Abstract
Cadmium (Cd) is a heavy metal widely used in industry, and the skin is an important target of this metal. Taxifolin (Tax), a natural source of bioflavonoids found in various conifers, exerts multiple biologic effects on skin cells. However, the mechanisms by which Tax protects keratinocytes against Cd are currently unclear. We investigated the cytoprotective effects of Tax against Cd-induced apoptosis in the human HaCaT keratinocyte. The water-soluble tetrazolium salt (WST-1) assay and Annexin V/propidium iodide double-staining assay results showed that Cd-induced cell death was lower in cells treated with Tax (0–100 μM) than in cells treated with Cd alone. Additionally, a reduction of Cd-induced DNA fragmentation by Tax was shown by terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling assay. The levels of reactive oxygen species were also lower in Cd/Tax-treated cells than in Cd-treated cells. We employed a two-dimensional electrophoresis-based proteomic analysis to identify treatment-related alterations in protein expression. Tax downregulated cathepsin B and D and upregulated hsp27, cyclophilin A, and peroxiredowin-1. Western blotting confirmed the downregulation of cathepsin B and D and the upregulation of hsp27. The cytoprotective effects of Tax against Cd-induced apoptosis were also characterized by the changes in the activity of caspase 3, -7, poly ADP-ribose polymerase, the cellular proliferation-related ERK1/2, and AKT. Furthermore, the levels of cell cycle-related proteins, such as SP1 and p21, decreased, whereas p53 level increased. We concluded that Tax reduced Cd cytotoxicity and Cd-induced apoptosis by inhibiting the apoptotic pathway.
Introduction
Cadmium (Cd), a highly toxic chemical and major industrial pollutant in developed countries, is widely distributed in the air, food, soils, sediments, and water. 1,2 Cigarette smoke, food, Cd mining industry, and nickel–Cd battery production are sources of Cd exposure for humans. 2,3 Cd has been listed as a high-priority chemical by the US Environmental Protection Agency. Additionally, in 1992, the International Agency for Research on Cancer (IARC) classified Cd as a human carcinogen. 4
Cd targets the liver, lungs, kidney, cardiovascular system, immune system, and reproductive system. 5 Exposure to Cd negatively affects a number of organs, including the kidney. 6 –8 Cd has been reported to be associated with the epidemics of skin diseases, such as contact hypersensitivity and dermatitis. 9,10 However, unlike the association of Cd with respiratory diseases, the association of Cd with skin inflammation has not been studied much.
Epidermal permeability barrier plays an important role in protecting against environmental toxic stress and preventing from dehydration. 11 From the basal layer of the epidermis, epidermal keratinocytes differentiate into keratinocytes in the epidermal keratin layer to form a protective skin barrier. Cd, an environmental toxic substance, disrupts the epidermal barrier and causes inflammatory skin diseases, such as atopic dermatitis. 12 In response to the penetration of various stimulants, epidermal keratinocytes produce a variety of pro-inflammatory cytokines and autacoids to regulate intercellular communication and affect the epidermal permeability barrier.
Taxifolin (Tax) is a compound found mostly in the Siberian larch (Larix sibirica). 13 This substance exhibits antioxidant and metal-chelating activities. 14 Tax is not mutagenic and has a lower toxicity than other flavonoids. 15 It is characterized by a number of biological activities, including antioxidant, anti-inflammatory, and antiapoptotic activities. 16 Tax also acts as an immune enhancer and it is likely to act through various immune modulatory mechanisms. 17
There have been numerous investigations that focused on the therapeutic effects of medicinal plant extracts against Cd-induced cytotoxicity. Tax is a widely distributed dietary antioxidant that exerts preventive effect against Cd-induced cytotoxicity in vivo. 18 However, little is known about the protective effect of Tax on Cd-induced cytotoxicity in vitro. HaCaT cells are sensitive targets of Cd-induced skin damage. 2 Herein, this study was designed to the effects of Cd and Tax on the intracellular events of HaCaT cells in order to better understand the protective mechanism of Tax on Cd-induced cytotoxicity.
