Abstract
Introduction
Doxorubicin (Dox) is a potent and effective antineoplastic antibiotic widely used for the treatment of various malignancies. The clinical use of Dox is limited by its dose-dependent cardiotoxic effect.1–3 Although the exact mechanism of the cardiotoxic effect is not yet clarified, it was reported that an increased production of reactive oxygen species (ROS) plays an initial role during the cardiotoxicity.4–6 It was shown that by cytochrome P450 reductase and xanthine oxidase enzymes, the quinone ring of the doxorubicin structure is reduced to the semiquinone radical7,8 and continues with reduction of oxygen molecules to superoxide and hydrogen peroxide which increases oxidative stress, lipid peroxidation, and finally cell damage.8,9 Lipid peroxidation is an indicator of oxidative stress, and is assessed by measuring the malondialdehyde (MDA) concentration which is formed as a secondary product by hydrolysis of lipid peroxides in an acid medium and reacts with thiobarbituric acids (TBA) to yield a colorful compound. 10 It was reported that doxorubicin-induced oxidative stress leads to necrosis, autophagy, apoptosis, ferroptosis, and necroptosis in cardiomyocytes.9,11–16
Necroptosis is a programmed cell death which is found in many diseases. Receptor interaction proteins 1 (RIP1), receptor interaction proteins 3 (RIP3), and mixed lineage kinase domain-like (MLKL) are key proteins in the necroptosis pathway. It was shown that overproduction of ROS enhanced the expressions of RIP1, RIP3, and MLKL.13–16 Necrostatin-1 has been shown to prevent necroptosis by inhibition of RIP1 kinase activity and reduction of RIP1-RIP3 interaction and oxidative stress-induced cell damage.16,17 It was highlighted that Nec-1 inhibits neuronal death by preventing RIP3 upregulation after ischemia reperfusion (I/R) injury. 16 The protective effect of Nec-1 was demonstrated in a mouse model of lipopolysaccharide-induced acute lung injury, indicated by reduced RIP1, RIP 3, and attenuated oxidative stress. 18 Similarly, it was reported that treatment with Nec-1 protected brains against ischemic necroptosis by reducing the ischemia-induced increase of receptor-interacting protein kinases‐1 (p-RIPK1) expression and receptor-interacting protein kinases‐3 (RIPK3), MLKL, and p-MLKL expression levels in ischemic brains. 19 In a recent study, the protective effects of Nec-1 against renal I/R injury were shown by inhibiting necroptosis, oxidative stress, and inflammation. 20
Since the protective effects of Nec-1 has been shown in different pathological process, in this study, we aimed to investigate the protective effects of necrostatin-1 on doxorubicin-induced cardiotoxicity by using the Langendorff model and to evaluate the pathways involved in that process.
Materials and methods
Pharmacological analysis
Male Sprague Dawley rats were divided into four groups. Groups were defined as: 1. Control group; received the injection of 1 mL (I.P) sterile saline. 2. Doxorubicin (Dox) group; single dose of 10 mg/kg (I.P) Dox injection. 3. Dox + Necrostatin-1 (Dox + Nec-1) group; a single dose of Doxorubicin (10 mg/kg, I.P) + Necrostatin-1 (1.65 mg/kg, I.P, for 7 days).
21
4. Necrostatin-1 group (Nec-1); Necrostatin-1 (1.65 mg/kg, I.P Nec-1 for 7 days)
After 7 days, in all groups, rats were heparinized (500 IU/kg) from the femoral vein and anesthetized with 100 mg/kg ketamine+15 mg/kg xylazine intramuscularly. Hearts were isolated by the ascendant aorta and immediately cannulated through the aorta according to the method of Langendorff. 22 Coronary perfusion was achieved by using the Krebs solution (aspirated with 5% CO2 and 95% O2 mix) with constant flow. After the 30- to 45-minute stabilization period that is required to reach maximum cardiac function values, the left ventricular developed pressure (LVDP) was measured in the left ventricular with a water-filled latex balloon. Also, the LV (dP/dt) max and LV (dP/dt) min values which show cardiac contractility and relaxation were recorded. In addition, the heart rate (HR) was recorded using electrodes attached to the hearts. In all groups, recordings taken by a Biopac MP 30 amplifier were analyzed on the computer.
