Abstract
Background
Radiotherapy is widely used in the treatment of tumors. However, while killing tumor cells, radiation may also cause damage to the surrounding normal tissues. Therefore, it is very important to find safe and effective radiation protection agents.
Purpose
To investgate the radiation protection effect of Lanzhou Lily polysaccharide fragments (LLP). Methods: The crude polysaccharides of Lanzhou Lily were extracted from the dried bulb powder of Lilium lilium by ultrasonic-assisted hot water method, and then five different fragments were separated from the polysaccharides by DEAE-52-cellulose column. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide assay, neutral comet and immunofluorescent staining were used to investigate the effect of LPe fragment on Human Umbilical Vein Endothelial Cells (HUVEC) survival and the possible radioprotective mechanism.
Results
The LPe fragment (composing of mannose and glucose, with a ratio of 5.5:2.9, and the average molecular weight is 8629.8 Da), significantly promoted the proliferation of HUVECs and protected cells from X-ray-induced double-strand breaks (DSBs) in DNA, in which pretreatment with the LPe fragment at 100 μg/mL showed the most pronounced protection. In addition, the occurrence of X-ray-induced γH2AX foci was significantly reduced by treatment with the LPe fragment at 50, 100, and 200 μg/mL. Furthermore, caffeine or wortmannin in combination with the LPe fragment at 25 μg/mL significantly reduced the number of X-ray-induced γH2AX foci, indicating phosphoinositide-3 kinases (PI3K) is involved in H2AX phosphorylation in HUVECs.
Conclusion
These results indicate the LPe fragment has a protective effect against radiation-induced DSBs and may be used as a natural antioxidant agent.
Introduction
Radiotherapy is widely used in the treatment of tumors; however, ionizing radiation (IR) is considered a “double-edged sword.” Although IR can effectively kill tumor cells, it can also cause damage to the normal tissues surrounding the tumor. 1 Several studies have reported that IR can directly or indirectly induce DNA damage, including DNA double strand breaks (DSB) and single strand breaks (SSB).2–5 Double-strand breaks caused by IR are lethal. 6 Double strand breaks can phosphorylate histone H2AX at serine 139, the site of γ-phosphorylation, which is one of the earliest events in the cellular response. H2AX phosphorylation is mediated by PI3K, which include ataxia telangiectasia mutated protein (ATM), DNA-dependent protein kinase (DNA-PKcs) and ataxia telangiectasia and Rad3 related protein (ATR).7–10 The number of γH2AX foci is an indicator of the extent of DSBs. Different kinase inhibitors, such as caffeine and wortmannin, have been used to differentiate between the involvements of the different PI3K family members in a variety of cellular settings. Caffeine is widely used to inhibit ATM and ATR, whereas wortmannin is a relatively potent inhibitor of DNA-PK and ATM. 11
Although many radiological advancements have been made in the treatment of cancer, normal tissues are damaged to varying degrees after radiotherapy. Therefore, protecting healthy tissues from damage caused by IR is the key to improving the quality of life of radiotherapy patients. 12 Radioprotective agents are substances that can remove active oxygen produced by radiation and prevent gene mutations, thereby protecting healthy tissues. The use of radioprotective agents before and after exposure to radioactive substances can prevent and reduce the clinical symptoms of radiation sickness and promote the early recovery of patients. Radioprotective agents are important in safeguarding healthy tissues during radiation exposure, and thus, natural products can serve as radioprotective agents because they show good effects and possess low toxicity.13,14
Lanzhou lily (Lilium davidii var. unicolor) is the only type of Lilium in China that has both culinary and medicinal value. 15 Its bulbs contain a variety of bioactive compounds, such as polysaccharides, saponins and colchicine. Lily polysaccharides (LPs) are one of the major bioactive compounds of lily bulbs, which are composed of D-glucose, D-mannose, galactose and other polysaccharides that are held together by pyranoside bonds. 16 The pharmacologic effects of LPs include regulating immune function, lowering blood sugar, functioning as an antioxidant, suppressing carcinogenesis, alleviating fatigue symptoms, and arresting bacterial growth. 17 Numerous studies have reported that bioactive compounds extracted from plants and foods, such as polysaccharides and phenols, can effectively reduce radiation-induced damage,18–21 although the radioprotective effects of LPs have not been reported. Here, we hypothesize that LPs can mitigate radiation-induced damage. We prepared LP fragments by chemical modification and explored their protective effects on radiation-induced damage in human umbilical vein endothelial cells (HUVECs) to obtain more evidence to support their application in the food industry.