Tax exerts antioxidant, radical-scavenging, and metal-chelating activities. Other studies have also examined the protective effect of Tax against Cd-induced toxicity in human keratinocytes. Hence, this study aimed to identify the toxicological effects of Cd on human HaCaT keratinocytes and the protective activities of Tax on Cd-treated keratinocytes.
Materials and methods
Reagents
Tax was purchased from Sigma-Aldrich (St Louis, Missouri, USA). Tax was dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich) to generate a stock solution (25 mM) and stored at −20°C. The purity of Tax is 85%. Cadmium chloride (CdCl2; Sigma-Aldrich) stock solution (25 mM) was prepared using distilled water, sterilized by filtration, and stored at 4°C.
Cell culture
HaCaT cells were purchased from the Korean Cell Line Bank (Seoul, Korea). The cells were maintained under controlled conditions (37°C, 95% humidity, and 5% carbon dioxide) and cultured in high glucose Dulbecco’s modified Eagle’s medium (Gibco, Grand Island, New York, USA) containing 1% penicillin/streptomycin (Gibco) and 10% heat-inactivated fetal bovine serum (Gibco). For the experiments, the cells were harvested at 70% confluence using 0.25% (w/v) trypsin-EDTA (Sigma-Aldrich) without exceeding 90% confluence. Before harvesting, the cells were rinsed with phosphate-buffered saline (PBS, Biomedic, Gyeonggi-do, Korea) twice. Cell pellets were collected using centrifuge (1350 r/min, 5 min).
Cell treatment with Cd and Tax
HaCaT cells were seeded in 25 cm2 flasks and 100 mm culture dishes 24 h before the treatment with Tax. Twenty-four hours after seeding, the medium was replaced with fresh medium containing Tax (50 or 100 µM) and the cells were incubated for 24 h (final DMSO concentration is 0.4%). The following day, 30-µM Cd was added to the medium. Next, the cells were harvested. A control with no treatment was systematically included in this experiment.
Cell viability assay
The water-soluble tetrazolium salt (WST-1) assay was performed according to the manufacturer’s protocol. Cell viability was determined using the WST-1 assay and EZ-Cytox Enhanced Cell Viability Assay Kit was purchased from DoGenBio (Seoul, Korea). HaCaT cells at a concentration of 5 × 104 cells/ml were seeded in a 96-well plate. The cells were treated with Tax and CdCl2 (Sigma-Aldrich), mixed with 10 µl of WST-1 solution, and incubated at 37°C for 1 h. After gentle shaking for 5 min, cell viability was determined by measuring the absorbance at 450 nm using an EnSpire Multimode Plate Reader (PerkinElmer, Waltham, Massachusetts, USA). All results are expressed as the mean percentage ± standard deviation (SD) of three independent experiments.
TUNEL assay
We performed the terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling (TUNEL) assay using a fluorometric TUNEL system (Promega, Madison, Wisconsin, USA) to detect fluorescence in apoptotic cells. We conducted experiment according to the manufacturer’s protocol. This assay is based on the detection of single-strand breaks within the DNA. HaCaT cells were seeded in six-well plates at a density of 1.0 × 105 per well and incubated for 48 h. After incubation, the cells were treated with Tax for 24 h. After then, Cd was added in media, respectively. After 24 h of treatment, the cells were fixed with 4% methanol-free formaldehyde in PBS for 20 min and incubated in permeabilization solution (0.2% Triton X-100 in PBS) for 5 min. After each well was washed twice with PBS, 100-µl equilibration buffer was added and the cells were incubated for 5 min at room temperature. The cells were treated with 50-µl TdT enzyme buffer (equilibration buffer 45 µl, nucleotide mix 5 µl, TdT enzyme 1 µl). Cell nuclei were dyed by Hoechst Stain Solution (Sigma-Aldrich). The cells were stained with 2-μg/ml Hoechst 33342 for 15 min. Finally, DNA fragmentations were detected by a fluorescence microscope (Nikon Eclipse TE 2000-U, Tokyo, Japan).
