Abstract
Background
Epilepsy is a common disorder affecting approximately 50 million people worldwide. Oxidative stress is known to play an important role in the pathophysiology of diseases, including epilepsy. In this study, we investigated the effects of sennoside B on PTZ-induced seizures in rats.
Method
The rats were grouped into Group Electroencephalography and Group Behavioral. Both Groups were divided into eight subgroups, and these subgroups were compared in terms of the time of first myoclonic jerk, Racine’s Convulsion Scale, malondialdehyde levels, and brain superoxide dismutase activity. The experimental seizure model was performed with pentylenetetrazol.
Results
The spike percentage was significantly lower in groups that received sennoside B, and this beneficial effect was shown to be associated with the dose of sennoside B received. The RCS score was lower and the FJM onset time was higher in the sennoside B-administered groups. Additionally, brain MDA and brain aquaporin-3 levels were lower and brain SOD activity was higher in the sennoside-administered groups.
Conclusions
The present study shows the beneficial effects of sennoside B on PTZ-induced convulsion in rats. It is considered that sennoside B which is a natural and safe product would be a good candidate for strengthening the management of epilepsy without serious side effects.
Introduction
Epilepsy is a common neurological disease that affects approximately 50 million individuals worldwide.1,2 Epilepsy causes increased mortality as well as comorbidity and impairment in all aspects of life.3,4 Despite its high rate of occurrence and significant treatment efforts, available antiepileptic agents provide symptomatic relief from seizures with considerable side effects, and merely 30% of people’s seizures are reported to be controlled.5,6 Furthermore, there is no prevention strategy for the onset of the disease. Seizures and status epilepticus can result in neurodegeneration in many brain areas. 7 Thus, the new agents should not only prevent epilepsy candidates from experiencing seizures, but they should also repair the damage caused by the seizures.8,9 To create such drugs, it is essential to understand the pathophysiological factors of seizure. Nonetheless, the etiopathogenesis of epilepsy is quite complex.10,11
It is well-established that oxidative stress plays a significant role in the pathophysiology of neurological diseases, such as epilepsy. 12 It was discovered that pentylenetetrazol (PTZ) acted as an antagonist of the GABA A receptor. Acute and chronic experimental seizures can be induced with PTZ. For instance, a threshold dose of 60–100 mg/kg administered intravenously as an acute injection of PTZ results in myoclonic jerks, clonus, and tonic extensions in rodents. 13 Thus, higher doses of PTZ create a seizure model defined as epilepsy with repetitive seizures. While an experimental model of seizure was produced by PTZ in mice, it was reported that a significant reduction occurs in thiols. Thiols have the status of an antioxidant, as increased levels of thiol disulfides, lipid peroxidation, and protein oxidation are associated with oxidative status in the hippocampus of mice. 14
Natural products (NPs) are commonly used in clinical practice and act important roles in drug development. The agents which mimic the effects of NPs are also significant for clinical practice and as well as research. Sennoside is produced from senna and is commonly used as a laxative. The senna was reported as safe and effective in clinical settings. 15 Besides its laxative effects, there is little data on other effects of sennosides. It was demonstrated that sennoside B could able to block platelet-derived growth factors in osteosarcoma cells. 16 Additionally, sennoside B was reported to inhibit SARS-CoV-2 main protease activity 17 and TNF-α activity. 18 . However, there is not any data that explored the effects of sennosides in the treatment of seizures.
In this research, we aimed to assess the effects of sennoside B on rats with PTZ-induced seizures.
Materials and methods
Animal and laboratory
The present study was approved by Animal Ethics Committee (Science University, Number: 20221116). All experiments were performed in the line of the Guide for the Care and Use of Laboratory Animals, as confirmed by the National Institutes of Health (U.S.)
Experimental procedures
Two groups of 48 rats were assigned to Group A for EEG recording and Group B for behavioral assessment. The rats in both groups were randomly and equally selected. In the EEG and behavioral groups, we experimentally established the first subgroup as the control group, the second subgroup as the placebo, and the third and fourth subgroups as the sennoside B dose determination subgroups, receiving 5 mg/kg and 10 mg/kg, respectively.
