Abstract
Clerodane diterpene, a class of bicyclic diterpenoids, is found in hundreds of plant species. 16-hydroxycleroda-3,13-dien-15,16-olide (CD) can be isolated from the plant Polyalthia longifolia and has been applied against oral cancer and glioma by xenograft model. In this study, we aim to explore its antitumour action by examining its histone deacetylase (HDAC) activity and integrin-associated intracellular signalling pathway on T24 human bladder cancer (BC) cells. Our results revealed that CD-inhibited colony formation, HDAC activity, HDAC (1, 2 and 3) mRNA and cell spreading on fibronectin-coated surfaces in a concentration-dependent manner. Furthermore, decreased cFLIP and increased caspase-8 cleavage accompanied CD-induced cell death. At non-toxic concentrations, CD blocked the migration and invasion of T24 cells. CD hindered migration and invasion by the downregulation of fibronectin, integrin α5β1, β-catenin, FAK, vinculin and Rho A, as well as by reduction of phosphorylated glycogen synthase kinase 3β (pGSK3β), pSrc, pstat3 and pNFκB. We observed that the MMP9 gene was closely linked with prognosis of patients with bladder cancer. MMP9 protein levels and activity were largely attenuated by CD in a concentration-dependent manner. In conclusion, CD-induced caspase-8-dependent apoptosis and suppressed migration and invasion by blocking several intracellular signalling pathways, including downregulation of HDAC activity and integrin–FAK and MMP9 pathways.
Introduction
Bladder cancer (BC) is the most common and recurrent neoplasm in the urinary system. 1 According to GLOBOCAN2020 database, 573,278 new cases of BC will be diagnosed, causing bladder cancer as the 10th diagnosed cancer in the world. 2 Despite the current advances in treatment, the 5-years survival rate of patients with advanced stages of BC remains poor and unsatisfactory. 3 More than half of the newly diagnosed patients with low-grade BC will suffer from tumour recurrence and progression, despite local therapy. 4 However, options for approved targeted therapies for BC are currently limited. 4 Further investigations on new treatments for the inhibition of BC progression are urgently needed. Recently, multi-target kinases inhibitors have offered relevant insights.5-7 The approach has become a novel strategy for cancer management. Two main pathways are involved in BC. Low-grade BC most often involves the phosphoinositide 3-kinase (PI3K)/Akt/ mammalian target of rapamycin (mTOR) and mitogen activated protein kinase kinase (MEK)/extracellular signal-regulated kinase (ERK) pathways. 8 As for high-grade BC, aberrant expressed genes are involved in cell adhesion molecules (N-cadherin and E-cadherin), matrix metalloproteinase (MMP) and angiogenesis [vascular endothelial growth factor] in comparison with normal tissues. 9
16-Hydroxycleroda-3,13-dien-15,16-olide (CD), a type of clerodane diterpene, is a small-molecule compound isolated from the leaves of Polyalthia longifolia Benth. & Hook. f. var. pendula. 10 It has been demonstrated that CD presents various pharmacological activities against tumour and cancer growth, inflammation and infection. 10 Besides, CD has been studied for its activity on lipid-lowering,11,12 antihyperglycaemic, 13 anticancer,14,15 antileishmanial, 16 and colitis inhibition 17 by using in vivo studies without noticeable adverse effects. In addition, at a concentration of 20 μM, CD is not harmful to human normal bronchial epithelial cells 15 and 3T3-L1 preadipocytes. 12 CD induces tumour cell death through intrinsic and extrinsic apoptotic pathways, autophagy, and cell-cycle arrest. 10 Moreover, CD impedes colon and renal cancer cell migration and invasion through the focal adhesion kinase (FAK) and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) pathways.18,19 Nevertheless, the detailed mechanism of CD on inhibition of tumorigenesis in human BC cells remains largely unknown. This study aimed to clarify the mechanisms involved in CD-induced apoptosis and suppression of migration and invasion in T24 BC cells by determining histone deacetylase (HDAC) activity, integrin–FAK signalling pathway involvement and MMP9 expression and activity.
Materials and Methods
Reagents
List of primary antibodies.

The HDAC activity assay kit and trichostatin A were obtained from Biovision Inc. (Burlingame, CA, USA). HDAC primers were synthesised by GeneDireX Inc. (Taoyuan, Taiwan). McCoy’s 5A medium, foetal bovine serum (FBS) and penicillin–streptomycin were purchased from Gibco (Grand Island, NY, USA). Matrigel® was obtained from BD Biosciences (San Jose, CA, USA). The donkey anti-mouse- and goat anti-rabbit–conjugated horseradish (HRP) peroxidase IgG secondary antibodies were purchased from GeneTex (Irvine, SC, USA). A Bradford protein assay kit was purchased from Bio-Rad Laboratories, Inc. (Hercules, CA, USA). Transwell inserts (8 μm) were obtained from SPL Life Sciences (Pocheon-si, South Korea).
