total Hg dose each person received from amalgam surfaces was determined by multiplying the β-coefficient (as determined in Table 3) by the number of amalgam surfaces. Then, the resulting value was multiplied by the inverse of the portion of Hg excretion via urine based upon the number of amalgam surfaces (ranging from 0.10 for one amalgam surface to 0.40 for 128 amalgam surfaces). The computed value was then divided by the person’s weight in Kg. The resulting value was each person’s exposure to µg of mercury from amalgam surfaces/Kg bodyweight/day.
A summary of the number of pregnant women receiving estimated daily Hg vapor doses from amalgams in excess various Hg vapor safety limits. a

Kg = kilogram, Hg = mercury, µg = microgram.
aThe value for each person’s exposure to µg of Hg from amalgam surfaces/Kg bodyweight/day was divided by each Hg vapor safety limit (expressed as µg of Hg/Kg bodyweight/day). Those persons with computed values >1 were in excess of the safety limit, whereas all others were not.
Discussion
The present study of NHANES provides the first, nationwide insights into the direct and significant contribution of amalgams on Hg vapor exposure among American pregnant women. Overall, it was observed that about 36% of all American pregnant had at least one amalgam surface. In addition, the median urinary excretion of Hg per day among American pregnant women with amalgams was about 2.5-fold higher compared to American pregnant women with no amalgams. Further, daily urinary Hg excretion significantly correlated with the number of amalgam surfaces. Finally, it was observed that many pregnant women were estimated to receive doses of Hg vapor from amalgams in excess of multiple Hg vapor safety limits and among American pregnant women with at least one amalgam, 100% were in excess of at least one Hg vapor safety limit.
The results of the present NHANES study build-upon and extend our previous NHANES research. 4 We observed in the present study about 36% of all pregnant women had ≥1 amalgam surface, whereas in our previous study, we observed that about 60% of all American adults had ≥1 amalgam surface. This may indicate that variables such as gender and age are important factors in the choice of using amalgams or not. In both studies, we observed significant β-coefficients correlating the number of amalgams surfaces to daily excretion of urinary Hg, but interestingly there was an almost 2-fold increase in the β-coefficient observed in this study (0.074) as compared to our previous study (0.041). This difference may indicate that pregnancy status has an important physiological impact on increasing exposure/distribution of Hg vapor from amalgams. More American pregnant women exceeded the US EPA Hg vapor safety limit (30% vs 10%) and the US ATSDR Hg vapor safety limit (32% vs 21%) in this study as compared to American adults in our previous study.
Outside of our own previous NHANES research, 4 the results observed in this study are most similar to those observed in a study by Richardson. 8 The Richardson study is similar to the present NHANES study, because both studies examined nationally representative samples of health data to evaluate the impact of Hg vapor from amalgams. The Richardson study is differentiated from the present study because data from the 2007-2009 Canadian Health Measures Survey (CHMS) was examined and no specific examination was undertaken to evaluate the impact of Hg vapor exposure from amalgams among pregnant women.
The observation of significant and ongoing elevated exposure to Hg vapor from amalgams among pregnant women in this study is particularly concerning in light of multiple studies demonstrating a significant dose-dependent relationship between maternal amalgams and the amount of Hg in fetal tissues. Studies in sheep and rats demonstrated a significant dose-dependent relationship between maternal amalgams and the amount of Hg in various fetal tissues.9,10 In addition to animal studies, a number of studies were undertaken to evaluate the Hg burden in human fetuses as compared to the number of maternal amalgams during pregnancy. For example, investigators observed significant correlations between maternal amalgams during pregnancy and the amount of Hg present in various fetal tissues. 11 Other researchers demonstrated a significant correlation between maternal amalgams and Hg in the cord blood.12,13 Still other researchers observed that maternal amalgams correlated with the concentration of Hg in amniotic fluid. 14
It is important to recognize that the present study is the first to evaluate Hg vapor exposure from amalgams among all American pregnant women. The uniqueness of NHANES data allowed us to be able to undertake such an ambitious study. NHANES data is collected using an observational cross-sectional study design, but NHANES is unique because it combines interviews and physical examinations. The US CDC describes that the NHANES is one of the greatest public health resources to learn about the health and nutritional status of American adults and children. Given the small subsample of the whole US population (only pregnant women) and all of the data needed to be examined for each pregnant women (many of the variables examined in this study were only collected for a fraction of the whole NHANES population), the present study utilized the continuous collection of NHANES data, over a multi-year period from 2015 to 2020, so as to be able to obtain data on sufficient numbers of pregnant women to evaluate the potential impacts of Hg vapor exposure from amalgams. It is recommended that future studies be undertaken in other populations to evaluate their consistency with the results observed in this study.
