Abstract
This study aimed to determine the effects of 13-week bisphenol A (BPA) and octylphenol (OP) exposure on the liver, kidney, and spleen of growing male rats. A total of 29 male Wistar rats aged 4–5 weeks were divided into five groups. The treatment groups were given low-dose (125 mg/kg bw/day) or high-dose (250 mg/kg bw/day) BPA or OP. These compounds were dissolved in corn oil and given via oral route for 13 weeks. Rats in the control group received corn oil for 13 weeks, as well. After 13 weeks of treatment, blood samples were analyzed for biochemical parameters. Tissue samples from the liver, kidney, and spleen were histopathologically and histomorphometrically examined. Liver tissue specimens were also stained by immunohistochemically; the number of apoptotic cells was counted, and the apoptotic indices were calculated. There were significant differences between the control and treatment groups with respect to the following parameters: body weight, relative left kidney weight, and total protein, glucose, and alkaline phosphatase levels. Edema and parenchymal degeneration in the liver and tubular degeneration in the kidney were more frequent in the treatment groups. The control and treatment groups were comparable with respect to the frequency of histopathological lesions in the spleen. Glomerular histomorphometry revealed no significant differences between the control and treatment groups. No significant differences existed between the control and treatment groups with respect to the number of apoptotic cells and apoptotic indices. Subchronic exposure to BPA and OP induced functional and structural changes in the liver, kidney, and spleen of growing male rats.
Introduction
With numerous reports describing high and increasing prevalence of testicular cancer and low and declining semen quality, 1 –3 endocrine-disrupting chemicals have been the focus of scientific research. Among those, bisphenol A (BPA) is used as the monomer to manufacture epoxy, polycarbonate, and corrosion-resistant unsaturated polyester–styrene resins required for food-packaging materials in industrial processing. 4 –6 In 2003, the global BPA capacity alone was reported to be 2,214,000 metric tonnes, with 6–10% growth in demand expected per year 7 ; about 100 tonnes is released into the atmosphere each year. 8 On the other hand, octylphenol (OP) is a degradation product of OP ethoxylates and is widely used in cleaners, detergents, emulsifiers, and pesticides. 9,10 Despite the efforts to minimize the use of these chemicals, high concentrations of these chemicals are still measured in surface waters, wastewater, and even in drinking water.
The endocrine-disrupting effects of BPA and OP in male and female reproductive systems have been thoroughly investigated, and it has been recognized that congenital anomalies of the reproductive tract or reproductive dysfunctions arising in the later periods of life may be associated with the exposure to endocrine-disrupting chemicals during the developmental stages. However, only little is known concerning the toxic effects of BPA and OP in the liver, kidney, and spleen and their potential to increase the risk of metabolic disorders. 11
The liver is a target tissue for endocrine-disrupting chemicals. Specific estrogen receptors exist in the liver, and cellular responses involving estrogen interactions have been identified.
12
Moreover, the metabolisms of BPA and OP have been described in detail. It has been shown that BPA is converted to glucuronide, a major metabolite, in the isolated rat hepatocytes.
13
BPA semiquinone has been shown
In this study, we evaluated the effects of these two endocrine-disrupting chemicals in the liver, kidney, and spleen of growing male rats by examining tissue damage in these organs and alterations in the biochemical parameters, and we aimed to evaluate the potential for the altered metabolism after treatment with BPA or OP for 13 weeks.
Methods
Chemicals
OP (CAS number 140-66-9, purity >97%) and BPA (CAS number 80-05-7, purity >99%) were purchased from Merkolab Chemistry (Ankara, Turkey) and dissolved in corn oil as stock before administration.
Animals and experimental design
A total of 29 male Wistar albino rats aged 4–5 weeks, weighting between 100 and 125 g, were obtained from the Experimental Animals Production Center, Hacettepe University (Ankara, Turkey) and were maintained under controlled conditions of temperature (20 ± 2°C) under 12 h light/dark cycle and humidity and were fed with standard rat diet and tap water
The study was designed as a 90-day subchronic toxicity study and conducted within the context of a master’s thesis. The dose levels used in this study were determined according to the previous studies investigating the endocrine-disrupting potency of OP and BPA in rats, 20,21 and the report of the European Council for Alkylphenols and Derivatives that defined the doses, by route of administration, at which metabolic capacity can be expected to be exceeded based on the previous toxicokinetic and experimental findings.
