Abstract
Regulation of endoplasmic reticulum stress (ER) stress-induced apoptosis and nerve regeneration is a hopeful way for acute spinal cord injury (SCI). Sitagliptin (Sita) is one of dipeptidyl peptidase-4 (DPP-4) inhibitor, which is beneficial neurons damaged diseases. However, its protective mechanisms of avoiding nerve injury remain unclear. In this study, we further investigated the mechanism of the anti-apoptotic and neuroprotective effects of Sita in promoting locomotor recovery from SCI. In vivo results showed that Sita treatment reduced neural apoptosis caused by SCI. Moreover, Sita effectively attenuated the ER tress and associated apoptosis in rats with SCI. A striking feature was the occurrence of nerve fiber regeneration at the lesion site, which eventually led to significant locomotion recovery. In vitro results showed that the PC12 cell injury model induced by Thapsigargin (TG) also showed similar neuroprotective effects. Overall, sitagliptin showed potent neuroprotective effects by targeting the ER stress-induced apoptosis both in vivo and vitro, thus facilitating the regeneration of the injured spinal cord.
Introduction
Spinal cord injury (SCI) is a severe trauma that usually results in high morbidity rates and limited treatment options.1,2 The pathological process of traumatic SCI involves two stages: first, the primary injury as a mechanical insult causes hemorrhage and ischemia, and second, the secondary injury is a complex cascade of pathological changes, including ischemia, immune responses, apoptosis, and necrosis, resulting in continued damage to the spinal tissue.3,4 Although the spinal cord have a limited self-healing ability after SCI, avoiding neuronal apoptosis is helpful for neural regeneration. 5 However, the exact mechanisms underlying during SCI remain unclear.
Regulation of endoplasmic reticulum stress (ER) stress-induced apoptosis may facilitate functional recovery in SCI.6,7 The endoplasmic reticulum is an important to maintain cellular homeostasis. 8 Accumulating evidence has pointed out that ER tress contributes to degeneration and neuronal dysfunction of the central nervous system, such as brain injury 9 and SCI. 10 After SCI, excessive ER tress initiates an overstressed unfolded protein response, eventually leading to neuronal apoptosis. 11 Meanwhile, excessive ER tress increased the expression of glucose-regulated proteins (i.e.,GRP78/BiP), which in turn activated the unfolded protein response. 12 Thus, inhibiting the ER stress-induced neuronal apoptosis during secondary injury is a promising strategy for treating SCI.
Sitagliptin (Sita)is a selective dipeptidyl peptidase-4 (DPP-4) inhibitor used to treat adult type 2 diabetes (T2D) in more than 130 countries worldwide. 13 Apart from its antihyperglycemic effects, Sita may fight oxidative damage, inflammation, and apoptosis in cardiovascular, neurodegenerative, and neuronal diseases.14–16 Several studies have demonstrated that Sita can reduce cell death by restoring cellular calcium homeostasis and inhibiting ER tress. 17 It was also reported that Sita protected rats from cisplatin-induced neurotoxicity and exhibited neuroprotection in restoring locomotor and rotarod activities, thus, showing a potential application in promoting neuronal growth after injury. 18 Recently, increased attention has been paid to the protective role of DPP-4 inhibitors, including Sita, in nerve injury repair. 19 However, the potential mechanisms by which Sita promotes locomotor recovery after SCI, especially in attenuating ER stress-related apoptosis, have not yet been investigated.
In this study, we hypothesized that Sita exerts neuroprotective effects in rats with SCI by suppressing ER stress-related apoptosis during secondary injury and improving locomotor recovery after SCI in rats. Further, we investigated the mechanism underlying the inhibition of ER stress-induced apoptosis and neural regeneration to restore locomotion after SCI in vivo.
Materials and methods
Experimental animals and surgical procedures
Seventy female Sprague-Dawley rats (weighing 220–250 g) were purchased from Chinese Academy of Science (Shanghai, China). All animal studies were performed in accordance with the procedures and principles defined by the Laboratory Animal Ethics Committee of Wenzhou Medical University (WYDW2022-0602).
