Abstract
Objective
The treatment of tuberculosis with isoniazid and rifampin is associated with hepatocellular damage. Therefore, the study was designed to evaluate the hepatoprotective potential of diosmin against hepatotoxic effect of isoniazid and rifampin in Wistar rats.
Methods
Hepatotoxicity was induced by administering isoniazid and rifampin (100 mg/kg), whereas diosmin was given as treatment control. Markers of liver function (ALT, AST, ALP and bilirubin), inflammatory cytokines (TNFα, IL-6 and IL-1β), apoptosis (caspase-3), oxidative stress parameters (LPO, GSH, CAT and SOD) and histological changes in liver were assessed in normal, hepatotoxic control and treatment groups.
Results
The administration of isoniazid and rifampin significantly increased markers of liver dysfunction (ALT, AST, ALP and bilirubin), cytokines (TNFα, IL-6 and IL-1β) and apoptosis (caspase-3). However, daily dosing of diosmin significantly reduced these markers of liver dysfunction, inflammatory cytokines and apoptosis to near normal levels. Additionally, markers of hepatocellular oxidative stress parameters were significantly altered as evident from increased LPO level and decreased endogenous antioxidants such as GSH, SOD and CAT in isoniazid-and rifampin-treated hepatotoxic group. It was observed that diosmin treatment reduced high levels of LPO and demonstrated significant improvement in antioxidant levels. Histological studies of liver also supported our biochemical findings, which are also manifested as diosmin treatment exhibited protection against hepatocellular degeneration and inflammation.
Conclusion
Results of the present study demonstrate hepatoprotective potential of diosmin against isoniazid-and rifampin-treated hepatotoxicity. Thus, we conclude that diosmin may be used along with anti-tubercular drugs (isoniazid and rifampin) in tuberculosis patients to overcome their hepatotoxic adverse effect.
Introduction
Tuberculosis (TB) is one of the serious communicable diseases, caused by the bacillus Mycobacterium tuberculosis. It is one of the leading infectious diseases, claiming 1.5 million lives every year Worldwide. 1 The incidence of TB always pose a serious health problem and is highest among poor people and Human Immunodeficiency Virus (HIV) infected patients. 2 It is a curable disease, if it is properly treated with anti-tubercular drugs. Isoniazid (INH) and rifampin (RIF) are commonly used in combination regimen as first-line anti-tubercular drugs due to their unique physicochemical properties and efficacy. Clinical uses of first line anti-tubercular drugs causing hepatotoxicity as the most common adverse effect, which is further enhanced when combinations of first line drugs are used.3,4 So, when TB patients are treated with anti-tubercular drugs for long time, there is increased risk of adverse reactions, causing irregularity or discontinuation of treatment as well as increased resistance. 5 The majority of the adverse reactions associated with anti-tubercular drugs are mild to moderate, but sometime there can be severe or even fatal toxic hepatocellular effects. 6 Liver injury associated with these anti-tubercular drugs has always been a significant clinical concern throughout the world.
The exact mechanism of liver injury associated with INH and RIF have not been investigated thoroughly, however it is reported that drug metabolites promote formation of reactive oxygen species (ROS) and augment cellular oxidative damage, inflammation and apoptosis of hepatocytes.7,8 Most of the anti-tubercular drugs are lipid soluble and need hepatic phase I and phase II metabolism into more water-soluble metabolites that can be easily excreted. In this context, INH is metabolized by N-acetyltransferase in the liver to toxic intermediates such as acetylhydrazine and hydrazine, which are subsequently metabolized by cytochrome P450 2E1 (CYP2E1) to toxic reactive metabolites.9,10 Similarly, RIF is also metabolized by microsomal enzymes (CYP450) into toxic intermediates and their accumulation results in enhanced production of ROS, which acts as an initiator of lipid peroxidation (LPO) and subsequent hepatocellular damage. 11 At the same time CYP2E1 is induced by RIF. The induction of CYP2E1 results in increased production of toxic metabolite of INH (hydrazine), which potentiates INH induced liver damage. 12 Previous research studies have suggested possible role of increased formation of toxic reactive metabolites of INH and RIF, which covalently bind to tissue and result in hepatocellular oxidative injury, activation of inflammatory signaling pathways and impairment of mitochondrial functions.13,14
Diosmin is an important flavonoid glycoside found amply in citrus fruits, vegetables, and tea. Flavonoids are plant components that protect the body against free radicals and other dangerous substances. Diosmin has important pharmacological activity such as anti-oxidant, anti-inflammatory, nephroprotective, anti-hyperglycemic, anti-mutagenic, anti-rheumatic, anti-allergic.15,16 A recent study has reported that diosmin has protective effect against cadmium induced liver damage. 17 It is also reported that diosmin is commonly used to treat hemorrhoids, varicose veins, venous insufficiency, leg ulcers, and other circulatory problems due to inadequate blood supply. 18
There is no appropriate drug available in the market to treat anti-tubercular drug induced hepatotoxicity. Protective effect of diosmin against anti-tubercular drug (INH and RIF) induced hepatotoxicity has not been investigated until now. Therefore, the present research study was carried out to evaluate the hepatoprotective potential of diosmin against hepatotoxic effects of isoniazid and rifampin in experimental animals.
