Abstract
Incidence and mortality rates of gastrointestinal (GI) and oral cancers are among the highest in the world, compared to other cancers. GI cancers include esophageal, gastric, colon, rectal, liver, and pancreatic cancers, with colorectal cancer being the most common. Oral cancer, which is included in the head and neck cancers category, is one of the most important causes of death in India. Cadmium (Cd) is a toxic element affecting humans and the environment, which has both natural and anthropogenic sources. Generally, water, soil, air, and food supplies are reported as some sources of Cd. It accumulates in organs, particularly in the kidneys and liver. Exposure to cadmium is associated with different types of health risks such as kidney dysfunction, cardiovascular disease, reproductive dysfunction, diabetes, cerebral infarction, and neurotoxic effects (Parkinson’s disease (PD) and Alzheimer’s disease (AD)). Exposure to Cd is also associated with various cancers, including lung, kidney, liver, stomach, hematopoietic system, gynecologic and breast cancer. In the present study, we have provided and summarized the association of Cd exposure with oral and GI cancers.

Introduction
Globally, gastrointestinal cancers (GI) are among the most common cancers and have high mortality rates.1–3 GI cancers include esophageal, gastric, colon, rectal, liver, and pancreatic cancer, while colorectal cancer is the most common. 4 GI cancers are more common among men than women, and their ratio is reported as 2:1. 3 Various risk factors contribute to their development, such as alcohol consumption, tobacco smoking, diet, obesity, sedentary lifestyle, age, gender, race, and personal or family medical history.3–6 Associated with both natural and anthropogenic sources, Cadmium (Cd) is a toxic element affecting humans and the environment. 7 Generally, water, soil, air, and food supplies are reported as some sources of Cd. 8 People who do not smoke are most likely to be exposed through oral ingestion. An individual is likely to consume between 30 and 50 (Mg/day) of cadmium, but only a small portion (1–10%) is absorbed by the body. Nonetheless, there are areas of the world where cadmium levels in the soil are very high. People living in these areas may be exposed to significant levels of cadmium through their diets due to crop uptake of the metal. 9 In Japan, there are areas where soil is contaminated with cadmium, for example, in the Jinzu and Kakehashi river basins. Itai-Itai disease, a serious kidney and bone disorder that mainly affects women, is caused by chronic consumption of cadmium-contaminated rice.10,11 Food from plants can have more Cd (a type of toxic pollution) than meat, eggs, milk, and dairy products, depending on how polluted the soil is. Some plants, like rice, wheat, green leafy vegetables, potatoes, carrots, and celery, can have more of this metal in them than other plants. People who only eat vegetables and those who eat shellfish may take in more cadmium than people who eat everything. 12 Additionally, smoking is another primary source of Cd exposure. Nearly 2–10% of the cadmium in cigarette smoke is transferred to the lungs. The cadmium burden in smokers is typically double that of non-smokers.9,13 Tobacco leaves gather a high accumulation of Cd metal. 14 The Cadmium- Nickel battery (with high output and rechargeable) is another critical application of Cd.15,16 The toxicity of cadmium has led to increased efforts to regulate its exposure at both the national and international levels. Due to the extensive knowledge of cadmium’s toxic and health effects, occupational, health, and environmental standards can be established based on a large database. Exposure standards have been established by many health agencies to protect the general public from excessive exposure to cadmium. According to the World Health Organization (WHO), cadmium intake should not exceed 7 micrograms per kilogram of body weight per week. 17 Cadmium in bottled water is limited to 0.005 mg/L by the USA Food and Drug Administration (FDA). Chronic durational oral minimal risk level (MRL) for cadmium based on kidney effects of 0.1 g/kg/day determined by the Agency for Toxic Substances and Disease Registry (ATSDR). In accordance with this standard, chronic oral consumption of cadmium is not associated with adverse health effects. The recommended dose is 1 mg/kg/day for food by the Environmental Protection Agency (EPA). Human exposure to water is estimated at 5 × 10−4 mg/kg/day. An estimate of a daily dose that is likely to be free from appreciable deleterious effects during the lifetime of an individual (including sensitive subgroups) is an estimate of a reference dose (Rfd). 18
Since the chronic Cd poisoning known as itai-itai occurred in the nineteenth century in Japan, scientists and other people have started paying attention to this harmful element. 8 Cd has a long biological half-life (15–20 years). 19 It accumulates in organs, particularly in the kidneys and liver. Concentrating on this transition metal causes different health risks and disorders such as kidney dysfunction, cardiovascular disease, reproductive dysfunction, diabetes, cerebral infarction, and neurotoxic effects (Parkinson’s disease and Alzheimer’s disease).20,21 Exposure to Cd is also associated with various cancers, including lung, kidney, liver, stomach, hematopoietic system, gynecologic and breast cancer.7,22 Some effective antioxidants are suggested to decrease Cd’s poisonous effects, such as curry leaf, garlic, onion, green tea, ginger, and soybean, including vitamins that can have a role in this action. 23
In the present study, we have provided and summarized the background of Cd along with the association of Cd exposure with oral and GI cancers. In the following sections, we have discussed the brief summary of each cancer, and the underlying mechanism by which Cd induces cancer. Finally, we have explained the impact of Cd on tumor angiogenesis, which can help readers understand this metal’s carcinogenicity.