Materials and methods
Materials and chemicals
Dulbecco’s Modified Eagle’s Medium and fetal calf serum (FCS) were purchased from Hyclone (Thermo Fisher Scientific, Waltham, MA, USA). The anti-phospho-histone H2AX (Ser-139) monoclonal antibody was obtained from Upstate (Lake Placid, NY, USA). The fluorescein isothiocyanate (FITC)-conjugated anti-mouse goat F(ab′)2 fragment, and the FITC-conjugated anti-rabbit goat F(ab′)2 fragment were purchased from DAKO (Carpinteria, CA, USA). Caffeine and wortmannin were purchased from Sigma (St Louis, MO, USA). All other chemicals were purchase from Sigma (St Louis, MO, USA), and were of the highest grade available.
Extraction of LP and preparation of LP fragments
The polysaccharides were extracted from the bulbs of Lilium davidii var. unicolor Salisb by the ultrasound-assisted method. The supernatant was concentrated to one-fourth of the initial volume, and the crude LP fraction was obtained by adding anhydrous ethanol up to 80% and storing for 12 h at 4°C. LPs were purified by the Sevage method (Vchloroform:Vn-butanol = 5:1) to remove the protein. 22
The LP fragments were isolated from LPs using a DEAE-52-cellulose column (80 × 610 mm, Beijing Solario Science and Technology Co., Ltd., Beijing China), which were eluted with ultra-pure water, 0.04 mol/L NaCl and 0.1 mol/L NaCl, respectively. The eluent was detected by phenol sulfuric acid-UV Spectrophotometry (UV-1800PC, Shanghai Meipada Instruments Co., Ltd., Shanghai China), and the major fractions were collected according to their elution patterns. Then the major fractions were hydrolyzed with 0.1 mol/L HCl under the protection of N2 flow in a water bath. After hydrolysis, 20% NaOH was added to the hydrolysates to adjust the pH to 7.0, and then dialyzed (there is no Cl− in and out of the dialysis bag), concentrated, and precipitated by anhydrous ethanol. The dry sample was separated by a Sephadex G-50 column (50 × 600 mm, from Sigma Chemical Co., USA), followed by eluting with 3 L ultra-pure water, purifying with a Sephadex G-100 column (3.5 × 600 mm, from Sigma Chemical Co., USA) and eluting with 1.8 L ultra-pure water. The extraction procedure of LP and preparation of LP Fragments are shown in Figure1 and the fragments of LP a, b, c, d, e were obtained. Extraction of LPs and preparation of LP fragments.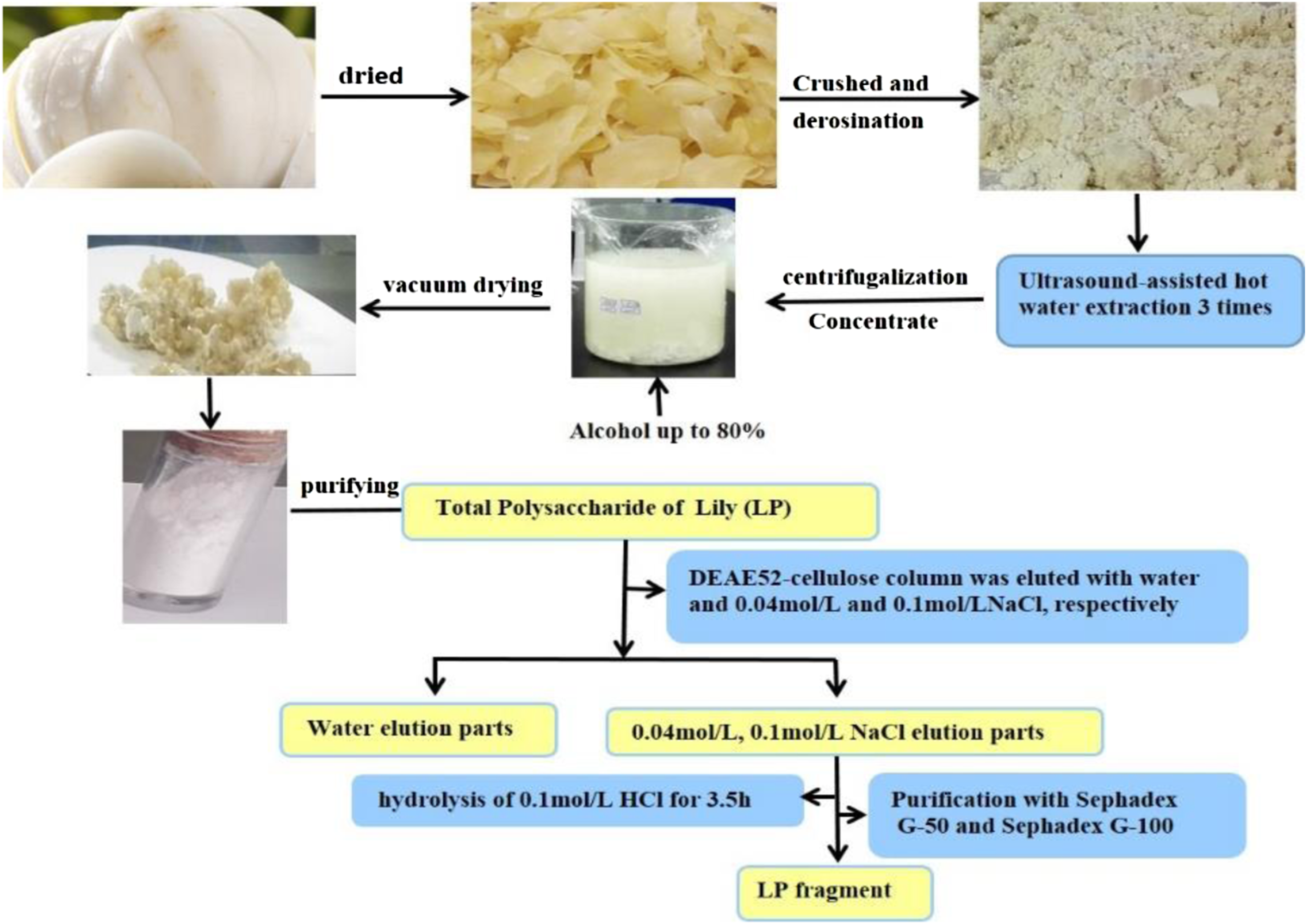
