Abstract
Kidney is a major target organ in preclinical studies. In recent years, intense research has been undertaken to characterize novel renal toxicity biomarkers. In this context, we studied nephron segment specific antibodies against aquaporin-1 (AQP-1), α-glutathione-S-transferase (alpha-GST), Tamm-Horsfall protein (TH), calbindin-D28K (CalD), and aquaporin-2 (AQP-2), using an immunoperoxidase method on formalin-fixed paraffin-embedded kidney tissues of dogs, rats, mice, and Cynomolgus monkeys. AQP-1 was specific for proximal tubules and thin descending limbs of Henle’s loops and AQP-2 for connecting and collecting ducts in dogs, rats, mice, and Cynomolgus monkeys. Alpha-GST stained the straight part of proximal tubules in dogs and proximal convoluted tubule and straight part of proximal tubules in rats. TH was specific for thick ascending limbs of Henle’s loops in mice, rats, dogs, and Cynomolgus monkeys and stained additionally scattered cells in cortical connecting/collecting ducts of dogs. CalD was found in distal convoluted tubules and cortical connecting and collecting ducts of dogs, rats, and mice and in distal convoluted tubules, connecting ducts, and cortical and medullary collecting ducts of Cynomolgus monkey. This panel of antibodies may be a helpful tool to identify renal tubules by light microscopy in preclinical studies and to validate new biomarkers of renal toxicity.
Keywords
Introduction
Drug-induced nephrotoxicity is a recurrent finding in preclinical studies. The tubular epithelium is especially sensitive to toxic compounds because of water and solute absorption and active transport systems, which result in concentration of toxicants in the tubular lumens or in tubular cells, and because of the high energy needs of these very active cells (Ferguson, Vaidya, and Bonventre 2008).
Early detection of nephrotoxicity by traditionally used markers like serum creatinine or blood urea nitrogen is not possible and prevents precise monitoring of kidney function. Precocious and reliable detection of renal toxicity in preclinical safety assessment has therefore stimulated intense research to develop novel biomakers (Boudonck et al. 2009). Among studied molecules, N-acetyl-β-glucosaminidase (NAG), glutathione S-transferases (GST), β2-microglobulin, cystatin-C, kidney injury molecule-1 (KIM-1), neutrophil gelatinase-associated lipocalin (NGAL), interleukin-18 (IL-18), fatty acid binding protein (FABP) (Ferguson, Vaidya, and Bonventre 2008; Togashi et al. forthcoming), Calbindin-D28K (Hoffmann et al. 2010), and Tamm-Horsfall protein (Lau et al. 2008) have given promising results; and seven of them (Kim-1, albumin, total protein, β2-microglobulin, cystatin C, clusterin, and trefoil factor-3) have been qualified in 2008 for renal safety by the Food and Drug Administration and the European Medecines Agency (see http://www.c-path.org/pdf/PSTC_nephro_VXDS_summary_final.pdf).
To evaluate sensitivity and specificity of tested renal biomarkers, correlation of measured variations with histopathology analyses of experimentally induced lesions is still the “gold standard.” But identifying the nephron segment targeted by a nephrotoxic agent either in the context of preclinical toxicological studies or of biomarker research using routine paraffin-embedded HE-stained sections is often challenging for the pathologist. Therefore, immunohistochemical staining may be a complementary helpful tool either to identify nephron segments targeted by a toxic compound or to localize directly the protein used as a biomarker if it is normally localized in the kidney.
The goal of this study is to develop an antibody panel to identify specific nephron segments immunohistochemically using five antibodies (against aquaporin-1, aquaporin-2, α-glutathione-S-transferase, CalD, and Tamm-Horsfall protein) by single, double, or multiple immunostainings on paraffin sections in commonly used preclinical study animal species: the Beagle dog (Canis familiaris), the Sprague-Dawley (SD) rat (Rattus norvegicus), the CD1 mouse (Mus musculus), and the Cynomolgus monkey (Macaca fascicularis).
Materials and Methods
Tissue Sampling
Kidney tissues were obtained from paraffin-embedded kidney tissues of control animals from 2- to 4-week toxicity studies. The species studied are those commonly used for studies in toxicology, i.e., CD1 mice (Crl:CD1[ICR]) (Les Oncins, Saint Germain sur L’Arbresle Cedex, France), Sprague-Dawley rats (Crl:CD[SD]) (Les Oncins, Saint Germain sur L’Arbresle, France), Beagle dogs (Marshal Farms, North Rose, NY, USA), and Cynomolgus monkeys (Noveprim, Mahebourg, Mauritius).
Animal housing and necropsies were conducted in accordance to AAALAC guidelines. All renal tissues were fixed in 10% neutral buffered formalin for 24 hours, shortly processed using a vacuum inclusion processor, and then to paraffin blocks. Four-micrometer-thick sections were cut from these blocks.
Antigen Retrieval, Primary Antibodies, Detection Systems
Antigen retrieval was achieved by immersion at 100°C for 20 minutes in a pH 6 citrate buffer. Endogenous peroxidase activity was quenched by incubation in 3% hydrogen peroxide in distilled water for 10 minutes. Endogenous biotin was blocked using a biotin blocking system obtained from Dako (Carpinteria, CA, USA).
All primary antibodies were incubated 1 hour at room temperature, except AQP-1 antibody for dog tissues which was incubated 12 hours at 4°C and alpha-GST antibody for rat tissues which was incubated 30 minutes at room temperature.
The primary antibodies and dilutions used were: rabbit anti-aquaporin-1 polyclonal antibody (Chemicon, Temecula, CA, USA) diluted at 1/500; rabbit anti-rat alpha-GST polyclonal antibody and rabbit anti-dog alpha-GST polyclonal antibody (Argutus, Dublin, Ireland) diluted at 1/500; sheep anti-human Tamm-Horsfall protein polyclonal antibody (Chemicon) diluted at 1/1,000; mouse anti-calbindin-D28K monoclonal (IgG1) antibody (Sigma, St. Louis, MO, USA) diluted at 1/1,000; and rabbit anti-water channel aquaporin-2 polyclonal antibody (Sigma) diluted at 1/1000.
Slides were incubated 30 minutes at room temperature with species-specific biotinylated secondary antibodies. Detection was achieved using an avidin-biotin-peroxidase method (StreptABcomplex/HPR detection system, Dako, Glostrup, Denmark). Two peroxidase substrates were used as chromogen: DAB brown chromogen (Dako, Carpinteria, CA, USA) and purple Vector VIP peroxidase substrate kit (Vector Laboratories, Burlingame, CA, USA). Slides were counterstained with hemalun, dehydrated, and mounted under glass coverslips.
Double and Multiple Immunostainings
Double stainings were performed as a first staining with DAB used as chromogen (brown) with a final wash in distilled water, followed by a second staining with Vector VIP peroxidase substrate (purple) used as chromogen, counterstaining in hemalun, dehydration, and mounting.
Combinations of primary antibodies used were: AQP-1 staining followed by AQP-2 staining on rat kidney tissues; AQP-2 staining followed by CalD staining on dog kidney tissues; AQP-2 staining followed by TH staining on dog kidney tissues; and CalD staining followed by TH staining on dog kidney tissues.
A multiple immunostaining procedure using a mixture of AQP-1, AQP-2, TH, and CalD primary antibodies used at the same time was performed to check the specificity of the antibodies. In this case, DAB chromogen was used.
Controls
For each immunohistochemistry procedure, isotypic and negative controls were included.
Negative controls were performed by omission of the primary antibody. Isotypic positive controls were achieved by substitution of primary antibodies by a rabbit anti-GFAP polyclonal antibody (Dako, Carpinteria, CA, USA).
Results
For each antibody, results obtained in the dog are exposed first and then compared to results in the rat, the mouse, and the Cynomolgus monkey.
Aquaporin-1
In the dog, AQP-1 positive cells were present in the cortex and in the medulla (Figure 1a).