All rats were cared for 7 days before the study in an animal facility with free access to food and water. The SCI model was established according to the modified Allen’s method. 20 All rats were anesthetized with 1% sodium pentobarbital (40 mg/kg, i.p.). After anesthesia, skin preparation and precise positioning of the rats, laminectomy was performed. After laminectomy, the dura was cut approximately 15 mm using microsurgical scissors to expose the T10 spinal cord. Weight impact contusions (susceptible energy, 50 g/cm) were performed on the exposed posterior spinal cord surface using a 15 g weight dropped from a height of 3.0 cm. After the impact, successful SCI was verified by body trembling, swaying of the tail, and a fluttering retraction of the hind limbs and body. The presence of rats showing flaccid paralysis of the hind limbs and where edema can be observed on the dural surface is indicative of a successful model. Sham group rats were exposed to the T10 spinal cord by the same method, but without weight impact treatment. All rats received postoperative care, including washing the spinal cord with saline, suturing the incision, and three consecutive days of intraperitoneal injections of penicillin sodium (50 mg/kg) and bladder emptying twice-daily. Rats fulfilling these SCI model criteria were randomly divided into two groups, the SCI group and the Sita group (n = 8/group). Sham operated rats served as the control (n = 8/group). Sita group rats received daily treatment with Sita (10 mg/kg, i.p.), whereas sham and SCI groups rats were injected with an equivalent dose of saline.
Behavioral testing
The Beattie, Bresnahan, and Basso (BBB) locomotor scale was used to assess the recovery of hindlimb motor function as the previous report. 21 These evaluations were performed on 1, 3, 5, 7, 14 and 28 days after the injury to the time of euthanasia.
Histological experiments
At 4 weeks after surgery, the T6-T12 spinal cord tissues were collected and placed in paraformaldehyde for fixation. These tissues were embedded in paraffin and cut into 4-μm-thick sections. The sections were stained with hematoxylin-eosin (HE) and Nissl dyes. Finally, images were acquired using a Pannoramic 250/MIDI scanner (3D HITECH Slide, Hungary). The percent of preserved tissue were quantified by calculating the area of HE stained images with Image J. The number of VMN were counted by calculating the number of Nissl stained images with Image J.
PC12 cell culture and treatment
The PC12 cells were cultured in DMEM medium with 10% FBS and incubated at 37°C in incubator containing 5% CO2. PC12 cells were treated with a concentration gradient of TG (0, 0.5, 1, 2.5, 5, and 10 μm) or a concentration gradient of Sita (0, 1, 5, 25, 50, and 100 μm). Moreover, cells were treated with TG (5 μm) with or without a concentration gradient of Sita (10, 50, and 100 μm). After the cells were co-cultured in 96-well plates for 24 h, the cell viability was detected by Cell Counting Kit-8 (CCK-8, Dojindo, Japan). In brifly, CCK8 reagent was added with 10 μl per well. After incubated at 37°C for 2 h, the absorbance of each well was measured at 450 nm using a microplate reader (Bio-Rad 680). The results are presented as a percentage of the values measured for untreated control cells.
In addition, Exposure to TG was used as an in vitro model of SCI. PC12 cells were divided into four groups. Control group: no treatment; TG group: 5 μM TG treatment; TG + Sita group: 5 μM TG with 50 μM Sita treatment; Sita group: 50 μM Sita treatment. After 24 h, the immunostaining assays were performed. For western blot analysis, the PC12 cells were treated with 5 μM TG with or without Sita (0, 50, and 100 μM) for 24 h.
Immunofluorescent assays
For tissues, the sections were placed in citric acid buffer for heat-mediated antigen retrieval. For cells, after Remove the supernatant, the treated cells were wash three times with PBS. 4% paraformaldehyde was used to hold the cells for 15 min. Then these cell or tissue samples are blocked with normal horse serum/PBST (phosphate-buffered saline-Tween 20, 1:20) for 15 min. The Sample were incubated overnight at 4°C with primary antibody and wash 3 times using PBST. The following primary antibodies were used: anti-NeuN (1:500, ab177487, Abcam), Anti CHOP (1:500, MA1-250, Gibco), and Anti GRP78 (1:100, MA5-27687, Gibco). The sections were then incubated with the secondary antibody (Alexa Fluor 568, 1:800; Alexa Fluor 488, 1:400; Alexa Fluor 647, 1:400; Invitrogen) for 2 h at room temperature. Photographs were taken under a fluorescent microscope after washing using PBST. Image-Pro Plus software (Media Cybernetics LP, Maryland, USA) was used to quantify immunostaining-positive signals by selecting at least five sections or fields per sample at the lesion center. Fluorescent intensity was determined by the ImageJ software.