Cell culture
Human Umbilical Vein Endothelial Cell were obtained from the Shanghai Institute of Biochemistry and Cell Biology (Shanghai, China). The cells were cultured in RPMI 1640 medium supplemented with 10% FCS and 1% penicillin/streptomycin at 37°C in a humidified atmosphere containing 5% CO2.
Cell viability assay
The effect of LP fragments (a, b, c, d, e) on HUVEC viability were assessed with the 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay. The cells were seeded at a density of 2 × 103 cells/well in 96-well microtiter plates and incubated for 24 h. At each time point, the wells were washed three times with phosphate buffer solution (PBS) and 20 μL of MTT solution (stock concentration, 5 mg/mL) was added to each well. The cells were incubated again for another 4 h. After removing the culture medium, 150 μL of DMSO was added to each well to dissolve the precipitates. The absorbance was measured at 570 nm with a Biotech Synergy 2 plate reader (Biotech; Winooski, VT, USA).
Molecular weight determination of LPe fragments
The molecular weight of Lily polysaccharide fragment (LPe) was determined by gel high performance liquid chromatography (HPLC, Agilent 1200, Agilent 1200 Refractive Index Detector (RID), Column: PL aquagel-OH Guard 8 μm, 50 × 7.5 mm, and PL aquagel-OH MIXED-M 8 μm, 300 × 7.5 mm, and PL aquagel-OH 30, 8 μm, 300 × 7.5 mm; Agilent Co. USA) using Dextran-4000, 5000, 12,000, 40,000, 100,000, and 200,000 (≥99%, Sigma Chemical Co., USA) as standards. The standard and the sample concentration was 20 mg/mL, and the injection volume was 20 μL. The temperatures of the column and RID were 25°C and 35°C, respectively. The flow rate was 1 mL/min, and eluted with ultra-pure water. 23
X-ray irradiation and treatment with LPe fragment
Exponentially growing cells were seeded at 2 × 104 cells/100 mm dish. The cells were pretreated with the LPe fragment at concentrations ranging from 12.5 to 200 μg/mL for 24 h, followed by exposure to 1 Gy of X-ray IR. Ionizing radiation was performed with a Faxitron Cabinet X-ray System (RX-650, Faxitron Bioptics, Tucson, AZ, USA) operated at 100 kVp. The dose rate was approximately 1.38 Gy/min, and the doses used for each irradiation were 0 and 1 Gy. The cells were irradiated once at room temperature on the same day.
