Abstract
As a potent neurotoxic agent, acrylamide (ACR) is formed in food processing at higher temperature. Taurine (TAU), a nonessential amino acid, is used to cure neurodegenerative disorders, followed by activation of the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) signaling pathway. In this article, we certified that antiapoptotic efficacy of TAU in vivo and vitro. ACR-treated rats received TAU by drinking water 2 weeks after ACR intoxication. The results showed that in treated rats, TAU alleviated ACR-induced neuronal apoptosis, which was associated with the activation of PI3K/AKT signaling pathway. TAU attenuated apoptosis caused by ACR through observing terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL)-positive cells, measure of protein expression of Bcl-2, Bax, and caspase 3 activity. TAU-induced antiapoptotic effect is PI3K/AKT-dependent, which was proved in ACR-intoxicated ventral spinal cord 4.1 cells in the presence of AKT inhibitor, MK-2206. Therefore, our results demonstrated that TAU-attenuated ACR-induced apoptosis in vivo through a PI3K/AKT-dependent manner provided new sights in the molecular mechanism of TAU protection against ACR-induced neurotoxicity.
Introduction
Acrylamide (ACR), a reactive and water-soluble chemical, is widely used in large spectrum of areas. 1 It has been proved that the main sources of ACR exposure are dietary and occupational exposure. High amount of ACR produced in food having starch and protein, during roasting, frying, grilling, and baking. 2 The symptoms of sweating hands, limb pain, and numbness 3 are observed in human exposed to ACR. At the same time, low body weight and high consumption of snacks can affect infants and children easily compared to adults. 4 Previous studies proved that the effects on rats depend on the different concentrations of ACR as shown by weight loss, peripheral neuropathy, and uncoordinated motor movements. 5 Many studies demonstrated that ACR has neurological and reproductive toxicities. 6,7 In addition, ACR causes damage in peripheral (PNS) and central nervous systems (CNS), which is induced by losing the number of neurons in the distal swellings of large axons. 8
Morphological observation suggested that ACR induced apoptosis in various models including in vitro studies 9 –12 (e.g. neuroblastoma SH-SY5Y and human liver embryo L-02) and in vivo studies (e.g. CNS and PNS tissues in Wistar rats and Sprague-Dawley (SD) rats). It was proved that the main damage induced by ACR is embodied in neurons. 13 The expression levels of apoptotic genes including Bax, Bcl-2, and caspase 3 are increased in ACR-intoxicated Leydig (TM3) and Sertoli (TM4) cells. 14 As a result, apoptosis is the most important features of ACR-induced neurotoxicity.
Taurine (2-aminoethane sulfonic acid, TAU) is considered to be a free intracellular β-amino acid which exerts beneficial health effects through regulating cellular functions.
15,16
Due to its antiapoptotic property, TAU has attracted much attention and is utilized to treat neurodegenerative diseases.
17
Clinical age-related diseases were developed once the TAU gene is knocked out from the rats.
18
Previous study indicated that S-Methyl-N, N-diethylthiocarbamate sulfoxide (DETC-MeSO) (an
In the present study, we demonstrated that TAU treatment promoted neuronal survival and attenuated ACR-induced apoptosis in the spinal cord of rats. Besides, activation of phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway was proved to be responsible for TAU-elicited protection, which subsequently increased the expression of Bcl-2 and decreased the expression of Bax and the activity of caspase 3. TAU attenuates ACR-induced apoptosis through a PI3K/AKT-dependent manner, which could supply a novel strategy for treating ACR-induced neuropathy.
Materials and methods
Treatment of animals and tissue preparation
In this experiment, 40 adult SD male rats (210–230 g) were purchased from the Experimental Animal Center of Dalian Medical University No.9 West Section Lvshun South Road Dalian Liaoning Province China, P.C. 116044. They were housed in polycarbonate boxes and maintained on a 12-h light/dark cycle at 22–24°C with sufficient drinking water and food. Forty rats were randomly divided into four groups (
Co-staining TUNEL and immunofluorescence
Spinal cord sections (10-μm axial sections) were immersed in 0.4% Triton X-100 for 30 min and 5% donkey serum for 1 h at room temperature. Subsequently, they were incubated with mouse anti-NeuN monoclonal antibodies (Sigma) overnight at 4°C. The secondary antibodies (Alexa Fluor 488 donkey anti-mouse IgG, Sigma, St Louis, Missouri, USA) were added to incubate the sections at room temperature for 2 h. Then, TUNEL assays were performed according to the protocol of the manufacturer (In Situ Cell Death Detection Kit, Roche, Germany). Sections were observed under a fluorescence microscope (×400 magnification).