Western blot analysis
For tissues, at 4 weeks after surgery, a 1-cm T9-T10 spinal cord segment containing the injury site was collected. The tissue samples were ground using liquid nitrogen. For cells, the treated cells were collected with a cell scrape. Then these cell or tissue samples are lysed with radioimmunoprecipitation assay lysis buffer for 30 min. The samples were then separated on a sodium dodecyl sulfate-polyacrylamide gel and transferred to a polyvinylidene difluoride membrane. They were then blocked with bovine serum albumin solution for 1 h. The samples were incubated overnight at 4°C with primary antibodies against C-caspase 3, GRP78, PDI, Caspase 12 and CHOP (Cell Signaling Technology, Danvers, MA, USA). Then incubated with horseradish peroxidase-conjugated secondary antibodies for 2 h at room temperature. Final the bands visualized by autoradiography. All immunoblots were independently repeated at least three times. The quantifying protein band numbers was done using ImageJ software.
Statistical analysis
Statistical results are expressed as the mean ± standard deviation. Statistical significance was tested using repeated one-way analysis of variance (ANOVA), followed by a Tukey test for multiple comparisons using an SPSS software package (IBM, version19). Differences were considered statistically significant at *p < 0.05 and **p < 0.01.
Results
Functional recovery
BBB scores, inclined plane test, and footprint analysis were used to evaluate the locomotor functional recovery of rats during the 28-days post-surgery period. During the observation period of 14–28 days, Sita-treated rats showed continuous improvement in locomotion function. Following 28 days, Sita rats could carry weight on their hindlimbs (Figure 1(a) and (b)). At 28-days post-injury, the Sita group reached a level of 11.3 ± 1.25, while the SCI group merely achieved a poor BBB score of 6.12 ± 0.80 (p < 0.01, Figure 1(c)). The results of the inclined plane test shown in Figure 1(c) were similar to the BBB scores, and the angle values of the Sita group were significantly better than those of the SCI group at all time points after 2 weeks (p < 0.01, Figure 1(d)). In the footprint analysis (Figure 