Double-strand break detection by the neutral comet assay
After exposing the cells to 1 Gy of IR for 4 h, the neutral comet assay was performed as previously described. 24 The cells were trypsinized, and a single suspension of 1.5 × 104 cells was embedded in 0.75% low-gelling-temperature agarose and quickly pipetted onto a pre-coated microscope slide. Cell lysis was performed by incubating slides for 4 h at 50°C in 0.5% SDS, 30 mM EDTA (pH 8.0). After rinsing overnight at room temperature in Tris/borate/EDTA buffer (pH 8.0), slides were electrophoresed at 0.6 V/cm for 25 min and then stained with propidium iodide (PI). A fluorescence microscope with a charge-coupled device camera was used to view the slides, and a total of 150 individual comet images for each sample were analyzed for tail moment (TM) and olive tail moment (OTM). Three independent trials were performed.
Immunofluorescence microscopy for γH2AX
Immunofluorescence staining was performed after exposing the cells to 1 Gy of IR for 0.5 h, as previously described with some modifications. 25 The cells were seeded at 2 × 104 cells/35 mm dishes containing a cover slip in each well. After irradiation, the slides were air-dried and the cells were fixed in 2% paraformaldehyde in Tris buffered saline (TBS) for 0.5 h. Followed by incubation in methanol at −20°C for 1 min and TBS containing 1% bovine serum albumin and 0.2% Tween-20 (TTN) for 20 min. The cells were incubated with an anti-phospho-histone H2AX (Ser-139) monoclonal antibody (1:500 in TNN) for 2 h. The cover slips were rinsed and the cells were incubated with FITC-conjugated anti-mouse goat F (ab′)2 fragment (1:200 in TTN, DAKO, Carpinteria, CA) for 1 h at room temperature. Then slides were rinsed and immersed in DAPI (0.05 mg/mL) for 15 min, washed and mounted with cover slips using 10 μL Fluorogard (Bio-Rad) as the antifade mounting medium, and then sealed. Over 800 randomly selected cells were counted. Cells with three or more foci were classified as positive. All trials were repeated in triplicate.
For the kinase inhibitor experiment, the cells were pretreated with caffeine (8 μmol/L) or wortmannin (20 nmol/L), followed by treatment with 1 Gy of IR for 1 h to inhibit the functions of ATM, ATR, and DNA-PK. Immunofluorescence staining was performed as described above.
Statistical analysis
Statistical analysis was conducted using SPSS 19.0 software (SPSS Inc, Chicago, IL, USA). Data were expressed as mean ± standard deviation (SD). A two-tailed Student’s t-test was performed to assess the differences between any two groups. A value of p < .05 was considered statistically significant.
Results
MTT assay of LP a, b, c, d, e fragments
Cells were treated with different concentrations of LP a, b, c, d, e for 24, 48, and 72 h. The results of the MTT assay showed that, although all the LP fragments promoted HUVEC proliferation to varying degrees, only the LPe fragment showed a significant effect (Figure 2). The effects of LP fragments on HUVEC proliferation. (a). 24 h, (b). 48 h, and (c) 72 h after LP treatment. Mean ± standard deviation (SD), n = 6. *p < .05, **p < .01 vs control at the same time point.