Annexin V/PI staining
Fluorescein (FITC)/Annexin V apoptosis detection kit (BD Bio-Sciences, Franklin Lakes, New Jersey, USA) was used to detect the cytoprotective effect of Tax against Cd toxicity in HaCaT cells. We performed experiment according to our previous study. 19 The cells were seeded in a six-well plate at a density of 1 × 105 cells per well. After 24 h, the cells were treated with different concentrations of Tax (0, 50, and 100 μM) and further treated with 30-μM Cd for 24 h. Next, the cells were washed with PBS, resuspended in binding buffer, and labeled with FITC/Annexin V for 20 min at room temperature in the dark. Next, the cells were analyzed by flow cytometry.
Determination of reactive oxygen species level
Intracellular reactive oxygen species (ROS) generation was assessed using the stable nonpolar dye 5-(and-6)-carboxy-2′,7′-dichlorofluorescein diacetate (DCF-DA) (Sigma-Aldrich), which readily diffuses into cells. We performed experiment according to our previous study. 20 Tax-pretreated HaCaT cells were treated with Cd for 24 h, incubated with 30-μM DCF-DA at 37°C for 30 min, and then washed twice with cold PBS. ROS production was measured by flow cytometry (Beckman Coulter Gallios, Fullerton, California, USA).
Two-dimensional gel electrophoresis
We performed experiment according to our previous study. 21 Cd- and Cd/Tax-treated HaCaT cells (6 × 106 cells per well) were harvested and lysed using lysis buffer (7 M urea, 2 M thiourea, 2% CHAPS, 20 nM Dithiothreitol (DTT), 0.5% pharmalyte, and protease inhibitor). After centrifugation in 28,000 × g, the supernatant was transferred to a fresh tube and the protein content was determined using a two-dimensional (2-D) Quant Kit (GE Healthcare, Little Chalfont, UK). Protein samples were cleaned using a 2-D cleanup kit (GE Healthcare). For the first-dimension separation, isoelectric focusing (IEF) was performed on an Immobiline DryStrip (18 cm, pH3–10; GE Healthcare) in a Multiphor II IEF system (GE Healthcare) at 24°C. After IEF, the IPG strips were incubated for 13 min in equilibration buffer (75 mM Tris–HCl, pH 8.8, containing 6 M urea, 29.3% glycerol, 2% sodium dodecyl sulfate (SDS), and 0.02% bromophenol blue) containing 1% DTT for reduction. Next, the proteins in the strips were alkylated with equilibration buffer containing 2.5% iodoacetamide for 13 min. Next, the proteins were electrophoresed using 12.5% SDS polyacrylamide gels and an Ettan DALTsix vertical electrophoresis system (GE Healthcare). Proteins in the gels were visualized by Coomassie blue staining, and the spot patterns of the control and Tax-treated cells were compared. Relevant protein spots were extracted from the two-dimensional electrophoresis (2-DE) gels and identified by matrix-assisted laser desorption/ionization–time of flight (MALDI-TOF).
Western blotting
HaCaT cells were harvested and lysed using lysis buffer containing RIPA buffer, protease inhibitor (Sigma-Aldrich), and phenylmethylsulfonyl fluoride (Sigma-Aldrich). The protein concentration of the cell lysate was measured using a Qubit™ Fluorocytometer (Invitrogen, Carlsbad, California, USA). The proteins were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) at 100 V for 2 h and electrophoretically transferred to a nitrocellulose membrane at 50 V for 2 h. The membranes were blocked with 5% bovine serum albumin (Biosesang, Korea). After blocking, the membrane was incubated with primary antibodies for cathepsin D, cathepsin B, hsp27, peroxiredoxin-1, cyclophilin A, caspase 3, -7, poly (ADP-ribose) polymerase (PARP), cleaved PARP, ERK, pERK, AKT, pAKT, p53, SP1, p21 (Cell Signaling Technology, Danvers, Massachusetts, USA), and b-actin (Santa Cruz Biotechnology, Santa Cruz, California, USA) at 4°C overnight. Next, the membranes were washed with TBS-T and incubated with HRP-labeled secondary antibodies for 1 h. Detection was performed using a ChemiDoc detection system (Bio-Rad Laboratories, Hercules, California, USA). Primary antibodies were purchased from Cell Signaling Technology and Santa Cruz Biotechnology. We recorded their catalog numbers and lot numbers as follows. Cell Signaling Technology: (Actin: #4970, 14), (Akt: #9272, 27), (pAkt: #4056, 11), (ERK: #4696, 3), (pERK: #4370, 7), (PARP: #9542, 10), (cleaved PARP: #9541, 6), (caspase 3: #9662, 12), (caspase 7: #9492, 6), (P53, #9282, 4), (P21, #2947, 9), (SP1, #9389, 4), (cathepsin D, #2284, 2), (cathepsin B, #3383, 2), (hsp27, #2402, 3), (cyclophilin A, #2175, 2), and (peroxiredoxin-1, #8499, 2). Secondary antibodies were purchased from Cell Signaling Technology (Rabbit, #7074, 27) and Santa Cruz Biotechnology (Mouse, sc-2005, D0116).