Real-time polymerase chain reaction (PCR)
RNA Isolation and RT-PCR Analysis: Total RNA was extracted from non-perfused rat cardiac tissues using MagNa Lyser Green Beads (Roche, #03358941001) and a High Pure RNA Isolation Kit (Roche, #11828665001). cDNA was synthesized with a Transcriptor First Strand cDNA synthesis kit (Roche, #04896866001). For relative quantification real-time PCR assay, a FastStart Essential Probe Master Kit (Roche, #06402682001) was used in a LightCycler® 96 Real-Time PCR System (Roche). Predesigned primer probes including caspase-3 (5′-AAACCTCCGTGGATTCAAAA,AGCCCATTTCAGGGTAATCC-3′), bcl-2 (5′-GTACCTGAACCGGCATCTG,GGGGCCATATAGTTCCACAA-3′), bax (5-CGAGCTGATCAGAACCATCA,GGGGTCCCGAAGTAGGAA-3′), Nox-2 (5′-TGCAGACAAAATCAAAGAATGG,AACATGGGACCCACTATCCA-3′), and B-actin (5′-CTAAGGCCAACCGTGAAAAG,GCCTGGATGGCTACGTACA-3′) genes were measured. B-actin was used as the endogenous reference gene. Denaturation was performed as 1 cycle for 10 min at 95°C; then, the amplification step was performed as 5 s at 95°C, 20 s at 54°C, and 1 s at 72°C for 55 cycles; and finally, cooling was performed as 1 cycle for 30 s at 40°C.
Histopathological analysis
The left ventricle of the heart was fixed in formalin 10%. 4- to 5-μm thick cross-section of the left ventricle was stained with hematoxylin-eosin, Azan, and Masson trichrome dyes. Finally, heart samples were examined under the Nikon Eclipse 80i microscope (Nikon, Japan), and micrographs were taken.
Biochemical analysis