Molecular weight analysis of LPe fragment
Linear regression was performed with molecular weight (lgMw)-Retention time (TR) plotting (see Figure 3). The equation of linear regression was as follows: The HPLC gel chromatogram of the LPe fragment.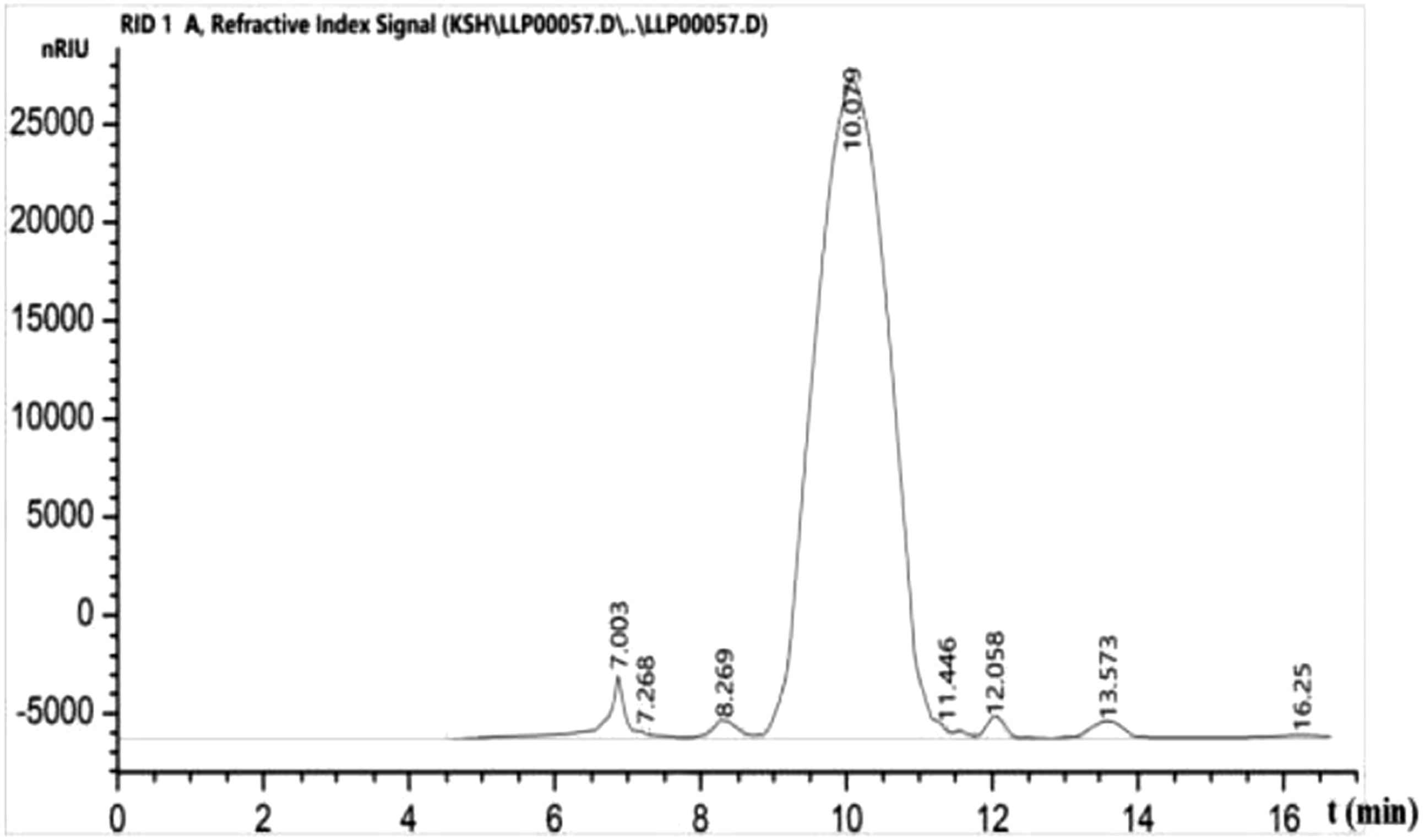
The molecular weight of LPe fragment was 8629.8 Da and the HPLC gel chromatogram is shown in Figure 3. Through the cell viability test results, it was found that fragment e had the strongest activity. Then we analyzed the structure of fragment e and finally determined that fragment e was composed of mannose and glucose, with a ratio of 5.5:2.9.