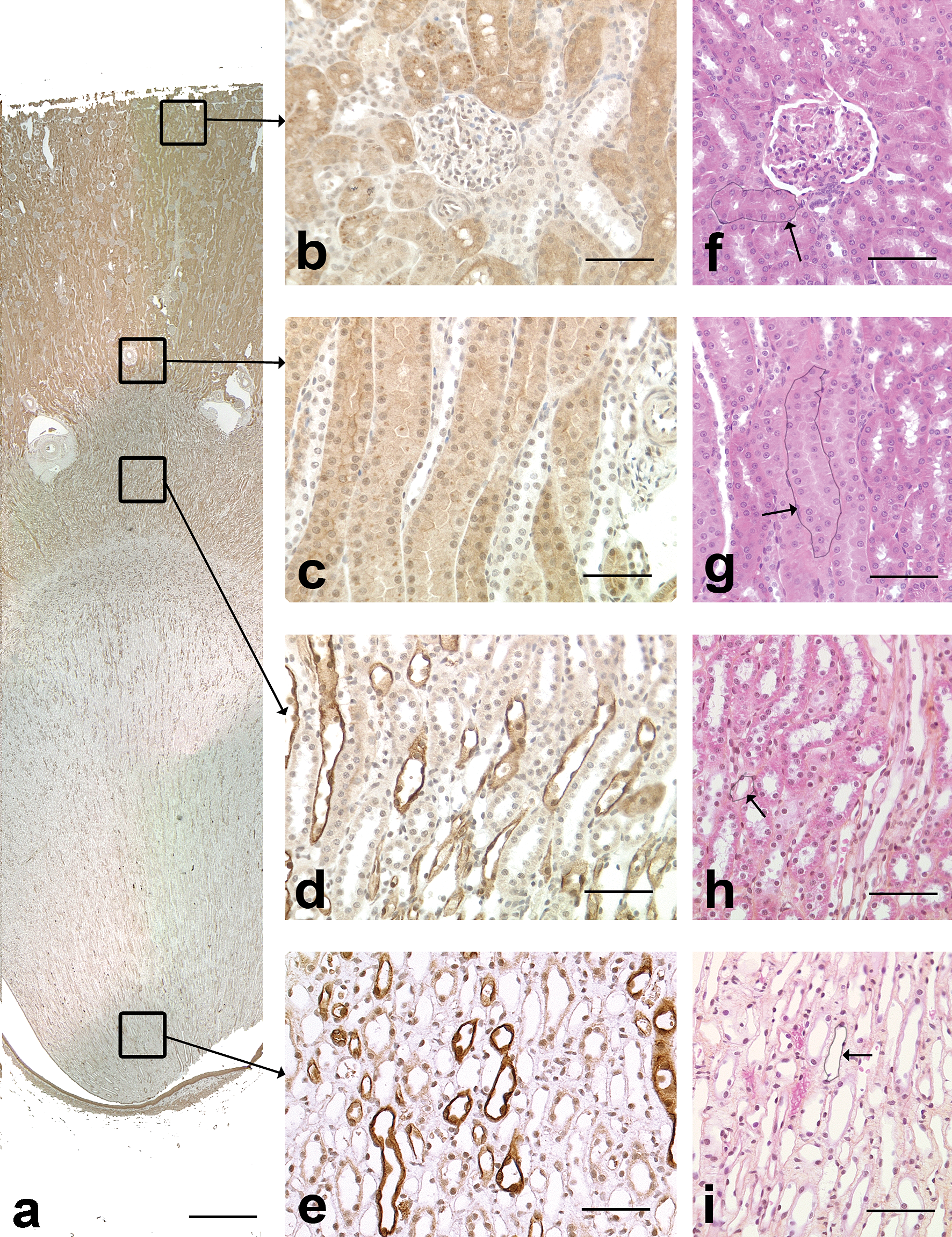
AQP-1 immunohistochemical staining in the dog kidney. a: Overview of AQP-1 staining. Note that positive cells were present in the cortex and in the medulla. Scale bar: 200 µm. b: Cortical labyrinth. Most of the tubules stained positive for AQP-1 and are compatible with proximal convoluted tubules. Scale bar: 70 µm. c: Medullary rays. The positive tubules in longitudinal section are compatible with straight part of proximal tubules. Scale bar: 70 µm. d: Outer medulla. The positive tubules in cross and longitudinal sections are compatible with thin descending limbs of Henle’s loops. Scale bar: 70 µm. e: Inner medulla. The positive tubules in cross and longitudinal sections are compatible with thin descending limbs of Henle’s loops. Scale bar: 70 µm. f: Cortical labyrinth, HES. Arrow: tubule with similar morphology as AQP-1 positive tubule, i.e. proximal convoluted tubule. Scale bar: 60 µm. g: Medullary rays, HES. Arrow: tubule with similar morphology as AQP-1 positive tubule, i.e. straight part of proximal tubule. Scale bar: 60 µm. h: Outer medulla, HES. Arrow: tubule with similar morphology as AQP-1 positive tubule, i.e. thin limb of Henle’s loop. Scale bar: 60 µm. i: Inner medulla, HES. Arrow: tubule with similar morphology as AQP-1 positive tubule, i.e. thin limb of Henle’s loop. Scale bar: 60 µm.
In the cortical labyrinth (Figure 1b) and medullary rays (Figure 1c), most of the tubules stained positive for AQP-1. These tubules, mainly observed as cross-sections (labyrinth) or in longitudinal orientation (medullary rays), had a narrow lumen and were lined by large polygonal epithelial cells with a striated cytoplasm and a central nucleus. The staining was diffuse in the cytoplasm and stronger along the apical membrane which had a brush border. The morphology and localization of these tubules were compatible with proximal convoluted and straight tubules with a stronger staining of the apical membrane and of the brush border.
In the outer and inner medulla (Figures 1d and 1e), scattered tubules stained positive for AQP-1. The affected tubules were mainly observed in cross-section, although a few were oriented longitudinally. These tubules were lined by simple squamous epithelial cells with scant cytoplasm and an oval nucleus and had a lumen of approximately 30 µm in diameter. The staining was diffuse in the cytoplasm. The morphology and localization of these tubules were compatible with thin descending limbs of Henle’s loop.
Endothelial cells of scattered small blood vessels in the renal cortex and medulla stained positive with AQP-1, especially in the glomeruli and in the vascular plexus of the outer medulla.
In rats, mice, and Cynomolgus monkeys, the pattern of AQP-1 staining was similar to that in dogs.
Aquaporin-2
In dogs, AQP-2 (AQP-2) positive tubules were present throughout the kidney from the cortex to the medulla (Figure 2a).