Rats were randomly divided into five groups as follows: BPA125 (received 125 mg/kg bw/day BPA;
Biochemical analysis
After obtaining blood samples from the heart of each rat, the samples were centrifuged at 3000 r/min for 10 min at 4°C, and the serum thus obtained was stored at −20°C until further analysis. The serum samples were assayed for total protein, total cholesterol, glucose, triglyceride, calcium, albumin, bilirubin, creatinine, urea, phosphor, lactate dehydrogenase (LDH), gamma-glutamyltransferase (GGT), aspartate transaminase (AST), creatine kinase (CK-MB, MB = Miyocard band), alanine transaminase (ALT), and alkaline phosphatase (ALP) using standard diagnostic kits (Audit diagnostic, Business and Technology Park-Carrigtwohill-Co-Cork, Ireland) with a clinical spectrophotometer (Shimadzu, Duisburg, Germany).
Histopathological examination
At necroscopy, the liver, kidney, and spleen were dissected and weighed in order to calculate organ/body weight ratio for each animal. After weighing, tissue samples were fixed in Bouin’s solution and then proceed to routine histopathological process. The 4-mm-thick sections were stained with hematoxylin and eosin (H&E), in accordance with the standard procedures. All slides were evaluated under a light microscope (Olympus, Hamburg, Germany).
Immunohistochemical staining
Cells undergoing programmed cell death were assessed using terminal deoxynucleotidyl transferase–mediated dUTP nick-end labeling (TUNEL) method. In brief, 4-μm-thick paraffin sections were incubated with poly-dU, followed by incubation with antibody to poly-dU and developed using an ABC immunoperoxidase reaction. TUNEL staining was carried out according to the manufacturer’s instructions (ApopTag™, in situ Apoptosis Detection Kit, Intergen, New York, NY, USA). TUNEL-positive cells were counted under a light microscope (Olympus, Hamburg, Germany). At least 1000 hepatocytes were counted for each slide, and the apoptotic index was calculated as the percentage of apoptotic cells of total counted hepatocytes.
Histomorphometry
Histomorphometric measurement of glomeruli was carried out in all groups. At least a hundred glomeruli for each group were selected; the shortest and the longest diameters for each glomerulus were measured using the Bs200prop program in an Olympus BX51 system light microscope. The glomerular volume was calculated using the following formula: 4
Statistical analysis
Statistical analysis was carried out using the Statistical Package for the Social Sciences (version 17.0, SPSS Inc., Chicago, Illinois, USA) and the Statistica 8.0 software. Data are presented as mean ± standard deviation or
Results
Comparison of the body and organ weights of the rats in the control and treatment groups revealed that the BPA125 group had lower final body weight when compared to the control group (
Body weight and selected absolute and relative organ weights of rats in the control and treatment groups

BPA: bisphenol A; OP: octylphenol.
aKruskal–Wallis test.
b
c
Significant differences were noted between the control and treatment groups in the following biochemical parameters: total protein, glucose, and ALP (
Biochemical parameters of rats in the control and treatment groups

BPA: bisphenol A; LDH: lactate dehydrogenase; GGT: gamma-glutamyltransferase; AST: aspartate transaminase; CK-MB: creatine kinase; ALT: alanine transaminase; ALP: alkaline phosphatase; OP: octylphenol.
*Kruskal–Wallis test.
a
b
c
d
e
Histopathological examination of the liver and kidney revealed significantly increased frequency of edema and parenchymal degeneration in the liver and tubular degeneration in the kidney in the treatment groups when compared with the control group (
Frequency of exposure-related pathological lesions in rats in the control and treatment groups

BPA: bisphenol A; OP: octylphenol.
aMonte Carlo statistics.