Cell Culture and Drug Treatment
The human T24 BC cell line was obtained from the Bioresources Collection and Research Center (Hsinchu, Taiwan). T24 cells were cultured in McCoy’s 5A medium with 10% FBS, 100 units/ml penicillin and 100 μg/mL streptomycin. Cells were then incubated and maintained at 37°C with 5% CO2. CD stock solution was prepared at a concentration of 30 m
Colony-formation assay
After 24 h of CD treatment, each group of 100 T24 cells were harvested and seeded on 35-mm dishes in a CO2 incubator with 5% CO2 and 95% air at 37°C for additional 10 days. Cells were fixed with 6% glutaraldehyde and stained with 0.5% crystal violet for 30 min at room temperature. The colony was defined to consist of at least 50 cells. Colonies were counted under a phase-contrast microscope (Leica Biosystems, Wetzlar, Germany).
Cell spreading assay
T24 cells were treated with or without CD for 24 h. After treatment, cells were seeded and grown onto fibronectin-coated 24-well plates for an additional 3 h. Cells were then washed three times with phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde for 5 min and stained with 0.25% Coomassie blue R250 for 30 min at room temperature. Images were captured under a phase-contrast microscope at 100x magnification (Leica Biosystems). Staining dye was extracted by DMSO and optic density value was analysed by a Synergy HTX microplate reader (BioTek Instruments, Winooski, VT, USA).
Cell survival assays
Cell viabilities were analysed by MTT assays. Briefly, T24 cells were cultured in 96-well plate at density of 7.5 × 103 cells in triplicates overnight. Cells were then pretreated with or without a caspase-8 inhibitor FMK (10 and 20 μM) for one hour prior to CD 24 h treatment. Control group was treated with 0.1% DMSO. The medium of each well was removed and growth medium containing 1 mg/mL MTT were added for 3.5 h. The purple formazan crystals were solubilized by DMSO and optic density values were measured at 570 nm by a Synergy HTX microplate reader (BioTek).
Migration and Invasion Assays
Cell migration assays were conducted as described previously. Briefly, a Transwell assay was conducted 24 h after drug treatment. In this case, cells (3 × 105/8-μm well) were seeded onto the upper chamber of a Transwell insert for 2 h of incubation and then placed into a 24-well plate. Next, 10% FBS in McCoy’s 5A medium was added to the lower invasion chamber of each well. For the invasion assay, the cells were added on cross-linking Matrigel® (2 mg/mL). A cotton-tipped applicator was used to carefully remove cells that had not migrated from the top of the membrane. The membrane of the insert was washed twice with PBS, fixed with 4% paraformaldehyde and stained with 0.25% Coomassie blue solution for 30 min. Cells were observed and photographed under a light microscope.
HDAC Activity Assay
After treatment, nuclear extracts were collected and assayed with the HDAC activity assays (BioVision, Inc., Waltham, MA, USA). According to the manufacturer’s instructions, the HDAC activity assay was conducted using HDAC substrates and lysine developer. The optical density values were measured using a Synergy HTX microplate reader (BioTek Instruments).
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
List of primer sequences.

Western blot analysis
Cell lysates were prepared in a cell lysis buffer (0.15% Triton X-100, 2 m
Bioinformatics
The MMP9 mRNA expression between tumour and normal tissue were extracted from The Cancer Genome Atlas (TCGA) using UALCAN website (http://ualcan.path.uab.edu/ (accessed on 2 May 2022)). 23 The criteria for the significant mRNAs in the analysis were a Transcript per million (TPM) of expression level and a p-value <0.05, which was calculated using the UALCAN website. The Kaplan Meier plotter (http://kmplot.com/analysis/ (accessed on 2 May 2022)) 24 was used to analyse the association of the mRNA expression with overall survival rate.
Statistical analyses
Quantitative data represented at least three experiments, and the values were expressed as means ± standard deviations (SDs). Statistical analyses were conducted by using GraphPad Prism 9.0 software (San Diego, CA, USA). Statistical differences in multiple groups were determined by one-way analysis of variance (ANOVA), followed by Dunnett’s post-hoc test (control and CD treatment). 25 For cell survival assays, statistical difference between two groups were determined by one way ANOVA, followed by Tukey’s post-hoc test. 25 p < 0.05 was considered significant.
Results
CD Induced Anoikis/Apoptosis in T24 Cells
Previously, we observed that CD induces cytotoxicity through the intrinsic apoptotic pathway and causes the T24 cell shape to become round-up at 30 and 40 μM.