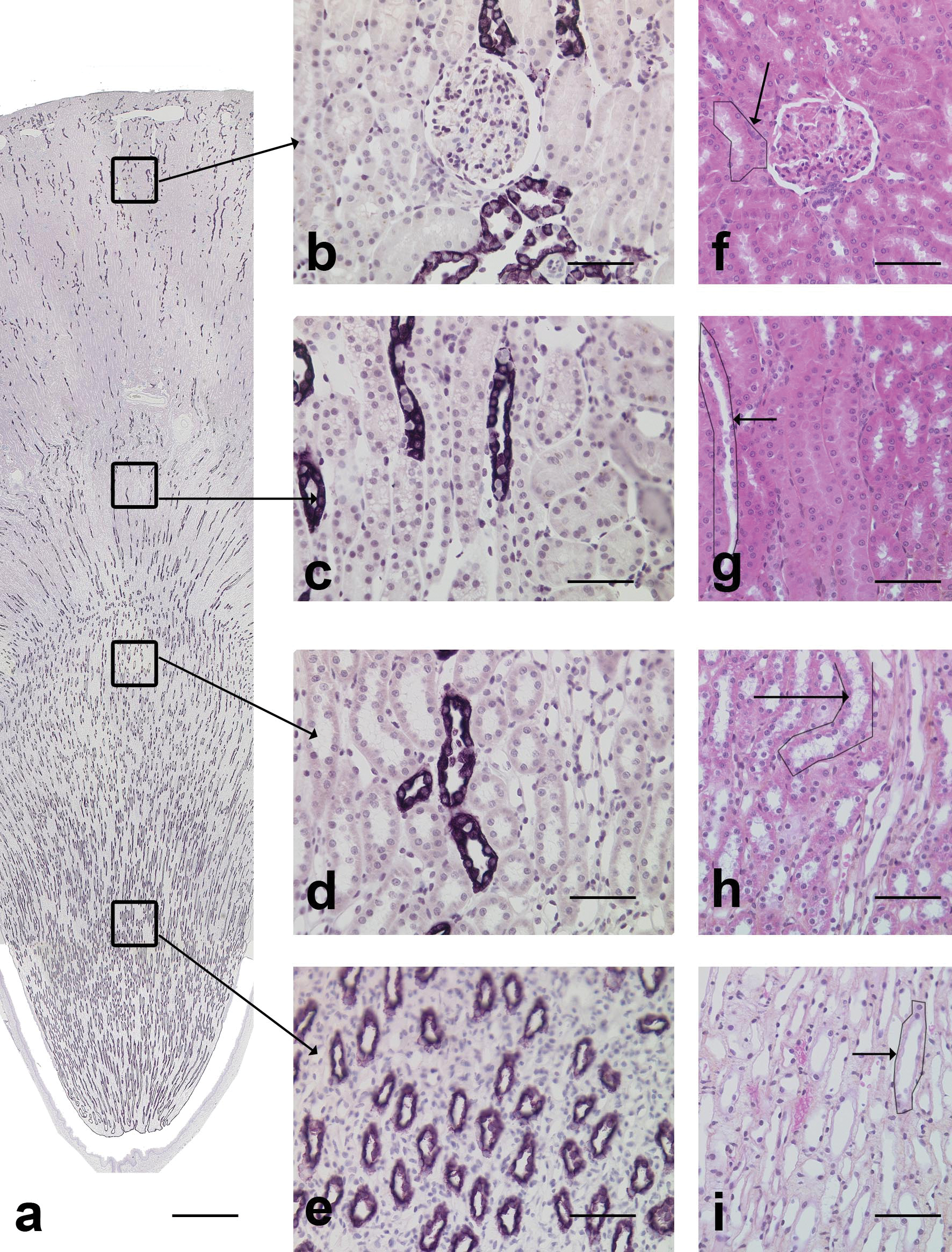
AQP-2 immunohistochemical staining in the dog kidney. a: Overview of the AQP-2 staining. Stained tubules were present from the cortex to the medulla mainly in longitudinal sections. Scale bar: 200 µm. b: Cortical labyrinth. The stained tubules are compatible with collecting ducts. Scale bar: 70 µm. c: Medullary rays. The stained tubules in longitudinal sections are compatible with collecting ducts. Scale bar: 70 µm. d: Outer medulla. The stained tubules in longitudinal sections are compatible with collecting ducts. Scale bar: 70 µm. e: Inner medulla. The stained tubules in cross sections are compatible with collecting ducts. Scale bar: 70 µm. f: Cortex labyrinth, HES. Arrow: tubule with similar morphology as AQP-2 positive tubule, i.e., collecting duct. Scale bar: 60 µm. g: Medullary rays, HES. Arrow: tubule with similar morphology as AQP-2 positive tubule, i.e., collecting duct. Scale bar: 60 µm. h: Outer medulla, HES. Arrow: tubule with similar morphology as AQP-2 positive tubule, i.e., collecting duct. Scale bar: 60 µm. i: Inner medulla, HES. Arrow: tubule with similar morphology as AQP-2 positive tubule, i.e., collecting duct. Scale bar: 60 µm.
In the cortical labyrinth, AQP-2 positive tubules were mainly noted in cross-sectional profiles and sometimes in close association with glomeruli (Figure 2b). In medullary rays, tubules were radiating and mainly oriented longitudinally (Figure 2c). Stained cortical tubules were lined by round to oval cells, delineating a large lumen. In each tubule, there were mainly AQP-2 stained cells and a few unstained cells. The staining was cytoplasmic and diffuse. In the medulla (Figures 2d and 2e), stained tubules were identical to cortical tubules, but the proportion of unstained lining cells gradually decreased from the corticomedullary junction to the papilla. The morphology and the localization of the tubules stained with AQP-2 were compatible with connecting and collecting ducts.
The pattern of AQP-2 staining in Cynomolgus monkeys, rats, and mice was similar to that in dogs. Stained tubules were present in the cortex and medulla and extended to the papilla. These tubules corresponded to connecting and collecting ducts. In Cynomolgus monkeys, unstained lining cells were observed in cortical and medullary collecting ducts. The identification of positive AQP-2 tubules was confirmed in Cynomolgus monkeys by the presence of multinucleated syncytial cells in medullary tubules, which are present in medullary collecting ducts in this species.
Calbindin-D28K
In dogs, CalD positive cells were observed in the cortex and in the medulla (Figure 3a). The outer two-thirds of the cortex contained the largest number of positive tubules. Only a few scattered tubules stained positive for CalD in the medulla. In the cortical labyrinth, scattered tubules stained positive and were mainly observed in cross-section (Figure 3b); while in the medullary rays, and in the outer and inner medulla, fewer tubules were stained with CalD (Figures 3c, 3d, 3e), and they were mainly noted in longitudinal profiles.
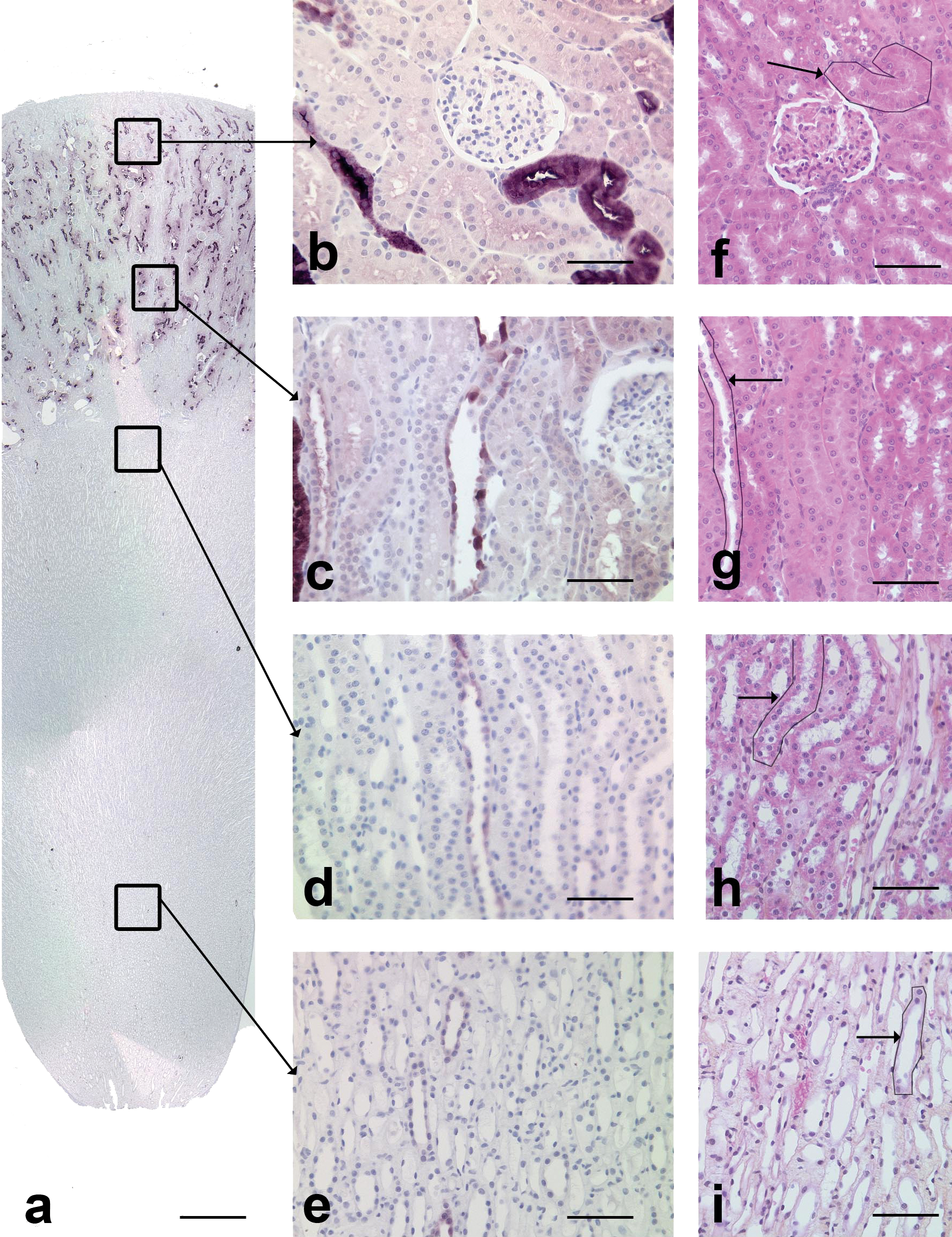
Calbindin-D28K immunohistochemical staining in the dog kidney. a: Overview of the CalD staining. Most of the positive cells were present in the cortex, and a few were observed in the medulla. Scale bar: 200 µm. b: Cortical labyrinth. The stained tubules are compatible with distal convoluted tubules. Scale bar: 70 µm. c: Medullary rays. Note the few scattered stained cells in tubules in longitudinal sections. These tubules are compatible with collecting ducts. Scale bar: 70 µm. d: Outer medulla. Few scattered lightly stained tubules are compatible with collecting ducts. Scale bar: 60 µm. e: Inner medulla. Few scattered lightly stained tubules are compatible with collecting ducts. Scale bar: 60 µm. f: Cortical labyrinth, HES. Arrow: tubule with similar morphology as CalD positive tubule, i.e., distal convoluted tubule. Scale bar: 60 µm. g: Medullary rays, HES. Arrow: tubule with similar morphology as CalD positive tubule, i.e., collecting ducts. Scale bar: 60 µm. h: Outer medulla, HES. Arrow: tubule with similar morphology as CalD positive tubule, i.e., collecting ducts. Scale bar: 60 µm. i: Inner medulla, HES. Arrow: tubule with similar morphology as CalD positive tubule, i.e., collecting ducts. Scale bar: 60 µm.