1(e)), Sita-treated rats showed relatively consistent hindlimb coordination and little toe dragging. In contrast, highly inconsistent footprints with extensive toe dragging were observed in rats with SCI. All these phenomena indicated that Sita administration significantly reversed the damage and improved locomotor function after SCI in rats. Sita improved locomotor recovery in SCI rats. (a-b) Displaying a profile view of rats at 4 weeks in the SCI, Sita and sham groups. Arrows showed the weight bearing action of right hindlimb in three groups. (c-d) The BBB walking scores and inclined plane test results in three groups. The Sita group showed significant improvement in locomotion compared with the SCI group for all time points from 2 weeks post injury. (e) Representative footprints at 28 days after operation; Statistically significant results were indicated as **p < 0.01.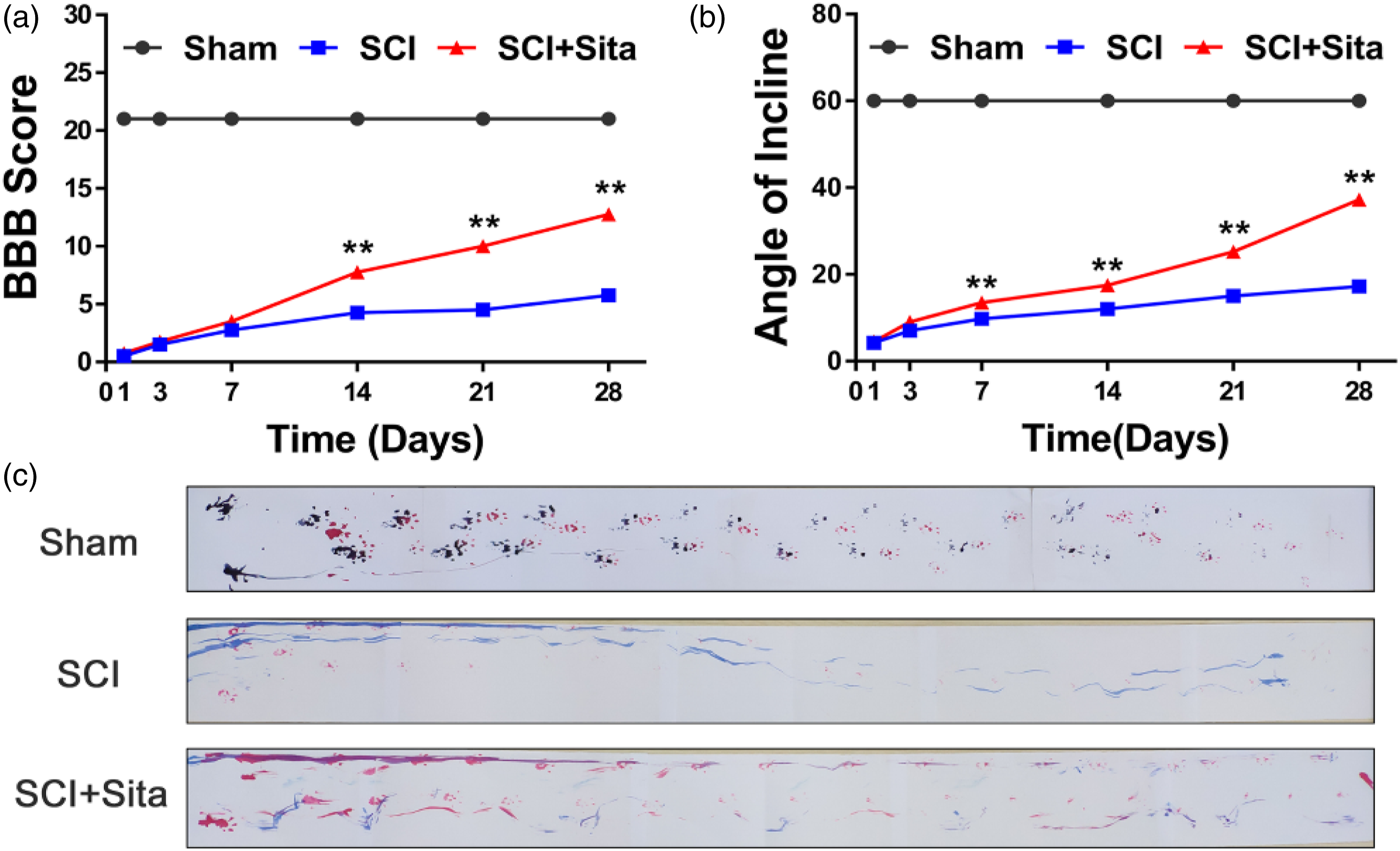
Tissue structural damage and loss of ventral motor neurons
To further demonstrate the protective effect of Sita after SCI, histological analyses of the spinal cord tissues were performed 28 days after SCI. Transverse sections of the spinal cords containing the injured sites were used to evaluate the lesion volume by HE staining. As shown in Figure 2(a), compared with the sham group, the SCI group exhibited severely damaged central gray and dorsal white matter. In contrast, remarkably attenuated structural damage was observed in the Sita-treated rats. The statistical data further confirmed that the cavity areas were least in the Sita group (1.63 ± 0.43 mm2; n = 12) compared with the SCI group (4.04 ± 0.83 mm2; n = 12) (Figure 2(b); p < 0.05). Meanwhile, the number of ventral motor neurons (VMNs) was counted through the Nissl staining images (Figure 2(c)). Compared to the SCI group, VMNs were significantly found in the Sita group (Figure 2(d); p < 0.05), which suggested that Sita treatment can maintain the VMNs in rats after SCI. These results indicated that Sita administration accelerated neuronal rehabilitation after SCI, which could facilitate the recovery of locomotor function in rats with SCI. Sita attenuated tissue structural damage, VMNs loss in SCI rats. (a) Representative H&E