Materials and methods
Experimental animals
Standing Committee for Scientific Research Ethics of Jazan University has approved all experimental methods to be carried out in this research study (ethical approval no. REC-42/01/138). Wistar rats of male sex weighing 180–200 g were procured from Medical Research Center (MRC), Jazan University. The animals were acclimatized for 12 h light: dark cycle, temperature, 25 ± 2°C in the animal house of MRC for one week and provided good nutrition and water. All dosing schedule and drug treatment were carried out in the MRC animal house.
Drugs and chemicals
All materials and drugs used in this experiment were carefully purchased from reliable sources in terms of quality and purity. And with this, drugs such as isoniazid, rifampin and diosmin were purchased from Sigma-Aldrich. Biochemical kits for liver function test (LFT) such as alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (TBI), direct bilirubin (DBI) and alkaline phosphatase (ALP) were purchased Randox, UK. In addition, ELISA kits for cytokines such as interleukin-6 (IL-6), tissue necrosis factor alpha (TNFα), interleukin 1 beta (IL-1β) and Caspase-3 were procured from MyBioSource, USA. Other chemicals used in the study were purchased from different companies.
Experimental design and treatment
40 Wistar rats in weight range (180–200 g) were assigned into 5 different groups, having equal numbers in each group (n = 8). The animals were evenly distributed into different groups. Isoniazid, rifampin and diosmin were dissolved in 1% carboxymethyl cellulose (CMC).
Group I → considered as normal control (NC), received saline (0.5 mL/kg, orally) daily for 3 weeks (21 days).
Group II → considered as hepatotoxic control (HC), received isoniazid + rifampin (100 mg/kg, orally) daily for 3 weeks (21 days).
Groups III → considered as diosmin treated, received diosmin (100 mg/kg, orally) and isoniazid + rifampin (100 mg/kg, orally) daily for 3 weeks (21 days).
Group IV → considered as diosmin treated, received higher dose of diosmin (200 mg/kg, orally) and isoniazid + rifampin (100 mg/kg, orally) daily for 3 weeks (21 days).
Finally, group V → considered as diosmin control, received higher dose of diosmin (200 mg/kg) for 3 weeks (21 days) as a dose-independent group to ensure that diosmin itself does not cause any hepatotoxicity. 19
Blood and tissue collection
Blood from each animal were collected in a serum separating vacutainer tube on the last day of drug treatment. All tubes filled with blood were centrifuged at 3000×g for 10 min and serum was separated. The serum was used for the estimation of ALT, AST, ALP, TBI, DBI, TNFα, IL-6, IL-1β and caspase −3. After blood withdrawal, all animals were sacrificed and their liver was removed, washed with normal saline (ice cold) and used for further investigations of LPO, glutathione reduced (GSH), catalase (CAT) and superoxide dismutase (SOD).
Liver homogenate preparation
The homogenate of liver was prepared in phosphate buffer and centrifuged at 800×g for 5 min. The supernatant (S1) was separated from the homogenate and used for the analysis of LPO and GSH. Post-mitochondrial supernatant (PMS) was obtained after centrifugation of remaining homogenate at 10,500×g for 15 min to analyze CAT and SOD.
Determination of markers for liver function test
The markers for liver function tests such as ALT, AST, bilirubin and ALP were determined in serum using diagnostic kits from Randox, UK. The absorbance of test sample and standard were used to calculate values for each parameter according the formula given in the kit.
Oxidative stress parameters assay
Method of Lowry et al., 20 was used to determine protein content in each sample. The assay for LPO was carried out according to Islam et al., 21 method, whereas the assay for GSH performed according to Jollow et al., 22 method. The methodology of Claiborne 23 was used to determine the activity of CAT, whereas the methodology of Marklund 24 was assayed to determine the activity of SOD.