Two patterns of CalD staining were observed: In the cortical labyrinth, especially in the outer two-thirds, there were numerous strongly stained tubules. The staining was cytoplasmic, diffuse, and homogeneous between cells. The positively stained tubules were lined by rather large polygonal cells and were characterized by a narrow lumen. Multifocally, they were in close contact with glomeruli. This localization and morphology were compatible with distal convoluted tubules. In the deep cortex, more lightly stained tubules were observed. They were often localized in medullary rays and were lined by round to oval cells with a basal nucleus. The staining was cytoplasmic and diffuse, and there was variable staining intensity between cells. The localization and morphology of these tubules were suggestive of connecting and collecting ducts.
Scattered tubules were slightly stained in the medulla. As in the deep cortex, their localization and morphology was compatible with collecting ducts.
This was further confirmed by AQP-2/CalD double immunostaining as AQP-2 positive ducts were also positive with CalD (especially in the outer cortex).
In Cynomolgus monkeys, CalD positive tubules were observed in both the renal cortex and medulla (Figure 4a) and in higher numbers than in dogs. Two types of tubules were stained: distal convoluted tubules and connecting/collecting ducts. Distal convoluted tubules were noted in the cortical labyrinth as large tubules, often in cross section and adjacent to glomeruli. They were lined by large polygonal cells delineating a narrow lumen and characterized by a diffuse and homogeneous cytoplasmic staining. Connecting/collecting ducts were observed in the deep cortex as numerous longitudinal sections of radiate tubules (especially in medullary rays) and in medulla. These tubules were lined by round or oval cells; the staining was cytoplasmic and variable among cells (Figure 4b).
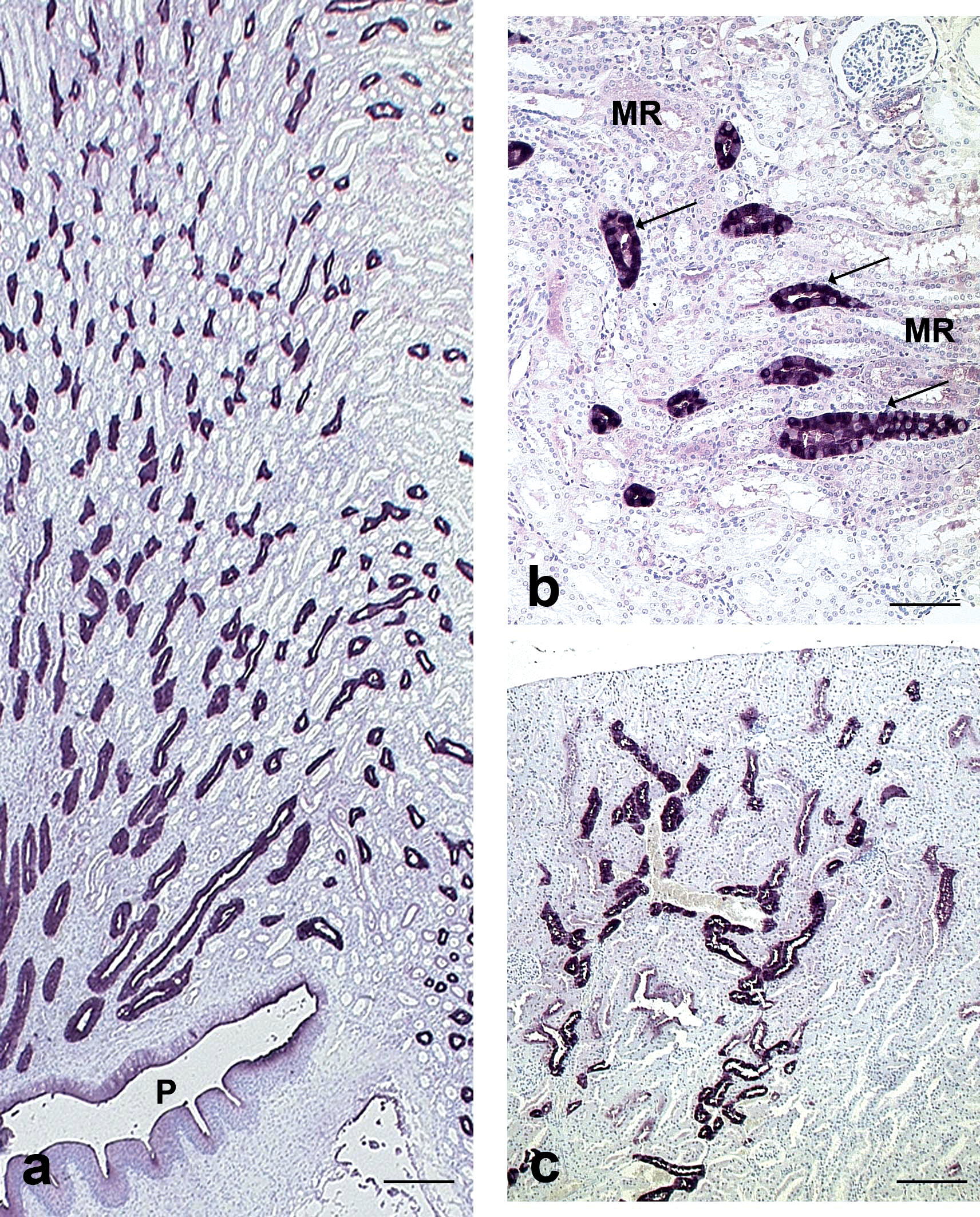
Calbindin-D28K immunohistochemical staining in Cynomolgus monkey and rat kidneys. a: Cynomolgus monkey, cortex, medulla, and papilla. Note the radiate stained tubules in longitudinal sections extending from the cortex to the pelvis (P) compatible with collecting ducts. Scale bar: 300 µm. b: Cynomolgus monkey, cortex. Note the stained tubules (arrows) in medullary rays (MR) composed of variably stained cells, compatible with collecting ducts. Scale bar: 30 µm. c: Rat, cortex. Note the stained tubules in cross and longitudinal sections compatible with distal convoluted tubules and cortical collecting ducts. Scale bar: 100 µm.
In rats and mice, the pattern of staining and the types of tubules stained were similar to dogs (Figure 4c).
Tamm-Horsfall Protein
In dogs, TH-positive cells were present in the cortex and in the medulla (Figure 5a). In the cortical labyrinth (Figure 5b), scattered tubules, often in cross-section, stained positive with TH protein. In medullary rays (Figure 5c), approximately one-third of the tubules, mainly those in short longitudinal sections, stained positive for TH. Most tubules of the outer medulla (Figure 5d) stained positive with TH. In the inner medulla (Figure 5e), only rare scattered cells in a few tubules stained positive for TH.