26
As shown in Figures 1(a) and 1(b), colony formation largely diminished after 24 h of CD treatment. This indicates that CD inhibits T24 BC cell proliferation. Loss of contact from the extracellular matrix (ECM) stimulates another type of programmed cell death, anoikis.
27
Two characteristics of anoikis are the downregulation of cellular FADD-like interleukin (IL)-1β-converting enzyme-inhibitory protein (cFLIP) and the activation of caspase-8.
27
We observed that CD decreased cFLIP expression but elicited caspase-8 cleavage (Figure 1(c)). Consistently, CD-induced cell death was reversed by FMK (Figure 1(d)). CD Induced Caspase-8-Dependent Apoptosis in T24 Cells. (a and b) Colony-formation assay. (c) Western blots of cFLIP and cleaved caspase-8. GAPDH was used as an internal control. (d) MTT assay. FMK was added into media for 1 h prior to CD treatment. (e and f) Cell spreading assay. Results (mean ± SD) are from three independent experiments. The figures shown are representative of one experiment. *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared with the control (0 μM).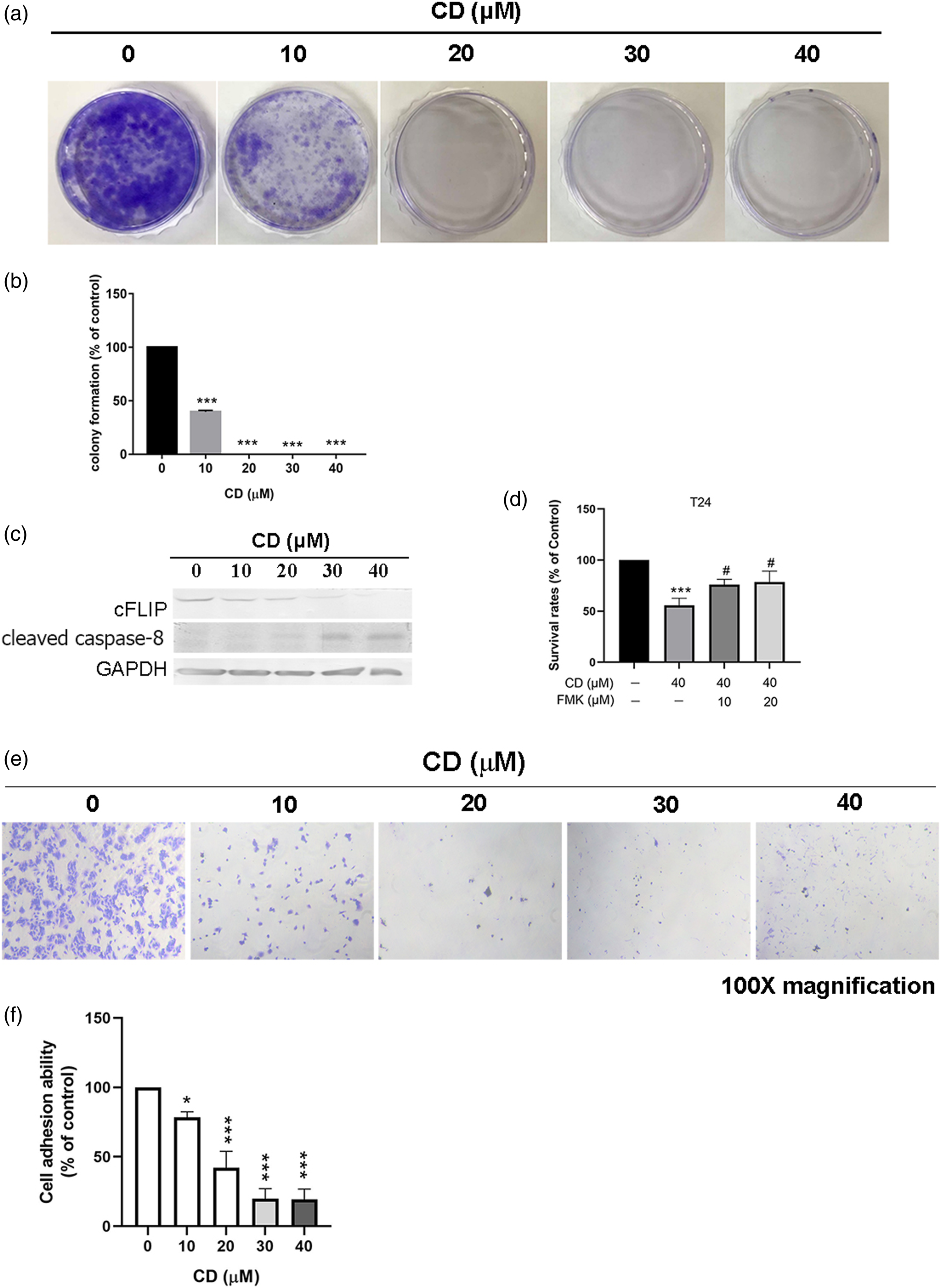
To investigate whether CD-induced anoikis/apoptosis is an irreversible event in BC cells, CD-treated T24 cells were harvested onto fibronectin-coated 24-well plates. Following CD treatment for 24 h, T24 cells could not re-adhere to fibronectin-coated cultured wells (Figures 1(e) and 1(f)). Compared to the control, CD-inhibited T24 cell adhesion at a concentration of 10, 20, 30, and 40 μM with 21.61%, 57.89%, 80.03% and 80.71%, respectively (Figure 1(f)).