Malondialdehyde (MDA) levels were measured by using the thiobarbituric acid method in non-perfused heart samples from all four groups. 10
Statistical analysis
Statistical Package for the Social Sciences (SPSS) software was used to compute statistical data. All results were expressed as means ± standard deviation. One-way analysis of variance (ANOVA) and post-hoc Tukey tests were used for comparisons of differences between groups. A
Results
Pharmacological and biochemical results
In the Dox group, PP significantly increased as compared to the control group (
Malondialdehyde levels significantly increased in the Dox group as compared to the control group ( Perfusion pressure (PP), heart rate (HR), left ventricular developing pressure (LVDP), MDA, and LV (dP/dt) max and LV (dP/dt) min in all groups (a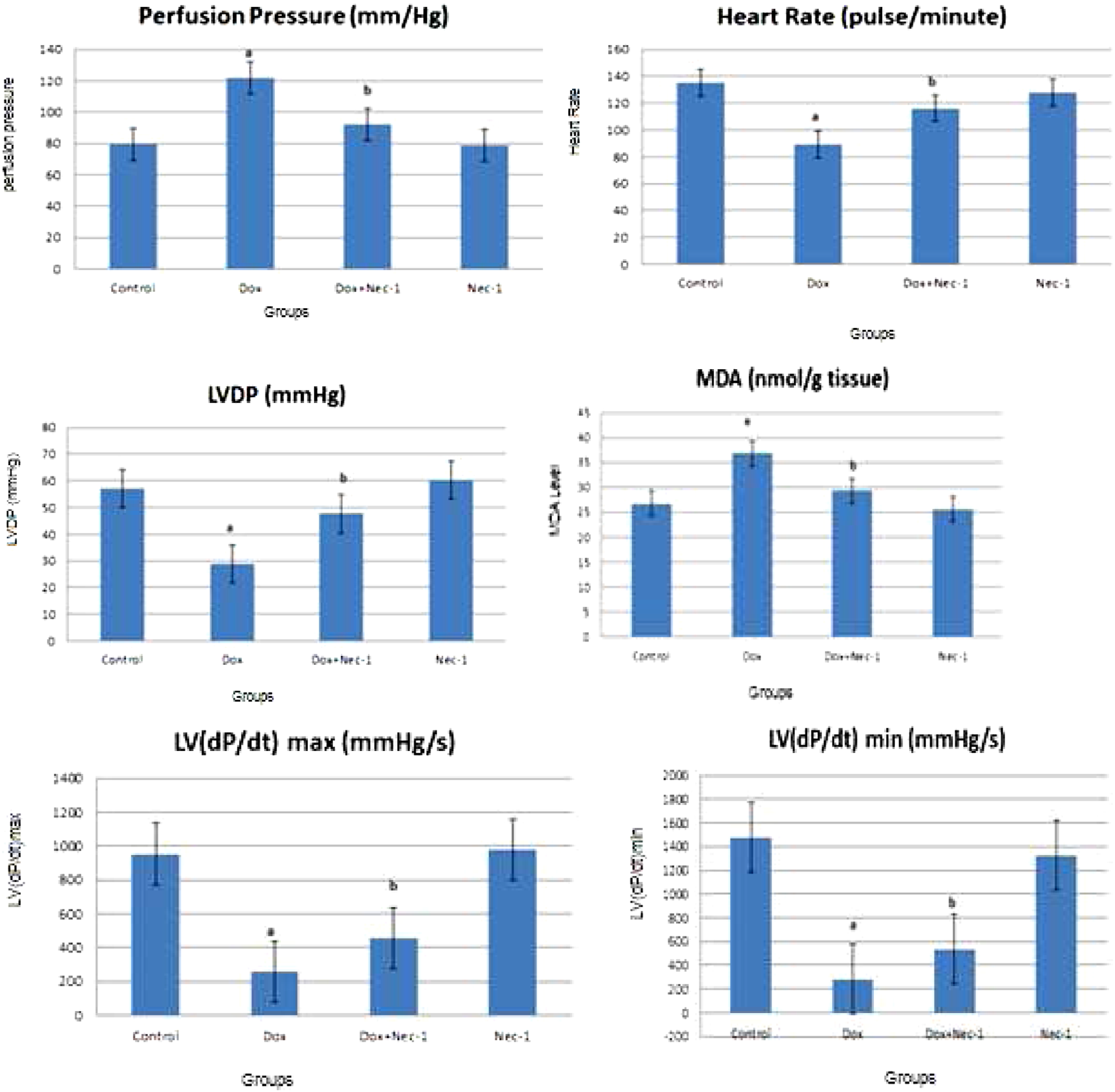
Histopathologic results
Control group: (A) small capillary structures (→) and endomysium (*) (Azan, Bar: 50 μm). (B) Oval euchromatic and central nuclei (
Dox group: (A) widespread hemorrhage (H-E, Bar: 50 μm) (B) in muscle fibers focal edema (Δ) hyalinization (H), and different forms of muscle cell nucleus (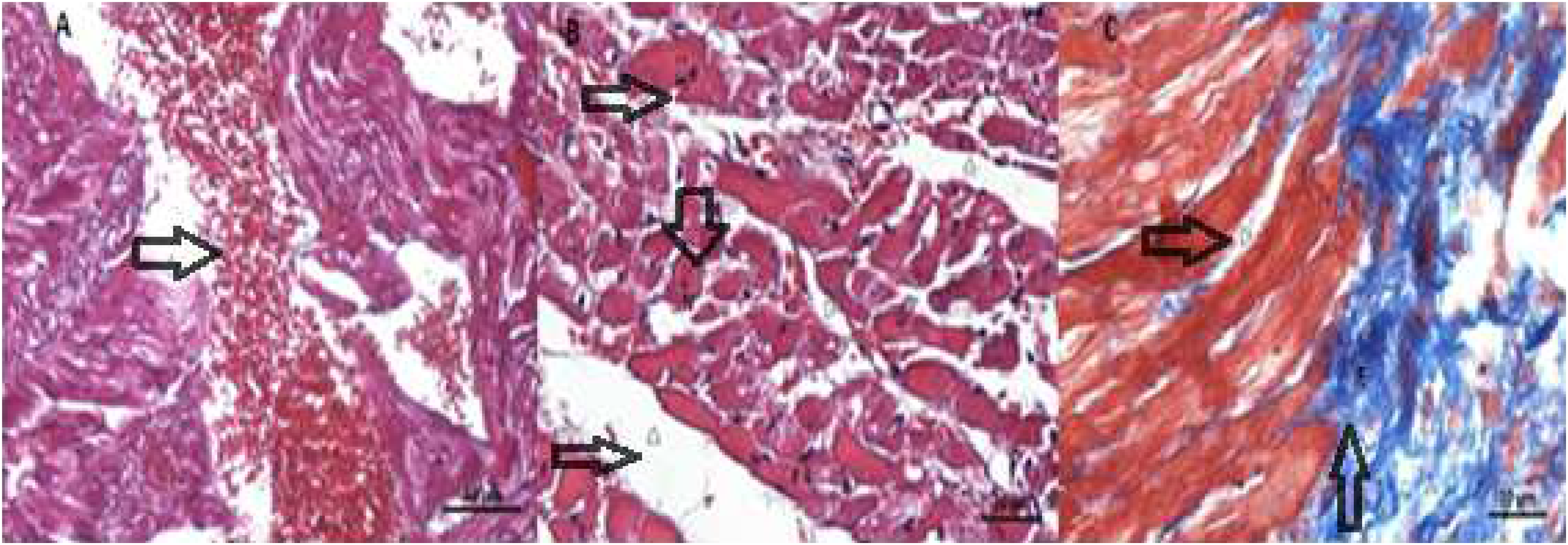
Dox+Nec-1 group: (A) focal interstitial edema (Δ), pyknotic nuclei (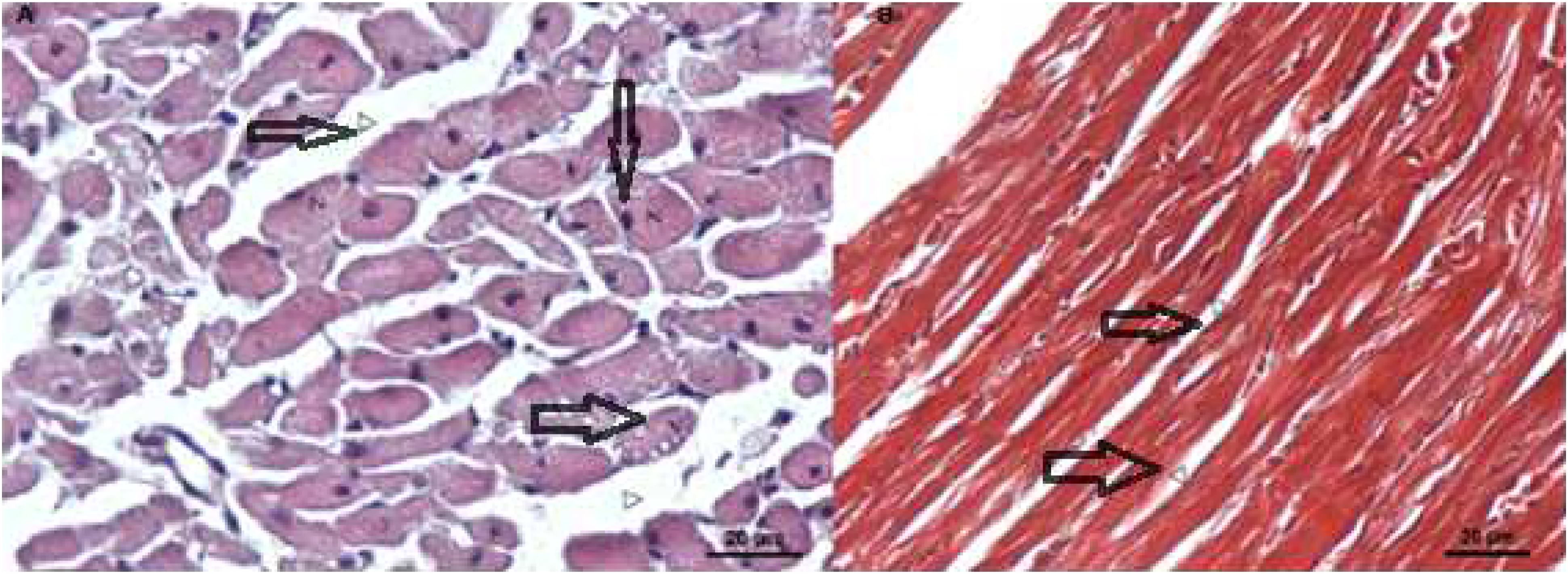
Nec-1 group: (A) normal striation and intercalated discs, and heart muscle cells with central positioning and oval and euchromatic nuclei (
Real-time PCR results
Bcl-2 expression levels significantly decreased in the Dox group as compared to those of the control group ( Bcl-2, bax, caspase-3, and Nox-2 gene expression levels belonging to the groups were given as % actin. a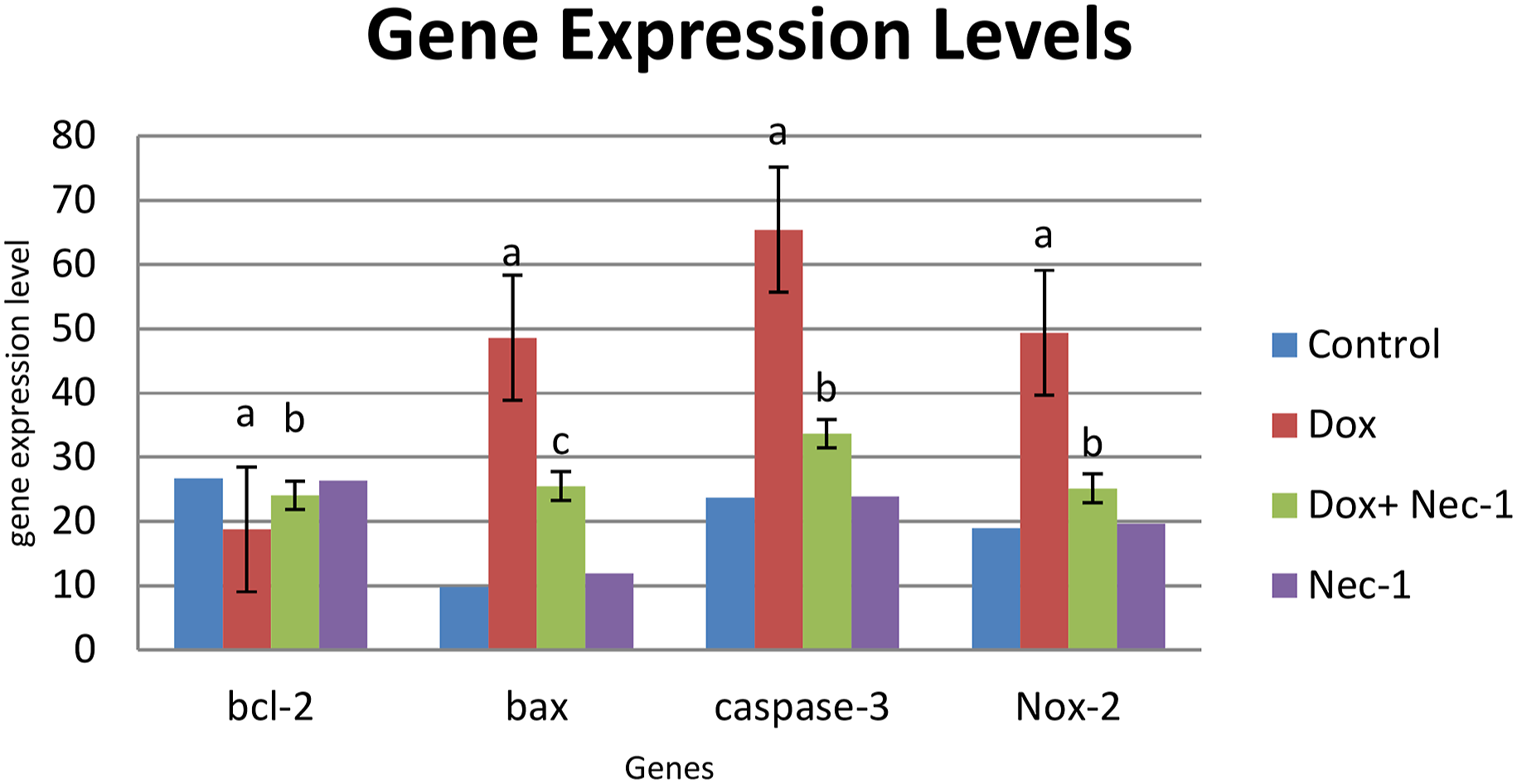
