Abstract
Bisphenol A (BPA), a carbon-based synthetic polymer compound, was newly classified as an environmental toxicant and an endocrine-disrupting chemical leading to abnormalities in cell proliferation, apoptosis, or migration that contributes to cancer development and progression. This study aims to evaluate the effect of the elevation of γ- radiation dose and BPA on the liver and ovaries of female rats. In this study, eighty female albino rats (130–150 g) were used in this work. Rats in this experiment received BPA in ethanol (50 mg/kg b. wt.) for 30 days, day after day, and in the irradiated groups, animals were administered BPA and then exposed to γ- radiation in doses (2, 4, and 6 Gy) one shot dose. Several members of the cytochrome family were examined. Exposure to γ-radiation and BPA showed an increase in cytochrome P450 and b5 fold change. Further, BPA and γ-radiation activate α and β estrogen receptors and also downregulate aromatase (CYT19) fold change. The current results also revealed that BPA and/or γ-radiation regulate the protein expression of the PI3K/Akt signaling pathway. The steroidogenic acute regulatory protein (StAR) appeared to be targeted by BPA and γ-radiation and its relative expression was elevated significantly by raising the γ-radiation dose. In conclusion, exposure to BPA, an endocrine-disrupting chemical, leads to marked toxicity. Additionally, toxicity is heightened by increasing the γ-radiation dose, either alone or in combination with BPA.

Introduction
Bisphenol A (2, 2-bis-(4-hydroxyphenyl)-propane; BPA) is a monomer widely used to manufacture polycarbonate plastic and epoxy resins.1,2 It can be found in many daily consumable items such as plastic bottles, beverage containers, medical devices, dental sealants, eyeglass lenses, PVC gloves, recycled paper, printing inks, consumer electronics, and the surface of internal water pipes.
3
BPA is a well-characterized endocrine-disrupting chemical (EDC)
4
that can affect hormone synthesis, metabolism, and function.
5
BPA is a derivative of diphenylmethane comprising two hydroxyphenyl groups that make its structure similar to estrogen,
6
it is known as a xenoestrogen as it shows estrogenic actions by binding to estrogen receptors, and thereby, disrupts endocrinological roles in living organisms, including humans.
1
Pupo et al.
7
reported that BPA activates the GPER/ERK1/2/EGFR signaling pathway in cancer cells by inducing target gene expression and coupling with the G protein receptor. Beyond the classical role played by the liver in detoxification, liver enzymes are responsible for glucuronidation (the main route of BPA metabolism) mediated by the UDP-glucuronosyltransferase (UGT) enzymes and sulfation by sulfotransferase,
8
which produces BPA glucuronide, and BPA sulfate, respectively, both conjugated metabolites with little or no estrogenic activity. Ionizing radiation (IR) is one of the harmful external environmental factors. The living cells absorb IR that makes biological and chemical alterations,
9
either directly by ionization and formation of DNA adduct or indirectly via water radiolysis and then generation of free radicals and reactive oxygen species (ROS), such as hydroxyl radical OH•, the superoxide anion (O2
Aims of the current work
This study aims to examine the harmful role of different doses of γ-radiation combined with BPA on the liver and ovary of female rats.
Material and methods
Chemicals
Bisphenol A (BPA) from Sigma Chemical Co., Cairo, Egypt was obtained in the form of rutin hydrate. All other chemical substances and reagents used in this analysis were of analytical grade. BPA was dissolved in ethanol, 3 and then complete the volume by corn oil (ethanol is 5% of the total volume).
Animals
Eighty female Wister rats (120–150 g) were used, got from the National Centre for Radiation Research and Technology (NCCRT), Cairo, Egypt. Rats were kept in an environmentally controlled clean-air room with standard temperature and maintained a 12 h light/dark cycle. They were permitted to adapt to the environmental conditions for 1 week before beginning the experiment and were fed on standard food pellets containing all nutritive elements and liberal water ad libitum. All animal procedures complied with the guidelines of the National Centre for Radiation Research and Technology, Egypt, and with guidance for the proper treatment and use of laboratory animals (NIH Publication No. 85–23, updated 1985), and managed by the animal facilities, National Center for Radiation Research and Technology, Egyptian Atomic Energy Authority (Serial No.21A/20).