CD-Induced Anoikis/Apoptosis and –Inhibited cell spreading Associated with HDAC Inhibition
HDACs are enzymes that contribute to tumorigenesis by removing the acetyl groups from histones and/or non-histone proteins.
28
To evaluate whether CD-inhibited cell proliferation and spreading occur through the regulation of HDAC activity, T24 cells were treated with CD (0–40 μM) for 24 h and nuclear extracts were assayed by using a pan-HDAC activity assay. Expectedly, CD treatment (10–40 μM) significantly downregulated HDAC activity by 23.87%, 34.17%, 45.66% and 45.22% (Figure 2(a)). Furthermore, total RNA was analysed to determine the subtype of HDAC most affected by CD treatment. Figure 2(b) shows that CD attenuated HDAC1, HDAC2 and HDAC3 mRNA expression (Figure 2(b)). The expression of HDAC1 mRNA was significantly decreased at a 20-μM concentration of CD, whereas HDAC2 and HDAC3 mRNA expression were blocked at a 40-μM concentration of CD (Figures 2(c)–2(e)). CD Suppressed HDAC Activity and HDAC1, 2 and 3 mRNA Expression. (a) Pan-HDAC activity assay. TSA (trichostatin A, 10 μM) was used as a negative control. Pos (positive nuclear extract) was applied as a positive control. Results are from four independent experiments. (b) RT-PCR analyses for HDAC1, 2 and 3 mRNA expression. GAPDH was used as an internal control. (c) Bar graphs show the relative density of each expression band (mean ± SD). The figures shown are representative of one experiment. Results are from three to four independent experiments. *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared with the control (0 μM).
CD Inhibited migration and invasion by integrin-dependent signalling pathways