Possible carcinogenic mechanism of cadmium
The carcinogenic mechanism of Cd is multifactorial. 24 Cd is a factor that indirectly causes genotoxicity. The mechanism of toxicity by Cd is not well understood, but it has been reported that Cd causes cell damage through the induction of oxidative stress.25,26 Cd can induce the production of hydroxyl radicals, superoxide anions, nitric oxide, hydrogen peroxide, and reactive oxygen species. Cd-induced biochemical changes (including aberrant gene expression, E-cadherin dysfunction, disruption of DNA repair, and cell death) may play roles in all stages of carcinogenicity.26,27 Cd effects may be through (1) reactive oxygen species (ROS)/free radicals or oxidative stress, (2) changes in apoptotic pathways, (3) epigenetic changes, or (4) a combination of effects in association with multiple pathways. Cadmium has been identified as a cation that exhibits redox inactivity, with its chief mechanism of toxicity being attributed to the act of binding with zinc fingers or sulfhydryl groups. Oxidative stress is described as a situation in which an imbalance between the systemic action of reactive oxygen species and the ability of biological systems to detoxify the reactive intermediates or repair the resulting damage in cells is happening. 28 Numerous reports and reviews have demonstrated the toxicity caused by ROS mediated by exposure to Cd. Research studies investigating the impact of Cd-induced oxidative stress have employed various cell lines or in vitro models. 28 Cd is known to be able to act as a catalyst in the formation of reactive oxygen species. 29 By increasing lipid peroxidation, Cd induces the production of reactive oxygen species and leads to the inhibition of antioxidants. 29 Previous studies have shown that the production of reactive oxygen species reduces cell antioxidants and decreases cell defense against oxidative stress. 30 The impact of Cd promotes malignancy and plays a crucial role in Cd-mediated carcinogenicity, which is Cd-induced reactive oxygen species (ROS) formation, by activating multiple signaling players such as the Mitogen-activated protein kinases (MAPKs).31,32 Cd has been reported to mimic the actions of estrogen in biological systems. Cd has been positively associated with hormone-dependent cancers such as endometrial and breast cancers.33,34 Normal apoptosis is needed to keep the integrity of cells. Throughout apoptosis, damaged cells are repaired or eliminated. Some proteins are tumor suppressors attempting to keep the cycle moderately. One of the harmful mechanisms of Cd is by suppression of caspase activity which leads to apoptotic inhibition. It will increase the proliferation of deficient cells. Also, molecular changes in apoptotic proteins such as p53 have been reported to result in a similar process of cell damage which appears through unfettered cellular growth.28,35,36
Oral cancer
Oral cancer refers to cancers of the lips, buccal mucosa, teeth, gums, tongue, hard palate, and retromolar trigone. 37 The number of new oral cancer cases in the world in 2020 was 377,713 and the number of deaths was 177,757. This cancer is highly prevalent in South Central Asia, Eastern and Western Europe, Australia, and New Zealand. It is also the leading cause of cancer death among men in India. 38 Some signs and symptoms of oral cancer include ulceration, mouth pain, dysphagia, sore throat, white or gray spots, numbness, and tooth pain. 39 Oral cancer usually is diagnosed by a complete head and neck examination, panendoscopy, and biopsy. Treatment decisions for oral cancer are based on the cancer stage and expected side effects. The primary treatment is surgery with or without lymph node dissection. Radiation therapy, chemotherapy, and immunotherapy are other options for patients with oral cancer. 40 Alcohol and tobacco use, human papillomavirus, hereditary syndromes, and the toxic metal Cd have been linked to the occurrence of this cancer.40,41
In 2010, Kazi et al.
42
measured the levels of Cd and zinc in blood and scalp hair of male oral cancer patients and referents. In comparison to healthy referents, male oral cancer patients had lower zinc levels and higher Cd levels in their samples. A higher level of Cd was found in the biological samples of referent subjects who smoked or chewed tobacco, compared to those of referent subjects who did not do either of these habits, while Zn levels were also low in both samples, but were not significantly different. Also, the risk of developing oral cancer increases when people have poor oral hygiene, ill-fitting dentures, and use tobacco and alcohol. This study supports the hypothesis that deficiency of essential elements such as Zn causes mortality from oral cancer due to exposure to Cd oxide fumes associated with tobacco smoking or chewing. In addition, Arain et al. (2015)
43
compared Cd-exposed patients with oral cancer with healthy referents in different cities of Pakistan. Their study suggested that Cd exposure from smokeless tobacco products may have synergistic effects with other oral cancer risk factors. According to Zhang et al. (2019),
44
male patients with oral squamous cell carcinoma who chewed betel-quid and smoked had significantly higher levels of Cd in their dental calculus than healthy individuals without those habits. There appears to be a positive association between Cd and oral squamous cell carcinoma risk. In a study, cadmium levels in saliva were higher in smokers than in non-smokers, but some patients with oral cancer do not show signs of heavy metal poisoning in their oral mucosa, while others are smokeless or non-drinkers.