Irradiation facility
Whole-body ionizing gamma-irradiation (IR) was carried out at the National Centre for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Authority, Cairo, Egypt, using Canadian Gamma Cell-40 biological irradiator (137 Caesium) produced by Canada Limited Atomic Energy, Ontario, Canada. During the time of exposure, the radiation dose was 0.61 Gy/min. The cumulative dose of radiation was 2.0, 4.0, and 6.0 Gy as a single dose.
Experimental design
Animals were randomly classified into eight groups (n = 10) as follows control(C); female rats were supplied distilled water by gastric intubation daily for 4 weeks. 2- Bisphenol A (BPA) positive control; rats received bisphenol A (50 mg/kg b.wt)for 30 days day after day, for 4 weeks (BPA) according to Pinafo et al 3 and Kobroob et al. 12 3- The two Gray irradiated group (R 2); rats were exposed to a single dose of one shot (2 Gy) γ-radiation.4- The four Gray irradiated group (R4); rats were exposed to a single dose of one shot (4 Gy) γ-radiation. 5-The six Gray irradiated group (6 Gy); Rats were irradiated with a single dose of one-shot γ-radiation (R6).6- BPA + R2 group; rats were orally administrated BPA as group 2 and then exposed to a single dose of IR as group 3. 7- BPA + R4 group; rats were orally treated with BPA as group 2 and then irradiated as group 4. 8-BPA + R6 group; Rats were orally treated with BPA as group 2 and then exposed to IR group 5. After radiation, rats fasted overnight. Intracardiac blood samples were collected, and serum was separated by centrifugation at 3000 r/min for 15 min and stored at −80°C until analysis. The ovary and liver tissues were immediately dissected, rinsed in ice-cold saline, plotted to dry, and weighed. Ovary and liver tissue’s left lobes were fixed in 10% formalin prepared in phosphate-buffered saline (PBS) histopathological examination. A weighted part of each ovary and liver was homogenized with ice-cooled PBS to prepare 20% w/v homogenate. The homogenate was centrifuged at 4000 r/min for 5 min, at 4°C. The aliquots were then kept at −80°C until use.
Biochemical investigation
Serum estrogen (ES), progesterone (PGS), Follicle-stimulating hormone (FSH), and Luteinizing hormone (LH) levels were studied according to standard methods using available commercial kits.
Quantitative real-time PCR
Western blot analysis
The protein expression of AKT (Cat. No.: ab65786, ∼60 kDa) and PI3k (Cat No: ab302958∼126 kDa) was protein assessed in the ovary and liver tissues homogenate. β-actin was used as a housekeeping arbitrary unit.
Histopathological examination
Liver and ovaries were collected from the different experimental groups and routinely processed. The paraffin-embedded blocks were sectioned at the 5-micron thickness and stained with Hematoxylin and Eosin 16 for histopathological examination by a light microscope (Olympus BX50, Japan).
Grading of histopathological alterations
Histopathological alterations of the liver and ovaries were graded as (0) indicating no changes, (+), (++), (+++), and (++++) indicating mild, moderate, less severe, and severe changes, respectively 17
Statistical analysis
The statistical package SPSS (Statistical Program for Social Science) version 20 was used to apply a one-way ANOVA test followed by a post hoc test for multiple comparisons. All data expressed SE (n = 6) and the difference between means was considered significant at p < .05.