Lily polysaccharide fragment mitigated radiation-induced DSBs in HUVEC
The neutral comet assay was use to assess the rejoining of DSBs in HUVECs exposed to X-ray IR for 4 h. Representative comet images are shown in Figure 4(a), and the proportions of residual damage are shown in Figure 4(b). Substantial residual damage was observed in HUVECs after X-ray IR. Ionizing radiation causes DNA to leave the nucleus and to form a tail of different lengths, and TM and OTM are important indicators that reflect DNA damage in neutral comet assays. Treatment with the LPe fragment reduced TM and OTM in a dose-dependent manner, indicating that the LPe fragment reduces DNA damage caused by X-ray IR. The protective effects of the LPe fragment on the rejoining of double strand breaks in HUVECs irradiated for 4 h. (a) Representative images from the neutral comet assay showing the protective effects of the LPe fragment on HUVECs irradiated for 4 h. Scale bar, 50 μm. (b) Statistical analysis of TM and OTM.Mean ± standard deviation (SD), n = 3. #p < .05 vs control, *p < .05, **p < .01 vs IR.
Lily polysaccharide fragment reduced the phosphorylated H2AX foci
Phosphorylated H2AX was observed by staining with an anti-γH2AX antibody (green), and the nuclei were observed by staining with DAPI (blue). Representative images of X-ray-induced γH2AX foci are shown in Figure 5(a). The γH2AX foci of X-ray-induced DSBs were counted in HUVECs at 0.5 h after IR, and it was observed that 63.30% of cells showed γH2AX foci after IR. However, treatment with the LPe fragment at different concentrations decreased the occurrence of X-ray-induced γH2AX foci, and the effects were significant after treatment with the LPe fragment at 50, 100, and 200 μg/mL (52.67% ± 1.83%, 48.48% ± 4.93%, and 50.25% ± 4.33%, respectively; Figure 5(b)). The LPe fragment decreased X-ray-induced H2AX phosphorylation in HUVECs. (a) Digitized images of γH2AX foci in HUVECs. (b) The LPe fragment in combination with PI3K inhibitors decreased X-ray-induced H2AX phosphorylation in HUVECs. Mean ± standard deviation (SD), n = 3. #p < .05, IR vs controls, Δp < .05, caffeine or wortmannin in combination with IR vs IR alone, Φp < .05, caffeine or wortmannin in combination with the LPe fragment and IR vs the LPe fragment in combination with IR. *p < .05, ** p < .01, the LPe fragment in combination with IR vs IR alone.
Treatment of cells with caffeine or wortmannin alone did not affect γH2AX expression (45.57% ± 3.64%, 44.27% ± 4.09%, and 41.93% ± 4.16% in untreated, caffeine-, and wortmannin-treated HUVECs, respectively). Although caffeine and wortmannin could significantly suppress γH2AX expression in HUVECs exposed to 1 Gy of IR, except for the cells treated with the LPe fragment at 25 μg/mL, the other groups treated with the LPe fragment in combination with caffeine or wortmannin showed no effect on γH2AX expression (Figure 5(b)).
Discussion
The widespread application of radiation in the fields of medicine and health makes it particularly important to identify safe and effective radioprotective agents. 26 Previous studies have examined the protective effects of certain synthetic drugs,27,28 although new directions in radioprotective screening involve a variety of natural products due to their pharmacological properties and low toxicity. Different kinds of radioprotective products have been discovered, including polyphenols, anthocyanins, and polysaccharides.18,29,30 In China, L. lancifolium Thunb. is widely grown for its edible bulbs, which is considered a vegetable with high nutritional value. 31 In the past few years, polysaccharides from the edible bulbs of lily have attracted much interest due to their inoxidizability, hypoglycemic properties, immunomodulatory properties, and antitumor activities.17,32,33 However, convincing data on the biological potential of LPs are still lacking, and there are no studies on the benefits of LPs in radiotherapy patients.
The molecular weight of a polysaccharide has a significant influence on the activity of that particular polysaccharide. Polysaccharide fragments or oligosaccharides with high molecular weight can be obtained by molecular modification, which can improve their biological activities across multiple cell membranes. 34 Five LP fragments were isolated and their effects on cell proliferation were evaluated in vitro. The results showed that the LPe fragment had the most pronounced positive effect on HUVEC proliferation, and its molecular weight was 8629.8 Da, as determined by UV spectrophotometry. Compared with the other fragments, molecular weight may be a key factor affecting activity.