Estimation of inflammatory cytokines marker
Inflammatory cytokines (TNFα, IL-6 and IL-1β) were estimated in the serum samples using simple ELISA method according to the procedure provided in the kit. The samples were added into wells of micro-plates pre-coated with specific cytokines (TNFα, IL-6 and IL-1β) antibodies. Then biotinylated detection antibodies and HRP conjugates were added into each well. It was then incubated for 5 min and washed to remove free components. Finally, TMB substrate reagent was added and yellow colour appeared in the samples were measured by ELISA plate reader at 450-nm wavelength. The concentrations of these cytokines in the test samples were calculated from the standard curve.
Estimation of caspase-3
Marker of apoptosis (caspase-3) was estimated as per the procedures provided by the manufacturer using ELISA sandwich method. Chromophore p-NA was produced after cleavage from the substrate of DEVD-p-NA. The light emitted from p-NA was measured spectrophotometrically at 405 nm using micro plate reader. The absorbance of p-NA from apoptotic samples and untreated control samples were used to determine the concentrations of caspase-3.
Histological examination of liver tissue
Liver was immediately removed after the sacrifice of each animal. It was then washed with normal saline and kept in formalin solution (10%v/v). The tissue was cut into appropriate size to make paraffin blocks, which were further used to make sections of 3–5 µm thickness using a microtome. The slides were stained with hematoxylin & eosin (H&E) and observed using microscope at ×40 magnification for histopathological examination.
Biostatistical analysis
Biostatistical analyses of the results were performed using GraphPad Prism software 8. The results obtained were analyzed by one-way ANOVA and Tukey’s post-hoc test to ascertain statistical significance. The results were compared among each other and presented as ± standard errors mean (SEM). The minimum criterion for the results deemed to be statistically significant was set at p < 0.05.
Result
Liver marker parameters
Effect of diosmin on ALT, AST and ALP level
Effect of diosmin treatment on liver marker enzymes (ALT, AST and ALP) in isoniazid and rifampin induced hepatotoxicity in Wistar rats.

## p < 0.001 → group II compared with group I; **p < 0.001 → group II compared with groups III & IV; nsP > 0.05 → group I compared with group V.
Effect of diosmin on bilirubin (total and direct) level
Effect of diosmin treatment on bilirubin (total and direct) in isoniazid and rifampin induced hepatotoxicity in Wistar rats.

## p < 0.001 → group II compared with group I; **p < 0.001 → group II compared with groups III & IV; nsP > 0.05 → group I compared with group V.
Effect of diosmin on histological assessment of liver
The liver of the NC rats (group I) was examined histologically and indicated normal hepatocytes without any inflammatory and necrosis changes (Figure 1A). However, hepatocytes degeneration, inflammation and necrosis were found in the liver of hepatotoxic control (Figure 1B). Lower dose of diosmin (100 mg/kg) with INH and RIF (Figure 1C) showed mild degenerative changes with slight decrease in hepatocytes, inflammation and necrosis. However, higher dose of diosmin (200 mg/kg) with INH and RIF (Figure 1D), p]rotected the hepatocytes to near normal morphology without any inflammatory and necrosis changes. Moreover, diosmin control group (200 mg/kg) showed normal morphology of hepatocytes without any inflammation and necrosis (Figure 1E). Photomicrograph of normal control, hepatotoxic control and diosmin treated groups are shown in fig.1. The liver of the normal control (1A) and diosmin control (1E), showed normal hepatocytes without any inflammatory and necrosis changes. INH and RIF treated hepatotoxic control (1B), showed hepatocytes degeneration, inflammation and necrosis. Lower dose of diosmin (100 mg/kg) with INH and RIF (1C) showed mild degenerative changes in hepatocytes with inflammation and necrosis. However, higher dose of diosmin (200 mg/kg) with INH and RIF (1D), protected the hepatocytes to near normal without any inflammatory and necrosis changes.