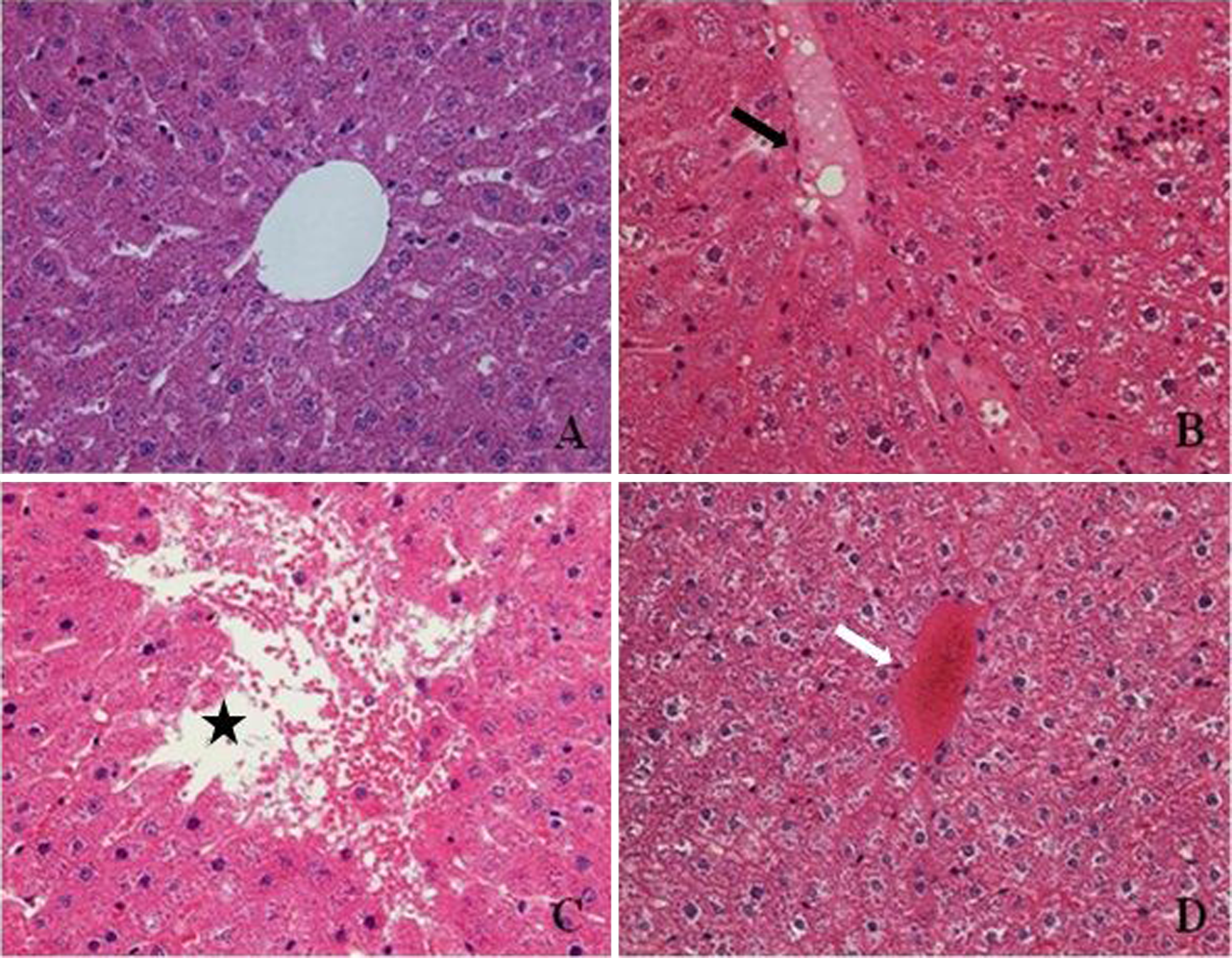
Photomicrographs showing liver tissues of control (a), 250 mg/kg BPA-treated group (b and c), and 125 mg/kg OP-treated group (d). In the liver of 250 mg/kg BPA-treated group, edema (b,
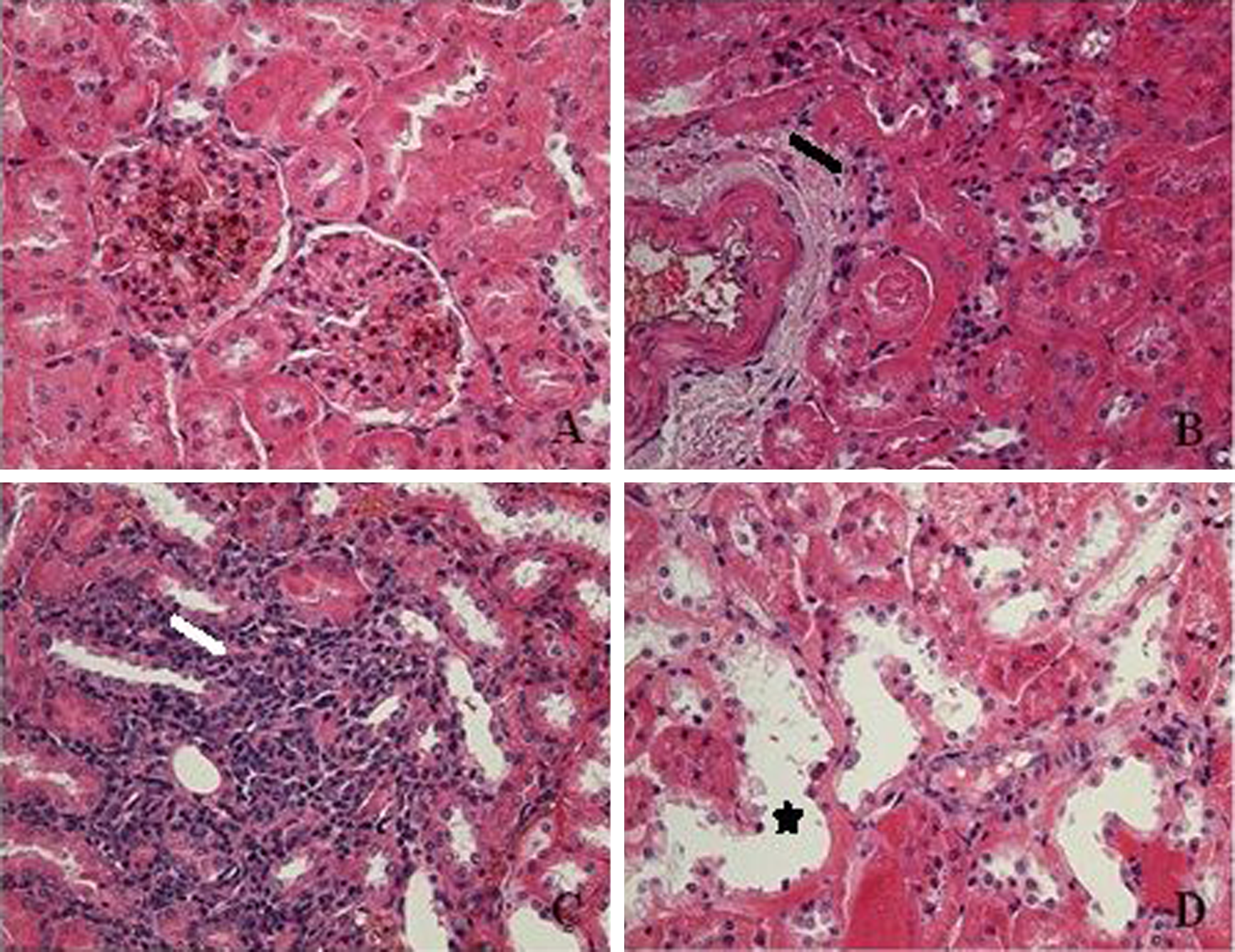
Photomicrographs showing kidney tissues of control (a), 250 mg/kg BPA-treated group (b), and 250 mg/kg OP-treated group (c and d). In the kidney of 250 mg/kg BPA-treated group, fibrosis (b,

Photomicrographs showing spleen tissues of control (a), 125 mg/kg BPA-treated group (b), 250 mg/kg BPA-treated group (c), and 125 mg/kg OP-treated group (d). In the spleen of 125 mg/kg BPA-treated group, increased number of megakaryocytes (b,
Glomerular histomorphometry revealed no significant differences between the control and treatment groups with respect to the analyzed glomerular parameters (Table 4).
Histomorphometric measurement of glomeruli in the treatment and control groups

BPA: bisphenol A; OP: octylphenol.
aKruskal–Wallis test.
Immunohistochemical staining with TUNEL method revealed no significant differences between the control and treatment groups with respect to the number of apoptotic cells counted and apoptotic index (

The number of apoptotic cells stained with TUNEL among the study groups. TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling.

Apoptotic index is defined as the percentage of apoptotic cells of total counted hepatocytes among the study groups.