Results
Exposure to ionizing radiation and BPA triggers toxicity in many tissues. The liver and ovaries are among the most sensitive tissues that have been affected. The present study showed a marked increase in FSH and LH hormone levels in rats administrated BPA to the respective control. Increasing the radiation dose leads to an elevation in both hormones, alone or combined with BPA (Figure 1(a) and (b)). Similarly, the estradiol level increased by increasing the radiation dose from 2 to 6 Gy compared to the control. There was a significant elevation in estradiol levels in groups that received BPA and were exposed to γ-radiation related to radiation groups (BP + R2, BP + R4, and BP + R6) (Figure 1(c)). Meanwhile, the study of progesterone levels revealed a marked rise in the BPA group linked to control, while there was no significant change between the radiation groups. A combination of BPA and γ-radiation showed a significant elevation in PGS levels compared to the corresponding radiation groups (Figure 1(d)). Gamma radiation and BPA impact on female hormones: (a) LH, (b) FSH, (c) estradiol, and (d) progesterone in examined rat groups. Each value represents the mean ± SE (n = 6). Values with different superscript letters are considered significantly different at p < .05.
For exploring the toxic effect of BPA and IR on the liver and ovarian tissues, mRNA expression of phase one monooxygenase enzymes CYT P450-2E1 was estimated (Figure 2). The data in this work revealed that CYT P450-2E1 mRNA expression was significantly elevated in the BPA group corresponding to the control group. Also, there was a potent increase in mRNA expression in rats exposed to radiation regarding the control and BPA group (R2, R4,&R6). Interestingly, by elevating the radiation dose combined with BPA, CYT