Ventral spinal cord 4.1 cell culture and treatment
Ventral spinal cord 4.1 (VSC4.1) motor neurons were cultivated in dulbecco's modified eagle medium (DMEM) medium with 15 mM pyridoxine, HEPES, and sodium bicarbonate (Sigma), supplemented with 2% Sato’s components, 1% penicillin and streptomycin (Beyotime, Shanghai, China), and 15% fatal bovine serun (FBS) (Hyclone, Logan, Utah, USA) at 37°C. The cells were treated with ACR (1.0 mM) or saline vehicle for 24 h and then were treated with TAU (5.0 mM) for additional 24 h, with or without LY294002 (Abcam, China Sigma), or MK2206 1 h pretreatment followed by previous studies. 23,24
Western blot
Ice-cold RIPA tissue protein extraction reagent (Beyotime, China) with 1% proteinase inhibitor was used to homogenize tissue and cells. Separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and then electrotransferred to polyvinylidene fluoride (PVDF) membrane (Millipore, France), proteins were extracted with protein extraction reagent (Bio-Rad Biotechnology, Hercules, CA, USA). After blocking in 5% nonfat milk at room temperature for 2 h, the membranes were incubated with appropriate primary antibodies at 4°C overnight. Antibodies utilized were Akt (1:1000, Sigma), p-Akt (ser-473; 1:1000, Sigma), Bax, Bcl-2, caspase 3 and cytochrome c (cyt-c; 1:500, SantaCruz, California, USA), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:1000, Abcam, San Francisco, USA). Then, membranes were incubated with the secondary antibody, horseradish peroxidase-conjugated antibody (1:5000, Sigma) and detected by enhanced chemiluminescence reagents. Moreover, UVP BioSpectrum Multispectral Imaging System (Ultra-Violet Products Ltd, Upland, CA, USA) was used to measure quantified densitometric analysis.