Rats in Group A were completely sedated. Next, a small hole was stereotaxically opened using a drill. The reference electrode was implanted over the cerebellum (1.5 mm posterior to the lambda, on the midline) and the electrodes (Polyamide-coated stainless-steel wires, 0.1 mm diameter, and electrical resistance 1/10 mm) were placed on the dura over the left frontal cortex (2.0 mm lateral to the midline, 1.5 mm anterior to the bregma). Ketamine (80 mg/kg) and xylazine (4 mg/kg) intraperitoneally (i.p.) were used to completely anesthetize rats. While 70 mg/kg consistently caused obvious behavioral changes but EEG readings have a limited signal-to-noise ratio to see differences in drug concentrations, 35 mg/kg is suitable for observing changes in EEG spikes but does not consistently produce observable behavioral changes. 24 rats were randomly separated into four groups (n = 6) as: Groups A1, A2, A3, and A4 after the electrodes had been set for 12 days.
Group A1 was designated as the control group and was not treated. Saline was given intravenously to Group A2, 5 mg/kg Sennoside B (Senokot 15 mg) was given intravenously to Group A3, and 10 mg/kg Sennoside B was given intravenously to Group A4. Before injecting pentylenetetrazol (PTZ) (35 mg/kg, i.p.), the medications were given. Except for Group A1, all groups received 35 mg/kg PTZ while having their EEGs recorded. After administering PTZ for 5 min, awake rats were placed in a separate container where EEG recordings were made. The previously outlined behavioral assessment procedures and all EEG recordings were carried out. 8
Then, 24 new rats were added to the groups (Group B), and these rats were split into four groups (n = 6): Groups B1, B2, B3, and B4. Group B1 was designated as the control group and was not administered any medication. Saline was given intravenously to Group B2, Sennoside B at a dose of 5 mg/kg to Group B3, and 10 mg/kg to Group B4. The medications were given 30 min before injecting PTZ (70 mg/kg, i.p.). The seizures were assessed by Racine’s Convulsion Scale (RCS) and “first myoclonic jerk” (FMJ) onset timings. 8
Measurement of brain lipid peroxidation (MDA)
Lipid peroxidation was determined in tissue samples by measuring malondialdehyde (MDA). Levels were calculated from the standard calibration curve using tetra ethoxy propane and expressed as nmol/gr protein.
Determination of brain SOD activity
Total SOD activity was determined according to the method of Sun et al. 19 SOD activity was given as units per milligram protein (U/mg protein).
Brain aquaporin three analysis
With the aid of rat enzyme-linked immunosorbent assay (ELISA) kits that are readily available in the marketplace, the concentrations of aquaporin 3 (MyBioSource Co. Ltd.) in the brain tissue supernatants were measured. (MultiscanGo, Thermo Fisher Scientific Laboratory Equipment, NH, US).
Statistical analysis
The acquired data was evaluated SPSS rendition 26.0 for Windows. Shapiro-Wilk test is utilized to decide whether a populace of values has a typical conveyance. The Kruskal-Wallis test and Mann-Whitney test were used to compare the nonparametric data. The parametric numeric data was evaluated through a one-way analysis of variance (ANOVA). For the pairwise comparisons, the Post-hoc Tukey test was used. It was considered that p < 0.05 was statistically significant.
Results
Assessment of spike percentage
Comparisons of spike percentages between groups.
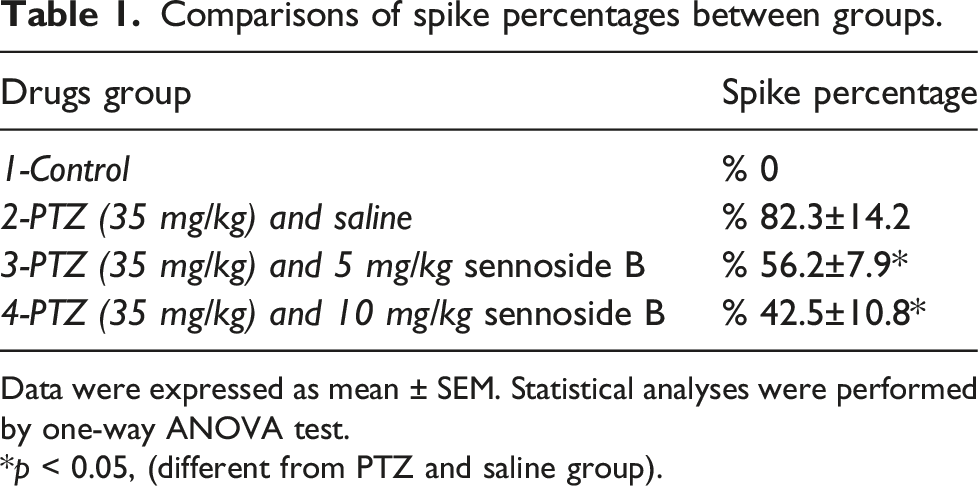
Data were expressed as mean ± SEM. Statistical analyses were performed by one-way ANOVA test.