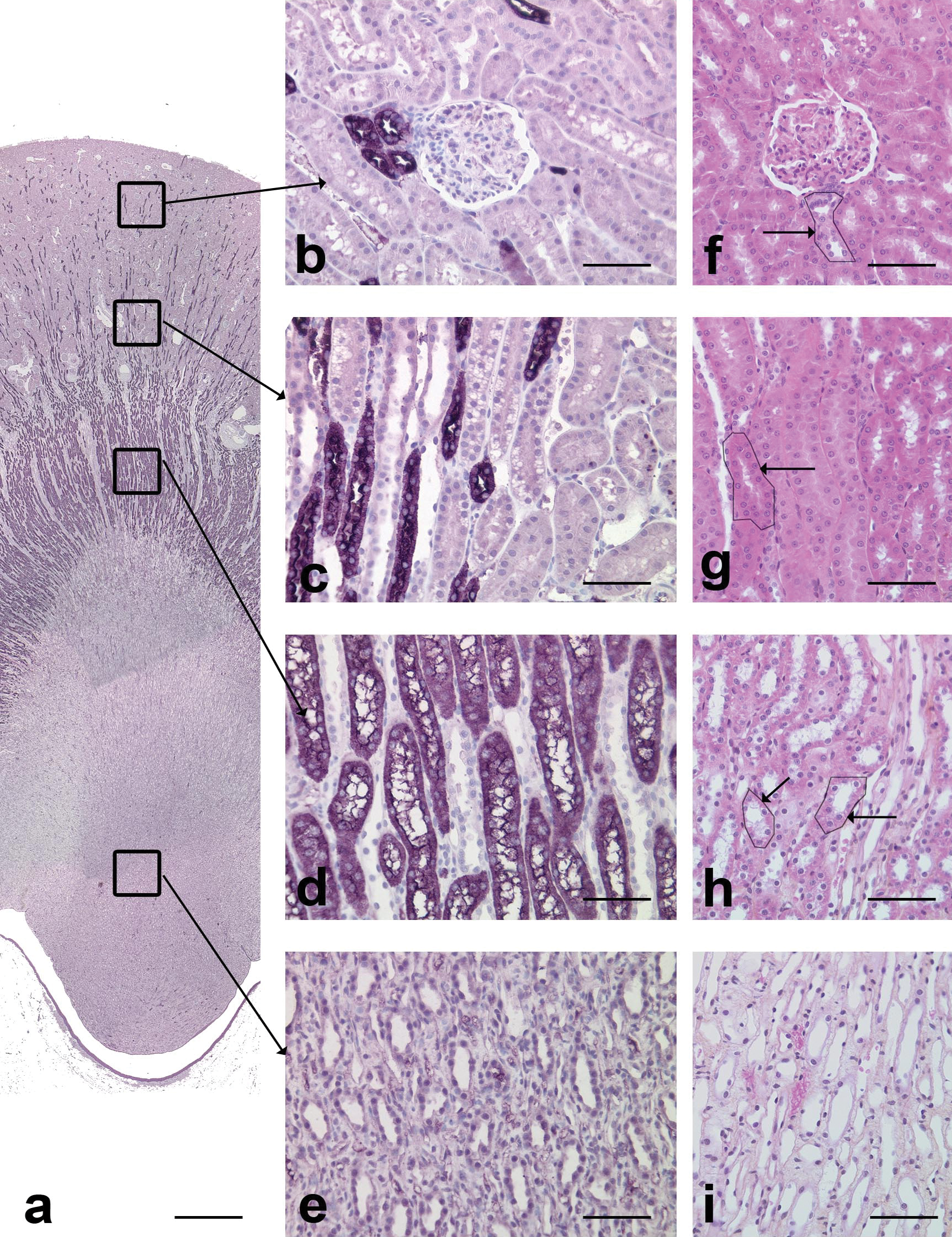
TH immunohistochemical staining in the dog kidney. a: Overview of TH protein staining. Note that positive cells were present in the cortex and in the medulla. Scale bar: 200 µm. b: Cortical labyrinth. Scattered TH protein positive tubules are compatible with thick ascending limbs of Henle’s loops. Scale bar: 70 µm. c: Medullary rays. Positive tubules in longitudinal sections are compatible with thick ascending limbs of Henle’s loops. Scale bar: 70 µm. d: Outer medulla. The numerous positive tubules in longitudinal sections are compatible with thick ascending limbs of Henle’s loops. Scale bar: 70 µm. e: Inner medulla. Note the absence of positive tubules. f: Cortical labyrinth, HES. Arrow: tubule with similar morphology as TH protein positive tubule, i.e. thick ascending limb of Henle’s loop. Scale bar: 60 µm. g: Medullary rays, HES. Arrow tubule with similar morphology as TH protein positive tubule, i.e. thick ascending limb of Henle’s loop. Scale bar: 60 µm. h: Outer medulla, HES. Arrow: tubule with similar morphology as TH protein positive tubule, i.e. thick ascending limb of Henle’s loop. Scale bar: 60 µm. i: Inner medulla, HES. Scale bar: 60 µm.
In the superficial cortex, the pattern of TH protein staining was different from that in the other parts of the kidney, as only scattered positive cells were observed (Figure 6a). These cells were present in tubules that had a relatively large lumen and that were lined by cuboidal to round epithelial cells. In order to identify these tubules, double staining was performed (i.e., TH/AQP-2 [Figure 6b] and TH/CalD [Figure 6c]). Tubules containing scattered TH positive cells stained positive for AQP-2 and slightly positive for CalD indicating that they were connecting/collecting ducts. In these tubules, the TH positive cells were not stained either with CalD or with AQP-2.
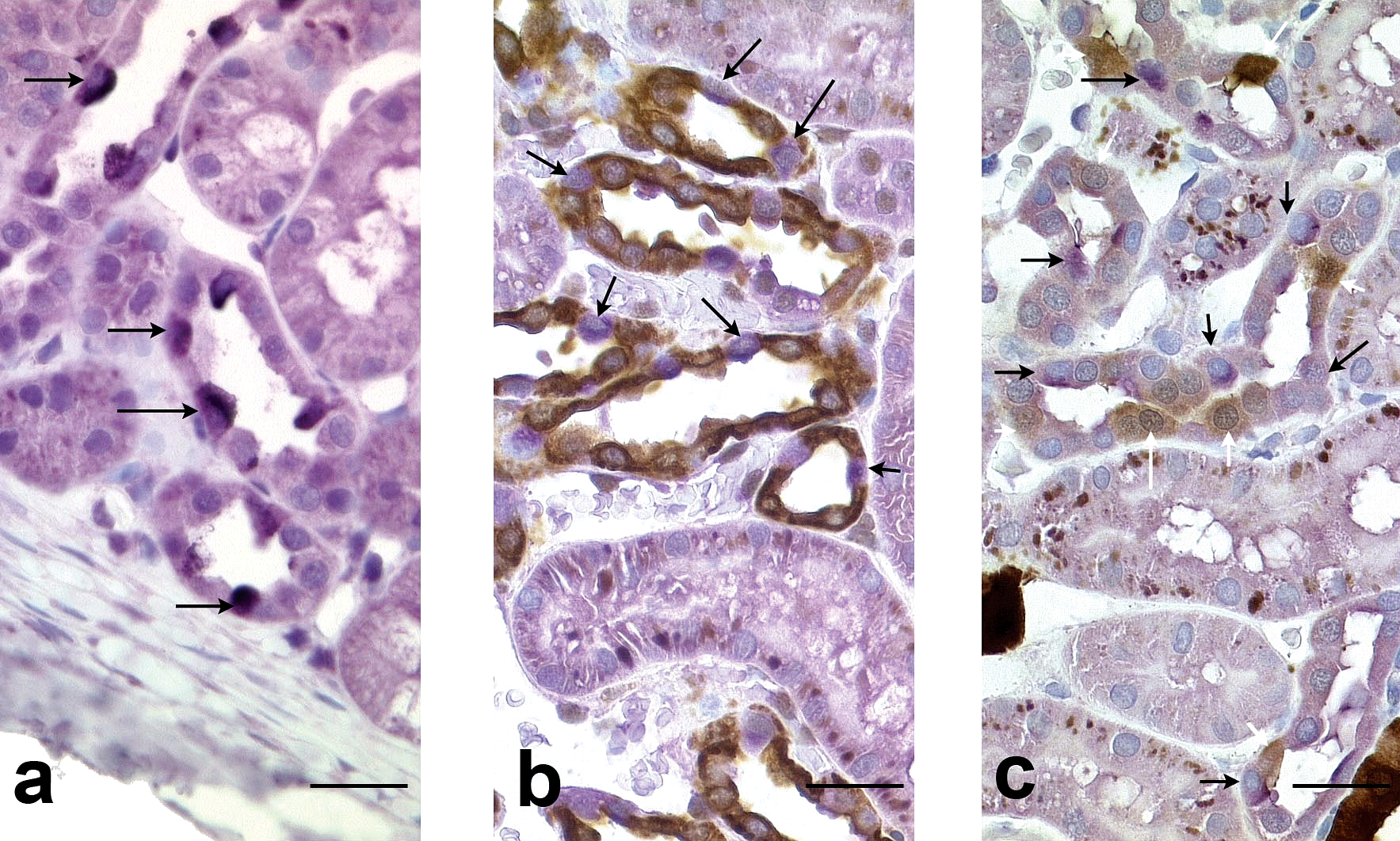
Kidney, cortex, dog. a: TH immunohistochemical staining. Note tubules with scattered positive cells (arrows). Scale bar: 50 µm. b: TH and AQP-2 double immunohistochemical staining. Note the tubules lined by positive AQP-2 cells (brown) with few scattered TH positive lining cells (purple, arrows). Scale bar: 50 µm. c: TH and CalD double immunohistochemical staining. Note the tubules lined by scattered CalD cells (brown, white arrows) and scattered TH positive (purple, black arrows) cells. Scale bar: 50 µm.