P450-2E1mRNA expression increased (BP + R2, BP + R4, & BP + R6) in both tissues. Effect of different doses of gamma radiation and/or BPA in different female rat groups (a) ovary cytochrome p450-2E1 and (b)liver cytochrome p450-2E1. Each value represents the mean ± SE (n = 6). Values with different superscript letters are considered significantly different at p < .05.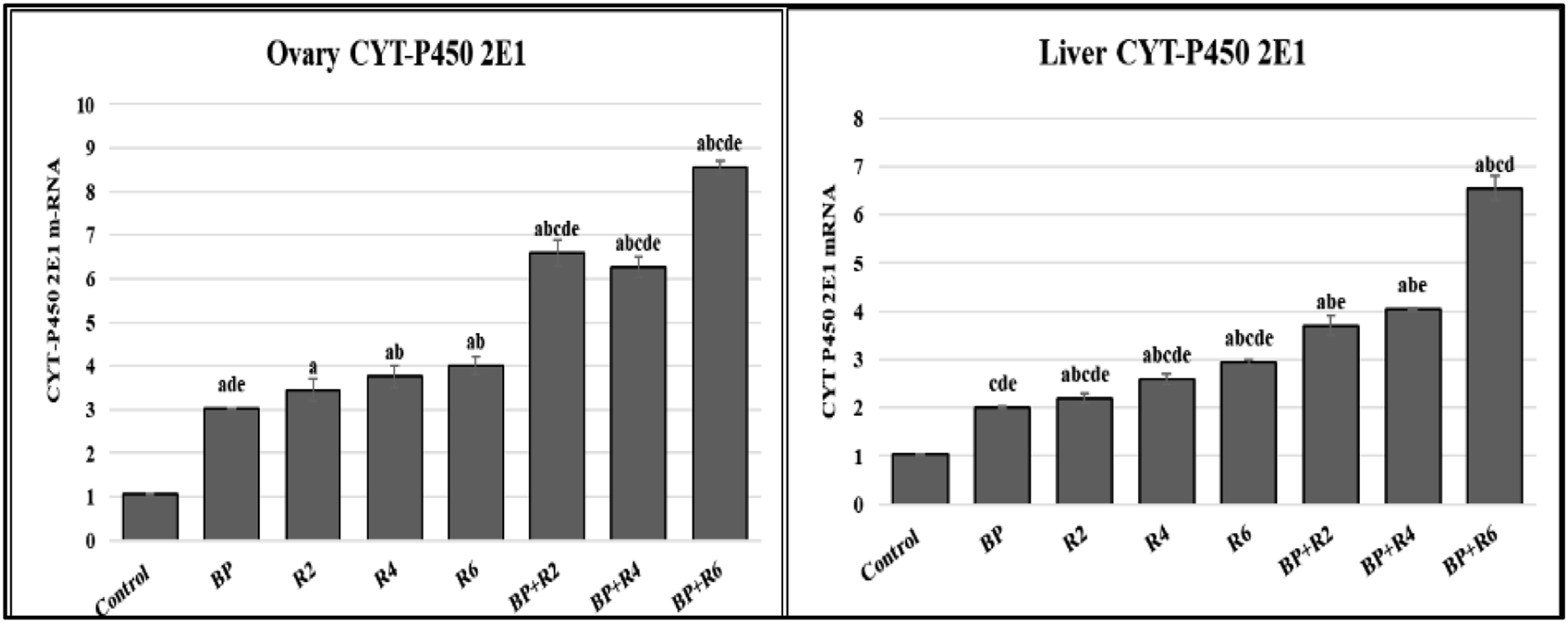
CYT19 and CYTB5 are other members of the cytochrome P450 family, their mRNA expression is illustrated in Figure 3. They exhibited different attitudes CYTB5 m-RNA expression increased by radiation, while declined by BPA administration. Otherwise, aromatase mRNA expression was significantly diminished in the BPA group compared to control. Furthermore, there was a potent decrease in the group administrated BPA and exposed to IR compared to the BPA or IR alone. Impact of different doses of γ-radiation and BPA in different tested groups (a) cytochrome B 5 and (b) aromatase. Each value represents the mean ± SE (n = 6).). Values with different superscript letters are considered significantly different at p < .05.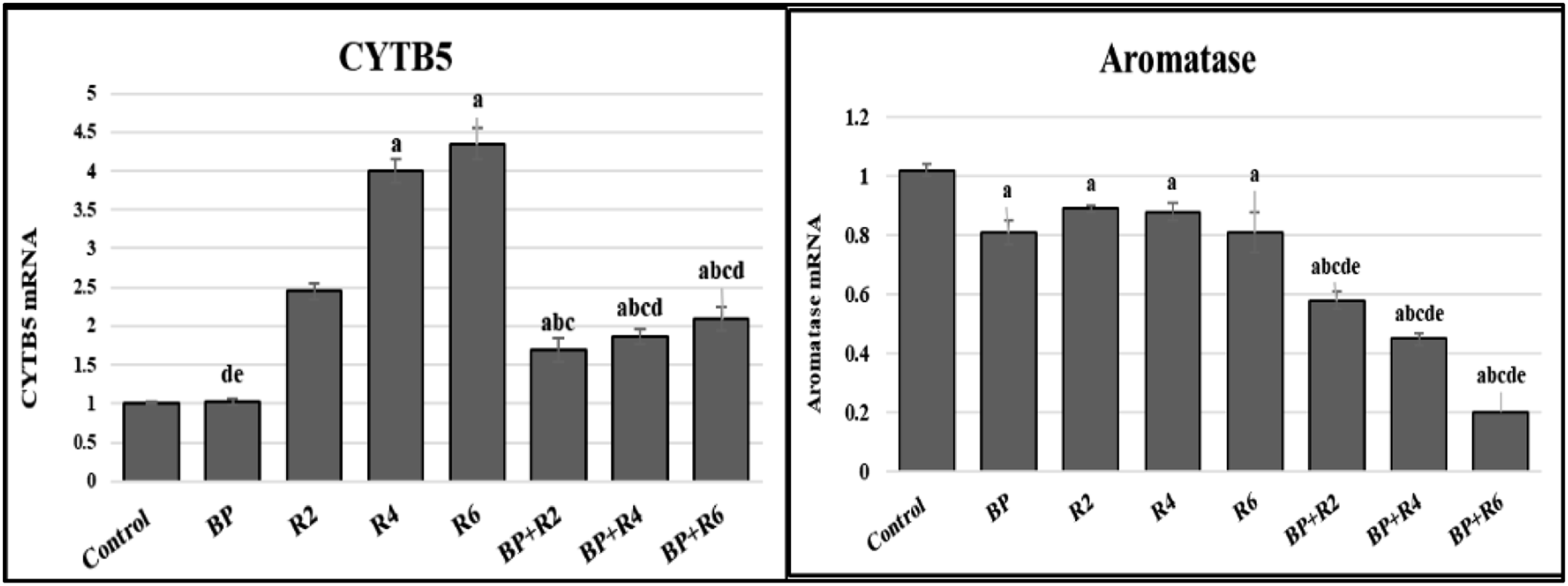
The mRNA expression of StAR (steroidogenic acute regulatory protein) was calculated and represented in Figure 4. Administration of BPA triggered a significant increase in StAR mRNA expression (BPA other) compared to the control. Additionally, rats exposed to different doses of radiation either alone (R2, R4, & R6) or in combination with BPA (BP + R2, BP + R4, & BP + R6) showed a significant elevation regarding control. Different doses of γ-radiation and BPA influences StAR in different tested rat groups. Each value represents the mean ± SE (n = 6). Values with different superscript letters are considered significantly different at p < .05.