*p < 0.05, (different from PTZ and saline group).

EEG recording. (a): Control group, (b): PTZ and saline group, (c): PTZ and 10 mg/kg Sennoside B group, (d): PTZ and 10 mg/kg Sennoside B group.
Assessment of the RCS
The median convulsion scale scores were 0, 5.5 ± 0.2, 4.1 ± 0.3, and 3.6 ± 0.3 in Figure 2 the Control Group, Saline group, Sennoside 5 mg/kg group, and Sennoside 10 mg/kg group, respectively. The Saline group had a higher RCS score than the control group and both groups (p < 0.0001, p < 0.05, and p < 0.05). Similarly, the control group had a lower RCS score than both of the sennosides groups (p < 0.0001 and p < 0.0001) Sennoside 5 mg/kg group had a higher RCS score than the sennoside 10 mg/kg group (p < 0.05) (Table 2). Comparisons of seizure outcomes. Comparisons of RCS scores and FJM onset time between groups. Data were expressed as mean ± SEM. Statistical analyses were performed by one-way ANOVA test. *p < 0.05, **p < 0.01 (different from PTZ and saline group).

Assessment of FMJ onset time
The mean FMJ onset times were 0 s, 63.8 ± 7.1 s, 106.2 ± 8.5 s, and 159.3 ± 12. Four in the Control Group, Saline group, Sennoside 5 mg/kg group, and Sennoside 10 mg/kg group, respectively. The FMJ onset time was significantly lower in the saline group compared with the control group (p < 0.0001). Mean FMJ onset time was also significantly lower in the saline group compared with Sennoside 5 mg/kg group and Sennoside 10 mg/kg group (p < 0.005 and p < 0.001). Similarly, the control group had a lower FMJ onset time than both of the sennosides groups (p < 0.0001 and p < 0.0001) (Table 2).