45
The process of chewing betel quid, as well as when no betel quid is chewed, can cause the oral mucosa to be constantly stimulated by toxic heavy metals, such as cadmium. Because dental calculus is porous and cellular in structure, heavy metals deposited in dental calculus, such as Cd, can leach from the calcified calculus. Due to the constant release of toxic trace heavy metals from dental calculus over long periods of time and their stimulation of the gums, inner mouth lining, and tongue border, these substances may play a role in the pathogenesis of oral cancer.
44
In light of this, there might be some differences in the cadmium content of dental calculus between patients with and without oral cancer. Cd may cause toxicity and cancer by replacing zinc in zinc finger DNA binding domains.
46
Some elements, such as zinc, compete for transport mechanisms, according to in vitro studies.
47
Cd has also been shown to displace zinc from several DNA repair enzymes (Figure 1). It may be a possible mechanism of Cd co-carcinogenicity.
48
The underlying mechanisms by which Cd exerts its effect on the initiation of oral cancer and maybe other GI cancers. Cd may cause toxicity and cancer by replacing zinc in zinc finger DNA binding domains. Elements like zinc compete for transport mechanisms and it has been shown that Cd could displace zinc from several DNA repair enzymes. Moreover, Cd could lead to an inflammatory state by stimulating the production of reactive oxygen species. It could inhibit glutathione and protein-bound sulfhydryl groups, producing reactive oxygen species and disrupting the pathway of oxidative metabolisms. Further, Cd induces DNA strand breaks and sister chromatid exchanges. It also enhances the mutagenicity of ultraviolet light in the cell.
Studies have reported that Cd causes an inflammatory response by stimulating the production of reactive oxygen species.46,48–51 A transcription factor called nuclear factor-B (NF-B) plays an important role in inflammation, as it regulates the expression of pro-inflammatory genes including cytokines, enzymes, adhesion molecules, and receptors, all of which contribute to leukocyte recruitment and survival. Inflammatory responses are activated by copper by upregulating the NF-B protein and reducing the expression of anti-inflammatory proteins. It is intriguing that activated NF-κB signaling is connected with the unfolded protein response (UPR) in Cd-induced apoptosis. 51 Also, It has been shown that Cd inhibits glutathione and protein-bound sulfhydryl groups, producing reactive oxygen species and disrupting the pathway of oxidative metabolisms (Figure 1).46,49 Studies have reported that Cd induces DNA strand breaks and sister chromatid exchanges. In addition, ultraviolet light is also made more mutagenic by it.52–54 Biologically relevant concentrations of cadmium interfere with DNA repair and do not induce oxidative DNA damage base modifications. 54 Metals like Cd, cobalt, arsenic, and nickel have been reported to directly disrupt the process of DNA repair. Importantly, they can interfere with the base excision repair (BER) or nucleotide excision repair (NER) pathways. Thus, they are associated with more negative consequences following DNA destruction. The zinc-finger motifs, which have a significant role in DNA repair mechanisms, are proposed to be the targeted component during the process of DNA damage induced by the aforementioned metals. 55
Esophageal cancer
The seventh most common cancer in the world and the sixth most common cause of cancer-related death is esophageal cancer. 38 According to the American Cancer Society, by 2022, there will be approximately 20,640 new esophageal cancer cases in the United States and 16,410 deaths. This cancer accounts for about 1% of all cancers diagnosed in the United States, but it is more common in Iran, southern Africa, northern China, and India. 56 In the early stages, esophageal cancer is often asymptomatic. However, progressive dysphagia, unintentional weight loss, and odynophagia are some symptoms in the advanced stages. Of these presentations, the most common symptom in patients is dysphagia. 57 Patients with these clinical conditions should undergo upper endoscopy as the first diagnostic assessment. 58 In endoscopy, chromoscopy and narrow-band imaging are commonly used to help the identification of suspicious lesions. A biopsy of a suspicious lesion should be done, but if this is not possible, brush cytopathology can also be used. 59 Esophageal cancer treatment options vary based on the stage of diagnosis. Curative surgery, chemotherapy, and chemoradiotherapy have all been shown to prolong survival and improve the patient's quality of life. 60 Some studies suggest that low fruit and vegetable intake, smoking, drinking alcohol, and hot food and beverages may be significant risk factors for the high incidence of esophageal cancer. 61 Some metals, such as Cd, widespread in environmental pollution, may also cause esophageal cancer. 62
In 2016, Mamyrbayev et al.
63
found that a high incidence of esophageal cancer in Kazakhstan was associated with inhalation and oral intake of Cd. Also, Yang et al.
64
2020 showed that Cd could induce upregulating of CDK6 in human esophageal cells, which can result in inhibited apoptosis and, finally, esophageal cancer (Figure 2). Jiang et al.
65
2021 studied cancer trends in China and focused on long-term exposure to soil Cd pollution. They noted that soil Cd level is positively associated with increased cancer mortality, especially in esophageal cancer. Chen et al.
66
found an association between chronic Cd exposure and the progression of esophageal squamous cell carcinoma in 2022. They also demonstrated that Cd could promote tumor cells’ chemo-radioresistance. The underlying mechanisms by which Cd exerts its effect on the initiation of esophageal cancer. Cd could induce upregulating of CDK6 in human esophageal cells, which can result in inhibited apoptosis and, finally, esophageal cancer. Upregulation of CDK6 expression by Cd leads to hyperphosphorylation of PKM2, inhibits rate-limiting glycolytic enzymes, directs some carbon to the pentose phosphate pathway, promotes the production of NADPH and glutathione to neutralize reactive oxygen species, and finally inhibits apoptosis. Eventually, epithelial cell lines with an increased apoptotic resistance can be host for the accumulation of powerful mutations, which are essential factor in the occurrence and development of esophageal cancer.