Oxidative stress parameters of liver
Effect of diosmin on LPO
Levels in the liver tissue homogenate of normal, hepatotoxic control and diosmin treated groups are shown in Figure 2. Hepatotoxic control (group II) displayed significantly (p < 0.001) high level of LPO compared to normal control, indicating substantial evidence of INH and RIF induced hepatocellular oxidative damage. However, LPO levels were reduced significantly (p < 0.001) after 21 days of diosmin (100 & 200 mg/kg) treatment in comparison to hepatotoxic control (group II). No significant difference in the values of LPO was observed in diosmin control group (200 mg/kg) compared to NC. Effect of diosmin on LPO levels against hepatotoxic effect of INH and RIF in Wistar rats. ##p < 0.001 versus group I (normal control), **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control).
Effect of diosmin on GSH
Hepatotoxic control (group II) demonstrated a significant (p < 0.001) decrease in GSH contents compared to normal control, indicating depletion in the GSH value which protects hepatocytes from deleterious effects of INH and RIF (Figure 3). However, GSH contents were increased significantly (p < 0.001) after 21 days of diosmin (100 & 200 mg/kg) treatment as compared to hepatotoxic control (group II). No significant difference in the values of GSH was observed in diosmin control group (200 mg/kg) compared to NC. Effect of diosmin treatment on GSH contents against hepatotoxic effect of INH and RIF in Wistar rats. ##p < 0.001 versus group I (normal control), **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control).
Effect of diosmin on CAT and SOD
Enzymatic activities of CAT and SOD in homogenate of normal, hepatotoxic control and diosmin treated groups are shown in Figures 4 and 5. Hepatotoxic control (group II) demonstrated a significant reduction in the CAT and SOD activities compared to NC (p < 0.001). The administration of diosmin (100 & 200 mg/kg) demonstrated significant