To discover the impact of increasing the dose of γ-radiation on the liver and ovary, AKT/PI3K pathway protein expression was investigated (Figure 5). Remarkably, there was harmony elevation in this pathway in both tissues in the BPA group compared to the control. Likewise, γ-radiation exposure showed a significant increase regarding corresponding control. The combination exposure to both BPA and IR exhibited a potent augmentation comparison to their corresponding R groups. Protein expression of AkTand PI3K in the ovary and liver tissues of different examined female rats. Each value represents the mean ± SE (n = 6). Values with different superscript letters are considered significantly different at p < .05.
Histopathological findings
Liver from the control negative group showed normal hepatic parenchyma with polygonal hepatic cells, normal blood sinusoids, central veins, and portal areas, (Figure 6(a)). In contrast, the liver from the BPA group revealed moderate vacuolar degenerated hepatocytes and congested central vein (Figure 6(b)). Furthermore, livers from the R2 exposed group showed mild hepatocyte swelling with cytoplasmic granulation (Figure 6(c)), moderate vacuolar degeneration was replaced with focal areas of necrosis was reported in the R4 exposed group (Figure 6(d)), and less severe vacuolar degeneration replaced with multifocal areas of necrosis were noted in the R6 exposed group (Figure 6(e)). Moreover, Bisphenol A and Radiation exposed groups showed variant damage degree in hepatic parenchyma, there were moderate, less severe, and severe vacuolar degenerated hepatocytes with focal, multifocal, and diffuse necrotic areas in the BPA + R2 group (Figure 6(f)), BPA + R4 exposed group (Figure 6(g)), and BPA + R6 exposed group (Figure 6(h)) respectively. Photomicrographs of liver from different experimental groups stained with H&E X400 showing: (a) Control negative group with normal polygonal hepatic cells, normal blood sinusoids, central veins, and portal areas (0), (b) BP intoxicated group with moderate vacuolar degenerated hepatocytes (*) and congested central vein (arrow) (++), (c) R2 exposed group with mild hepatocytes swelling and cytoplasmic granulation (*) (+), (d) R4 exposed group with Moderate vacuolar degeneration replaced with focal areas of necrosis (arrow) (++), (e) R6 exposed group with less severe vacuolar degeneration replaced with multi focal areas of necrosis (arrow) (+++). (f) BP + R2 exposed group with Moderate vacuolar degeneration replaced with focal areas of necrosis (arrow) (++), (g) Bp + R4 exposed group with less severe vacuolar degeneration replaced with multi focal areas of necrosis (arrow) (+++), (h) BP + R6 exposed group with Severe Vacuolarly degenerated hepatocytes replaced with diffuse areas of necrosis (arrow) (++++).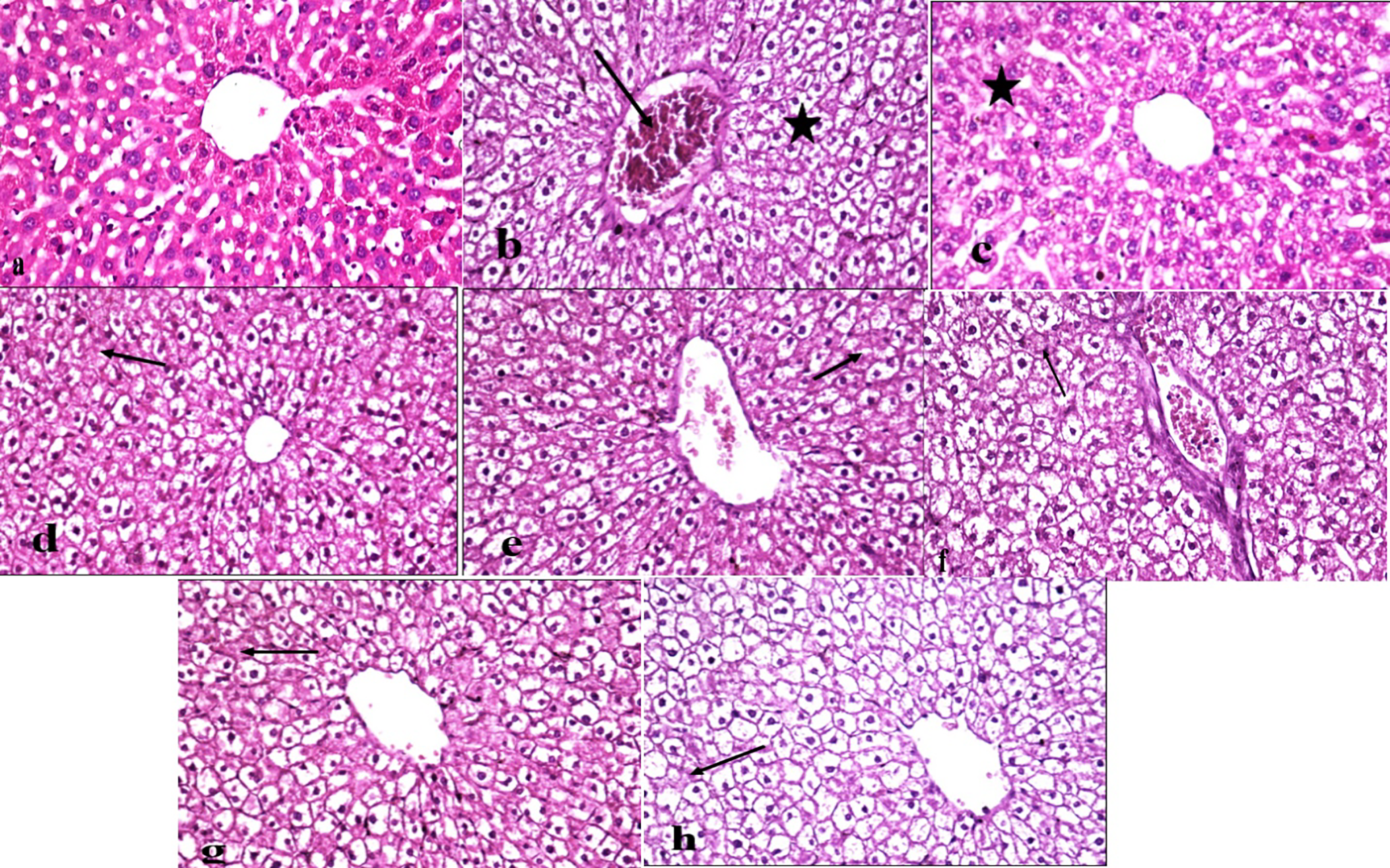