staining images of spinal cord tissue at 4 weeks after SCI; Images of HE (10×) and HE (20×) correspond to the square marked area in HE (4×); Boxes of the same color within the same group represent the same site; (b) Quantification data of the percentage of preserved tissue at 4 weeks; (c) Nissl staining images of the spinal cord tissue at 4 weeks; Red arrow: ventral motor neurons (VMNs); (d) Quantification analysis of the number of Nissl-stained VMNs. Statistically significant results were indicated as *p < 0.05, **p < 0.01. Scale bars: 200 μm (4×), 100 μm (10×) and 50 μm (20×).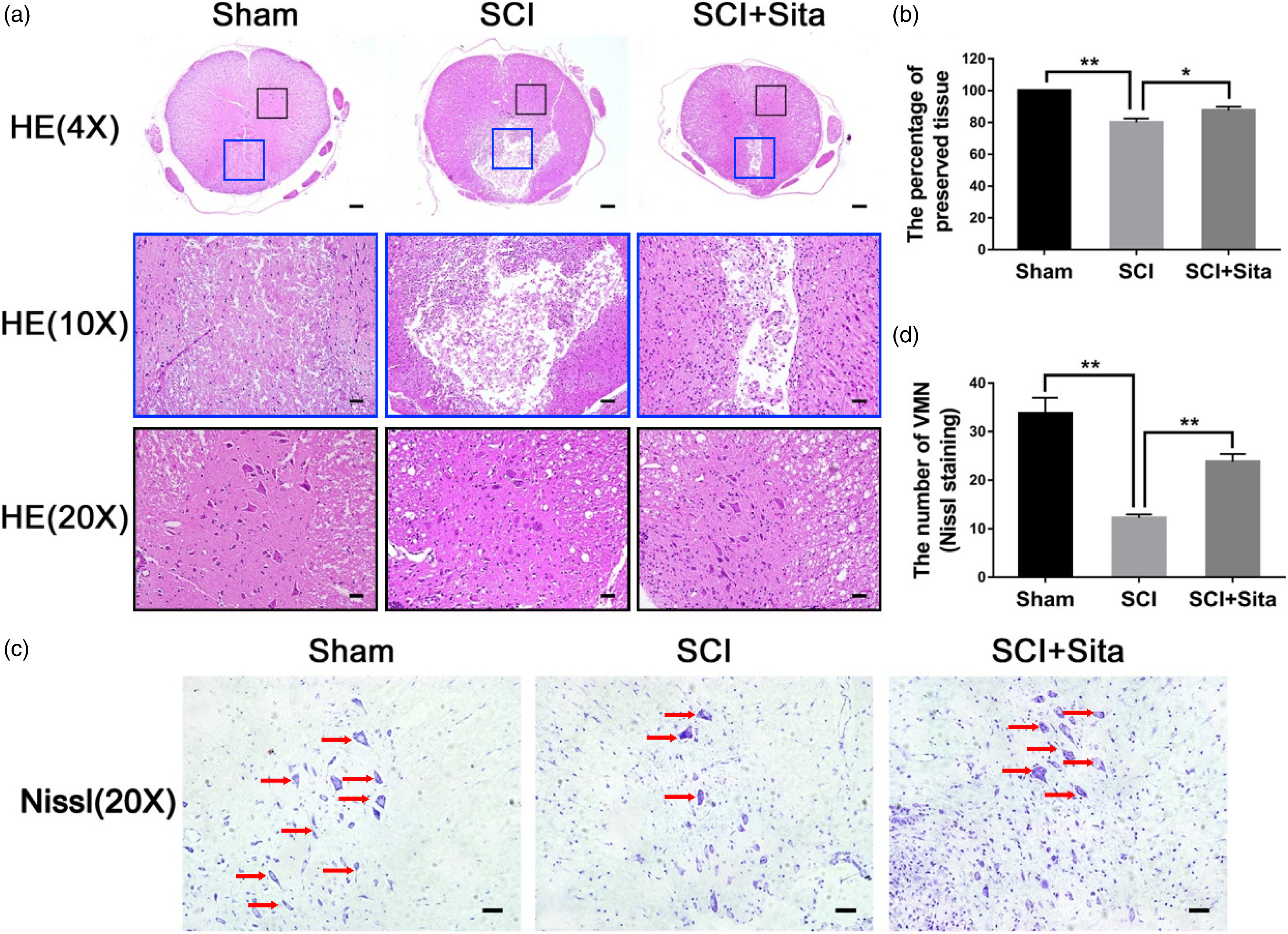
Neuronal apoptosis
Apoptosis-related proteins expression, such as C-caspase 3 and TUNEL assays, were performed to examine whether the neuronal protective effect of Sita is related to apoptotic regulation in rats with SCI. As shown in Figure 3(a), much stronger positive staining of C-caspase 3 existed in the SCI group when compared to the sham group, while the Sita group exhibited a much lower expression of C-caspase 3, which suggested that apoptosis can be attenuated by treatment with Sita after SCI. TUNEL assay was also used to evaluate the apoptotic levels of SCI. From Figure 3(b), we can see that SCI induced higher levels of apoptosis than the sham group. Similarly, the apoptosis level was reduced by Sita treatment. Moreover, western blotting results (Figure 3(c) and (d)) showed that the relative protein level of C-caspase 3 to Pro-caspase 3 was significantly reduced in the Sita group compared to that in the SCI group. These results suggest that Sita treatment can ameliorate neuronal apoptosis in the spinal cord tissue, which plays an important role in restoring locomotor function after SCI in rats. Sita alleviated neuronal apoptosis at 28 days in SCI rats. (a) Immunostaining images of NeuN (red) and Cleaved-caspase 3 (green) in spinal cord in different groups; (b) TUNEL staining results at 28 days after operation; (c) Western blot analysis showing Pro-caspase 3 and C-caspase 3 in three groups. (d) Bar chart showing the level of Pro-caspase 3 and C-caspase 3 protein in the three groups. Statistically significant results were indicated as **p < 0.01. Scale bar: 20 μm.