In rats, mice, and Cynomolgus monkeys, the staining pattern was almost identical to that in dogs and corresponded to the ascending limbs of the Henle’s loops. However, unlike in dogs, no scattered positive cells were observed in the superficial cortex in these species.
Alpha-GST
In dogs, alpha-GST positive cells were only present in cortical medullary rays (Figure 7a), where approximately half of the tubules, primarily those in longitudinal profile, stained positive for alpha-GST (Figure 7c). Positive tubules had a narrow lumen and were lined by large polygonal epithelial cells with a striated cytoplasm and a central nucleus. The staining was cytoplasmic and diffuse. The localization of the tubules and their morphology were compatible with the straight part of the proximal tubules. The epithelial cells of the urothelium also stained positively for alpha-GST.
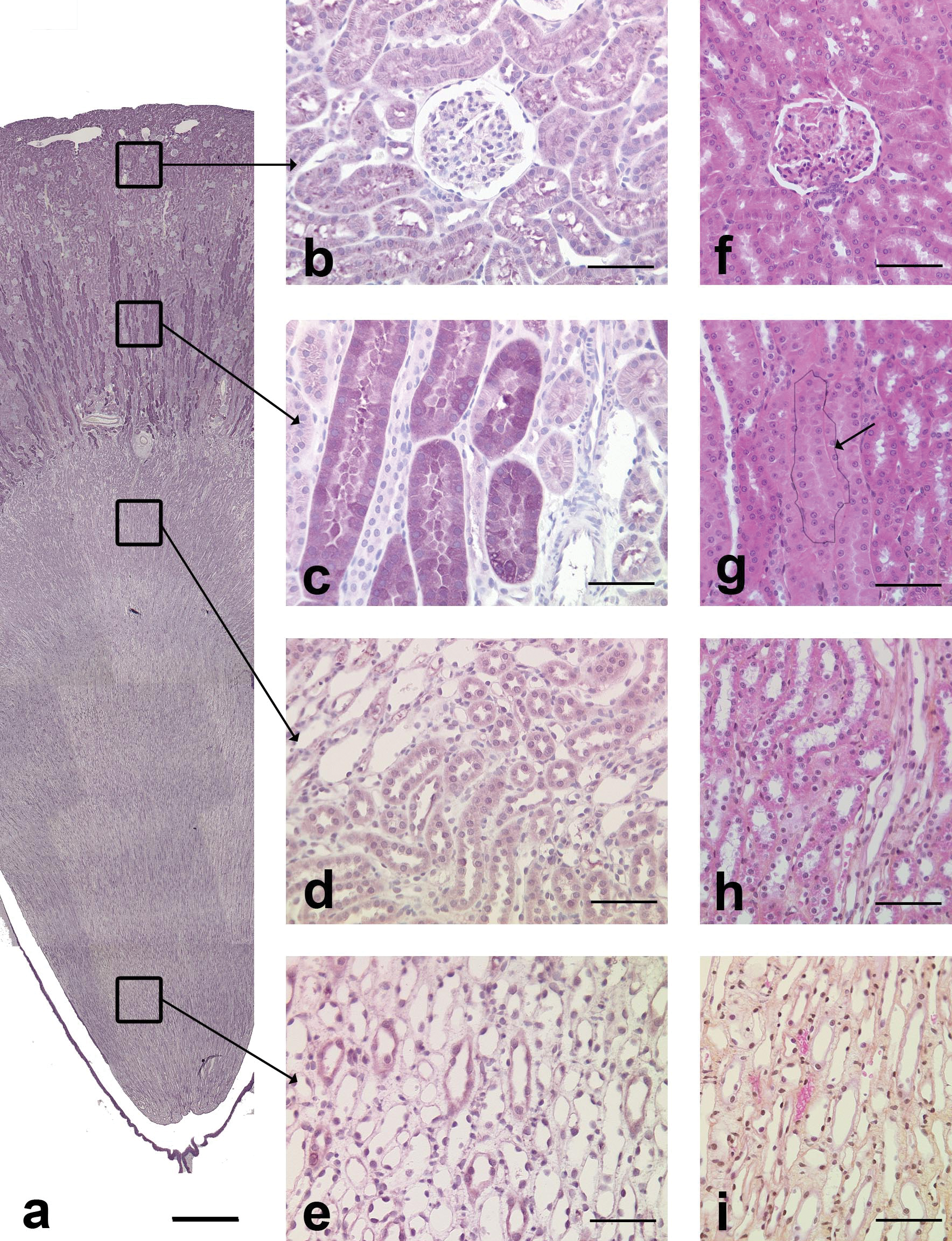
Alpha-GST immunohistochemical staining in the dog kidney. a: Overview of alpha-GST staining. Note that positive cells were only present in the cortex. Scale bar: 200 µm. b: Cortical labyrinth. Absence of staining. Scale bar: 70 µm. c: Medullary rays. The positive tubules in longitudinal section are compatible with straight part of proximal tubules. Scale bar: 70 µm. d: Outer medulla. Absence of staining. Scale bar: 70 µm. e: Inner medulla. Absence of staining. Scale bar: 70 µm. f: Cortical labyrinth, HES. Scale bar: 60 µm. g: Medullary rays, HES. Arrow: tubule with similar morphology as alpha-GST positive tubule, i.e., straight part of proximal tubule. Scale bar: 60 µm. h: Outer medulla, HES. Scale bar: 60 µm. i: Inner medulla, HES. Scale bar: 60 µm.
In rats, alpha-GST positive cells were observed in the cortex and in the outer stripe of the outer medulla. In the cortex, stained tubules were mainly in cross-sectional profile; whereas in the outer medulla, they were mainly in longitudinal profile. Positive tubules had similar morphology to the dog and by their location were compatible with proximal convoluted tubules and straight parts of proximal tubules. No cross-reactivity was observed in the mouse and Cynomolgus renal tissues with the anti-alpha-GST antibodies.
Multiple Immunostaining and Identification of Thin Ascending Limbs of Henle’s Loops in the Inner Medulla
The panel of antibodies used allowed identification of proximal tubules from the cortex to the medulla (AQP-1), of thick ascending limbs of the Henle’s loops (TH), of distal convoluted tubules (CalD), and of collecting ducts (AQP-2).
To identify the thin ascending limb of the Henle’s loop and to check the specificity of the antibodies, multiple and double AQP-1/AQP-2 stainings were performed.
In the medulla (Figure 8), collecting ducts were stained by AQP-2 and thin descending limbs of the Henle’s loop and endothelial cells of small blood vessels by AQP-1. Positive small blood vessels can be differentiated from thin limbs of the Henle’s loop in the medulla by their small diameter and by occasional red blood cells in the lumen. In the multiple (Figure 8a) or double staining (AQP-1/AQP-2) (Figure 8b), thin unstained tubules were observed in the inner medulla and more rarely in the inner stripe of the outer medulla. They were small, lined by a squamous epithelium delineating a narrow lumen. By their localization, morphology, and lack of AQP-1 staining, these tubules could be identified as thin ascending limbs of the Henle’s loops.