Ovaries from control negative group showed normal ovarian follicles and interstitial tissue (Figure 7(a)), while ovaries from Bisphenol A intoxicated group revealed moderate atretic follicles with fragmentation of granulose cell layer with interstitial tissue reaction and congested blood vessels (Figure 7(b)). Ovaries from R2 exposed group showed mild atretic follicles with nuclear pyknosis (Figure 7(c)), Moderate atretic follicles with fragmentation of granulose cell layer and focal areas of clusters of pale interstitial cells interspersed among follicles were reported in R4 exposed group (Figure 7(d)), and less severe atretic follicles with fragmentation of granulose cell layer and multi focal areas of clusters of pale interstitial cells interspersed among follicles were reported in R6 exposed group (Figure 7(e)). Bisphenol A and Radiation exposed groups showed variant damage degree in ovaries, there were moderate, less severe, and severe ovarian damage with focal, multifocal, and diffuse areas of clusters of pale interstitial cells interspersed among follicles in Bisphenol A + R2 exposed group (Figure 7(f)), Bisphenol A + R4 exposed group (Figure 7(g)), and Bisphenol A + R6 exposed group (Figure 7(h)) respectively. Photomicrographs of ovaries from different experimental groups stained with H&E X200 showing: (a) control negative group with normal ovarian follicles and interstitial tissue (0), (b) BP intoxicated group with moderate atretic follicles with fragmentation of granulose cell layer and interstitial tissue reaction (*), and congested blood vessels (arrow) (++), (c) R2 exposed group with mild atretic follicles and nuclear pyknosis (*) (+), (d) R4 exposed group with Moderate atretic follicles and fragmentation of granulose cell layer, and focal areas of clusters of pale interstitial cells interspersed among follicles (*) (++), (e) R6 exposed group with less severe atretic follicles and fragmentation of granulose cell layer, and multi focal areas of clusters of pale interstitial cells interspersed among follicles (*) (+++). (f) BP + R2 exposed group with moderate atretic follicles with fragmentation of granulose cell layer and congested blood vessels (arrow) (++), (g) BP + R4 exposed group with less severe atretic follicles and fragmentation of granulose cell layer, and multi focal areas of clusters of pale interstitial cells interspersed among follicles (*) (+++), (h) BP + R6 exposed group with Severe atretic follicles with fragmentation of granulose cell layer and diffuse areas of interspersed clusters of pale interstitial cells among follicles (*) (++++).
Histopathological lesion scoring
Histopathological criteria and grading of the lesions found in liver.

Discussion
Grading of histopathological lesions in the liver and ovaries in all groups.