ER stress-related apoptotic protein expression
The levels of ER stress-related proteins, including GRP78, PDI, CHOP, and Pro-/C-caspase 12, were determined to investigate whether the anti-apoptotic effect of Sita was associated with the inhibition of ER tress in rats. Immunostaining showed that Sita remarkably reduced the levels of CHOP and GRP78 compared to those in the SCI group (Figure 4(a) and (b); p < 0.05). Meanwhile, western blot analysis also confirmed that the expression of ER stress-related proteins GRP78, CHOP, Pro-/C-caspase 12, and PDI was remarkably increased 3 days after the surgery. However, Sita treatment significantly reduced the expression levels (Figure 4(c)). Western blotting indicated that the levels of GRP78, CHOP, Pro-/C-caspase 12, and PDI proteins at the lesson sites in the Sita group were lower than those in the SCI group (Figure 4(d)–(g); p < 0.05). This indicates that ER tress caused by SCI could be reversed by Sita. Sita inhibited ER stress and related apoptotic protein expression at 28 days in SCI rats. (a) Immunofluorescence staining results of GRP78 (green) in spinal cord tissue; (b) Immunofluorescence staining results of CHOP (green) in spinal cord tissue; (c) Western blot results of GRP78, PDI, Caspase 12 and CHOP expression in spinal cord tissue. (d–g) Bar chart showing the level of GRP78, PDI, Caspase 12 and CHOP protein in the three groups. Statistically significant results were indicated as **p < 0.01. Scale bar: 20 μm.
Sita suppressed TG-induced ER tress in vitro
To verify the effect of Sita on inhibiting ER tress and related apoptosis in vitro, a cellular model was induced by a specific ER tress inducer of TG in PC12 cells. As shown in Figure 5(a), CCK-8 results showed that when TG concentration was 5 μM, the cell viability suddenly decreased to half value of control. Therefore, 5 μM of TG was selected for the following experiments. Besides, Sita treatment alone to PC12 cells for 24 h didn’t show any obvious cytotoxicity (Figure 5(b)). Moreover, Sita enhanced the cell viability significantly in a dose-dependent manner when cells were pre-treated by TG (Figure 5(c)). Exposure to 5 μM TG was used as an in vitro model of SCI. Immunofluorescence staining of GRP78 and CHOP showed that the significantly stronger positive expression of both GRP78 and CHOP caused by TG can be remarkably reversed by the treatment of Sita in PC12 cells (Figure 5(d) and (e)). The ER tress and apoptosis related proteins of GRP78, PDI and CHOP were also detected in TG-induced PC12 cells. The western blot results showed that GRP78 and PDI were remarkably decreased by Sita in TG-induced PC12 cells. The similar changing trend was observed in ER stress related apoptotic protein of CHOP in PC12 cells (Figure 5(f)–(i)). Sita suppressed TG-induced ER stress and related apoptosis in PC12 cells. (a) Cell viability results of PC12 cells treated with different doses of TG for 24 h; (b) Cell viability results of PC12 cells treated with different doses of Sita for 24 h; (c) Cell viability results with Sita and TG treatment; (d-e) Immunostaining results of GRP78 (green) and CHOP (green) in PC12 cell injury model. (f–i) Western blot results and quantitative data of GRP78, PDI and CHOP expression in different groups. Statistically significant results were indicated as *p < 0.05, **p < 0.01. Scale bar: 20 μm.
Discussion
Excessive neuronal apoptosis plays an important role in the cascade of secondary injury following SCI, as previous studies have noted, leading to locomotor paralysis in patients with SCI.3,22–24 However, no effective treatment has been developed for SCI. Therefore, there is an urgent need to further study the pathological mechanism and explore new treatment strategies to inhibit neuronal apoptosis and facilitate locomotor recovery in SCI.