Statistical analysis
All results are represented as the means ± standard deviation from three independent experiments with triplication. At the same time, the statistical data were analyzed by one-way analysis of variance according to the least significant difference (LSD) test, which was performed using SPSS 19.0 statistical software. The
Results
Body weight and clinical observations after TAU treatment
SD rats were intoxicated with ACR (50 mg/kg/day) or saline for 2 weeks and then were treated with (200 mg/kg/day) TAU or vehicle for another 2 weeks. From the beginning of TAU treatment, body weights of all groups were detected per 2 days. As shown in Figure 1, the body weights of control group and TAU control group get constant gain, while ACR-intoxicated rats showed attenuated increase in body weight and even lost weight during the last 4 days. Compared to only ACR-intoxicated rats, the weights of TAU -ACR co-treated rats increased gradually and were active than only ACR-intoxicated rats. On day 12, the body weights of ACR-TAU co-treated rats were significantly (
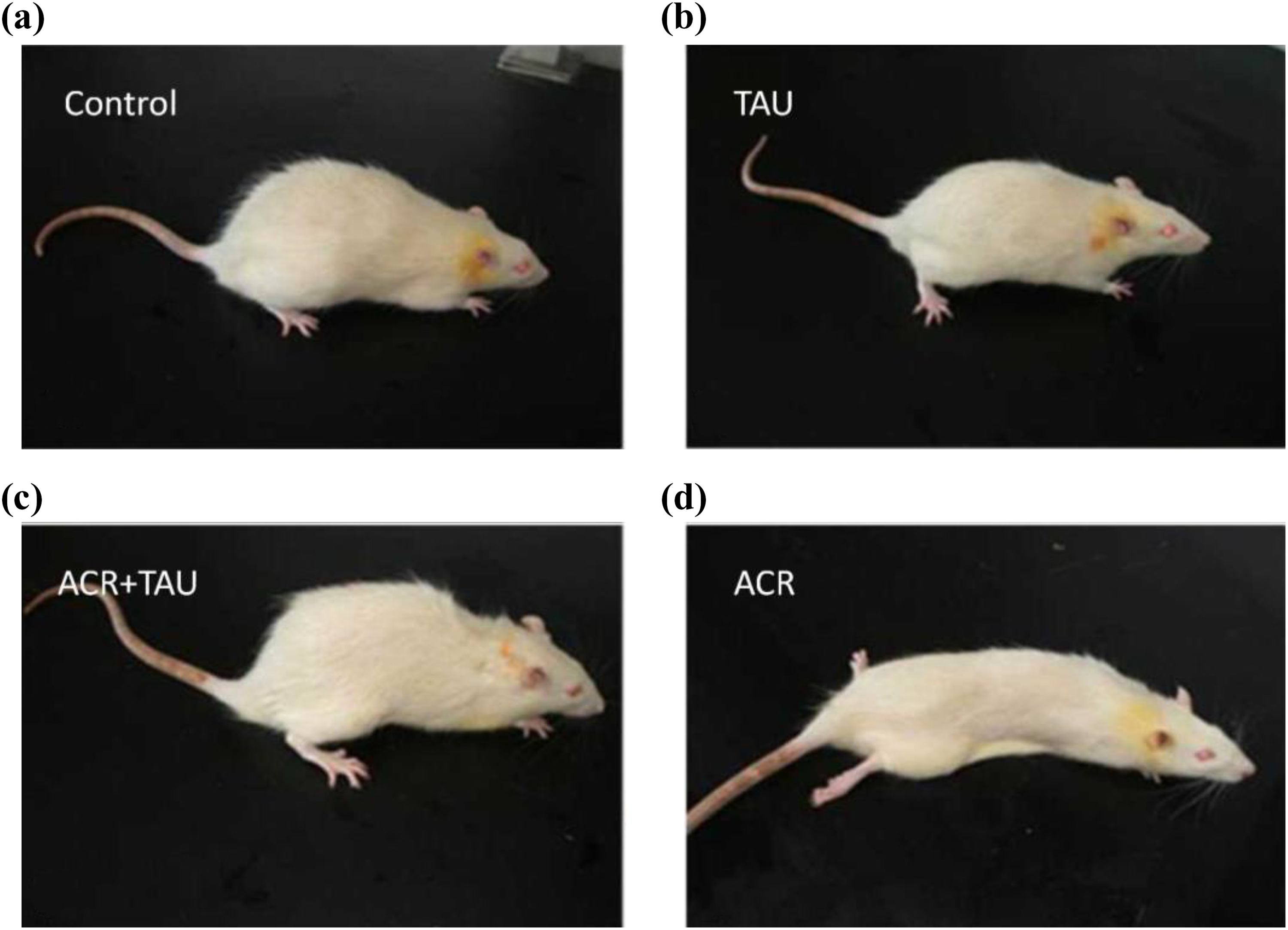
Clinical performance of each group on 28 days. (a) Control group: rats were treated normal feeding; (b) ACR: acrylamide group: rats were received ACR (50 mg/kg/day i.p.); (c) ACR+TAU: taurine group: rats were pretreated with ACR (50 mg/kg/day i.p.) for 14 days and then administered TAU (200 mg/kg/day i, g) intragastrically for 14 days; and (d) TAU group: rats were TAU (200 mg/kg/day i, g) for 28 days. ACR: acrylamide; TAU: taurine; i.p.: intraperitoneal.