Statistical analysis
The date shown are representatives of three or more independent experiments. The Student’s t-test and one-way analysis of variance followed by Tukey’s test were used for statistical analysis, and p-value <0.05 was considered statistically significant. IC50 value was calculated using nonlinear regression.
Results
Tax reduced Cd-induced cell death in HaCaT cells
The protective effect of Tax against Cd-induced cellular death in HaCaT cells was investigated by the WST-1 assay. We first examined the effect of Cd on the viability of HaCaT cells. HaCaT cells were treated with 0, 15, 30, 45, 60, 75, 90, or 105 μM Cd for 24 h, and then cell viability was analyzed by the WST-1 assay (Figure 1(a)). The results showed that cell proliferation was inhibited by up to 50% (IC50 value) by treatment with 36.6 μM for 24 h, and various concentrations of Cd significantly decreased the viability of HaCaT cells.

Protective effects of Tax on Cd-treated cell viability. (a) Cell viability was evaluated by the WST-1 assay. HaCaT Cells were exposed to 0 to 105 μM Cd for 24 h. Each bar represents mean ± SEM. *p < 0.05: significant differences than the control group. (b) HaCaT cells were cultured with or without Cd (0–75 μM) and Tax (0–300 μM). Each bar represents mean ± SEM. *p < 0.05, ***p < 0.001: significant differences than only the Cd-treated group. (c) Effect of treatment with Cd or Cd/Tax on morphological changes in HaCaT cells was observed under a microscope (×100). Cd: Cadmium; Tax: taxifolin; WST-1: water-soluble tetrazolium salt; SEM: standard error of the mean.
We also examined the effect of Tax on Cd-induced cell death (Figure 1(b)). Cells were treated with different concentrations of Tax (0, 100, 200, and 300 μM) for 24 h prior to treatment with Cd (0–75 μM) for 24 h. Afterward, the viability of HaCaT cells was measured by the WST-1 assay. We observed that 100 μM Tax most significantly inhibited 30 μM Cd-induced cell death. Thus, 30 μM Cd and 50 and 100 μM Tax were selected for the evaluation of the protective effect of Tax against Cd-induced cytotoxicity. Furthermore, we observed that Cd-induced morphologic collapses were significantly recovered in HaCaT cells treated with 100 μM Tax (Figure 1(c)). Our results showed that Tax exhibited protective effects against Cd-induced cytotoxicity in HaCaT cells.
Tax inhibited Cd-induced apoptosis in HaCaT cells
Next, we investigated the cytoprotective effect of Tax against Cd-induced apoptosis in HaCaT cells. Annexin V/propidium iodide (PI) double staining was performed to evaluate the apoptotic cell number in Cd- and Cd/Tax-treated HaCaT cells. We observed that Cd-induced increases in the levels of Annexin V+/PI−, and Annexin V+/PI + cells were decreased by Tax (Figure 2(a) and (b)). This finding indicated that Cd-induced apoptosis can be significantly inhibited by treatment with 100 μM Tax.