Endothelial cells play an important role in maintaining vascular function. Radiotherapy-impaired endothelial cells may be a leading cause of cardiovascular disease.35,36 Endothelial cells, as the most radiosensitive cell type in the vasculature, are undoubtedly a major target of radiation-induced cardiovascular injury. During radiotherapy, many free radicals are produced, and they can trigger oxidative reactions that target biological macromolecules, which can alter DNA, proteins, and lipids, causing a chain reaction of cellular damage. 37 Ionizing radiation can cause many kinds of DNA damage, and DSBs are the main factor causing cell death. The neutral comet assay can be used as tool to detect DSBs induced by X-rays.38–40 In the present study, DSBs induced by X-ray IR were detected in HUVECs by the neutral comet assay, and the results were consistent with others.41,42 Treatment with the LPe fragment at concentrations ranging from 12.5 to 100 μg/mL reduced the occurrence of DSBs caused by IR. Thus, these results indicate the distinct radioprotective effects of the LPe fragment on DSBs in HUVECs induced by X-ray IR.
Several reports support the conclusion that H2AX is specifically phosphorylated in response to DSBs caused by IR. There seems to be a direct linear correlation between the number of γH2AX foci and the number of DSBs, which suggests that each γH2AX focus may represent an individual DSB. 43 Our results showed that the number of γH2AX foci was significantly increased after X-ray IR. This finding is consistent with those of other studies, where IR impaired DNA dynamics and increased H2AX expression in HUVECs.26,44,45 The LPe fragment could protect cells from X-ray-induced DSBs, shining light on this issue. As reported here, the results of immunofluorescence assays showed that treatment with the LPe fragment at concentrations of 50, 100, and 200 μg/mL could inhibit γH2AX expression in HUVECs exposed to IR. These findings were consistent with those of neutral comet assays where the LPe fragment had a protective effect on X-ray-induced DSBs.
Double strand break activate DNA damage sensors, such as ATM and DNA-PK, which phosphorylate H2AX. γH2AX localizes to DSBs and recruits proteins involved in DSB repair.43,46 Given that both neutral comet and immunofluorescence assays proved that the LPe fragment could protect HUVECs from X-ray-induced DSBs, the question of which specific PI3K may be responsible for this process became of interest. Therefore, HUVECs were pretreated with 8 μmol/l caffeine or 20 nmol/l wortmannin, concentrations that would inhibit the actions of ATM, ATR, and DNA-PK. 47 Both caffeine and wortmannin decreased the number of γH2AX foci not only in the X-ray IR group, but also in the groups pretreated with the LPe fragment at 25 μg/mL. This observation was not surprising because caffeine and wortmannin can inhibit ATM, ATR, and DNA-PK, which are thought to catalyze γH2AX formation. However, it is unclear why higher concentrations of the LPe fragment in combination with caffeine or wortmannin could not further attenuate the γH2AX level in the X-ray IR group. The possible reason may be that higher concentrations of the LPe fragment significantly decreased the γH2AX level, and as a result, the inhibitory effects of the PI3K inhibitors were not obvious at this concentration. Although further studies are needed to investigate the mechanisms of action, these findings indicate that components of the PI3K signaling pathway, namely, DNA-PK, ATM, and ATR, may be partly involved in the protective mechanism of the LPe fragment on X-ray induced DSBs in HUVECs.
Conclusions
A fragment with a molecular weight of 8629.8 kD from LP crude extract could effectively reduce the occurrence of DSBs in HUVECs, possibly by suppressing the PI3K signaling pathway. These results indicate that the LPe fragment may be a radioprotective agent. However, further studies are required to obtain more insights into the structural features and to elucidate the protective mechanism of action and the physicochemical properties of the LPe fragment.
Footnotes
Author contribution
Zhong Guo, Shuhe Kang, and Jin Zhao: conceptualization, methodology, and software. Fei Zhao: data curation and visualization. Lei Song: software and validation. Lina Lu and Chenjing Wang: supervision. Zhong Guo, ZhiyuanLiu, and Jin Zhao: writing-reviewing and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81560508), Natural Science Foundation of Gansu Province (20YF8FA046), Fundamental Research for the Central University (Nos. 31920160048, 31920190210, 31920220012) and Key Laboratory of Environmental Ecology and Population Health in Northwest Minority Areas (No. MWZD202201).