Photomicrographs showing TUNEL-positive apoptotic cells (
Discussion
The current study was conducted in order to investigate the biochemical, histopathological, and immunohistochemical effects of BPA and OP in the liver, kidney, and spleen of the growing male rats subchronically exposed to these estrogenic compounds. Although the endocrine-disrupting effects of the estrogenic compounds in the male and female reproductive tracts have been thoroughly investigated, there are a limited number of studies concerning the effects of estrogenic compounds on the liver, kidney, and spleen. 20
Increased postnatal growth was observed in both male and female rats and mice treated with BPA at maternal doses between 2.4 and 500 µg/day. 24 Growing evidence suggests unexpected effects on the differentiation of adipocytes as well as postnatal growth, when estrogenic chemicals were administered during the critical periods in development. 25 Although some studies have found decreased body weight in response to developmental BPA exposures, 26,27 some have found no significant effects on body weight. 16,28,29 In the current study, we noted a decrease in body weight in all the treatment groups. However, none of the decrements reached the statistical significance, except the decrease in the BPA125 group, but this finding was not further supported by a significant difference between the control and treatment groups with respect to the percentage change in body weight. In a recent study, Barlas and Aydoğan 20 found no significant effects of 100 mg/kg OP (subcutaneous injection) on body weight when they examined the potential toxic effects of prenatal exposure to 4-tert-OP on the liver, kidney, spleen, and hematologic parameters of male and female rats in adult life. On the other hand, there was a significant increase in the body weight in male rats treated with 250 mg/kg OP during the prenatal period. These differences may be depending on the treatment route and doses. However, in that particular study, prenatal OP treatment had no significant effects on adult body weight of female rats. Nunez et al. 30 demonstrated that the decrease in the body weight in adult rats is due to the effects of BPA on metabolism. In their study, investigating whether BPA induced oxidative stress in the liver of rats, Bindhumol et al. 16 reported no significant changes in body weights and liver weights of the animals treated with BPA for 30 days. In their two-generation reproductive study on CD-1 mice, Tyl et al. 31 found no significant effects of BPA treatment on F0 male body weights throughout the 8-week prebreed period, the 2-week mating period, and the postmating period. The authors observed reduced body weights in F1 parental and retained F1 males at 3500 ppm doses. In that particular study, BPA treatment had no effect on F0 and F1 female body weights during the 8-week prebreed period. 31
Although the majority of scientific interest has focused on reproductive effects of endocrine-disrupting compounds, there is limited number of studies concerning the effects of these compounds on metabolism. 20 In the current study, either BPA or OP exposure for 13 weeks had no significant effect on relative or absolute liver weight. These findings are in accordance with the findings of Hernández-Rodríguez et al. 32 who conducted a multigenerational study evaluating the hepatic effects of estrogenic compounds, nonylphenol and OP and found no significant effect of OP on relative liver weight. On the other hand, in a recent study of Barlas and Aydoğan, 20 the authors reported increased relative liver weight in female rats treated with 250 mg/kg OP during the prenatal period. In their two-generation toxicity study, Tyl et al. 31 reported increased liver weight in F0/F1 adult male rats treated with 3500 ppm BPA. In the current study, the only significant change was noted in relative left kidney weight of rats treated with 125 mg/kg OP. Contrary to the lack of a significant change in the absolute kidney weight, in the current study, Barlas and Aydoğan 20 reported an increase in this parameter in male rats prenatally exposed to 250 mg/kg OP. Similarly, Chapin et al. 33 reported an increase in kidney weight of male Sprague-Dawley rats treated with 650 and 2000 ppm nonylphenol.