To elucidate whether non-toxic concentrations of CD exhibit anti-migration or anti-invasion activities, Transwell migration assays and Matrigel®-coated Boyden chamber assays were conducted after CD treatment. At a concentration of 20 μ CD Suppressed Migration and Invasion. (a) Migration assay. (b) Invasion assay. Results are from three independent experiments. Representative images display at 50× magnification. The bar graphs demonstrate the quantification of cell numbers that passed through the Transwell membrane. ***, p < 0.001 compared with the control (0 μM).
Integrin-α5β1-mediated adhesion to fibronectin results in a phenotype that includes reduced cell proliferation, increased E-cadherin localisation in cell–cell contacts and increased β-catenin localisation throughout the cell.
29
Consistently, fibronectin, integrin α5, integrin β1, phosphorylated GSK3β (Ser 9), E-cadherin, N-cadherin and β-catenin were downregulated after 24 h of CD treatment (Figures 4(a) and 4(b)). CD-Suppressed Migration and Invasion Occur through the Integrin-α5β1-Associated Signalling Pathway. (a) Immunoblots of fibronectin, integrin α5 and β1, pGSK3β (Ser9), E-cadherin, N-cadherin and β-catenin. GAPDH was used as an internal control. (b) The bar graphs demonstrate densitometric data (mean ± SD) from three to five independent experiments. The data are expressed as a percentage of the control value. Figures shown are representative results of one experiment. *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared with the control (0 μM).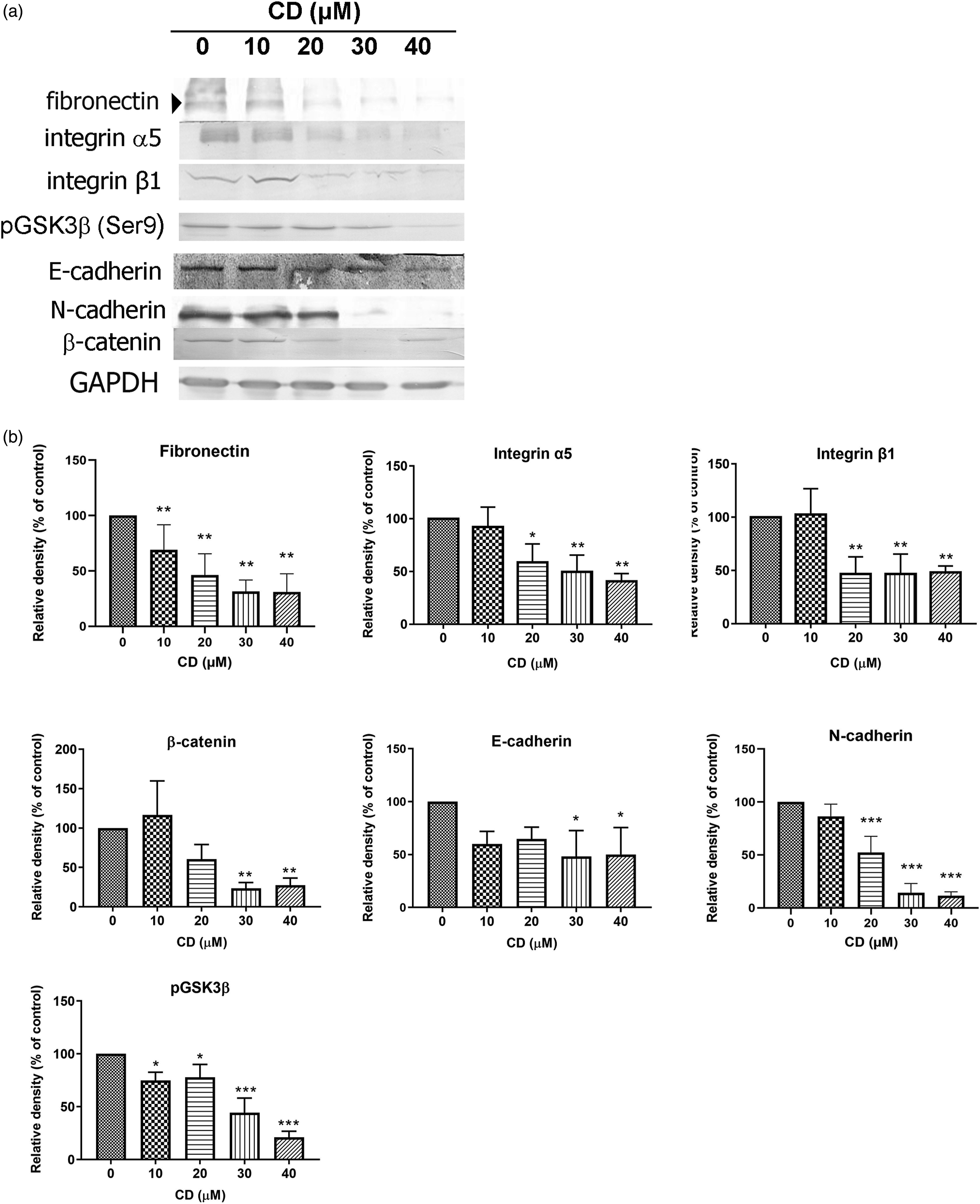