The Cytochrome superfamily is a set of membrane-bounded hemoproteins, that induce a series of crucial biological reactions in all living organisms. 26 CYP450s have central roles in some endogenous compounds’ biosynthesis; like steroids, fatty acids, vitamins, and eicosanoids 27 . Besides, they play a vital role in xenobiotics detoxification such as drugs. 28 On the other hand, they have a pivotal function in the bioactivation of many carcinogens. 29 The CYP450 family is categorized into two groups depending on their ability to substrate recognition. The first group stimulates specific reactions on specific endogenous substrates, and the other contains enzymes evolved in xenobiotic metabolism. 30 In this study, several members of the cytochrome family have been investigated. The mRNA expression of CYP450 2E1 in the tissues of the liver and ovary showed a marked increase in the BPA group. As well-known, BPA metabolized to 2,2-bis(4-hydroxyphenyl) propanol, o-hydroxy bisphenol, or bisphenol-o-quinone products by P450 oxidative action. 31 Also, Wang et al 32 emphasized that BPA toxicity is mitigated by the action of CYP450. Consequently, the role of γ-radiation was understandable by the significant elevation of P450 2E1 in both hepatic and ovarian tissues after the radiation exposure, which is in agreement with Chung et al, 33 who documented that relatively high irradiation-induced CYP2E1in the liver with an associated increase in its mRNA. Further, Chandra et al 34 stated that the induced activity of the cytochrome P450 system might aid in metabolizing the chemically reactive species produced from free-radical reactions. Interestingly, this significant elevation was confirmed by the action of sex hormones explored, this is confirmed by Lee et al 35 who stated that sex and growth hormones regulate the drug-metabolizing enzyme expression including CYP450. There is a complex relationship between CYP450 and CYb5; CYb5 can stimulate P450, not affect or even prohibit its action, 27 so in this study, P450 was not affected by CYb5 (Figures 2 and 3). Furthermore, the γ-radiation effect was clear in Figure 3(a) that it elevates CYb5 activity either alone or with BPA. Previously, it was documented by Rendic et al 36 that the γ-radiation increases CYb5 activity in a dose-dependent manner (up to 6 Gy). Aromatase (CYP19) is another member of the CYP450 family that stimulates the aromatization of androgens into estrogen which plays a crucial role in reproductive development. 24 The present data demonstrate that BPA administration declines aromatase mRNA levels in the ovary, which may be due to the activation of PARP γ which downregulates aromatase enzyme through NF-κB inhibition. 37 This results in harmony with Huang and Leung 38 who reported that BPA downregulates the mRNA expression of CYP19 in the placental cell. Looking at our data, CYP19 expression was also declined by exposure to γ-radiation, maybe through decreasing aromatase promoter II mRNA expression and COX-1 and COX-2 expression. 39 El Bakary et al 40 also reported that low doses of radiation can suppress CYP19 mRNA expression in breast cancer-bearing mice. Mukhopadhyay et al 21 suggested that exposure to BPA CYP19A1 downregulates that affect the conversion of androgen to E2.