Protective effects of Tax against Cd-induced apoptosis. (a) HaCaT cells were cultured with/without Cd (30 μM) and Tax (100 μM) for 24 h, harvested, stained by Annexin V and PI, and analyzed by flow cytometry. Tax (100 μM) inhibited 30 μM Cd-induced apoptosis. (b) Bar graph showed the percentages of late and early apoptotic cells (upper right and lower right). Each bar represents mean ± SEM and the results were analyzed by one-way ANOVA and Tukey’s tests. The difference in mean (p < 0.05) was considered significant. *p < 0.05: significant differences among control group, Cd-treated group, and Cd/Tax-treated group. #p < 0.05: significant differences between Cd-treated group and Cd/Tax-treated groups. Cd: Cadmium; Tax: taxifolin; PI: propidium iodide; SEM: standard error of the mean; ANOVA: analysis of variance.
Tax reduced Cd-induced DNA fragmentation in HaCaT cells
The protective effect of Tax against Cd-induced DNA fragmentation in HaCaT cells was further examined using the TUNEL assay (Figure 3). DNA fragment is a common feature of apoptosis, and damaged DNA strand is stained brighter than undamaged DNA. The enzyme TdT, which was used in the TUNEL assay, catalyzes the incorporation of fluorescent stains with the nicks of DNA, the 3′-OH ends. The percentage of green fluorescence-stained cells increased in Cd-treated HaCaT cells but decreased in Cd-treated HaCaT cells treated with 50 or 100 μM Tax. This finding suggested that Cd-induced apoptosis was inhibited by Tax treatment.

Detection of DNA fragmentation in apoptotic cells using TUNEL assay. HaCaT cells were treated with Cd or Cd/Tax for 24h, and DNA fragmentation was visualized by fluorescence microscopy (×100). Blue fluorescence indicates nuclei and green fluorescence indicates DNA fragmentation. The merged images show blue-stained nuclei with green-stained nicked DNA. Cd: Cadmium; Tax: taxifolin; TUNEL: terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling.
Tax reduced Cd-induced elevated ROS level in HaCaT cells
To evaluate the antioxidative effect of Tax against Cd-induced oxidative stress in HaCaT cells, we carried out an ROS detection assay using DCF-DA. HaCaT cells were treated with 0 or 100 μM Tax for 24 h and then exposed to 0, 30, or 50 μM Cd. Intracellular ROS levels were measured by flow cytometry (Figure 4(a)). The results showed that the percentage of cell population in the B gate was increased by 0.16%, 44.4%, and 72.06% in response to treatment with Cd at 0, 30, and 50 μM. However, the percentage of cell population in the B gate was increased by 0.39%, 29.98%, and 51.49% in Cd/Tax (100 μM)-treated HaCaT cells, indicating ROS reduction by Tax (Figure 4(b)). In addition, the mean fluorescence intensity was increased by 33.9, 158, and 228 in response to treatment with Cd at 0, 30, and 50 μM, but increased by 43.5, 132 and 179 in Cd/Tax (100 μM)-treated HaCaT cells, indicating ROS reduction by Tax (Figure 4(b)). Thus, our results indicated that Tax reduced Cd-induced increase in intracellular ROS level.

Changes in ROS level in Cd- and Cd/Tax-treated HaCaT cells. DCF-DA staining was conducted to measure ROS level. (a) The levels of intracellular ROS were measured in Cd- and Cd/Tax-treated HaCaT cells by flow cytometry. (b) The population of HaCaT cells entering the B gate was measured by cytometry. The MFI of ROS was measured by flow cytometry. *p < 0.05: significant differences from the control cells. ROS: reactive oxygen species; Cd: Cadmium; Tax: taxifolin; DCF-DA: 2′,7′-dichlorofluorescein diacetate; MFI: mean of fluorescence intensity.