Effect of TAU against acrylamide on body weight after TAU treatment.a

ACR: acrylamide; TAU: taurine; i.p.: intraperitoneal.
aData were shown as mean ± SEM. There were 10 animals in each group at each time point. Effect of TAU against ACR on body weight after TAU treatment. Control group: rats received normal feeding for 14 days; ACR group: rats received ACR (50 mg/kg/day i.p.) at the beginning of animal treatment; ACR+TAR group: rats were treated with ACR (50 mg/kg/day i.p.) and then administered TAU (200 mg/kg/day i, g) intragastrically for 14 days; TAR group: rats received TAU (200 mg/kg/day i, g) in the whole process of animal treatment.
b
Consistent with the body weights, clinical performance of rats in each group shows the influence of TAU. Rats treated with TAU alone were healthy and active presenting no difference from normal rats. By contrast, ACR-intoxicated rats showed reduced activity as well as were anorexic and listlessness. In addition, abnormal symptoms induced by ACR intoxication were also mitigated when ACR-intoxicated rats were treated with TAU (Figure 1).
TAU decreased apoptosis induced by ACR
To analyze whether TAU treatment decreased ACR-induced apoptosis, TUNEL staining was done. The results were shown by the different colors of the nuclei of normal cells and cytoplasms or nuclei of TUNEL-positive cells. Obviously, TUNEL-positive cells were significantly increased (

TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling staining in the spinal cord of rats and the representative images of each group were shown. Control group: rats received normal feeding for 14 days; ACR group: rats received ACR (50 mg/kg/day i.p.) at the beginning of animal treatment; ACR+TAR: taurine group: rats were pretreated with ACR (50 mg/kg/day i.p.) and then administered TAU (200 mg/kg/day i, g) for 14 days; TAR group: rats received TAU (200 mg/kg/day i, g) in the whole process of animal treatment. ACR: acrylamide; TAU: taurine; i.p.: intraperitoneal.
TAU-induced AKT phosphorylation through activating PI3K signal pathway
To evaluate whether TAU enhanced AKT phosphorylation, the level of phosphorylated AKT of each group was measured. According to Figure 3(a), ACR significantly decreased AKT phosphorylation, which was attenuated once ACR-intoxicated rats were received with TAU. To investigate whether PI3K dependence is involved in TAU-induced AKT phosphorylation, LY294002, which is an inhibitor of PI3K, was used. TAU-induced elevation of AKT phosphorylation in ACR-intoxicated VSC4.1 cultures was significantly decreased in the presence of LY294002, indicating that TAU-induced AKT phosphorylation is PI3K-dependent (Figure 3(b)).

Effect of TAU on ACR-induced Akt activation. (a) The effects of TAU on the levels of Akt and p-Akt in the spinal cord of ACR-intoxicated rats were detected with Western blot and the density of blots was quantified. (b) VSC4.1 cells were treated with ACR (1.0 mM) or saline for 24 h and then were treated with TAU (5.0 mM) in the presence or absence of LY294002 1 h pretreatment for additional 24 h. The levels of Akt and p-Akt were detected with Western blot. a
TAU treatment activated AKT pathway and subsequently regulated the expression of protein related to apoptosis
Based on Figure 4(a) and (b), ACR caused the increase in the protein expression of Bax, the activity of caspase-3, and the decrease in the protein expression of Bcl-2, which were all significantly (

TAU attenuates ACR-induced apoptosis in an AKT serine-threonine kinase-dependent manner. (a) The effects of TAU on the levels of Bcl-2 and Bax in the spinal cord of ACR-intoxicated rats were detected with Western blot and the density of blots was quantified. (b) Caspase 3 activity was detected in the spinal cord of ACR-intoxicated rats. (c) and (d) VSC4.1 cells were treated with ACR (0.1 mM) or saline for 24 h and then were treated with TAU (5 mM) in the presence or absence of MK2206 1 h pretreatment for additional 24 h. Bcl-2 and Bax (c) expression and caspase 3 activity (d) were detected with Western blot. a
Discussion
To explore whether TAU could attenuate the damage induced by ACR in rat spinal cord and the mechanism, the body weight, clinical state, and the number of TUNEL-positive cells were examined. Meanwhile, we determine the phosphorylation of AKT and the expression of protein associated with apoptosis in each group to clarify the mechanism of protection-induced TAU.