Using a rat model of SCI, we explored the therapeutic effects of Sita on apoptosis and functional recovery. In comparison to the SCI group, Sita can increase the recovery of hindlimb behavior in rats. Besides, more numerous new nerve fibers, and less glial scars were found in the Sita group than in the SCI group. In addition, fewer tissue cavities and lower neuronal apoptosis were observed in the Sita group than in the SCI group, which is consistent with the results of a previous study. For example, Sita plays a protective role by activating glucagon-like peptide-1 receptor (GLP-1R) to alleviate neuronal apoptosis. 19 It has also been found that Sita attenuates hypoxia-induced apoptosis and autophagy in mesenchymal stem cells. 25 Therefore, an increased supply of Sita can promote survival and stimulate the migration of stem cells and eventually improve functional recovery.
In contrast to previous studies, we noticed that ER tress plays a vital role in neural cell death caused by secondary damage after SCI. Many studies have demonstrated that prolonged ER stress-induced apoptosis plays an important role in neuronal cell death in the central nervous system.26–28 Therefore, targeting the inhibition of ER stress-induced apoptosis in neural cells and neurons would be a promising therapeutic strategy for SCI. As previously reported, ER tress-associated apoptotic proteins, including CHOP, GRP78 and Caspase-12, are activated after SCI. 29 CHOP, which is a crucial pro-apoptotic factor during ER stress-induced apoptosis, may contribute to the activation of Caspase-12 and provide a potential link among the three unfolded protein response branches. 30 In addition, GRP78 expression is upregulated under ER tress. 31 In this study, immunostaining showed that the levels of CHOP and GRP78 were remarkably decreased in the Sita group compared with those in the SCI group. Western blot analysis indicated that the expression of GRP78, PDI, cleaved-caspase 12, and CHOP at the injured sites in the Sita group was lower than that in the SCI group (Figure 3(c)–(g)), suggesting that Sita can protect rats from ER tress and ER stress-induced apoptosis and further help the locomotor recovery of rats suffering from SCI. Moreover, Sita administration can inhibit ER tress and related apoptosis both in vitro and in vivo, which helped cell survival, the restoration of tissue damage and the locomotor recovery of the SCI rats. The results indicated that the protective effects of Sita are related to its ability to inhibit prolonged ER tress and stress-induced apoptosis after SCI.
Apart from its anti-hyperglycemic effects, previous studies have demonstrated that Sita exerts anti-inflammatory, anti-reactive oxygen species, and anti-apoptotic effects in various diseases.32–34 For example, Sita attenuated the secretion of pro-inflammatory cytokines in the myocardium of a rat model of heart failure and ameliorated atherosclerosis by regulating the AMPK and MAPK pathways in apolipoprotein-E knock out mice.
35
It has also been found that Sita decreases central nervous system damage by inhibiting oxidative stress and inflammatory reactions in mice with chronic nerve injury and showed neuroprotective effects in a mouse model of traumatic central nervous injury.
36
Nevertheless, treatment with exendin9-39, a GLP-1R inhibitor, remarkably reversed the protective effects of Sita.
37
In the present study, Sita administration significantly decreased the apoptosis of VMNs and improved locomotor functional recovery after SCI in rats. Meanwhile, significantly reduced C-caspase 3 expression was found in these segments of the injured spinal cord, which suggested that Sita administration alleviated the inflammatory reaction in rats with SCI. Hence, these results indicate that Sita exerts a potent neuroprotective effect in rats after SCI. The mechanism of sitagliptin in alleviating spinal cord injury by inhibiting endoplasmic reticulum stress is shown in Figure 6. Schematic diagram of Sitagliptin treatment of spinal cord injury through inhibition of endoplasmic reticulum stress.
Based on the results of this study, the administration of Sita significantly increased neuronal survival and facilitated the fast locomotor functional recovery of SCI. Further, we have provided combined evidence to demonstrate for the first time that Sita plays protective roles by regulating ER stress-induced neuronal apoptosis both in vitro and in vivo. Additionally, we have shown that Sita administration may alleviate inflammatory reactions during SCI recovery. In conclusion, our study provides a foundation for future clinical studies on Sita or ER tress inhibitors for the treatment of spinal cord injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by General research program of Medical Science and Technology Project of Zhejiang Province (No. 2022KY354).