Studying StAR protein production is very important because it represents a rate-limiting step in steroidogenesis. The data of this study revealed that BPA elevates StAR mRNA expression.
Zhou 18 and his colleagues demonstrated that BPA caused an elevation in theca-interstitial cell testosterone levels, which was related to increased pregnenolone and E2 concentrations and increased expression of P450 c17, P450 from, P450scc, and StAR mRNA. Mukhopadhyay et al 21 emphasized that BPA administration causes an elevation in StAR protein that promotes progesterone production.
PI3K/Akt signaling is encompassed in the cell function regulation including cell proliferation, cell survival, and cell cycle progression. 41 Ionizing radiation (IR) can induce the abnormal activation of numerous signaling pathways. 42 Based on the aforementioned data, the current results revealed a remarkable increase in PI3K/Akt signaling in hepatic and ovarian tissues by BPA or γ -irradiation administration. The previous study stated that BPA activates primordial follicle progression mainly through the PI3K/Akt pathway. 43 Additionally, Xia et al 44 documented that BPA induces ROS production which stimulates the HIF-1a/VEGF/PI3K/AKT axis to promote the migration and invasion of human colon cancer cells. The estrogen elevation obtained from the present study also can induce the PI3K/Akt pathway, this is in the same context as 41 who indicated that estrogen stimulates the growth signal PI3K/Akt pathway through the mediation of its specific receptor (ERα) in breast cancer cells. Moreover, current results manifested that exposure to γ -irradiation stimulates PI3K/Akt signaling. Many previous studies documented that γ -irradiation induces both MAPK and PI3K/AKT pathways except ERK. 45 Furthermore, Zingg et al 46 postulated that the elevation of the PI3K/Akt signaling pathway by radiation occurring via ErbB receptor stimulation in tumor cells is activated through growth factor- and radiation-induced ErbB receptor stimulation. Additionally, Suman et al 47 suggested that radiation increases PI3K/Akt through the insulin growth factor/insulin receptor substrate pathway (IGF1/IRS1).
Conclusion
Although assessing every combination of stressors is unrealistic, we should not ignore the interaction of physical (γ-radiation) and chemical (bisphenol A) pollutants that share similar modes of action. For these reasons, these findings could provide support for enhancing knowledge and developing in silico models. Moreover, the current study reveals the risks resulting from exposure to radiation while chronically exposed to a pollutant like BPA. Both stressors can cause disruptions in female hormone levels and make changes in many members of the cytochrome family. Furthermore, they trigger the PI3K/Akt signaling pathway in hepatic and ovarian tissues, which can prevent apoptosis and potentially lead to the development of cancer. Further research is necessary to fully comprehend their combined impact on apoptosis prevention and cancer development.
Footnotes
Author contributions
A. A. Hassan (corresponding author): Validation. Methodology. Writing – review & editing.
S. Z. Mansour: Conceptualization. Supervision. Resources
S.S. Abdelgayed: Visualization.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Data is available as a request.