Cd can also affect P53 protein. P53 is a tumor suppressor protein and arrests cells in response to genotoxic stress. 67 Urani et al. (2014) 68 showed that Cd could change the structure and function of P53. Cd can bind to thiol groups and replace zinc in the structure of P53. Impairment and decreased cell ability to respond to DNA damage result from these functions. 67
Cd also can inhibit apoptosis. Upregulation of CDK6 expression by Cd causes hyperphosphorylation of PKM2, inhibits rate-limiting glycolytic enzymes, directs some carbon to the pentose phosphate pathway, promotes the production of NADPH and glutathione to neutralize reactive oxygen species, and finally inhibits apoptosis. The apoptotic resistance of epithelial cell lines can increase the accumulation of powerful mutants, which is an essential factor in the occurrence and development of esophageal cancer. 64
Gastric cancer
Gastric cancer is one of the most frequent cancers and will account for more than 26,000 new cases and 11,000 deaths in the United States in 2022.69,70 Poor appetite, nausea and vomiting, heartburn, and abdominal pain are some symptoms that patients with gastric cancer experience. 71 Upper endoscopy is the most often done for patients with these symptoms. During endoscopy, biopsy samples can be taken from abnormal areas. Computed tomography scans, endoscopic ultrasounds, and laparoscopy are some other tests that can be done for these patients. 72 Based on the cancer stage and the tumor’s location, the treatment of gastric cancer varies. Surgery is a good option for resectable tumors and offers the best survival. Chemotherapy, radiation therapy, targeted therapy, and immunotherapy are other cancer treatment options. 73 Aging, male gender, and genetic factors are some of the factors that contribute to the development of gastric cancer. But some preventable factors such as alcohol intake, smoking, and Helicobacter pylori infection are more critical. 74 Metals such as Cd are other preventable factors. 75 Ostadrahimi et al. 76 explored the correlation between blood Cd levels and the risk of gastrointestinal cancer in 2017. They noticed that blood levels of Cd in patients with gastrointestinal cancer were higher. Lin et al. 77 also investigated the association between Cd and gastrointestinal cancer in 2018. They showed that Cd in the blood might be a risk factor for gastrointestinal cancer, and higher levels of blood Cd may promote the initiation and development of these cancers. Kim et al. 78 2019 studied the effect of Cd intake on gastric cancer in the Korean population. They demonstrated a positive correlation between Cd intake and early gastric cancer.
Several mechanisms have been reported in which Cd can alter carcinogenic processes. Cd can lead to mutation and cancer initiation through induction of DNA damage, suppression of DNA repair system, abnormal DNA methylation, suppression of apoptosis, endocrine disruption, induction of oxidative stress, formation of reactive oxygen species, and destruction of antioxidant enzymes (Figure 3).79,80 Cd also inhibits p53 and p27, tumor suppressor genes, and activates the expression of the c-myc and c-Jun genes.
81
A summary of the underlying mechanisms by which Cd exerts its effect on the initiation of GI cancers. Cd can lead to mutation and cancer initiation through induction of DNA damage, suppression of DNA repair system, abnormal DNA methylation, suppression of apoptosis, endocrine disruption, induction of oxidative stress, formation of reactive oxygen species, and destruction of antioxidant enzymes. Cd can also inhibit the expression of p53 and p27, tumor suppressor genes, and activates the expression of the c-myc and c-Jun genes.
Cd can affect E-cadherin function, which results in breaking tight cell junctions (Figure 4). This affected function can reduce mucus thickness, content, and basal acid excretion, increase lipid peroxidation and destroy the gastric mucosal barrier. This may indicate a triggering step in both cancer occurrence and tumor development.
82
Of these mechanisms, the oxidative properties of cells are the most relevant. Cd can increase the number of reactive oxygen species, leading to weakened antioxidant defense. Cd also can replace membrane and/or cytoplasmic copper or iron and release unbound heavy metal ions involved in the Fenton reaction.
83
The underlying mechanism of Cd-induced gastric cancer. Cd can interfere with E-cadherin function, which leads to the destruction of the tight cell junctions. This affected function can decrease mucus thickness, content, and basal acid excretion, increase lipid peroxidation and destroy the gastric mucosal barrier. This may indicate a triggering step in both cancer occurrence and tumor development.