increase in the activities of these antioxidant enzymes compared to hepatotoxic control (p < 0.01–p < 0.001). We observed no significant difference in the activities of CAT and SOD in diosmin control group (200 mg/kg) when compared to NC. Effect of diosmin on CAT levels against hepatotoxic effect of INH and RIF in Wistar rats. ##p < 0.001 versus group I (normal control), *p < 0.01 - **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control). Effect of diosmin on SOD levels against hepatotoxic effect of INH and RIF in Wistar rats. ##p < 0.001 versus group I (normal control), *p < 0.01 - **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control).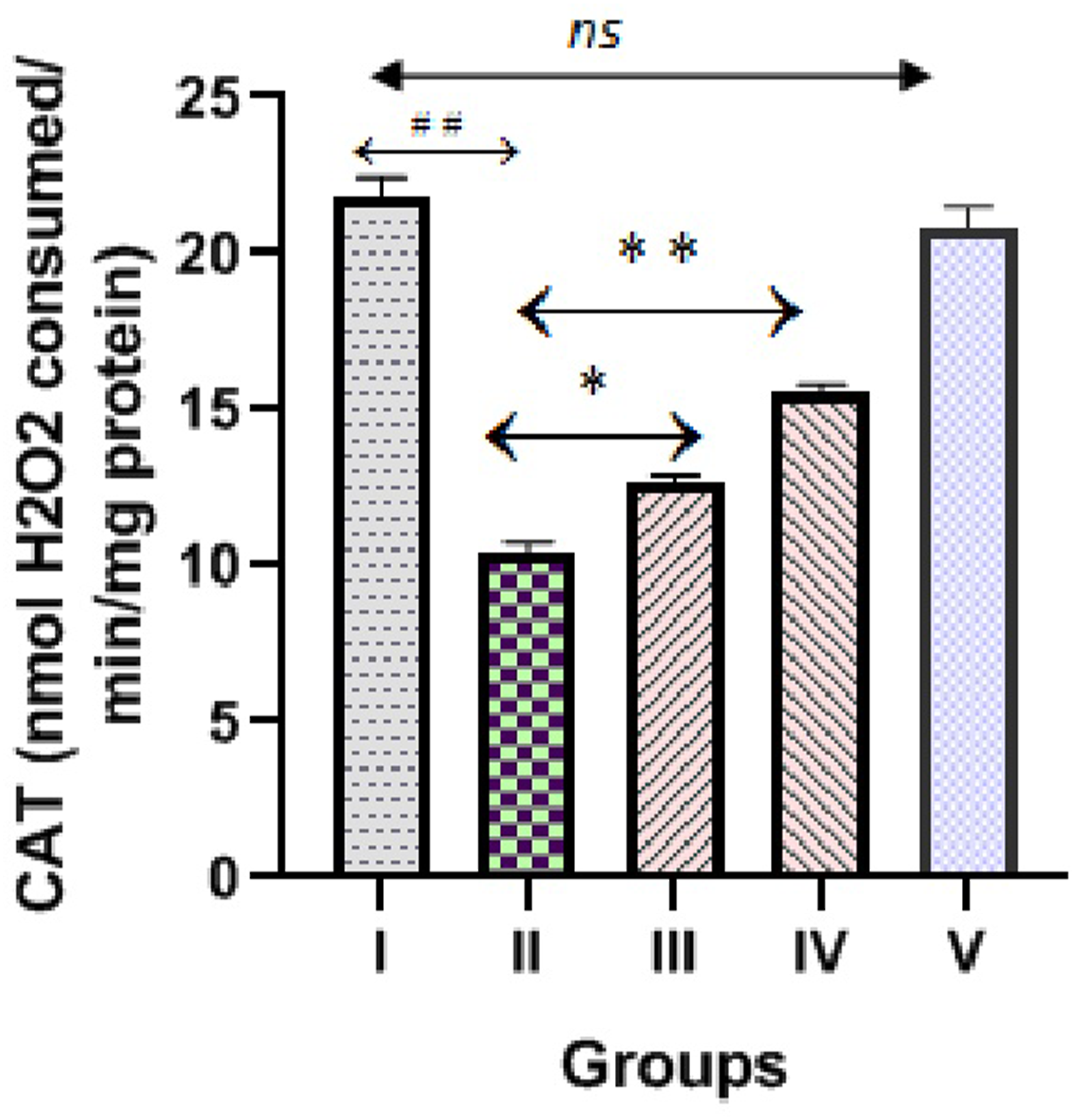

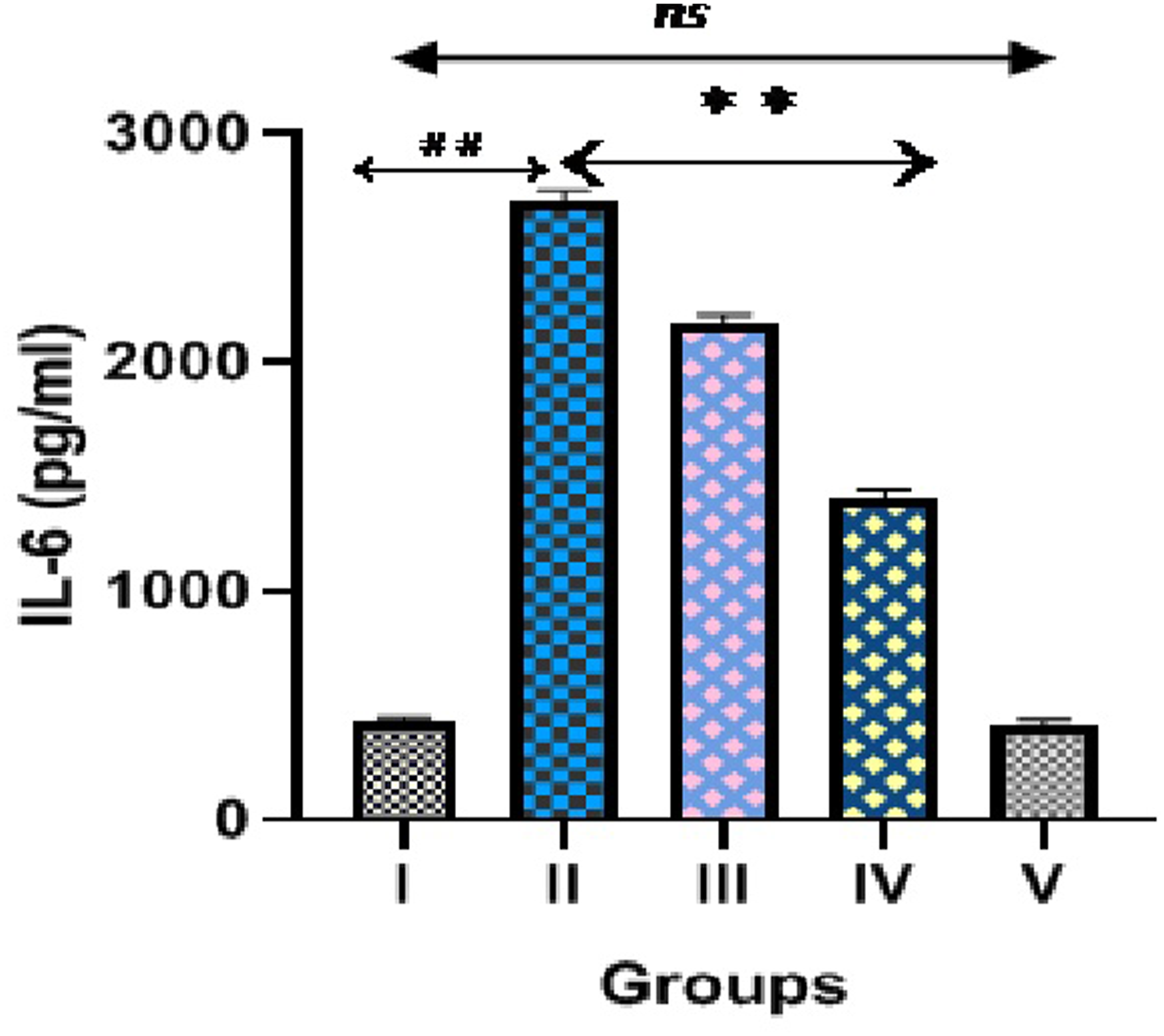
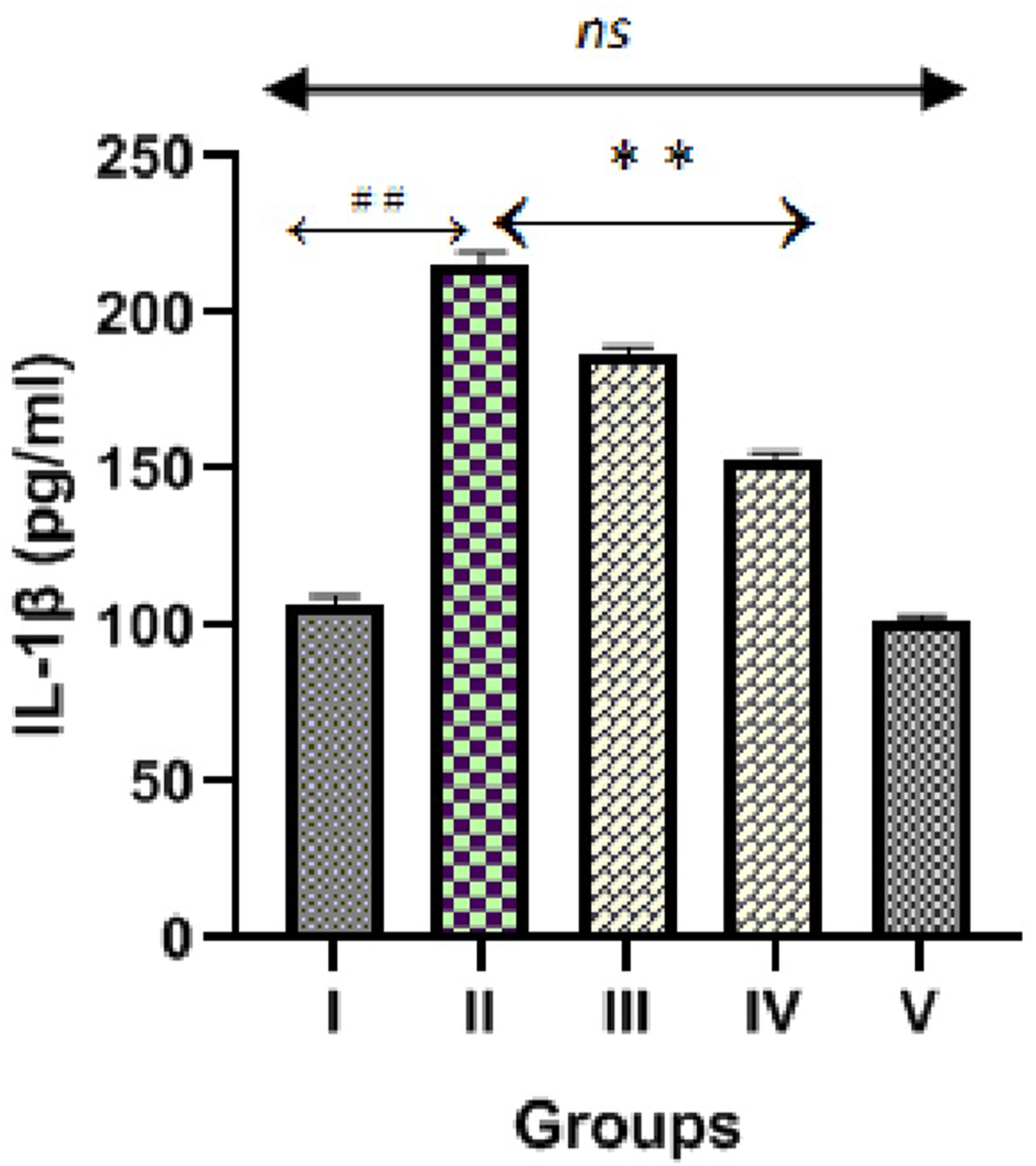
Effect of diosmin on inflammatory cytokines marker
Figures 6, 7 and 8 demonstrated markers of inflammation and cytokines (TNFα, IL-6 and IL-1β) in normal, hepatotoxic control and diosmin treated groups. INH and RIF treated hepatotoxic control (group II) significantly (p < 0.001) increased cytokines levels (TNFα, IL-6 and IL-1β) compared to normal control. We observed significant decrease in these inflammatory cytokines levels after administration of diosmin (100 & 200 mg/kg) compared to hepatotoxic control (p < 0.001), which clearly indicates that diosmin blocks the release of inflammatory cytokines. We observed no significant difference in cytokines levels in diosmin control group (200 mg/kg) compared to NC. Effect of diosmin on TNFα levels in isoniazid and rifampin induced hepatotoxicity in Wistar rats. ##p < 0.001 versus group I (normal control), **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control). Effect of diosmin on IL-6 levels in isoniazid and rifampin induced hepatotoxicity in Wistar rats. ##p < 0.001 versus group I (normal control), **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control). Effect of diosmin on IL-1β levels in isoniazid and rifampin induced hepatotoxicity in Wistar rats. ##p < 0.001 versus group I (normal control), **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control).