Discussion
The most important and serious side effect of doxorubicin is cardiotoxicity. Doxorubicin-induced cardiotoxicity is associated with several mechanisms, including oxidative stress; autophagy; calcium dysregulation; mitochondrial dysfunction; and the apoptosis, necroptosis, and ferroptosis pathways.4,7–9,12,15
In this study, cardiotoxicity was achieved by administering 10 mg/kg dose of doxorubicin. 23 We used an isolated perfused heart to evaluate the changes in doxorubicin-induced cardiac dysfunctions. Via Langendorff, isolated hearts were perfused, and PP, LVDP, HR, and LV (dP/dt) max and LV (dP/dt) min which shows cardiac contractility and relaxation were recorded. It was observed that in the Dox group, PP levels significantly increased; HR, LVDP, LV (dP/dt) max, and LV (dP/dt) min significantly decreased as compared to the control group. These findings indicate the doxorubicin-induced cardiomyopathy, and are consistent with the previous studies that reported the doxorubicin-induced cardiac dysfunction.2,3 Increased MDA levels in the Dox group also pointed out the involvement of ROS and oxidant stress in the cardiotoxicity. Similar studies also reported that doxorubicin increases oxidative stress and MDA levels.24,25 It is reported that an increase in lipid peroxidation and MDA level were also accompanied by a decrease in total antioxidant capacity of rats’ cardiac tissues. 26