To further determine the downstream signalling pathway in CD-induced apoptosis and/or CD-suppressed migration and invasion, FA complexes were analysed by Western blotting. As shown in Figure 5, CD reduced pFAK (Tyr 397), FAK, vinculin, paxillin and RhoA expression (Figures 5(a) and 5(b)). CD Inactivates FAK–Associated Signalling Pathway. (a) Immunoblots of pFAK (Tyr397), FAK, paxillin, vinculin and RhoA. GAPDH was used as an internal control. (b–g) The bar graphs demonstrate densitometric data (mean ± SD) from three to five independent experiments. Figures shown are representative results of one experiment. *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared with the control (0 μM).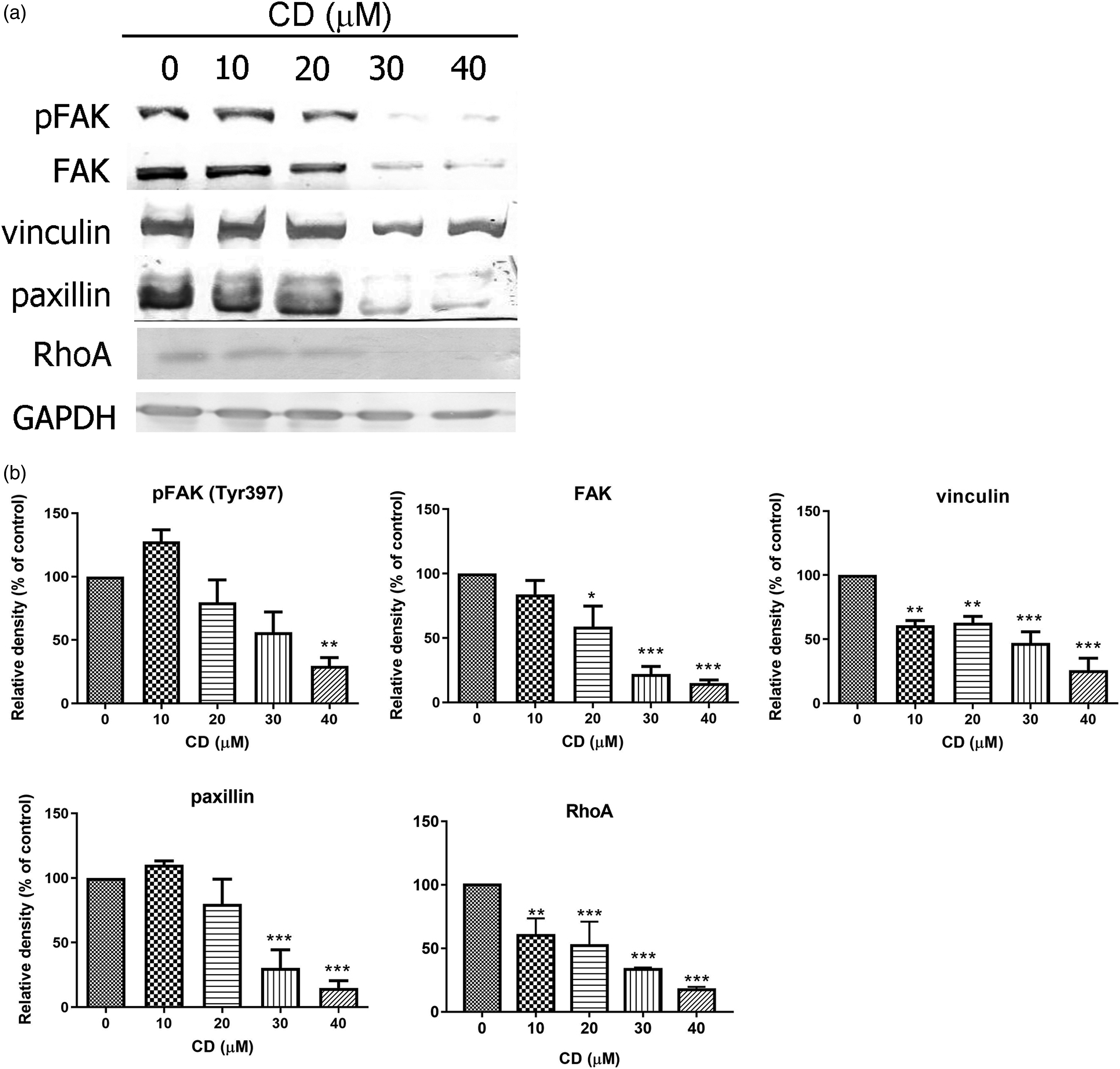
CD Suppressed Invasion of T24 BC cells Through MMP9
MMPs promote cancer metastasis by modulating ECM remodelling.
30
The upregulation of MMP9 in stage 2 and 3 BC tumours was demonstrated by Transcript per million levels in the TCGA cohort, compared with normal tissue (Figure 6(a)). Next, we analysed whether the expression level of MMP9 correlated with BC prognosis. Kaplan–Meier methods showed that higher MMP9 expression levels corresponded to poorer prognoses (Figure 6(b), p = 0.0019, hazards ratio = 1.68). CD Diminished MMP9 Expression and Activity. (a) MMP9 mRNA expressions among normal tissue and different stage of tumour samples were shown and analysed with the TCGA dataset. (b) Overall survival rates analysed by Kaplan–Meier plotter. (c and d) Functional analyses of cancer metastasis by gene set enrichment analysis (GSEA) with high or low MMP9 mRNA expression levels in TCGA. (e) Immunoblots of MMP9 in T24 cells. GAPDH was