Effect of diosmin on apoptosis marker
Hepatotoxic control (group II) demonstrated a significant (p < 0.001) augmentation in apoptosis marker (caspase-3) level compared to NC, which is displayed as Figure 9. However, the level of caspase-3 was significantly (p < 0.001) decreased after 21 days treatment of diosmin (100 & 200 mg/kg) as compared to hepatotoxic control (group II). We observed no significant difference in caspase-3 level in diosmin control group (200 mg/kg) compared to NC. Effect of diosmin on caspase-3 level in isoniazid and rifampin induced hepatotoxicity in Wistar rats. ##p < 0.001 versus group I (normal control), **p < 0.001 versus group II (hepatotoxic control), nsp > 0.05 versus group I (normal control).
Discussion
One of the important things in dealing with drugs is to study the nature of their effect on humans in the short and long term, which allows researchers and scientists to know the side effects of these drugs, which may contribute significantly to the occurrence of complications or serious diseases in the future. Tuberculosis is one of the leading diseases that require combination of drugs for their treatment and used for several months. 25 First line anti-tubercular drug (INH and RIF) induced hepatotoxicity has become one of the frequent adverse effects in the recent period and result in serious damage to the liver, gallbladder and hepatocytes. 26 Previous studies have confirmed that taking combination of isoniazid and rifampin for a long period of time definitely results in liver injury. 4 However, anti-tubercular drug regimen associated adverse drug reactions may be responsible for the discontinuation of treatment. 27