Tax changed protein expression in HaCaT cells treated with Cd
To identify the potential changes in protein expression in HaCaT cells after treatment with Cd (30 μM) and Cd (30 μM) + Tax (100 μM) for 24 h, the levels of several proteins were determined using 2-DE (Figure 5). We analyzed the intensity of protein spots using the image analysis and selected six protein spots for analysis with MALDI-TOF mass spectrometer (Table 1). Protein expression of cathepsin D and B was downregulated, whereas those of hsp27, cofilin-1, cyclophilin A, and peroxiredoxin-1 was upregulated. Differentially expressed proteins, such as cathepsin D and B, have various functions associated with programmed cell death. Moreover, cyclophilin A and peroxiredoxin-1 prevent apoptosis in response to oxidative stress. In addition, hsp27 supports cell survival.

Visualization of differentially expressed proteins of Cd- and Cd/Tax-treated HaCaT cells by 2-DE. Both panels show Coomassie-stained 2-DE gel containing proteins. Differentially expressed protein spots in the Cd- and Cd/Tax-treated groups are indicated by circles. Proteins were identified by LC-MS/MS (see Table 1). We measured the relative spot intensities in the Cd/Tax-treated group using three gels. Cd: Cadmium; Tax: taxifolin; 2-DE: two-dimensional electrophoresis; LC-MS: liquid chromatography–mass spectrometry.
Identification of differentially expressed protein spots in the Cd-treated groups and Cd/Tax-treated groups.

Cd: Cadmium; Tax: taxifolin; 2-DE: two-dimensional electrophoresis; MW: molecular weight.
aSpot number on 2-DE gel.
bSwiss-Prot accession number.
Tax inhibited Cd-induced increase in apoptotic proteins in HaCaT cells
Western blotting was performed to confirm the protein expression alteration in Cd-treated HaCaT cells and Cd/Tax-treated HaCaT cells. Following cell treatment with Cd or Cd/Tax for 24 h, protein levels of cathepsin D, cathepsin B, hsp27, peroxiredoxin-1, and cyclophilin A were measured, and the results were compared to those of the 2-DE analysis (Figure 6(a)). It was shown that the levels of cathepsin B and D were downregulated and that of hsp27 was upregulated