Colorectal cancer
Colorectal cancer (CRC) is one of the deadliest neoplasms worldwide; in 2020, there were more than 1.9 million new cases. 84 The colorectal cancer, known as adenocarcinoma, usually emerges from the glandular epithelial cells of the large intestine. Most of the time, CRC happens when specific epithelium cells acquire a series of genetic or epigenetic mutations. 85 The database on GLOBOCAN 2018 shows the grade of this cancer is the third most deadly, and besides that, in fourth grade, about the most commonly diagnosed cancer in the world.86,87 CRC exhibits a prevalence of 6.1% in terms of incidence and a mortality rate of 9.2%, ranking as the second most lethal cancer. According to estimates, the cumulative incidence of mortality due to colon and rectal cancer is projected to escalate by 71. 5% and 60%, correspondingly, by the year 2035.88,89 It is projected that in the year 2018, approximately 1,096,000 novel occurrences of colon cancer will be identified, alongside an anticipated total of 704,000 recent incidents of rectal cancer. Collectively, these findings correspond to a total of 1. 8 million fresh incidences of colorectal cancer. 90 In 10 of the 195 countries worldwide, CRC is the most prevalent cancer among men. There are several ways to diagnose, such as medical imaging, histopathology, and staging. Generally, diagnosis is performed by sampling areas of the colon suspicious of possible tumor development, and it depends on the location of the lesion, typically in the abdomen and pelvis. 91 The histopathology method works with tissues taken from a biopsy or surgery to search whether there is a tumor inside it or not. Eventually, the staging of the cancer is based on radiological and pathological findings. The estimation shows that there are different types of activity to treat CRC, such as surgery, chemotherapy, radiation therapy, and immunotherapy. The cancer stage is essential for the surgery, so it may be removed during a colonoscopy using several techniques if it is in an early stage. It can be in two methods, but it depends on factors in every person, and it is divided into laparotomy or laparoscopy. 91 In chemotherapy, it can be done using drugs like capecitabine, fluorouracil, irinotecan, oxaliplatin, and UFT. 92
There are several risk factors for this cancer which can be classified as health-related behaviors such as lifestyle, smoking, obesity, and exercise; another group of factors are social factors like education, income, and government expenditure on health. But between these factors, the increase in morbidity is influenced by lifestyle, body fatness, and dietary patterns. 84 This shows the impact of economic growth on the new way of life in developing countries that leads to low physical activity and consuming more animal fats, processed meats, refined grains or sweets, a low supply of dietary fibers, fruits, and vegetables. One of the significant impacts of increasing CRC is pollutants such as metals.
In one study in the Iranian population-based cancer registry of Golestan Province, north-eastern Iran there were 69 rice milling factories analyzed to review the amount of Cd. The method performed in this study was the inverse distance weighting (IDW) algorithm. They found that cadmium, with a risk of more than 60% for both women and men, can be a risk factor for colon cancer, but they declared no clear direction of cadmium in all models. 93 Since this situation is not discussed in their study, the reason for the lack of proper orientation of cadmium can be investigated in the sampling method and sample volume. Different studies illustrate that Cd acts as a mutagen by suppressing the DNA mismatch repair mechanism, and eventually, CRC appears. 94
Pancreatic cancer
Pancreatic cancer is currently the twelfth most common malignancy in men and the eleventh most common malignancy in women, and is reported as the seventh most frequent cause of cancer mortality. 95 The International Agency for Research on Cancer’s (IARC) GLOBOCAN estimated 458,918 new cases and 432,242 deaths from pancreatic cancer globally in 2018. The highest incidence rates of pancreatic cancer occur in Western Europe, North America, and Central and Eastern Europe. In contrast, the lowest incidence rates are reported in Africa and South-Central Asia.96,97 Generally, pancreatic cancer is not diagnosed in its early stages due to its non-specific symptoms, which can be considered some other diseases. Usually, patients present with advanced stages, most of which contain unresectable tumors at the moment of diagnosis. Patients' symptoms include epigastric or back pain, nausea, bloating, abdominal fullness or change in stool consistency, gastric outlet or bowel obstruction, weight loss, anorexia, depression, new-onset diabetes, venous thrombosis, jaundice, pruritus, and dark urine.98,99 Some diagnostic investigations which patients must follow to ensure the detection are suggested, such as histological examination, tumor biomarkers, endoscopic ultrasonography, Magnetic Resonance Imaging (MRI), Computed Tomography (CT), Positron Emission Tomography (PET), and endoscopic retrograde cholangiopancreatography. 100 Various treatment options are available depending on the stage of diagnosis and location of the tumor, reported as surgery, chemotherapy, radiotherapy, Targeted Therapy, Immunotherapy, Stem cell therapy, and Microbial Therapy.100,101 Risk factors of this cancer have been divided into two categories: modifiable and non-modifiable risk factors. Modifiable risk factors include smoking, obesity, alcohol, diet, occupational exposure and chronic infections. Non-modifiable risk factors include age, gender, family history of pancreatic cancers, genetic factors, diabetes mellitus, ethnicity, and blood group. 98 Some nutrients capable of preventing cancer have been suggested, such as omega-three fatty acids, polyphenols, folic acid, selenium, and zinc. 102
Occupational exposures have been found to have a link with pancreatic cancer. Exposure to some chemicals and metals are considered to be potential carcinogens. Cd is a heavy metal with broad toxicity, accumulating in some body organs and tissues, including the pancreas. Its exposure increases pancreatic cancer development and especially increases the risk of exocrine pancreatic cancer.98,103–105
The etiology of pancreatic cancer and associated pancreatic damage is closely linked to oxidative stress, which is characterized by the generation of ROS or free radicals. The prevalence of research pertaining to Cd-induced oxidative stress in the pancreas has been rather limited. There exists a multitude of reports evidencing the accumulation of Cd within the pancreas. Inflammation consequent to oxidative stress, which may serve as a precursor to possible oncogenesis, is an underlying etiologic factor of pancreatitis.