Anti-tubercular drug induced liver impairment has been reported previously by many researchers and is difficult to distinguish from viral hepatic disorder.28,29 In addition, the prevalence of hepatotoxicity is higher in patients receiving combination of INH and RIF as reported in previous clinical and experimental studies. 30 The synergistic potential of RIF on INH associated hepatotoxicity is possibly because of the induction of enzymes involved in drug metabolism. In this study, we observed about 2-3 folds increase in markers of liver dysfunction such as ALT, AST, ALP and bilirubin after INH and RIF treatment. The increased level of liver markers in serum is possibly due to their leakage from liver cells during INH and RIF associated hepatocellular damage. The administration of diosmin demonstrated significant protection against INH and RIF induced liver damage by attenuating high levels of ALT, AST, ALP and bilirubin in dose dependent manner. Our results substantiate previous research studies conducted on similar animal model to investigate the mechanism of anti-tubercular drugs associated hepatotoxicity and its protection from various synthetic and natural products.31,32
Liver enzymes are commonly thought to be the serum biomarker of INH and RIF induced hepatocellular injury, which is also evident from histopathological studies in our present research. The liver of the normal control group (1A) and diosmin control group (1E) were examined histologically and indicated normal hepatocytes without any inflammatory and necrosis changes. However, hepatotoxic control (1B) demonstrated degeneration in hepatocytes with vacuolation, inflammation and necrosis, which are also substantiated by previous researchers.7,8 The present study revealed that diosmin at lower dose (100 mg/kg) with INH and RIF (1C) demonstrated protection of liver as apparent from mild degenerative changes in hepatocytes with inflammation and necrosis. However, higher dose of diosmin (200 mg/kg) demonstrated liver protection (1D) which is evident as normal morphology of hepatocytes without any inflammatory and necrosis changes.