by Tax treatment. There was little difference in the expression of cyclophilin A and peroxiredoxin-1 between Cd- and Cd/Tax-treated HaCaT cells. Additionally, the inhibition of apoptosis-related protein expression was investigated in Cd- and Cd/Tax-treated HaCaT cells. The expression of cleaved PARP, an important modulator of DNA repair and apoptosis, decreased, whereas the expression of caspase 3 and -7, which are related to apoptosis, increased. Moreover, the levels of the cellular proliferation-related proteins Akt and ERK were consistent, whereas that of pAkt and pERK increased and decreased, respectively (Figure 6(b)). The expression of p53 and SP1, which are associated with cell cycle, increased, but p21 expression decreased (Figure 6(c)).
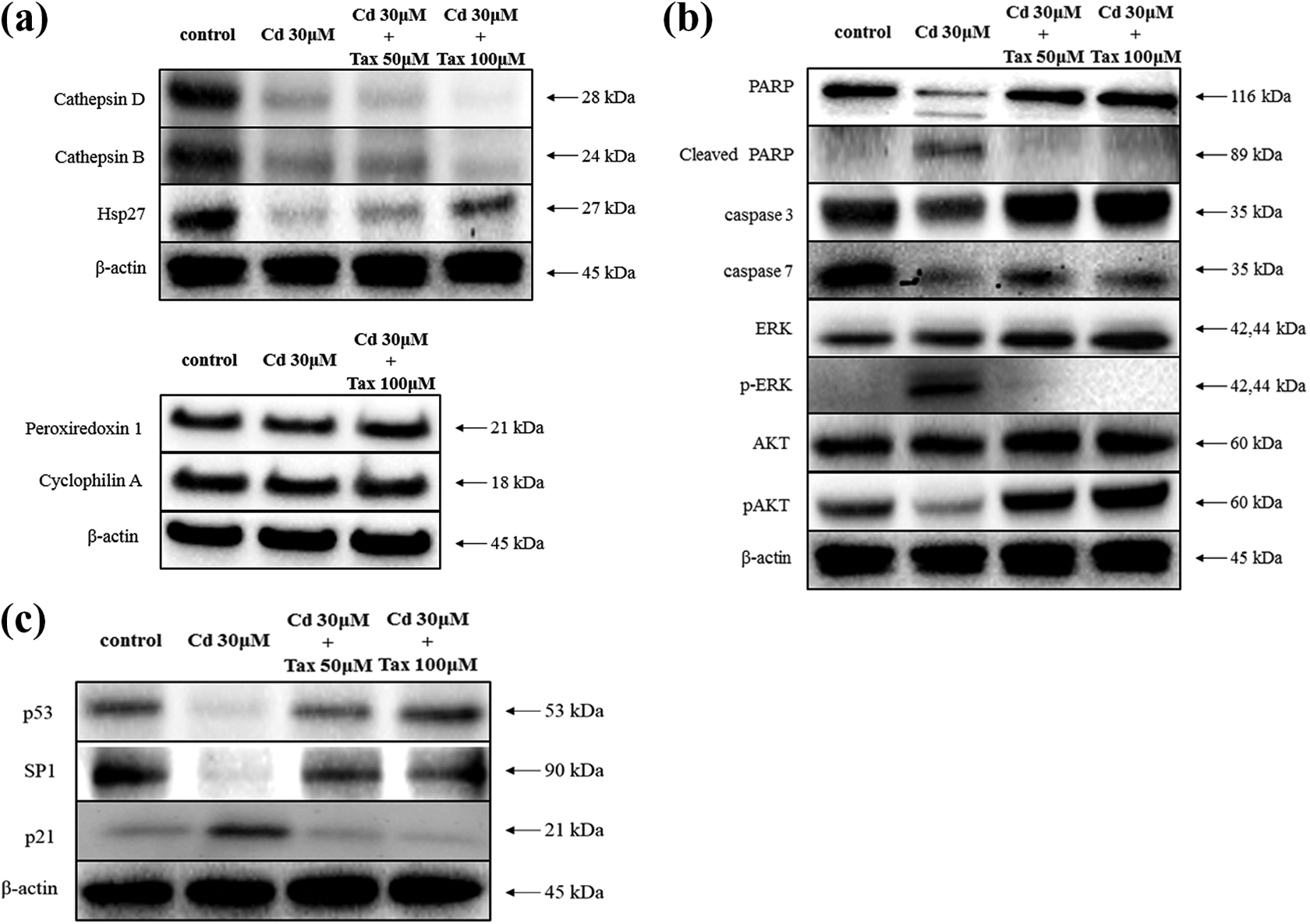
Validation of the 2-DE result and detection of proteins in Cd- and Cd/Tax-treated HaCaT cells. HaCaT cells were seeded into a culture dish and incubated for 24 h, and then treated with 0, 50, and 100 μM Tax for 24 h. The following day, HaCaT cells were treated with 30 μM Cd for 24 h. (a) The expression of cathepsin D, cathepsin B, hsp27, peroxiredoxin 1, and cyclophilin A in HaCaT cells was analyzed after 24 h of treatment. (b) The expression of PARP, cleaved PARP, caspase 3, caspase 7, ERK, p-ERK, AKT, pAKT, (c) P53, SP1 and p21 in Cd- and Cd/Tax-treated HaCaT cells was also analyzed after 24 h of treatment. The relative expression levels were normalized to β-actin. Cd: Cadmium; Tax: taxifolin; 2-DE: two-dimensional electrophoresis.
Discussion
A study on human myeloid leukemia (HL-60) cells showed that the IC50 of Cd is approximately 20 μM. 22 In addition, the IC50 of Cd in a human lymphoblastoid cell line is 350 μM. 23 However, our study on HaCaT cells showed that the IC50 for Cd was 33.54 μM/ml. Therefore, 30 μM Cd as well as 50 and 100 μM Tax were selected to be the optimal concentrations used in the WST-1 assay (Figure 1). Our results showed that 50 and 100 μM Tax inhibited cell death induced by 30 μM Cd. Primarily, the 24-h pretreatment with Tax before Cd treatment was selected for the study on the protective mechanism of Tax.