Epigenetics correlates to genetic alteration due to environmental or occupational effects, which can change the expression of inborn matter. Exposure to Cd over embryogenesis may cause changes and mutations in DNA methylation. It seems that Cd can effect via different molecular mechanisms such as DNMT activity suspension, DNA repair mechanism interference, or miRNA upregulation. 28 Mortoglou et al. 106 studied the role of Cd in miRNA expression. There is a linkage between specific miRs containing miR-221, miR-155, and miR-126 and the development of pancreatic ductal adenocarcinoma (PDAC). They found different results in each of the miRs. A considerable upregulation of mRNA expression was shown in miR-221 and miR-155, among the most oncogenic miRs in PDAC. Also, downregulation of miR-126 appeared. It can inhibit the ADAM metallopeptidase domain 9 (ADAM9) target gene, commonly expressed in PDAC.
Hepatocellular carcinoma
Hepatocellular carcinoma is a prevalent type of liver cancer. 107 Liver cancer is the sixth most common cancer globally, and it is more common in Southeast Asia and sub-Saharan Africa. 38 Based on the latest projections from the American Cancer Society regarding primary liver cancer and intrahepatic bile duct cancer in the United States in 2023, it is estimated that there will be approximately 41,210 newly diagnosed cases (27,980 in men and 13,230 in women). Additionally, the expected mortality rate for these cancers is approximately 29,380 individuals (19,000 men and 10,380 women). 108 Liver cancer often presents with weight loss, liver enlargement, jaundice, itching, and abdominal pain. However, these symptoms usually do not appear until the late stages. 109 Imaging tests (such as ultrasound and magnetic resonance imaging) and laboratory tests (such as alpha-fetoprotein blood tests, viral hepatitis tests, and liver function tests) will be performed on these patients. 110 Treatment options vary based on age, health condition, and cancer stage. Some therapeutic options include surgery, liver transplantation, targeted therapy, immunotherapy, chemotherapy, and radiation therapy.110,111 Male gender, race, chronic viral hepatitis, cirrhosis, heavy alcohol use, tobacco use, obesity, type 2 diabetes, aflatoxin, and exposure to Cd are known risk factors for hepatocellular carcinoma.26,110
In 2012, Satarug 112 reviewed the potential effects of long-term Cd exposure on the liver. He demonstrated that exposure to Cd, especially from the diet, is one of the least known risk factors for hepatocellular carcinoma. Zhang et al. 113 2018 investigated genes associated with Cd-induced liver carcinoma. They discovered three Cd-related genes (ITGA2, SLC7A11, and LRAT) that may play a role in liver carcinoma and may be necessary for exploring the mechanism of Cd-induced liver carcinoma. Men et al. 114 studied early-life Cd exposure in mice in 2021. They showed that early life Cd exposure leads to liver tumors which are larger in size, earlier in onset, and more significant. Also, Barsouk et al. 26 published a study on the chemical risk factors for primary liver cancer in 2021. They stated that Cd concentrates in organs such as the liver and adversely affects those organs. They also said that occupational chemicals like Cd might increase the risk of liver cancer.
It is essential to distinguish the effects of Cd under conditions of normal autophagy and impaired autophagy because autophagy plays a dual role in Cd-induced hepatotoxicity. Normal autophagy functions protect hepatocytes from Cd toxicity, whereas impaired autophagy causes more severe damage to the liver. Cd-induced impaired autophagic function occurs because of a lack of fusion of the autolysosome with the lysosome. As a result, autophagy cannot recycle damaged molecules. 115 Other mechanisms also contribute to Cd hepatocyte toxicity. Failure of DNA repair, DNA methylation, activation of protein degradation, loss of apoptosis, E-cadherin dysfunction, affecting cell cycle regulation, promote tumorigenesis by promoting genomic instability and mutation in critical genes, interfering with gene expression, induction of inflammatory signaling, cytokines up-regulation, and activation oncogenes such as c-fos, c-myc, and c-jun are some of these mechanisms.116–118
The impact of Cd on tumor angiogenesis
The process of angiogenesis is the growth of new blood vessels from preexisting vessels. By responding to proangiogenic stimuli, pericytes detach from endothelial cells (ECs) normally quiescent in basement membranes. The extracellular matrix (ECM) is formed by plasma proteins extravasating through EC junctions. By migrating, proliferating, and fusing with neighboring vessels, ECs form tubes. It is essential for the normal development of a fetus and the healing of wounds that angiogenesis occurs. Solid tumor growth and metastasis are also dependent on it. 119 Oxygen (O2) and nutritional supplements are provided by tumor angiogenesis, as well as tumor metabolites are disposed of via tumor angiogenesis. The diameter of the tumor cannot exceed 2 mm without neovascularization. Furthermore, tumor angiogenesis can result in twisted and hyperpermeable vasculature. An abnormal vasculature promotes tumor progression by aggravating regional hypoxia and acidosis. 120
Angiogenesis in tumors is primarily mediated by vascular endothelial growth factor (VEGF). Angiogenesis is promoted by VEGF even before a visible tumor is formed by malignant cells. Cancerous cells grow extremely fast, despite the already increased formation of vessels, creating hypoxia in solid tumors. When there is an oxygen insufficiency, hypoxia inducible factor-1α (HIF-1α) is activated. After translocating to the nucleus, HIF-1 increases expression of VEGF by binding to promoter regions. This procces promote EC survival and proliferation. Cancerous cells also release pro-inflammatory cytokines, which increase vascular permeability and promote EC migration. Several stimulators and inhibitors regulate angiogenesis. 121 It has been possible to improve cancer treatments by developing inhibitors of angiogenesis. Cancer patients' tumor size and invasiveness were reduced by combining VEGF antagonists with chemotherapy, for instance. However, environmental chemicals, such as toxic metals, may stimulate angiogenesis, thereby facilitating tumor growth. 122
Cd, a long-known carcinogen, influences tumor angiogenesis at all doses. On the other hand, Cd appears to have a bidirectional action regulated by Cd concentration.