Comparison of brain MDA levels between groups
The brain MDA level was significantly higher Figure 3 in the saline group compared with the control group and the sennosides group (p < 0.001, p < 0.05, and p < 0.01, respectively). It was also significantly higher in the control group compared with sennoside 5 mg/kg and 10 mg/kg groups (p < 0.001 and p < 0.001, respectively) (Table 3). Comparisons of brain MDA, SOD, and AQP3 levels. Comparisons of brain MDA levels, brain SOD activity, and brain aquaporin 3 levels between groups. *ANOVA test, **Kruskal Wallis test aControl&Saline: p < 0.001, Control&Sennoside 5mg: p < 0.05, Control&Sennoside 10mg: p < 0.01, Saline&Sennoside 5Mg: p < 0.001, Saline&Sennoside 10Mg: p < 0.001 bControl&Sennoside 5mg: p < 0.05, Control&Sennoside 10mg: p < 0.05, Saline&Sennoside 5Mg: p < 0.05, Saline&Sennoside 10Mg: p < 0.05, Sennoside 5mg&Sennoside 10Mg: p < 0.05 cControl&Saline: p < 0.0001, Saline&Sennoside 5mg: p < 0.001, Saline&Sennoside 10mg: p < 0.001, : p < 0.001, Control&Sennoside 5mg: p < 0.05, Control&Sennoside 10mg: p < 0.05, Sennoside 5mg&Sennoside 10Mg: p < 0.05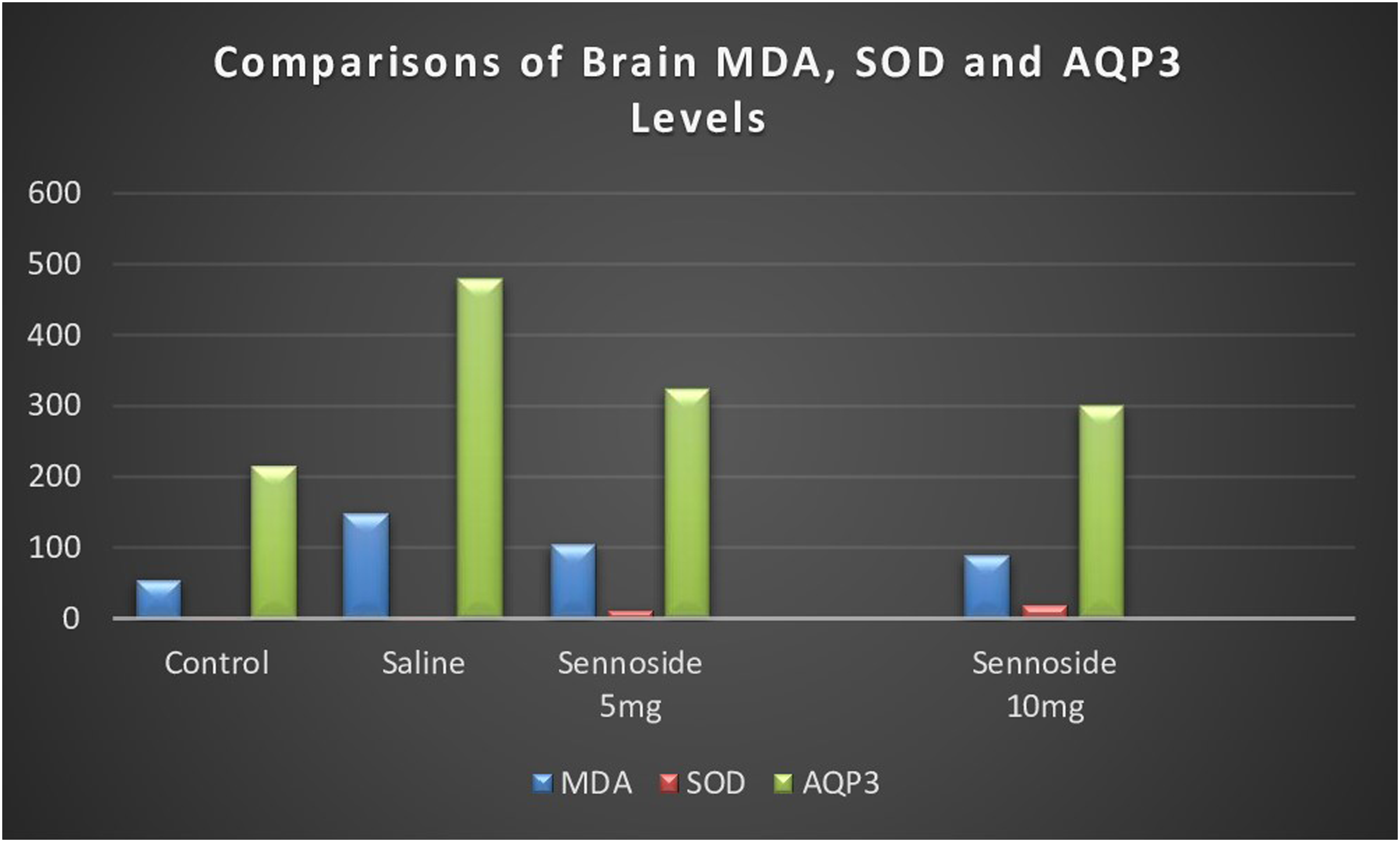

Comparison of brain SOD activity between groups
The brain SOD activity was significantly higher in sennoside groups compared with control and saline groups (p < 0.05 and p < 0.05). The brain SOD activity was similar between sennoside 5 mg/kg and 10 mg/kg groups. Sennoside 10 mg/kg group had higher brain SOD activity compared with the sennoside 5 mg/kg group (p < 0.05) (Table 3).