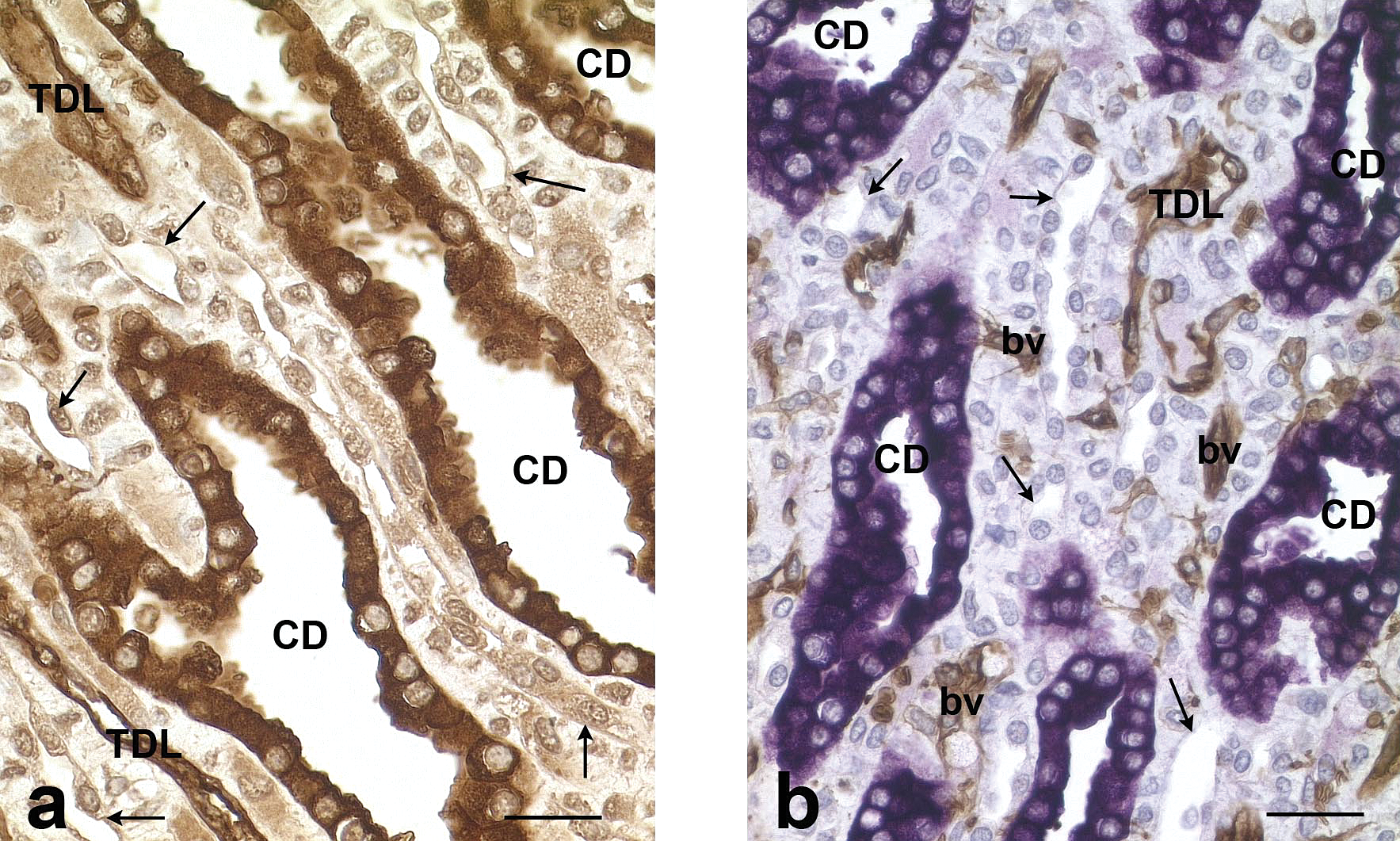
Rat, kidney, inner medulla. a: AQP-1, AQP-2, TH, and CalD multiple immunohistochemical staining. Note the unstained thin tubules (arrows) corresponding to thin ascending limbs of the Henle’s loops. CD: collecting duct. TDL: Thin descending limb of the Henle’s loop. Scale bar: 50 µm. b: AQP-1 and AQP-2 double immunohistochemical staining. Note the unstained thin tubules (arrows) corresponding to thin ascending limbs of the Henle’s loops. CD: collecting duct. TDL: Thin descending limb of the Henle’s loop. bv: blood vessels. Scale bar: 50 µm.
Discussion
In this study, five specific immunohistochemical markers were successfully used in our laboratory for identification of nephron segments in species commonly used in preclinical toxicity studies (i.e., the Beagle dogs, the SD rat, the CD1 mouse, and the Cynomolgus monkey) on paraffin sections: AQP-1, AQP-2, alpha-GST, TH, and CalD. Four of the five antibodies showed cross-reactivity between the studied species, while alpha-GST was species-specific. This is the first report of multispecies immunohistochemical identification of five nephron segments. Results are summarized in Table 1.
AQP-1, Alpha-GST, TH, CalD, and AQP-2 reactivity in dog, Cynomolgus monkey, rat, and mouse kidneys.

Identification of nephron segments in HE-stained sections is often challenging for the pathologist. In the renal cortex, proximal tubules characterized by an almost virtual lumen and lined by large polygonal cells with a central nucleus must be differentiated from distal convoluted tubules and ascending thick limb of the Henle’s loop, which are both lined by large polygonal cells and have respectively a narrow lumen and a medium-sized lumen, and from connecting and collecting ducts with their large lumen and oval to round lining cells. In the medulla, collecting ducts lined by oval cells must be differentiated from thin descending and ascending limb of the Henle’s loop, which are both lined by a simple squamous epithelium. Electron microscopy is currently the most reliable method to achieve this, but tissue processing is time-consuming and requires specific fixation, materials, and experience. In this context, immunohistochemistry may be a faster and simpler method to identify specific nephron segments in preclinical studies or in ongoing studies to discover and validate new biomarkers of renal toxicity. For this latest, it may allow, on one hand, to precisely localize proteins that are being developed as biomarkers (i.e., CalD, alpha-GST) and, on the other hand, to characterize the toxic target of compounds used in experimental studies to qualify new renal biomarkers.
Aquaporin-1 is a transmembrane protein that forms an aqueous pathway in the membrane (Schrier and Cadnapaphornchai 2003). It is present in the eye (photoreceptor cells and retinal pigmented epithelium), brain (choroid plexus), heart, pancreas, pain sensory cells, kidneys, and endothelial cells of the peripheral vasculature (Yool 2007). In the mammalian kidney, AQP-1 is reported to occur in proximal tubules and thin descending limbs of Henle’s loops (Bedford, Leader, and Walker 2003). In this study, we have confirmed these localizations in the dog, the rat, the mouse, and the Cynomolgus monkey. As we did not test any specific marker for the ascending thin limb of Henle’s loops, differentiation of these tubules from the AQP-1 positive descending thin limbs of Henle’s loops cannot be certain. However, in AQP-1/AQP-2 double staining, ascending thin limbs of Henle’s loops were identified in the inner medulla and, more rarely, in the inner stripe of the outer medulla (related to the occurrence of short loop nephrons in this kidney region) supporting the fact that AQP-1 was specific to descending thin limbs of Henle’s loops.
AQP-1 was also reported to be present in endothelial cells of the peripheral vasculature (Yool 2007). Positive staining was indeed observed in glomerular capillaries and in the vascular plexus in the outer medulla. AQP-1 is reported to be localized in the apical and basolateral cellular membranes (Bedford, Leader, and Walker 2003). In this study, diffuse staining was observed in the cytoplasm and was more prominent in the apical cell brush border and in the basolateral membranes confirming a membranous localization but associated with a cytoplasmic presence of the protein. In dogs, AQP-1 staining was consistently less intense than in rats, mice, and Cynomolgus monkeys despite longer incubation time (i.e., 12 h versus 1 h).