123
In both the wound healing assay and the chicken chorioallantoic membrane (CAM) experiment, Cd at ultra-low doses (0.5 μM) inhibits angiogenesis.
124
There is still a lack of understanding of the mechanisms behind these discoveries. The inhibition of eNOS activity (Figure 5) may contribute to Cd’s anti-angiogenic effects.
124
ENOS is an enzyme found in ECs that catalyzes the formation of nitric oxide (NO).
125
As a result, Cd may directly block eNOS phosphorylation, decreasing eNOS activation.
124
Meanwhile, when ECs are treated with a modest dosage of Cd, the bradykinin (BK) induced perinuclear translocation of eNOS is prevented (113). Ultra-low dosage Cd inhibits eNOS signaling by inhibiting eNOS phosphorylation and perinuclear translocation.
126
Hence, reduced NO from ultra-low dosage Cd exposure lowers vascular endothelial growth factor (VEGF) expression. Furthermore, hypoxia promotes the binding of cytochrome c oxidase to NO.
127
When triggered by NO deficiency, the protein hydroxylase domain (PHD) stimulates the proteasomal degradation of hypoxia-inducible factor-1 (HIF-1).
128
As a result, lower NO levels caused by ultra-low dosage Cd exposure result in reduced VEGF synthesis and impairment of angiogenesis.
127
Underlying pathways by which various concentrations of Cd affect tumor angiogenesis. Ultra-low dosage Cd inhibits eNOS signaling by inhibiting eNOS phosphorylation and perinuclear translocation. Hence, reduced NO from ultra-low dosage Cd exposure lowers VEGF expression. Low-dose Cd increases tumor angiogenesis by displacing endogenous Fenton metals such as Fe2+ from proteins, which may indirectly up-regulate ROS. Cd promotes PKB/Akt, NF-B, and MAPKs via boosting ROS production, resulting in endothelial cell activation and tumor angiogenesis. PKB/Akt signaling-induced VEGF synthesis and EC migration may contribute to low-dose Cd-induced tumor angiogenesis. High dosage Cd inhibits angiogenesis by inducing apoptosis. It can activate all three MAPK family members in mouse brain microvascular endothelial cells and cause apoptosis. A high Cd concentration dramatically reduces VEGF and VEGFR-2 expression, impairing VEGF signaling. Created using BioRender.com.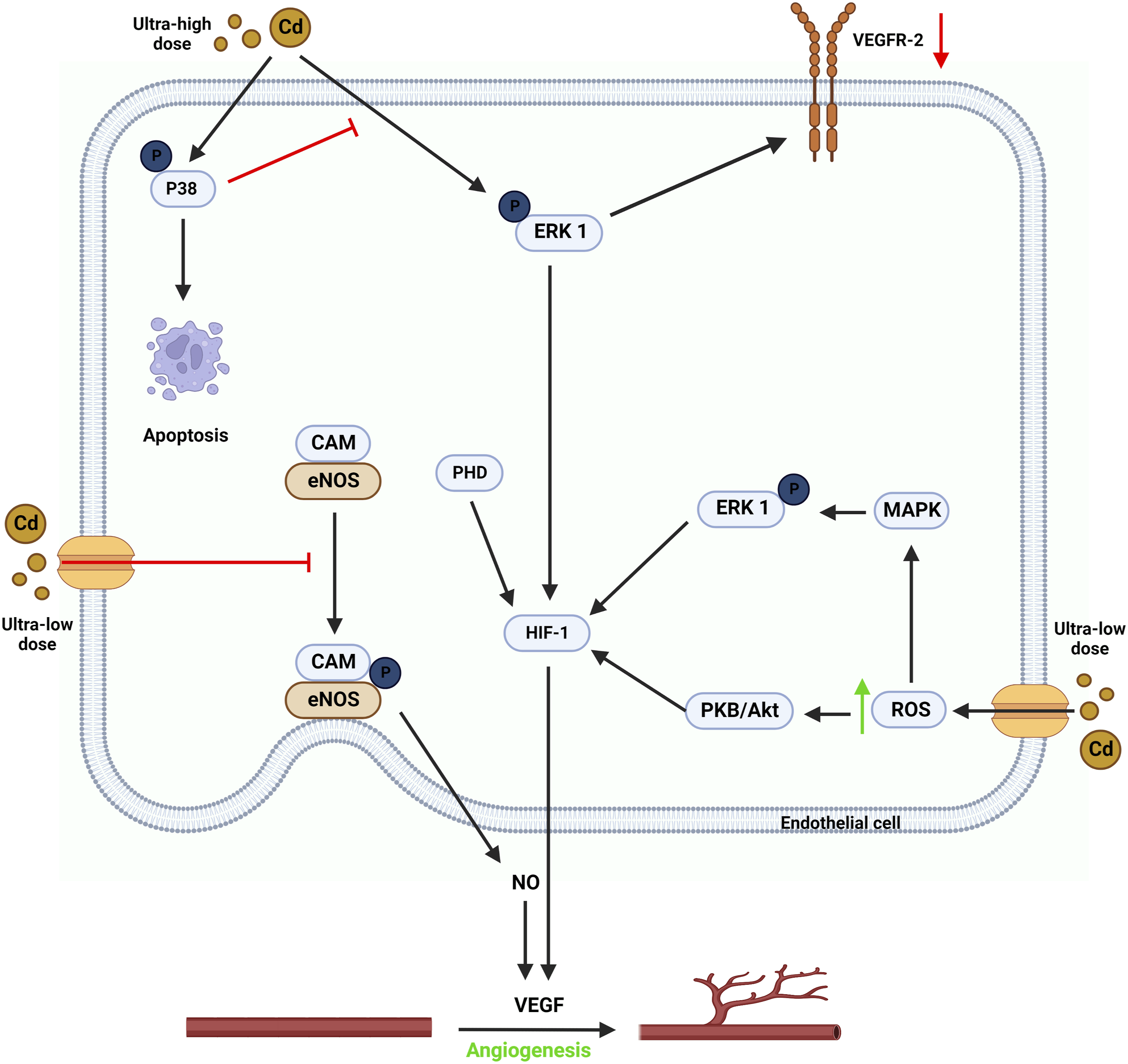
Low-dose Cd (1 μM–10 μM) increases tumor angiogenesis in various experimental settings (Figure 5). It may also affect the phenotypic of non-endothelial cells and ECs. 129 Furthermore, by displacing endogenous Fenton metals such as Fe2+ from proteins, Cd may indirectly up-regulate reactive oxygen species (ROS). 130 Cd promotes PKB/Akt, NF-B, and MAPKs via boosting ROS production, resulting in endothelial cell activation and tumor angiogenesis. 123 Protein kinase B (PKB), commonly known as Akt, is a serine/threonine-specific protein kinase that plays a role in the transcriptional regulation of protein expression. 