There are potential limitations to the results observed. First, there are environmental/social factors that would be more appropriately accounted for in longitudinal cohort studies and not quantified in the NHANES data examined. For example, daily behaviors such as eating or drinking of various foods, tooth brushing, bruxism, etc. may influence the release of Hg vapor from amalgams. 15 As another example, environmental sources of Hg exposure from fish consumption, occupational exposure, or medicinal exposure were not examined. Second, the NAHNES amalgam data examined did not provide the chemical composition or length of placement in the mouth for the amalgam surfaces. It is known that the amalgams containing high amounts of copper relative to amalgams containing low amounts of copper release significantly more Hg. 16 It is also known the contribution of amalgams to the excretion of urinary Hg is related to the time since placement (newer amalgams contributing more to urinary Hg than older amalgams). 17 Third, the Hg vapor safety limits utilized in this study were established by applying uncertainty factors ranging from 30-fold for Health Canada, the US ATSDR, and US EPA Hg vapor safety limits to 100-fold for the Richardson et al. Hg vapor safety limit to 300-fold for the California EPA Hg vapor safety limit derived from data collected on adult males receiving occupational exposure to Hg vapor. 18 The uncertainty factors were applied to establish Hg vapor safety limits ensuring that no negative health outcomes would occur in the populations covered. The consequence of the uncertainty factors is that the absolute risk of Hg intoxication for a given person receiving a dose of Hg vapor exposure from amalgams in excess of the safety limits is not known, but there is an increasing cause for concern with increasing quantities of Hg vapor exposure from amalgams in excess of the Hg vapor safety limits. Fourth, the present study did not measure Hg vapor directly released from amalgams among the persons examined, but instead utilized previously published techniques to estimate daily Hg vapor exposure from amalgams utilizing urinary Hg excretion measurements. The estimates of Hg vapor exposure from amalgams observed in this study are consistent with those reported in previous studies utilizing similar methodologies4,7,8 and are also consistent with daily Hg vapor doses from actual measurements of intra-oral Hg vapor exposure from amalgams. 19 Overall, when considering the aforementioned potential limitations of this study, it is presumed that they would have had a limited impact on our study results, but we recommend that they should be further examined in future studies.
Conclusion
This study is the first to assess the impact of Hg vapor exposure from amalgams among all American pregnant women by analyzing newly available 2015-2020 NHANES data. This study was able to successfully achieve its aims. Pregnant women with amalgams, as compared to those without amalgams, were observed to have significantly greater urinary Hg excretion. Among all pregnant women examined, many were estimated to have received daily Hg vapor doses from amalgams in excess of multiple Hg vapor safety limits, and among those pregnant women with at least one amalgam, all were in excess of at least one Hg vapor safety limit. The results observed in this study are of particular concern because of the demonstration of Hg accumulation in the fetus following Hg vapor exposure from amalgams and the known adverse effects on the developing fetus from significant ongoing Hg vapor exposure during pregnancy. 20 In keeping with the dental amalgam phase-down recommended by the international legally binding 2013 Minamata Convention on Mercury, 21 it is recommended that specific public health programs be introduced to reduce/eliminate amalgams in pregnancy and among women of reproductive age. It is also recommended that future epidemiological studies should be undertaken to evaluate the potential relationship between Hg vapor exposure from amalgams and adverse pregnancy outcomes.
Footnotes
Author contributions
All authors contributed to conceptualization, material preparation, data curation and formal analysis, investigation, methodology, and manuscript writing. All authors read and approved the final manuscript. Each author participated sufficiently in the work to take public responsibility for the content.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Both authors are directors of the nonprofit Institute of Chronic Illnesses, Inc. They are both shareholders in EmeraMed, Ltd (Dublin, Ireland), a company developing a compound to treat mercury toxicity.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Institute of Chronic Illnesses, Inc by a granted received from the International Academy of Oral Medicine and Toxicology (IAOMT).