Comparison of aquaporin 3 MDA levels between groups
The brain aquaporin three level was significantly higher in the saline group compared with the control group and the sennosides group (p < 0.0001, p < 0.001, and p < 0.001, respectively). It was also significantly higher in sennoside 5 mg/kg and 10 mg/kg groups compared with the control group (p < 0.05 and p < 0.05, respectively) Sennoside 10 mg/kg group had lower brain aquaporin three levels compared with sennoside 5 mg/kg group (p < 0.05) (Table 3).
Discussion
In the present study, we found that sennoside B had favorable results in terms of behavioral and neurochemical parameters on rats that had PTZ-induced seizures. Sennoside B has been shown to have antioxidant effects on brain tissue. Moreover, aquaporin three levels were significantly lower in both of the sennoside B groups compared with the saline group.
Several studies have reported that antioxidants were successful in reducing neuronal damage after prolonged seizures. However, they could not demonstrate a particular effect on the development of convulsions.20–22 Studies that reported the beneficial effects of the administration of antioxidants, such as NAC and sulforaphane (SFN), have also been carried out.23,24 Moreover, PTZ triggers various toxic conditions by possibly supporting oxidation that causes seizures. The abnormal redox state of the cells can be associated with several effects during epileptogenesis in different manners. The increase in oxidative stress is potentially related to mitochondrial dysfunction and increased neuronal excitability. 21 In the present study, PTZ-administered rats were shown to have increased amounts of brain MDA, which demonstrates increased lipid peroxidation compared with control rats. The amount of brain MDA was shown to be less in rats given 5 mg/kg and 10 mg/kg sennoside B compared with the saline- and PTZ-administered groups. Moreover, the level of brain MDA was less in the sennoside B 10 mg/kg group compared with the 5 mg/kg group. SOD is a metalloprotein and the first line of defense against oxidative stress in various tissues of the body. It catalyzes superoxide radicals and acts as a leading antioxidant. 25 We have found that SOD activity increased in sennoside B groups dependent on dose compared with the saline group. In our study, we demonstrated also that sennoside B administration decreased the spike percentage in PTZ-induced rats. While the dose of sennoside B was increased, the spike percentage also decreased. Additionally, the mean values of the RCS scores were significantly lower in the sennoside B-administered groups, and the group that received an increased dose of sennoside B had a significantly lower RCS score. Importantly, the sennoside B-administered groups had a longer FJM onset time in a dose-dependent manner. Our results are preliminary and the first to demonstrate the beneficial effects of sennoside B in an experimental seizure model and the first to show antioxidant effects in brain tissue. The probable antioxidant activity of sennoside B could be related to anti-seizure effects.
The mammalian aquaporins (AQPs) are a family of proteins and are placed in several tissues including the kidney, skin, and fat cells. 26 Aquaporin-3 was shown to regulate cellular uptake of H2O and affect intracellular signaling in mammalian cells. 27 Additionally, AQP-mediated water transport was reported to act important roles in brain volume regulation, cerebrospinal fluid movement, and metabolism. 28 It was reported that an increased level of AQP3 was associated with focal cerebral ischemia in experimental models. 29 Moreover, decreased levels of AQP were shown to protect brain tissue against oxidative stress and this effect is considered to be a candidate for clinical implications. 30 In the present study, we succeeded to demonstrate that the administration of sennoside B led to a decrease in the brain level of AQP3. Moreover, the decrease in the level of brain AQP3 was higher in the sennoside B 10 mg/kg group compared with the 5 mg/kg group. Our results are first to demonstrate decreasing effects of sennoside B on the AP3 level. The anti-seizure effect of sennoside B can be associated with reducing the level of brain AQP3.
The present study shows the beneficial effects of sennoside B on PTZ-induced convulsion in rats. Our study demonstrated these beneficial effects by utilizing objective tests, such as spike percentage and other tools, which assess convulsions. Moreover, we showed that sennoside B administration could reverse oxidative stress in brain tissue and decrease the level of brain AQP3. We argue that sennoside B, which is a natural and safe product will be a good candidate for strengthening the management of epilepsy without serious side effects. Further experimental and clinical studies are needed to confirm our preliminary results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This research was performed by the principles of the Helsinki Declaration.