The mechanism behind INH and RIF induced hepatocellular damage is multifaceted, including generation of ROS, hepatocellular oxidative damage, mitochondrial toxicity and production of toxic reactive metabolite. 33 The ROS which are overproduced after the administration of INH and RIF, impair oxidant - antioxidant system, damage cell membrane lipids and react/or bind covalently with cellular components resulting in hepatocytes damage. 34 Studies in the past have documented that LPO is an important marker of cellular oxidative stress that damages cell membrane lipids through generation of ROS which subsequently increases malondialdehyde (MDA) level, a metabolic product of LPO.35,36 However, experimental animals treated with anti-tubercular drugs have shown strong correlation between oxidative stress and liver toxicity. 37 In this study, we observed disruption in the oxidant – antioxidant system after INH and RIF administration, which is apparent from high levels of hepatic MDA, depletion in GSH contents and antioxidant enzymes (CAT and SOD). The potential therapeutic strategies are mainly focused on protecting liver cell from oxidative damage by reducing ROS generation and maintaining endogenous antioxidants. Catalase is an enzyme that catalyzes hydrogen peroxide to form water. SOD is an enzyme that breaks down superoxide radicals into hydrogen peroxide and molecular oxygen. Depleted antioxidant enzymes reduce clearance of hydrogen peroxide and facilitate production of toxic hydroxyl radicals leading to cellular oxidative damage. Several previous studies reported that endogenous antioxidant such as GSH, CAT and SOD are directly involved in the elimination of ROS.31,35 The administration of diosmin significantly reduced hepatic MDA levels, increased GSH contents and augmented activities of CAT and SOD. These observations suggest that diosmin protects INH and RIF induced hepatocellular oxidative damage by scavenging ROS, maintaining MDA, GSH, CAT and SOD levels.
In addition to cellular oxidative stress, inflammatory cytokines and apoptotic pathways activation also plays vital role in anti-tubercular drug associated liver injury. Cytokines are a type of protein that are linked to inflammation, immunological reactivity, tissue injury and organ dysfunction. Apoptosis is a programmed cell death carried out by the enzymatic pathway and involves caspases (caspase 3 & 9). These caspases are involved in the initiation of apoptosis, where it is completed with the help of downstream signaling pro-apoptotic (Bax) and anti-apoptotic (Bcl-2) protein. Previous studies reported over-expression of TNFα, IL-6, IL-1β and caspases in both experimental animals as well as in patients with isoniazid induced liver injury. 33 According to previous research patients with increased level of ALT also demonstrated an increase in inflammatory cytokines (IL-10, TNF‐α, IL‐1β and IL‐6), also implicated in the pathogenesis of liver injury. 38 In this study, we observed high levels of TNFα, IL-6, IL-1β and capase-3 in INH and RIF treated rats, suggesting involvement of inflammatory cytokines and apoptotic pathways that damage liver cells. Interestingly, diosmin treatment significantly attenuated the levels of these inflammatory cytokines and apoptotic pathway and thus demonstrating its protective effect in INH and RIF induced hepatotoxicity.
Conclusion
The results of this study exhibited hepatoprotective potential of diosmin against INH and RIF induced hepatotoxicity. This is possibly achieved by reducing liver markers, scavenging free radicals, up-regulating cellular antioxidant, alleviating inflammatory cytokines, apoptosis and restoring structural changes in the liver. Based on our findings, we suggest that diosmin may be used as a dietetic supplement to the patients on first line anti-tubercular (INH and RIF) drugs and are at risk of hepatotoxicity. However, further clinical investigations are required to support this research study.
Footnotes
Acknowledgements
The authors extend their gratitude to the Deputyship for Research and Innovation, Ministry of Education, Saudi Arabia for funding this research work through the project no – RUP20-01.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deputyship for Research and Innovation, Ministry of Education in Saudi Arabia (RUP20-01).