There are several studies investigating the cytoprotective and therapeutic effects of Tax. Recent research showed that Tax alleviated the effects of STZ-induced diabetes. 24 Moreover, Tax also has antioxidant effect in retinal pigment epithelium cells. 25 Furthermore, Tax alleviates DNA damage in vitro. 26 However, the cytotoxic effect of Tax in human keratinocyte HaCaT cells has not studied well. The purpose of this research was to investigate the cytoprotective effect of Tax in Cd-treated HaCaT cells. We aim to study the unknown mechanisms by performing various experiments, such as WST-1 assay, annexin/PI double-staining assay, TUNEL assay, ROS detection, 2-D gel electrophoresis, and Western blotting.
As measured by flow cytometry, Cd-induced apoptosis was prevented significantly by Tax (Figure 2). DNA strand breaks caused by Cd in HaCaT cells are well-documented phenomenon. 27 In our study, we also found DNA strand break in cells treated with Cd (Figure 3). The changing tendency of ROS expression in HaCaT cells was consistent with the result obtained by the flow cytometric analysis (Figure 4(a)). These results showed that the Tax treatment reduced Cd-induced apoptosis.
To understand the mechanism of protective effects of Tax in Cd-induced apoptosis in HaCaT cells, we performed 2-DE and Western blotting (Figures 5 and 6(a)). The levels of cathepsin D, cathepsin B, hsp27, peroxiredoxin-1, and cyclophilin A were measured to compare with the results of 2-DE analysis. However, there was little difference in the expression of cyclophilin A and peroxiredoxin-1 between Cd- and Cd/Tax-treated HaCaT cells.
Cathepsin D is a soluble lysosomal aspartic endopeptidase and has recently been implicated in apoptosis. 28 Cathepsin B, a lysosomal cysteine protease, is also associated with apoptosis. 29 Depending on the environment, cathepsin D and B can induce or inhibit apoptosis and through different mechanisms. 30 –33 The lysosomal efflux of cathepsin D and B can induce cytochrome c release from the mitochondria. The released cytochrome c induces the activation of downstream caspases, the degradation of cellular components, and programmed cell death. 34,35 In this study, we investigated cathepsin D and B as well as caspase 3 and -7 activity following treatment with Tax in Cd-treated HaCaT cells and further investigated the cleavage of PARP. Our results showed that Tax reduced the activity of cathepsin D, B, caspase 3, and -7, as well as the cleavage of PARP (Figure 6(a) and (b)).
Hsp27 is involved in the regulation of cellular responses to stimulation and is involved in a number of signaling pathways, including the MAPK and Akt pathways. 36 Furthermore, Hsp27 is also involved in the inhibition of apoptosis, protection against oxidative stress, and regulation of cytoskeleton. 37 Hsp27 modulates the activity of phosphatidylinositol 3-kinase and activates the protein kinase Akt to prevent apoptosis induced by the release of cytochrome c from the mitochondria. 38 Our results showed that the expression levels of hsp27 and pAkt were increased, whereas that of pERK was decreased (Figure 6(a) and (b)).
p21 protein is a key member of the p53 signaling pathway and it controls cell cycle progression at the G0/G1 checkpoint. 39,40 It is activated by p53 following DNA damage, which causes cell cycle arrest and apoptosis. 41 p21-induced cell cycle arrest can protect cells and inhibit apoptosis. However, many studies suggested that p21 also has proapoptotic function under certain conditions. 42,43 Our results showed that Tax increased the expression levels of Sp1 and P53 and decreased that of p21 in Cd-treated HaCaT cells.
In summary, we concluded that Tax protected HaCaT cells against Cd by suppressing the activity of cathepsin D, B, PARP, caspase 3, -7, and cell cycle-related proteins and by activating the hsp27. In addition, further protective mechanism of Tax against Cd-induced apoptosis in HaCaT cells has to be elucidated in future studies. We expected a present study to help demonstrate unknown mechanisms of Tax.
Footnotes
Authors’ contribution
SHM and CML contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by a grant from the Business for Cooperative R&D between Industry, Academy, and Research Institutes (Grant No. C0443066) funded by the Korean Small and Medium Business Administration in 2016 and the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (Grant No. NRF-2014R1A6A3A04054307).