used as an internal control. (f) The bar graph of densitometric data (mean ± SD) from four independent experiments. (g) Gelatinase zymography assay of MMP9. PC is the positive control. The bar graph shows densitometric data (mean ± SD) from four independent experiments. The figures shown are representative of one experiment.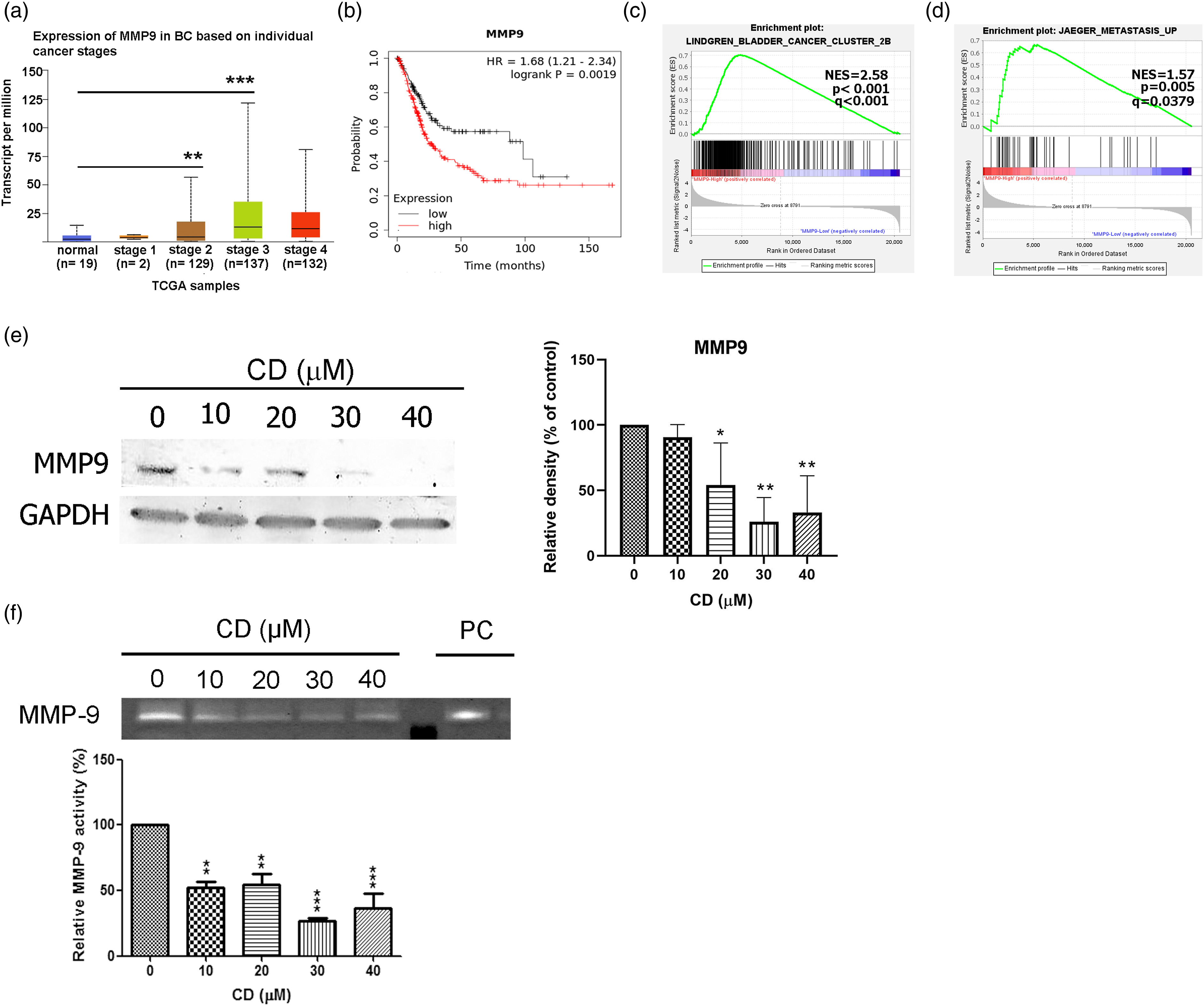
We also performed gene enrichment analyses to confirm that higher MMP9 expression enhances metastasis (Figures 6(c) and 6(d)). These results indicated that higher MMP9 levels provide a reasonable biological marker for poor prognosis in BC. Our study revealed that treatment with CD not only blocked migration and invasion activity (Figures 3(a) and 3(b)) but caused cells to lose their adhesion and spreading ability from the ECM and could suppress MMP9 protein expression and activity (Figures 6(e) and 6(f)).
To further explore the link between FAK and MMPs in T24 cells after exposure to CD, we examined the associated upstream protein levels in T24 cells. The results revealed that CD significantly reduced the expression of pSrc (Tyr418) and pNFκB (Ser536) in a concentration-dependent manner (Figures 7(a) and 7(b)). CD-Suppressed Migration and Invasion Involves Src, NFκB, and stat3 Inactivation. (a) Immunoblots of pSrc (Tyr418), src, pstat3 (Tyr705), stat3, pNFκB (Ser536) and NFκB. GAPDH was used as an internal control. (b) The bar graphs demonstrate densitometric data (mean ± SD) from three to five independent experiments. Figures shown are representative results of one experiment. *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared with the control (0 μM).