In our study, treatment with Nec-1 25 decreased PP and MDA levels, and attenuated Dox-induced myocardial damage, indicating the role of necroptosis in doxorubicin-induced cardiotoxicity and the protective effect of Nec-1.
Our histological findings were also found to be compatible with biochemical results. We observed Dox-induced marked injury—such as widespread hemorrhages, loss of muscular striations, and hydropic degeneration in cardiomyocytes. In a previous study, similar histopathological changes include interstitial myocardial fibrosis, and the appearance of vacuolated cardiomyocytes was reported during doxorubicin treatment. 27 Similarly, it was shown that Nec-1 attenuated necrotic cell death in the spinal cord of rats with neuropathic pain. 28
In this study, to evaluate the apoptotic pathways involved in doxorubicin-induced cardiotoxicity and effect of Nec-1, gene expression levels of bcl-2, bax, Nox-2, and caspase-3 were determined in the heart tissues by using real-time PCR. In the Dox-treated group, we observed an increase in expression levels of pro-apoptotic protein Bax, apoptotic marker caspase-3, Nox-2 (a cardiac isoform of the Nox family), and a decrease in the expression level of anti-apoptotic bcl-2. These results indicate doxorubicin-induced apoptosis, reversed by Nec-1 treatment.
A study, which evaluated the influence of Dox in apoptosis and oxidative stress in the breast cancer cell lines, reported that Dox induces apoptosis by upregulating bax, caspase-8, and caspase-3 and downregulation of bcl-2 protein expression. 11 Similarly, in another study, it was reported that Dox treatment increased bax expression and decreased bcl-2 expression, and triggered ROS generation and cell apoptosis. 29
In accordance with our results, it was reported that doxorubicin increases NADPH oxidase activity and Nox2/Nox4 mRNA expression, and NADPH oxidase-derived ROS have a major role in cardiac pathophysiology. 30 It was also demonstrated that Dox increased the levels of ROS and MDA in heart tissues and also reduced protein expression of glutathione peroxidase, and it was shown that RIP1 and RIP3 were significantly increased with Dox treatment. 31
These findings point out that ROS production, apoptosis, and necroptosis exist in doxorubicin-induced cardiotoxicity. Several studies suggest the protective effect of Nec-1 against various pathology including I/R injuries, myocardial infarctions, and inflammation.19,32,33
Nec-1 was found to prevent cardiac contractile dysfunction by the inhibition of RIP1-RIP3-MLKL signaling cascade and ROS formation. 34 In another study, it was reported that Nec-1 reduced H2O2-induced cell damage by the mechanisms involved in the inhibition of lysosomal protease and cathepsin D, but not of caspase-3 or calpain activities. 35
Our results clearly demonstrated that the protective effects of Nec-1 on doxorubicin-induced cardiotoxicity are not only based on inhibition of necroptosis, but also by suppressing apoptotic and proapoptotic gene expressions and increasing antiapoptotic gene expressions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