The current study clearly demonstrates that estrogenic compounds OP and BPA have adverse effects on the liver and kidney, as indicated by increased frequency of lesions in OP- and BPA-treated groups. In the multigenerational study of Hernández-Rodríguez et al., 32 in which the glycogen content in hepatocytes was determined using the anthrone reagent method, it was shown that estrogenic compound increased glycogen accumulation in rat liver. Although the precise mechanism has not yet been well understood, such an increase might be attributed to an indirect action mediated by pancreatic hormones. 32 This notion has been supported by recent studies, reporting the effects of estrogenic compounds on pancreatic β-cells. 34 Alonso-Magdalena et al. 35 found that treatment with BPA at a dose of 10 µg/kg stimulated pancreatic β-cells and increased insulin secretion. Moreover, the authors reported that insulin resistance and postprandial hyperinsulinemia developed in mice following 100 µg/kg BPA administration via oral feeding or injection for 4 days. In that particular study, analysis of pancreatic islets revealed increased insulin secretion in response to increased glucose concentrations. These findings have shown the causal link between BPA exposure and insulin resistance 35 and alteration of insulin biosynthesis and secretion in pancreatic β-cells of adult male mice. 36 Göktekin and Barlas 21 found that prenatal exposure to OP led to minimal congestion and cellular degeneration in Langerhans islet cells and associated these findings with increased β-cell activity and increased insulin concentrations. In the current study, we found that glucose levels were significantly decreased in the OP250 group when compared with the control group. However, the control and OP125 groups were comparable with respect to glucose levels. Moreover, our findings were also in contrast with those of Alonso-Magdalena et al. 35 who reported increased glucose concentrations in BPA-treated mice. On the other hand, in their study, Korkmaz et al. 17 did not find a significant effect of BPA, OP, and nonylphenol that were administered at a dose of 25 mg/kg/day three times a week for 50 days on serum glucose levels. Despite the lack of a significant effect of BPA and OP treatment on AST, ALT, and LDH in the current study, Korkmaz et al. 17 reported increased serum AST, ALT, and LDH levels in BPA- and OP-treated pubertal rats. These results demonstrated that effects of endocrine-disrupting chemicals may be depending on doses, administration route, and duration of treatment or age and sex of the treated animals.
Despite evidence suggesting increased fatty acid biosynthesis and accumulation of cholesteryl esters and triglycerides in mouse liver with BPA exposure, 37 in the current study, we found no significant changes in serum triglyceride levels.
In the current study, histopathological examination of the liver, kidney, and spleen of OP- and BPA-treated growing male rats revealed treatment-induced histopathological changes in these tissues. These findings are in accordance with the study of Barlas and Aydoğan 20 who demonstrated increased frequency of mononuclear cell infiltration and tubular degeneration in the kidney. Moreover, in their study, Barlas and Aydoğan 20 reported increased frequency of fibrosis in the spleen with OP treatment. On the other hand, in the present study, the control and treatment groups were comparable with respect to the frequency of treatment-induced histopathological changes in the spleen. Although we observed treatment-induced damage in the spleen, as also reflected by a slight anemic trend in the BPA125 group (data not shown), we found no significant differences between the groups in this respect. Decreased red blood count (RBC) and hematocrit values might indicate a disruption in erythropoiesis or an increase in the destruction of RBC. 20,38
Although the precise mechanism underlying the estrogenic compound-induced histopathological changes in tissues has not yet been fully elucidated, numerous studies have suggested estrogenic compound-induced cell death 39 –43 and increased production of reactive oxygen species. 16,44 In a study conducted by Kim et al. 42 on human embryonic stem cells, the authors suggested that nonylphenol and OP were able to trigger apoptosis in hES cells via a pathway dependent on caspase activation and Fas-FasL interaction. Recently, Li et al. 43 found that high-dose BPA induces apoptosis of Leydig and germ cells in the mouse testis through the Fas-signaling pathway. The findings of that particular study provide evidence that the BPA upregulates the expression of Fas, FasL, and active caspase-3 in the mouse testis. In a recent study, Asahi et al. 45 investigated the biochemical and ultrastructural changes in BPA-treated mouse hepatocytes to elucidate the molecular basis of BPA-induced liver damage and showed remarkable elongation of the endoplasmic reticulum and increased production of reactive oxygen species in BPA-treated hepatocytes. The authors concluded that the endoplasmic reticulum stress was involved in BPA-induced apoptosis, and BPA-induced apoptosis was independent of estrogenic actions. 45 In the current study, we evaluated apoptotic activity in the liver and found no significant differences between the groups. This might have resulted from the limited number of subjects. Small sample is the major limitation to this study. Therefore, to make a definitive conclusion on apoptotic activity in the liver and to elucidate the process and role of the Fas system as well as other molecular pathways in BPA- and OP-induced apoptosis, further studies are warranted.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research was partially supported by the Scientific Research Unit of Hacettepe University (Project No. 09 D05 601 002).