Discussion
Reports have stated that CD purified from P. longifolia leaves induces cell death through several mechanisms, including apoptosis, cell-cycle arrest and autophagy. 10 Moreover, CD inactivates oncogenic signalling pathways, such as MEK–ERK, mTOR–hypoxia inducible factor 2 alpha (HIF-2α) and Akt in tumour cells.17,20 However, the mechanisms of CD-inhibited proliferation, migration and invasion are not entirely understood in BC cells. This study illustrates that CD-induced cell death occurred through caspase-8 dependent apoptosis. Concomitantly, HDAC1, HDAC2 and HDAC3 mRNA expressions were attenuated by CD. CD-inhibited migration and invasion, with downregulation of the integrin–FAK signalling pathway and the inactivation of GSK3β, NFκB and signal transducer and activator of transcription 3 (stat3). In fact, the reduced expression and activity of MMP9 by CD contributed to block migration and invasion in BC. These results were validated by Kaplan–Meier plots, TCGA and GSEA cohort data.
We have illustrated that CD induces cell death in clear cell renal cell carcinoma by apoptosis and anoikis.19,20 In this study, we observed that CD affected T24 cell behaviour through several alternative pathways. At non-toxic concentrations (10 and 20 μM), CD-inhibited migration and invasion through integrin α5β1, pGSK3β, FAK, vinculin, RhoA, pstat3 and pNFκB. At cytotoxic concentrations (30 and 40 μM), CD-induced apoptosis by activating caspase-8 and decreasing cFLIP expression. Interestingly, CD-inactivated HDAC activity contributes to different effects. For anti-migration and anti-invasion activities, CD-inactivated HDAC activity occurs mainly through the regulation of HDAC1. As for apoptotic-inducing effects, the inactivated HDAC activity may act predminantly through HDAC1, 2 and three.
HDAC1–3 and 8 belong to class I HDACs, which are enzymes that perform post-translational modifications of amino acids on histones. 31 HDACs regulates gene expression, transcription and cell processes, including growth arrest, apoptosis and induction of cytotoxicity. 31 We did not detect HDAC8 expression because it is not known as a promising target in BC. 32 Lin et al. (2011) showed that CD treatment influences histone-modifying enzyme expression. 33 However, they did not display the quantification data of histone acetylation and methylation after CD treatment. According to our previous study, CD inactivated epidermal growth factor receptor (EGFR) and induced apoptosis and cell cycle arrest in T24 bladder cancer cells. 26 Bahl et al. (2021) demonstrated that the antiapoptotic function of HDAC1 was highly correlated with EGFR activity. 34 Therefore, the connection among the signalling pathways, HDAC inactivation and the histone modification following CD treatment to induce bladder cancer cell death needs more study to clarify.
Fibronectin, an ECM protein, triggers integrin α5β1 activation and the downstream signalling network to further increase MMP gene transactivation. 29 Among these, MMP9 is closely associated with BC metastasis and patient prognosis, as shown in the TCGA cohort and Kaplan–Meier plot results (Figures 6(a) and 6(b)). MMP9 can be stimulated by fibronectin and regulated by several signalling pathways, including Akt, mitogen activated protein kinases, FAK, Stat3, GSK3β and NFκB. 35 Our results are consistent with these observations. According to studies by Lin et al. (2011) and Hsu et al. (2015), CD has been evaluated by molecular docking methods with PI3K and FAK inhibitors. Whether CD inhibits other kinases by binding to the functional site would be another useful investigation.
Anoikis-inducing agents have been studied extensively in urothelial cancer. 36 Even so, cancer cells often develop resistance to cytotoxicity and apoptosis. 36 Epithelial–mesenchymal transition (EMT) is one of the pathway which implicates in cancer metastasis to achieve stem-cell properties and confers anoikis resistance. 36 The protein expressions of epithelial (E-cadherin) and mesenchymal (N-cadherin) markers were both decreased in CD-treated T24 cells, suggesting that CD might not stimulate the progress of EMT.
Limitation of the study
This study has some limitations. 1. Lack of direct evidence on specific HDAC on CD-induced anoikis/apoptosis in bladder cancer cells. 2. Authors only screen HDAC1, 2, and 3 mRNA expression. Other HDACs may also involve in CD-induced cell death. 3. The efficacy of CD against bladder cancer needs to be further evaluated in in vivo models.
Conclusion
In this work, we have demonstrated that CD induces caspase-8-dependent anoikis/apoptosis and suppresses migration and invasion in T24 BC cells through integrin–FAK and MMP9 pathway inactivation. CD could be a potential agent to treat BC because of its multiple antitumour functions and lack of EMT progression.
Footnotes
Abbreviation
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partially by the Research Center for Precision Environmental Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan and by Kaohsiung Medical University Research Center Grant (KMU-TC111A01 and KMUTC111IFSP01), and the E-Da Cancer Hosipital grant (EDCHP110006).
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