Tamm-Horsfall protein, also called uromodulin, is a glycosylphosphatidylinositol (GPI) protein: an anchor-linked protein with high carbohydrate content (Devuyst, Dahan, and Pirson 2005). Although TH is the most abundant protein excreted in urine at a rate of about 50 mg/day (Devuyst, Dahan, and Pirson 2005), its function remains unclear. It interferes with lymphocytes, competes with uroplakin receptors for the adhesion of type-1 fimbriated Escherichia coli, and is the major constituent of hyaline casts. It may play a role in binding to calcium and in prevention from urinary crystal formation (Devuyst, Dahan, and Pirson 2005). Knock-out mice for the Umod gene, encoding uromodulin, show an increased sensitivity to bacterial urinary infections and to calcium crystal formation in the collecting ducts of the medulla (Devuyst, Dahan, and Pirson 2005). TH is localized in the kidney, mainly in the thick ascending limb of Henle’s loop with extension to the early distal convoluted tubule in humans (Sikri et al. 1981), rats (Wong et al. 1998, Vekaria et al. 2006), and dogs (Raila, Neumann, and Schweigert 2003). In our study, TH was localized as reported in the ascending thick limb of the Henle’s loop in the outer medulla, in the medullary rays and in the cortex of dogs, rats, mice, and Cynomolgus monkeys, but no extension to early distal convoluted tubules was observed. The staining was cytoplasmic, although stronger along apical membranes as described by Devuyst, Dahan, and Pirson (2005). Very rare scattered TH-positive cells in a few tubules were observed in the inner medulla of dogs, probably due to TH protein deposits along the apical membrane of collecting duct cells as this protein is secreted in the urine. Scattered TH-positive cells were also observed in dogs in superficial cortical tubules that had a morphology that was different from the thick ascending limb of Henle’s loop tubules. Double staining showed that these tubules were mainly composed of AQP-2- and CalD-positive cells, suggesting that they were connecting/collecting ducts. The scattered TH positive cells were not stained by AQP-2 and CalD as are intercalated cells in rats, mice, and humans (Reilly and Ellison 2000; Bindels et al. 1991; Biner et al. 2002). By their localization and morphology and the absence of AQP-2 and CalD stainings, the positive TH cells in these latter tubules would be identified as intercalated cells.
Calbindin-D28K is a calcium-binding protein that plays a role in calcium homeostasis. It is present in avian intestines, mammalian and avian kidneys and pancreas, brain, bone, teeth, internal ear, and mammary gland of multiple species (Christakos et al. 2003; Lambers et al. 2006). In the kidney, CalD allows calcium intracellular transport from the apical membrane of epithelial cells to the basolateral membrane (Christakos et al. 2003).
Renal CalD expression is reported in distal convoluted tubules, connecting ducts, and outer medullary collecting duct in rats and mice (Bindels et al. 1991; Reilly and Ellison 2000; Rhoten and Christakos 1981; Roth et al. 1982) and in distal convoluted tubules, connecting tubules, and cortical collecting ducts in humans (Biner et al. 2002). The density of positive cells in tubules decreased from the distal convoluted tubule to the collecting duct in rats and humans (Roth et al. 1982).
In our study, positive tubules were primarily found in the cortex of dogs, mice, and rats and were identified as distal convoluted tubules and connecting/collecting ducts as reported. Scattered occasional collecting ducts were also stained with CalD in the medulla. Connecting and collecting ducts could be differentiated from distal convoluted tubules by the presence in the former of both stained and unstained cells identified as intercalated cells (Reilly and Ellison 2000; Bindels et al. 1991; Biner et al. 2002). These intercalated cells stained positive with TH in the dog kidney as mentioned previously. The staining in distal convoluted tubular cells, as in principal cells of cortical collecting ducts, was diffuse and cytoplasmic as previously reported (Roth et al. 1982). In Cynomolgus monkey, CalD staining was observed in distal convoluted tubules, connecting ducts, and cortical collecting ducts as in other species, but medullary collecting ducts also displayed a strong staining.
Aquaporin-2 is a kidney transmembrane protein that serves as a water channel and plays a central role in water reabsorption from collecting ducts (Schrier and Cadnapaphornchai 2003). At a cellular level, AQP-2 occurs in apical regions of the principal cells (Schrier and Cadnapaphornchai 2003). When vasopressin binds to a receptor in the basolateral membrane, there is activation of an adenyl cyclase-cyclic AMP-protein kinase that results in phosphorylation of AQP-2 and trafficking on the apical membrane.
The channel is localized in connecting and collecting ducts in rats and humans (Biner et al. 2002; Fenton et al. 2007) and is not reported in distal convoluted tubule (Reilly and Ellison 2000). As reported, AQP-2 positive tubules were compatible by their localization and morphology with collecting ducts. In our study, the staining was diffuse and cytoplasmic in all species, which did not confirm the membranous localization of AQP-2 reported by Schrier and Cadnapaphornchai (2003), but intracellular localization can be changed by fixation techniques (Bindels et al., 1991). Unstained cells were always present in AQP-2-positive tubules in the cortex and were also present in the medulla in Cynomolgus monkeys; these cells were identified as intercalated cells (Reilly and Ellison 2000; Bindels et al. 1991; Biner et al. 2002). The identification of the stained tubules was confirmed in Cynomolgus monkeys by the presence of multinucleated syncytial cells, which are described in the inner medullary collecting ducts in this species (Drevon-Gaillot et al. 2006).
Alpha-GST belongs to the large family of GST enzymes and is cytoplasmic (Rodilla et al. 1998). In humans, five types of alpha-GST are described: hGSTA1, A2, A3, A4, and A5 (Coles and Kadlubar 2005). hGSTA1, hGSTA2, and hGSTA4 are present in kidneys (Coles and Kadlubar 2005). There are numerous functions of GSTs; they include xenobiotic bioactivation, detoxification of oxidative stress products, aromatic amino acid catabolism, steroid hormone synthesis, eicosanoid synthesis and degradation, and intracellular signalling (Hayes, Flanagan, and Jowsey 2005). In human kidney, alpha-GST is localized in proximal convoluted tubules and sometimes in the thin descending limb of the Henle’s loop (Rodilla et al. 1998).
In our study, positive alpha-GST staining was localized in tubules with a morphology suggestive for the straight parts of proximal tubules (in medullary rays in dogs and in the outer stripe of the outer medulla in rats) and proximal convoluted tubules in rats. The staining observed was diffuse confirming its cytoplasmic localization (Rodilla et al. 1998). Furthermore, positive staining was observed in urothelial cells of the pelvis in dogs. The antibodies we used in dog and rat are species-specific; we did not observe cross-reactivity with mouse or Cynomolgus tissues.
Among the immunohistochemically detected antigens of this study, CalD, and alpha-GST gave promising results as biomarkers for early detection of nephrotoxicity. CalD shows to be a better indicator of gentamicine nephrotoxicity in rats than serum creatinine or blood urea nitrogen (Hoffmann et al. 2010) and was used as a biomarker for triple reuptake inhibitor nephrotoxicity in Cynomolgus monkey (Guha et al. 2011).
Detection of alpha-GST in urine is a sensitive method for detection of early kidney damage in humans (Sundberg et al. 1994) and in rats (Gautier et al. 2010). In dogs and rats, this antibody was developed by Argustus, to design ELISA tests to detect alpha-GST in urine (Kilty et al. 1998).
The knowledge of the microscopic renal localization of CalD and alpha-GST in these species may allow a better understanding of significance of their urinary levels and correlation with the severity of microscopic tubular lesions.
In conclusion, this panel of antibodies may be a helpful tool to identify nephron segments in normal kidney tissues or in tissues damaged by a nephrotoxic compound in preclinical studies. In addition, it can contribute to characterization and validation of novel renal biomarkers. As it concerned multiple species, it allows interspecies comparison and may help translational validation.
However, this panel has some limits. In case of nephrotoxicity, immunohistochemical expression of these antigens may be decreased or modified, and identification of the targeted nephron segment may become difficult. No antibody is dedicated to the identification of the thin ascending limb of the Henle’s loop, and AQP-1 staining does not permit to discriminate the different parts of the proximal tubule (convoluted part, straight part, and thin part). Alpha-GST antibody does not react with mouse and Cynomolgus monkey tissues.
Other markers of renal tubules could be included in this panel such as cyclooxygenase-2, which in humans was detected in cortical thick ascending limb of the Henle’s loop (medullary rays and macula densa), in some collecting ducts, and in endothelial cells (vasa recta and glomeruli) (Adegboyega and Ololade 2004). Similarly, cadherins that are specific for proximal tubule (N-cadherin) or distal tubules and collecting ducts (E-cadherin) in rats and humans (Prozialeck, Lamar, and Appelt 2004; Kirk et al. 2010) could be added in this panel.
Detection of exosomes is one of the new perspectives to monitor renal toxicity. These are late endocytic compartments known as multivesicular bodies secreted by various cell types and especially renal epithelial cells that contain membrane and cytosolic proteins (Sonoda et al. 2009).