ACR affected the growth of rats through examining the body weight and clinical performance. The body weight and clinical performance were regarded as the indicators to evaluate the effect of ACR on the rats' growth. Specifically, ACR not only significantly influenced weight gain but also damaged the motor function compared with control, which was attenuated when the rats were treated with TAU. The phenomenon of anomalous behavior induced by ACR accords with previous results. 25 ACR-intoxicated rats displayed slight uncoordinated motor movement almost they had inability to support the body. 26 Moreover, previous study had suggested that the intoxication of ACR induces deep sensory disturbance and ataxia. 27 The symptoms of weight loss and clinical abnormity were attenuated by TAU, indicating that taurine was considered as an effective remedy to treat ACR intoxication.
Since the spinal cord could regulate the motor activity and neuronal activity, 28 ACR cause abnormal clinical performance, suggesting that it can cause neurotoxicity and induce injury to spinal cord. It has been reported that the degeneration in spinal cord was the main matter of rats intoxicated with ACR. 29 Our studies proved that TAU exhibited effective protection from the neuronal damages induced by ACR to increase the number of nerve cells. The similar protection has been demonstrated through neurological recovery induced by TAU in closed head injury in rats. 30 The neuroprotection of TAU against ACR-induced damage in spinal cord suggests that TAU treatment may be a promising solution to halt the neuropathy of ACR.
As an important role in several biological processes, TAU exists in various types of cells. 31,32 In this study, we delve deeper into the investigation of the phosphorylation of AKT, exploring that ACR significantly inhibited the level of phosphorylated AKT. By contrast, TAU displayed an overwhelming capability to enhance the level of phosphorylated AKT. Previously, the researchers found that ACR-treated neurospheres of KT98/F1B-GFP showed decrease in phosphorylated AKT through analyzing AKT pathway. 33 At the same time, extracellular TAU induces angiogenesis by activating AKT-dependent signal pathways. 10 TAU promotes cognitive function of prenatally stressed juvenile rats by activating the AKT-CREB-PGC1α pathway, 34 proving that the level of phosphorylated AKT when the rats or the cells treated with TAU. In the presence of LY294002, TAU failed to increase the low level of phosphorylated AKT induced by ACR in VSC4.1 cells, which indicates that TAU-stimulated AKT phosphorylation is PI3K-dependent.
Apoptosis has been proved to be an important physiological process which regulated environmental adaptation of neurons in adult nervous system. The unbalance of this process is involved in a wide variety of neurodegenerative disease, including ACR-induced neuropathy. 35 –37 The results of the present data provided that TAU attenuated the degree of apoptosis induced by ACR through measuring by TUNEL detection. Admittedly, Bcl-2 is an antiapoptotic protein. 38 Comparatively, Bax is a proapoptotic protein. 39 Both of them affect apoptosis by regulating the levels of caspase 3. Caspase 3 is an important player in regulating apoptosis, 40 which activates the process of apoptosis. 41 Meanwhile, decreasing the expression of cyt-c led to an upgrade in the apoptotic once rates exposed to berberine, 42 which were blocked by the release of antiapoptotic protein Bcl-2. 43 Therefore, in this work, we observed that ACR intoxication decreased Bcl-2 as well as increased Bax and caspase 3, which was significantly attenuated once ACR-intoxicated rats treated with TAU. Consistent with influence induced by TAU, exposure to ACR for 48 h resulted in increased Bax/Bcl-2 ratios in primary astrocytes and U-87 MG cells, 36 which was similar to the previous studies. 35 In the model of glial cells, the relative expression level of Bax mRNA and Bcl-2 mRNA showed the similarity once the cells were cultured with TAU. 44 In our study, MK-2206, an inhibitor of AKT, was used to prove TAU-induced balance of apoptotic proteins.
In summary, this study convinced that TAU treatment ameliorates the neuronal apoptosis, the decline of Bcl-2, and the increase in Bax and caspase 3 activity induced by ACR in the spinal cord through a PI3K/AKT-dependent manner. Besides, the obvious protective effects suggest that TAU treatment could be a novel candidate against ACR-induced neuropathy. Future studies focusing on the neuroprotective effect of TAU in animal model of ACR-induced neuropathy should be guaranteed.
Footnotes
Authors’ contribution
GS and XW are the co-first authors and have contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (no. 81102160).