131 PKB/Akt signaling-induced VEGF synthesis and EC migration may contribute to low-dose Cd-induced tumor angiogenesis. 122 NF-B is a transcription factor made up of various combinations of Rel-family members, such as RelA (p65) and RelB. (p50). 132 It greatly impacts gene expression, regulating cell survival, proliferation, and differentiation. 132 Low-dose Cd (4 μM) activates NF-κB by promoting the degradation of IκBα in human renal glomerular endothelial cells. 133 ERK1/2 is a member of the MAPK family and plays a traditional function in pro-survival signaling. 134 Low-dose Cd (5 μM), like PKB/Akt, promoted ERK phosphorylation in human lung epithelial cells by causing ROS production. 135 Importantly, it appears that members of the MAPK family, ERK, JNK, and p38 MAPK, induce a slew of downstream signaling events in response to low-dose Cd activation. The net result is increased VEGF signaling and tumor angiogenesis. 123
High dosage Cd (>10 μM) inhibits angiogenesis by inducing apoptosis (Figure 5). Cd (30 μM) activated all three MAPK family members in mouse brain microvascular endothelial cells (bEnd.3), causing apoptosis, according to Jung et al. 136 However, inhibiting p38 MAPK improves survival, suggesting that p38 MAPK is the only active MAPK that causes high-dose Cd-induced EC apoptosis. 136 A high Cd concentration dramatically reduces VEGF and VEGFR-2 expression, impairing VEGF signaling. 137 In addition to boosting tumor angiogenesis, VEGF signaling protects against Cd-induced apoptosis via various pathways. 138 Variations in oxidative stress levels caused by varying Cd concentrations might explain Cd's dose-dependent influence on tumor angiogenesis. Antioxidant enzyme activities protect cells from low levels of oxidative damage. 139 Sub-apoptotic oxidative stress induces adaptive mechanisms that promote cell survival and growth. 130 Excessive oxidative stress caused by high-dose Cd induces EC apoptosis. 122 The mechanisms underpinning Cd’s impact on tumor angiogenesis remain unknown. Future research employing in vivo models is required to validate existing findings.
Conclusion
Since Cd is a toxic element affecting humans and the environment, which has both natural and anthropogenic sources, nations and authorities must be vigilant about the severe toxicity and carcinogenic effect of this metal. In the present study, we have provided and summarized the association of Cd exposure with GI, hepatocellular, and pancreatic cancers. We discussed the epidemiology of each cancer, the basic mechanisms by which Cd causes cancer, and the diagnosis and treatment of associated cancers. Finally, we described the effect of Cd on tumor angiogenesis, which may help readers understand the carcinogenicity of this metal. We hope that this study can give a deeper insight about Cd-related carcinogenicity and help the authorities to deal with it.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
